Summary
Background and objective
Cigarette smoking is one of the main causes of health problems in the world and can also lead to an increased risk of frailty. Our goal is to perform a systematic review and meta-analysis of the relationship between smoking and frailty.
Methods
Researchers searched PubMed, Scopus, Google Scholar and Research Gate by using keywords through December 2018. Eligible articles were merged and a meta-analysis was conducted using the random effects method. Finally an analysis was done based on smoking status, and publication bias was assessed as well.
Results
The population analyzed comprised 61,905 people. The risk ratio (RR) of frailty based on smoking was 1.22 with a confidence interval (CI) of 1.12–1.33 (p < 0.001). In current smokers, the RR was 1.63 (p < 0.001). No significant difference was found in former smokers. The results indicated a publication bias in the studies included into the meta-analysis.
Discussion
Smoking increases the risk of disability; therefore, the provision of cigarette smoking cessation treatment can reduce this risk. As the results also showed, former smokers are less likely to be frail.
Zusammenfassung
Hintergrund und Ziel
Zigarettenrauchen ist eine der Hauptursachen gesundheitlicher Probleme in der Welt und kann auch zu einem erhöhten Risiko für Gebrechlichkeit führen. Ziel der Autoren ist es, eine systematische Übersicht und Metaanalyse zum Zusammenhang zwischen Rauchen und Gebrechlichkeit zu erstellen.
Methoden
Dazu durchsuchten die Wissenschaftler anhand von Schlüsselwörtern die Datenbanken PubMed, Scopus, Google Scholar und Research Gate bis einschließlich Dezember 2018. Geeignete Artikel wurden zusammengeführt, und es erfolgte eine Metaanalyse unter Verwendung der Random-Effects-Methode. Schließlich wurde auf der Grundlage des Raucherstatus eine Auswertung vorgenommen und auch die potenziellen Publikationsfehler untersucht.
Ergebnisse
Die ausgewertete Population umfasste 61.905 Personen. Das relative Risiko (RR) für Gebrechlichkeit auf der Basis von Rauchen betrug 1,22 mit einem Konfidenzintervall (KI) von 1,12–1,33 (p < 0,001). Bei derzeitigen Rauchern lag die RR bei 1,63 (p < 0,001). Für frühere Raucher war kein signifikanter Unterschied festzustellen. Die Ergebnisse wiesen auf eine Publikationsverzerrung der in die Metaanalyse eingeschlossenen Studien hin.
Diskussion
Rauchen erhöht das Risiko einer gesundheitlichen Beeinträchtigung, daher kann das Angebot einer Therapie zur Beendigung des Rauchens dieses Risiko vermindern. Wie die Ergebnisse ebenfalls zeigten, ist die Wahrscheinlichkeit, gebrechlich zu sein, für ehemalige Raucher geringer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Frailty is a decline in various aspects of body’s physiological system which can result in increased people’s dependency, mobility, and mortality when confronted with stressors [1,2,3,4]. Frailty is associated with negative health consequences, including mortality [5, 6], loss of activities of daily living [6, 7], hospitalization [6, 8], physical limitations [6], falls [6, 9], multimorbidity [10], and fractures [6, 11]. In some studies, the prevalence of frailty was reported as 7.4%; for pre-frailty and robustness the reported rates have been 48.1 and 44.4% [12]. The age-adjusted prevalence of frailty in a population in China was reported to be between 3.3% and 9.1% [13]. Multiple physiological and psychological risk factors related to disability have been investigated, including depression and high risk of frailty [14], alcohol consumption, Mediterranean diet and low risk of frailty [15, 16]. An important factor in health behaviors in relation to the risk of frailty can be smoking.
Smoking is one of the major health risk factors that is known to be the second leading cause of early death and disability worldwide [17]. The prevalence of daily cigarette consumption worldwide is 25% for men and 5.4% for women; this represents a decline of 28.4 and 34.4%, respectively, in men and women since 1990 [18]. As widespread studies have shown, smoking is associated with a variety of health-threatening illnesses such as respiratory diseases [19, 20], multiple sclerosis [21], mortality and cardiovascular events [22], and sarcopenia [23]. Smoking is also a risk factor for mental health and is associated with suicide [24] and psychosis [25].
Various review studies have looked at the risk factors of frailty [26, 27]. A systematic review study conducted in 2015 has examined the effects of smoking on frailty [28]. According to the study, smoking was associated with increased frailty in a community-dwelling population [28]. But this review study merely included 5 studies in a systematic review, and the meta-analysis has not been done. Furthermore, the state of smoking including former smoking and current smoking has not been documented in relation to frailty. Based on this, the aim of the current study was to systematically review and analyze the effects of smoking on frailty, as well as investigate the status of smoking (former and current) in relation to frailty.
Materials and methods
PRISMA [29] is a well-known method of systematic review and meta-analysis that is used.
Searches
The MESH keywords were used to systematically search databases to find articles related to the topic of interest. These sequences were conducted in multiple databases including PubMed, Scopus, Google Scholar and Research Gate. In total, databases searches were carried out through December 2018. References of the eligible articles were also reviewed to retrieve more studies.
Eligibility criteria
Exposure variables of this study included smoking (former smoker, current smoker, ever smoker) which was measured by self-reporting or clinical evaluation. The outcome variable included frailty which has several subcomponents that are evaluated. The evaluation of frailty was based on self-reporting and clinical evaluation. Longitudinal studies as preferential designs were eligible. If several articles were reported from the same database, the one with the larger sample size or longer follow-up time, or with more adjusted variables or the one which reported more details was selected.
Data extraction
From each of the eligible articles, various information was independently extracted by the researchers, which was then merged. This extracted information included the following: the authors of the article, the country in which the data was collected, the year of publication of the article, the follow-up period, age, gender, statistical results and adjusted variables.
Evaluation of studies quality
Qualitative assessment of eligible studies was performed using EPHPPC [30]. It measures five qualitative dimensions in studies, including bias in selection, the amount of control of confounder’s variables, the quality of the metering of the exposure and the outcome variable and also the indicator withdrawals and dropouts.
Meta-analysis
The data analysis contained two parts: first integration of the studies with each other, and second the measurement of heterogeneity. To integrate the studies with each other, the results of each study were first extracted and logarithmically converted. In the following, the results of the studies were combined using random effects method. Several subgroup analyses were also conducted to obtain more comprehensive results. Several tests measured the degree of heterogeneity: Cochrane \(\chi ^{2}\) and I2 statistic [31, 32]; funnel plots, Beeg test and Egger test and the trim-and-fill method [33,34,35]. Stata-14 software was used for the meta-analysis (Stata Corp., College Station, TX, USA).
Results
Study selection
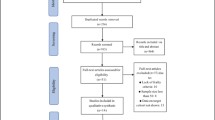
The flowchart shown in Fig. 1 illustrates the steps used to select eligible studies. In all, 2967 articles were achieved from four databases from which 2891 articles were left after removing duplicate articles. Following screening, 55 articles were evaluated for eligibility. Of the 42 articles entered in the qualitative synthesis, 22 articles were excluded based on the study design, 3 articles were excluded due to inadequate results and 3 articles due to use of the same databases. Thus, the 14 articles [36,37,38,39,40,41,42,43,44,45,46,47,48,49] listed in Table 1 were eligible to be entered into the meta-analysis.
Study selection flow diagram. © 2009 Moher et al. [29] This figure has been originally published Open Access in 2009 in the journal PLoS Med 6(7): e1000097 under https://doi.org/10.1371/journal.pmed.1000097
Quality of included studies
Table 1 shows the quality assessment of the 14 studies. Of the 14 studies, 3, 8, and 3 studies have low, moderate, and high bias in selection bias dimension, respectively. There were 3 low bias studies, 5 moderate bias studies and 6 high bias studies in the dimension of confounder’s control. In detection bias, we had 14 low bias studies. In performance bias, there were 9 and 5 low and moderate bias. In the fifth dimension, namely withdrawals and dropouts, 1 study was evaluated as low, 11 studies as moderate and 3 studies as high bias.
Smoking and frailty
A total of 61,905 people were analyzed. The risk ratio (RR) of frailty based on smoking in Fig. 2 is 1.22 and confidence interval (CI) is between 1.12–1.33 (p < 0.001).
Current and former smoking and frailty
Based on Fig. 3 in the former smokers, the RR was equal to 1.04 and CI was 0.94–1.15 (p = 0.456). In the current smokers, the RR was 1.63 and CI was 1.24–2.14 (p < 0.001).
Publication bias
The results of the funnel plot in Fig. 4 indicate asymmetries and publication bias. The Begg test (0.477) did not show any bias; the Egger test (0.002) showed bias and the trim-and-fill method imputed 8 missing studies [35]. I2 was 76.9% which indicates a high level of heterogeneity [50]. Analysis of the results according to current and former smoking status indicates a decrease in heterogeneity in the former smoking status.
Discussion
Current research was conducted with the aim of systematically reviewing and performing a meta-analysis of the relationship between smoking and frailty. Also, the status of smoking (current and former consumers) was examined in relation to frailty. Smoking increases the risk of disability up to 22%. This finding is in line with a previous study that examined the effects of smoking on frailty [28]. But the current study also looked at the state of smoking as current smokers and former smokers and the relationship with frailty. In current smokers, the risk of frailty was higher by 63%. However, this finding showed that this relationship was not significant in the former smokers. The reason why previous smokers are not significantly at risk of incapacity is related to several factors. The mechanism that puts smokers at risk of disability is unclear. As stated, smoking can affect a range of tissues and organs [51]. Therefore, the organ systems of people who have previously smoked and do not currently consume cigarettes may have the ability to compensate for the negative effects of cigarettes compared to those who are current smokers. Also, the relationship between current smoking and disability may be explained by the fact that smoking is associated with inflammation [52], which weakens muscle [53] and the body [3]. Thus, former smokers are not experiencing cigarette effects and as a result, the risk of frailty is lower. The factors leading to frailty in smokers are unknown. But its source can be multifactorial given that smoking affects a range of organs and tissues [51]. Smoking is associated with diseases such as cardiovascular disease [22], cancer [54], respiratory diseases [19, 20], multiple sclerosis [21], and sarcopenia [23]. These can have morbidities and disabilities which can lead to frailty [28]. Another possible mechanism that has been stated in this regard is that smoking involves substances that increase inflammatory mediators [52] which result in muscle loss, weight loss and fatigue—all factors engaged in frailty [3, 53]. This relationship has been confirmed [55,56,57]. In general, abandoning smoking can help reducing the risk of frailty, as smoking cessation is associated with weight gain [58, 59]. As stated, weight loss is one of the dimensions of frailty. Smoking is also associated with slow walk speed [60], which is also a component of frailty. Smoking may also reduce physical activity and, as a result, this reduced physical activity can increase the risk of frailty. As a study shows, sedentary behaviors are associated with the risk of frailty [27].
The strength of our research was that, in comparison with the previous systematic review, which only examined 5 studies, our research included 14 studies, and we also performed a meta-analysis. It also examined the status of current and former smokers in relation to frailty. Current research has introduced longitudinal studies into the systematic and meta-review that allows identification of a causal relationship between smoking and disability, while in cross-sectional studies it is not possible to investigate such a relationship. An important limitation in this research is that the degree of heterogeneity is high, which should be noted in the interpretation of the meta-analysis results. Another important limitation of the current research is that meta-analysis of different measures of frailty can affect the outcome, although, most of the studies used “Fried’s frailty scale” for the evaluation of frailty. Most of the studies that were included in the study were conducted in developed countries, so there may be restrictions on generalizing the results to other countries. The study of gender differences between men and women is another limitation that should be considered when interpreting the findings. On the other hand, the distinction between pre-frailty and frailty is also important.
References
Abate M, Di Iorio A, Di Renzo D, Paganelli R, Saggini R, Abate G. Frailty in the elderly: The physical dimension. Eura Medicophys. 2007;43(3):407–15.
Morley JE, Vellas B, van Kan GA, et al. Frailty consensus: A call to action. J Am Med Dir Assoc. 2013;14(6):392–7.
Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: Evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146–M56.
Mitnitski AB, Mogilner AJ, Rockwood K. Accumulation of deficits as a proxy measure of aging. ScientificWorldJournal. 2001;1:323–36.
Theou O, Brothers TD, Mitnitski A, Rockwood K. Operationalization of frailty using eight commonly used scales and comparison of their ability to predict all-cause mortality. J Am Geriatr Soc. 2013;61(9):1537–51.
Vermeiren S, Vella-Azzopardi R, Beckwee D, et al. Frailty and the prediction of negative health outcomes: A meta-analysis. J Am Med Dir Assoc. 2016;17(12):1163.e1161–1163.e1117.
Kojima G. Frailty as a predictor of disabilities among community-dwelling older people: A systematic review and meta-analysis. Disabil Rehabil. 2017;39(19):1897–908.
Kojima G. Frailty as a predictor of hospitalisation among community-dwelling older people: A systematic review and meta-analysis. J Epidemiol Community Health. 2016;70(7):722–9.
Kojima G. Frailty as a predictor of future falls among community-dwelling older people: A systematic review and meta-analysis. J Am Med Dir Assoc. 2015;16(12):1027–33.
Hanlon P, Nicholl BI, Jani BD, Lee D, McQueenie R, Mair FS. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: A prospective analysis of 493 737 UK Biobank participants. Lancet Public Health. 2018;3(7):e323–e32.
Kojima G. Frailty as a predictor of fractures among community-dwelling older people: A systematic review and meta-analysis. Bone. 2016;90:116–22.
Kojima G, Iliffe S, Taniguchi Y, Shimada H, Rakugi H, Walters K. Prevalence of frailty in Japan: A systematic review and meta-analysis. J Epidemiol. 2017;27(8):347–53.
Wu C, Smit E, Xue QL, Odden MC. Prevalence and correlates of frailty among community-dwelling Chinese older adults: The China health and retirement longitudinal study. J Gerontol A Biol Sci Med Sci. 2017;73(1):102–8.
Soysal P, Veronese N, Thompson T, et al. Relationship between depression and frailty in older adults: A systematic review and meta-analysis. Ageing Res Rev. 2017;36:78–87.
Kojima G, Liljas A, Iliffe S, Jivraj S, Walters K. A systematic review and meta-analysis of prospective associations between alcohol consumption and incident frailty. Age Ageing. 2018;47(1):26–34.
Wang Y, Hao Q, Su L, Liu Y, Liu S, Dong B. Adherence to the Mediterranean diet and the risk of frailty in old people: A systematic review and meta-analysis. J Nutr Health Aging. 2018;22(5):613–8.
Forouzanfar MH, Afshin A, Alexander LT, et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1659–724.
Reitsma MB, Fullman N, Ng M, et al. Smoking prevalence and attributable disease burden in 195 countries and territories, 1990–2015: A systematic analysis from the Global Burden of Disease Study 2015. Lancet. 2017;389(10082):1885–906.
Jayes L, Haslam PL, Gratziou CG, et al. SmokeHaz: Systematic reviews and meta-analyses of the effects of smoking on respiratory health. Chest. 2016;150(1):164–79.
Tamimi A, Serdarevic D, Hanania NA. The effects of cigarette smoke on airway inflammation in asthma and COPD: Therapeutic implications. Respir Med. 2012;106(3):319–28.
Degelman ML, Herman KM. Smoking and multiple sclerosis: A systematic review and meta-analysis using the Bradford Hill criteria for causation. Mult Scler Relat Disord. 2017;17:207–16.
Pan A, Wang Y, Talaei M, Hu FB. Relation of smoking with total mortality and cardiovascular events among patients with diabetes mellitus: A meta-analysis and systematic review. Circulation. 2015;132(19):1795–804.
Steffl M, Bohannon RW, Petr M, Kohlikova E, Holmerova I. Relation between cigarette smoking and sarcopenia: Meta-analysis. Physiol Res. 2015;64(3):419–26.
Poorolajal J, Darvishi N. Smoking and suicide: A meta-analysis. PLoS ONE. 2016;11(7):e156348.
Sankaranarayanan A, Mancuso S, Wilding H, Ghuloum S, Castle D. Smoking, suicidality and psychosis: A systematic meta-analysis. PLoS ONE. 2015;10(9):e138147.
Mello Ade C, Engstrom EM, Alves LC. Health-related and socio-demographic factors associated with frailty in the elderly: A systematic literature review. Cad Saude Publica. 2014;30(6):1143–68.
Kehler DS, Hay JL, Stammers AN, et al. A systematic review of the association between sedentary behaviors with frailty. Exp Gerontol. 2018;114:1–12.
Kojima G, Iliffe S, Walters K. Smoking as a predictor of frailty: A systematic review. BMC Geriatr. 2015;15:131.
Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Plos Med. 2009;6(7):e1000097.
Armijo-Olivo S, Stiles CR, Hagen NA, Biondo PD, Cummings GG. Assessment of study quality for systematic reviews: A comparison of the Cochrane collaboration risk of bias tool and the effective public health practice project quality assessment tool: Methodological research. J Eval Clin Pract. 2012;18(1):12–8.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–58.
Ioannidis JP, Patsopoulos NA, Evangelou E. Uncertainty in heterogeneity estimates in meta-analyses. BMJ. 2007;335(7626):914–6.
Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50(4):1088–101.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455–63.
Brothers TD, Kirkland S, Theou O, et al. Predictors of transitions in frailty severity and mortality among people aging with HIV. PLoS ONE. 2017;12(10):e185352.
Brunner EJ, Shipley MJ, Ahmadi-Abhari S, et al. Midlife contributors to socioeconomic differences in frailty during later life: A prospective cohort study. Lancet Public Health. 2018;3(7):e313–e22.
Chamberlain AM, St Sauver JL, Jacobson DJ, et al. Social and behavioural factors associated with frailty trajectories in a population-based cohort of older adults. BMJ Open. 2016;6(5):e11410.
Etman A, Kamphuis CB, van der Cammen TJ, Burdorf A, van Lenthe FJ. Do lifestyle, health and social participation mediate educational inequalities in frailty worsening? Eur J Public Health. 2015;25(2):345–50.
García-Esquinas E, Graciani A, Guallar-Castillón P, López-García E, Rodríguez-Mañas L, Rodríguez-Artalejo F. Diabetes and risk of frailty and its potential mechanisms: A prospective cohort study of older adults. J Am Med Dir Assoc. 2015;16(9):748–54.
Hoogendijk EO, van Hout HP, Heymans MW, et al. Explaining the association between educational level and frailty in older adults: Results from a 13-year longitudinal study in the Netherlands. Ann Epidemiol. 2014;24(7):538–544.e532.
Iwasaki M, Yoshihara A, Sato N, et al. A 5‑year longitudinal study of association of maximum bite force with development of frailty in community-dwelling older adults. J Oral Rehabil. 2018;45(1):17–24.
Kojima G, Iliffe S, Jivraj S, Liljas A, Walters K. Does current smoking predict future frailty? The English longitudinal study of ageing. Age Ageing. 2018;47(1):126–31.
Lee JS, Auyeung TW, Leung J, Kwok T, Woo J. Transitions in frailty states among community-living older adults and their associated factors. J Am Med Dir Assoc. 2014;15(4):281–6.
Myers V, Drory Y, Goldbourt U, Gerber Y. Multilevel socioeconomic status and incidence of frailty post myocardial infarction. Int J Cardiol. 2014;170(3):338–43.
Strandberg TE, Sirola J, Pitkala KH, Tilvis RS, Strandberg AY, Stenholm S. Association of midlife obesity and cardiovascular risk with old age frailty: A 26-year follow-up of initially healthy men. Int J Obes Relat Metab Disord. 2012;36(9):1153–7.
Thompson MQ, Theou O, Yu S, Adams RJ, Tucker GR, Visvanathan R. Frailty prevalence and factors associated with the frailty phenotype and frailty index: Findings from the north west Adelaide health study. Australas J Ageing. 2018;37(2):120–6.
Woods NF, LaCroix AZ, Gray SL, et al. Frailty: Emergence and consequences in women aged 65 and older in the Women’s Health Initiative Observational Study. J Am Geriatr Soc. 2005;53(8):1321–30.
Zheng Z, Guan S, Ding H, et al. Prevalence and incidence of frailty in community-dwelling older people: Beijing longitudinal study of aging II. J Am Geriatr Soc. 2016;64(6):1281–6.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Office on Smoking and Health, National Center for Chronic Disease Prevention and Health Promotion. Health effects of cigarette smoking. 2014. http://www.cdc.gov/tobacco/data_statistics/fact_sheets/health_effects/effects_cig_smoking/index.htm. Accessed 19 May 2015.
Gonçalves RB, Coletta RD, Silvério KG, et al. Impact of smoking on inflammation: Overview of molecular mechanisms. Inflamm Res. 2011;60(5):409–24.
Kanapuru B, Ershler WB. Inflammation, coagulation, and the pathway to frailty. Am J Med. 2009;122(7):605–13.
Macacu A, Autier P, Boniol M, Boyle P. Active and passive smoking and risk of breast cancer: A meta-analysis. Breast Cancer Res Treat. 2015;154(2):213–24.
Gale CR, Baylis D, Cooper C, Sayer AA. Inflammatory markers and incident frailty in men and women: The English longitudinal study of ageing. Age (Dordr). 2013;35(6):2493–501.
Barzilay JI, Blaum C, Moore T, et al. Insulin resistance and inflammation as precursors of frailty: The cardiovascular health study. Arch Intern Med. 2007;167(7):635–41.
Puts MT, Visser M, Twisk JW, Deeg DJ, Lips P. Endocrine and inflammatory markers as predictors of frailty. Clin Endocrinol (Oxf). 2005;63(4):403–11.
Tian J, Venn A, Otahal P, Gall S. The association between quitting smoking and weight gain: A systematic review and meta-analysis of prospective cohort studies. Obes Rev. 2015;16(10):883–901.
Pistelli F, Aquilini F, Carrozzi L. Weight gain after smoking cessation. Monaldi Arch Chest Dis. 2009;71(2):81–7.
North TL, Palmer TM, Lewis SJ, et al. Effect of smoking on physical and cognitive capability in later life: A multicohort study using observational and genetic approaches. BMJ Open. 2015;5(12):e8393.
Author information
Authors and Affiliations
Contributions
S. Amiri contributed to the formation of the subject, collecting and extracting data, analyzing data and writing the draft. S. Behnezhad contributed to database searches and data extraction and suggested some changes.
Corresponding author
Ethics declarations
Conflict of interest
S. Amiri and S. Behnezhad declare that they have no competing interests. All authors approved the final version of the article before submission.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amiri, S., Behnezhad, S. Systematic review and meta-analysis of the association between smoking and the incidence of frailty. Neuropsychiatr 33, 198–206 (2019). https://doi.org/10.1007/s40211-019-0315-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40211-019-0315-4