Abstract
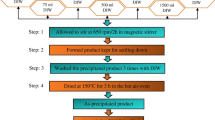
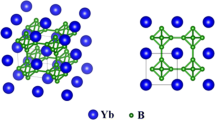
Nanocrystalline HfB2 powders were successfully synthesized by molten salt synthesis technique at 1373 K using B and HfO2 as precursors within KCl/NaCl molten salts. The results showed that the as-synthesized powders exhibited an irregular polyhedral morphology with the average particle size of 155 nm and possessed a single-crystalline structure. From a fundamental aspect, we demonstrated the molten-salt assisted formation mechanism that the molten salts could accelerate the diffusion rate of the reactants and improve the chemical reaction rate of the reactants in the system to induce the synthesis of the high-purity nanocrystalline powders. Thermogravimetric analysis showed that the oxidation of the as-synthesized HfB2 powders at 773–1073 K in air was the weight gain process and the corresponding oxidation behavior followed parabolic kinetics governed by the diffusion of oxygen in the oxide layer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fahrenholtz WG, Hilmas GE, Talmy IG, et al. Refractory diborides of zirconium and hafnium. J Am Ceram Soc 2007, 90: 1347–1364.
Gui KX, Liu FY, Wang G, et al. Microstructural evolution and performance of carbon fiber-toughened ZrB2 ceramics with SiC or ZrSi2 additive. J Adv Ceram 2018, 7: 343–351.
Ren XR, Feng PZ, Guo LT, et al. Synthesis of ultra-fine TaB2 nano powders by liquid phase method. J Am Ceram Soc 2017, 100: 5358–5362.
Guo SQ, Hu CF, Kagawa Y. Mechanochemical processing of nanocrystalline zirconium diboride powder. J Am Ceram Soc 2011, 94: 3643–3647.
Radev DD, Klissurski D. Mechanochemical synthesis and SHS of diborides of titanium and zirconium. J Mater Synth Process 2001, 9: 131–136.
Guo SQ, Ping DH, Kagawa Y. Synthesis of zirconium diboride platelets from mechanically activated ZrCl4 and B powder mixture. Ceram Int 2012, 38: 5195–5200.
Chen LY, Gu YL, Shi L, et al. Synthesis and oxidation of nanocrystalline HfB2. J Alloys Compd 2004, 368: 353–356.
Guo WM, Zhang GJ, You Y, et al. TiB2 powders synthesis by borothermal reduction in TiO2 under vacuum. J Am Ceram Soc 2014, 97: 1359–1362.
Ni DW, Zhang GJ, Kan YM, et al. Synthesis of monodispersed fine hafnium diboride powders using carbo/borothermal reduction of hafnium dioxide. J Am Ceram Soc 2008, 91: 2709–2712.
Ma L, Yu JC, Guo X, et al. Effects of HBO2 on phase and morphology of ZrB2 powders synthesized by carbothermal reduction. Ceram Int 2017, 43: 12975–12978.
Yang BY, Li JP, Zhao B, et al. Synthesis of hexagonal-prism-like ZrB2 by a sol-gel route. Powder Technol 2014, 256: 522–528.
Rabiezadeh A, Hadian AM, Ataie A. Synthesis and sintering of TiB2 nanoparticles. Ceram Int 2014, 40: 15775–15782.
Patra N, Nasiri NA, Jayaseelan DD, et al. Synthesis, characterization and use of synthesized fine zirconium diboride as an additive for densification of commercial zirconium diboride powder. Ceram Int 2016, 42: 9565–9570.
Ran SL, Sun HF, Wei YN, et al. Low-temperature synthesis of nanocrystalline NbB2 Powders by borothermal reduction in molten salt. J Am Ceram Soc 2014, 97: 3384–3387.
Bao K, Wen Y, Khangkhamano M, et al. Low-temperature preparation of titanium diboride fine powder via magnesiothermic reduction in molten salt. J Am Ceram Soc 2017, 100: 2266–2272.
Liu ZT, Wei YN, Meng X, et al. Synthesis of CrB2 powders at 800 ? under ambient pressure. Ceram Int 2017, 43: 1628–1631.
Zhang GJ, Ni DW, Zou J, et al. Inherent anisotropy in transition metal diborides and microstructure/property tailoring in ultra-high temperature ceramics—A review. J Eur Ceram Soc 2018, 38: 371–389.
Jalaly M, Gotor FJ, Sayagués MJ. Self-propagating mechanosynthesis of HfB2 nanoparticles by a magnesiothermic reaction. J Am Ceram Soc 2018, 101: 1412–1419.
Ren XR, Shang TQ, Wang WH, et al. Dynamic oxidation protective behaviors and mechanisms of HfB2-20wt%SiC composite coating for carbon materials. J Eur Ceram Soc 2019, 39: 1955–1964.
Liang H, Guan SX, Li X, et al. Microstructure evolution, densification behavior and mechanical properties of nano-HfB2 sintered under high pressure. Ceram Int 2019, 45: 7885–7893.
Fahrenholtz WG, Binner J, Zou J. Synthesis of ultra-refractory transition metal diboride compounds. J Mater Res 2016, 31: 2757–2772.
Toyoura K, Koyama Y, Kuwabara A, et al. First-principles approach to chemical diffusion of lithium atoms in a graphite intercalation compound. Phys Rev B 2008, 78: 214303.
Parthasarathy TA, Rapp RA, Opeka M, et al. A model for the oxidation of ZrB2, HfB2 and TiB2. Acta Mater 2007, 55: 5999–6010.
Zapata-Solvas E, Jayaseelan DD, Brown PM, et al. Effect of La2O3 addition on long-term oxidation kinetics of ZrB2-SiC and HfB2-SiC ultra-high temperature ceramics. J Eur Ceram Soc 2014, 34: 3535–3548.
Nickel KG. Corrosion of Advanced Ceramics: Measurement and Modelling. Dordrecht, the Netherlands: Kluwer Academic Publishers, 1994.
Harrison RW, Lee WE. Mechanism and kinetics of oxidation of ZrN ceramics. J Am Ceram Soc 2015, 98: 2205–2213.
Deal BE, Grove AS. General relationship for the thermal oxidation of silicon. J Appl Phys 1965, 36: 3770–3778.
Acknowledgements
We acknowledge financial support from the National Key R&D Program of China (No. 2017YFB0703200), National Natural Science Foundation of China (Nos. 51802100 and 51972116), Young Elite Scientists Sponsorship Program by CAST (No. 2017QNRC001), and the fund of the State Key Laboratory of Solidification Processing in NWPU (No. SKLSP201820).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, D., Fu, Q. & Chu, Y. Molten salt synthesis, formation mechanism, and oxidation behavior of nanocrystalline HfB2 powders. J Adv Ceram 9, 35–44 (2020). https://doi.org/10.1007/s40145-019-0345-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40145-019-0345-1