Abstract
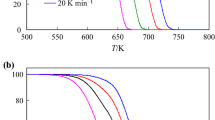
Thermal degradation and degradation kinetics of methylphenylphosphinic acid(MPPA) and diphenylphosphinic acid(DPPA) were investigated via thermogravimetric analysis(TGA) technique under non-isothermal conditions. The activation energies of the decomposition process for the two compounds were calculated through the Friedman and Kissinger-Akahira-Sunose(KAS) methods. The thermal decomposition mechanism was investigated by the Criado method based on a set of TGA data obtained at different heating rates. It was shown that the activation energies calculated from the decomposition reaction by different methods were consistent with each other. The results show that the probable model for the degradation of MPPA and DPPA agreed with the two-dimensional(D2) and three-dimensional(D4) diffusion models, respectively. Moreover, the thermodynamic functions(ΔH ≠, ΔS ≠, ΔG ≠) of the two decomposition reactions were also calculated.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Hartley F.R., The Chemistry of Organophosphorus Compounds, Vol 4, Wiley Press, New York, 1996, 145
Mitchell J., Galli D., Fire-resistant Branched Polyamide Composition, WO 2004014993, 2004
Korshak V. V., Gribova I. A., Kabachnik M. I., Krasnov A. N., Chumaevskaya A. N., Vinogradova O. V., Vinogradova S. V., Krongauz E. S., Berlin A. M., Self-lubricating Antifriction Materials, BE 834735, 1976
Simpkin J., Zinc Chloride, Titanium Dioxide, Organic Phosphinic Acid, US 627808, 1975
Titskii G. D., Stepko O. P., Litvinenko L. M., Zh. Org. Khim., 1975, 1, 1021
von Gentzkow W., Huber J., Kapitza H., Also Containing Aromatic Polyamine Hardener; for Prepregs and Composites, US 5587243, 1996
Huber J., Kapitza H., Kleiner H., Phosphorus-modified Epoxy Resins, Processes for Their Preparation and Their Use, US 5811188, 1998
Garsperini M., Ragaini F., Remondini C., J. Organomet. Chem., 2005, 690(20), 4517
Ragaini F., Cognolato C., Gasperini M., Cenini S., Angew. Chem. Int. Ed., 2003, 42(25), 2886
Ragaini F., Gasperini M., Cenini S., Adv. Synth. Catal., 2004, 346(1), 63
Gasperini M., Ragaini F., Cazzaniga C., Adv. Synth. Catal., 2005, 347(1), 105
Gasperov V., Lindoy L. F., Parkin A., Turner P., J. Mol. Struct., 2007, 839(1–3), 132
Shen W. N., Feng L. J., Feng H., Chem. J. Chinese Universities, 2012, 33(2), 353
Lü L., Xu K. Z., Qiu Q. Q., Wang G., Song J. R., Zhao F. Q., Chem. Res. Chinese Universities, 2012, 28(5), 878
Nguyen C., Lee M., Kim J., Polym. Adv. Technol., 2011, 22(5), 512
Friedman H. L., J. Polym. Sci., Part C, 1964, 6(1), 183
Kissinger H. E., J. Anal. Chem., 1957, 29(11), 1702
Akahira T., Sunose T., Res. Report Chiba Inst. Technol. (Sci.Technol.), 1971, 16, 22
Vyazovkin S., Burnham A. K., Criado J. M., Pérez-Maqueda L. A., Popescu C., Sbirrazzuoli N., Thermochim. Acta, 2011, 520, 1
Hu R. Z., Gao S. L., Zhao F. Q., Shi Q. Z., Zhang T. L., Zhang J. J., Thermal Analysis Kinetics, Science Press, Beijing, 2008, 151
Malek J., Thermochim. Acta, 1995, 267, 61
Cordes H. M., J. Phys. Chem., 1968, 72(6), 2185
Criado J. M., Pérez-Maqueda L. A., Sánchez-Jiménez P. E., J. Therm. Anal. Calorim., 2005, 82(3), 671
Du X. J., Li X. D., Yang R. J., Li Y. C., Pang S. P., Chem. Res. Chinese Universities, 2014, 30(1), 130
Neale E., Williams L. T. D., Moores V. T., J. Chem. Soc., 1956, 422
Mortimer C. T., Pure Appl. Chem., 1961, 2, 71
Singh A., Soni P. K., Singh M., Srivastava A., Thermochim. Acta, 2012, 548, 88
Wilkie C. A., Morgan A. B., Fire Retardancy of Polymeric Materials, 2nd Ed., CRC Press, Boca Raton, 2009, 120
Vlase T., Vlase G., Doca M., Doca N., J. Therm. Anal. Calorim., 2003, 72(2), 597
Vlase T., Jurca G., Doca N., Thermochim. Acta, 2001, 379, 65
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the Research Fund for the Doctoral Program of Shaanxi University of Technology, China[No.SLGQD13(2)-2].
Rights and permissions
About this article
Cite this article
Shao, X., Ji, X., Min, S. et al. Thermal kinetics and decomposition mechanism of methylphenylphosphinic acid and diphenylphosphinic acid. Chem. Res. Chin. Univ. 30, 1028–1031 (2014). https://doi.org/10.1007/s40242-014-4092-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-4092-0