Abstract
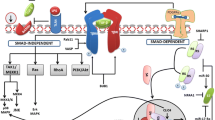
The activation of mesenchymal cells to matrix secreting myofibroblasts is central to the initiation and development of liver fibrosis. Understanding the mechanisms that govern this process is a vital step in the rational development of new anti-fibrotic therapies. Platelet-derived growth factors (PDGFs) and their cognate receptors play a key role in hepatic stellate cell and liver myofibroblast biology. In this review, we outline the major hepatic cellular sources of PDGFs and their modes of action during liver fibrogenesis. Furthermore, we examine how the PDGF pathway may represent a tractable anti-fibrotic target, and how the PDGF receptors may be harnessed to allow cell-specific delivery of therapeutics to help treat patients with liver fibrosis.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Mederacke I et al (2013) Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nat Commun 4:2823. doi:10.1038/ncomms3823
Iwaisako K et al (2014) Origins of myofibroblasts in the fibrotic liver in mice. Proc Natl Acad Sci U S A 111(32):E3297–E3305
Pinzani M et al (1989) Effects of platelet-derived growth factor and other polypeptide mitogens on DNA synthesis and growth of cultured rat liver fat-storing cells. J Clin Invest 84(6):1786–1793
Balk SD (1971) Calcium as a regulator of the proliferation of normal, but not of transformed, chicken fibroblasts in a plasma-containing medium. Proc Natl Acad Sci U S A 68(2):271–275
Kohler N, Lipton A (1974) Platelets as a source of fibroblast growth-promoting activity. Exp Cell Res 87(2):297–301
Ross R et al (1974) A platelet-dependent serum factor that stimulates the proliferation of arterial smooth muscle cells in vitro. Proc Natl Acad Sci U S A 71(4):1207–1210
Westermark B, Wasteson A (1976) A platelet factor stimulating human normal glial cells. Exp Cell Res 98(1):170–174
Betsholtz C (2003) Biology of platelet-derived growth factors in development. Birth Defects Res C Embryo Today 69(4):272–285
Andrae J, Gallini R, Betsholtz C (2008) Role of platelet-derived growth factors in physiology and medicine. Genes Dev 22(10):1276–1312
Borkham-Kamphorst E et al (2007) Pro-fibrogenic potential of PDGF-D in liver fibrosis. J Hepatol 46(6):1064–1074
Ogawa S et al (2010) Anti-PDGF-B monoclonal antibody reduces liver fibrosis development. Hepatol Res 40(11):1128–1141
Heldin CH, Eriksson U, Ostman A (2002) New members of the platelet-derived growth factor family of mitogens. Arch Biochem Biophys 398(2):284–290
Pinzani M (2002) PDGF and signal transduction in hepatic stellate cells. Front Biosci 1(7):d1720–d1726
Krampert M, Heldin CH, Heuchel RL (2008) A gain-of-function mutation in the PDGFR-beta alters the kinetics of injury response in liver and skin. Lab Invest 88(11):1204–1214
Wong L et al (1994) Induction of beta-platelet-derived growth factor receptor in rat hepatic lipocytes during cellular activation in vivo and in culture. J Clin Invest 94(4):1563–1569
• Henderson NC et al (2013) Targeting of αv integrin identifies a core molecular pathway that regulates fibrosis in several organs. Nat Med 19(12):1617–1624. This study demonstatres that PDGFRβ-Cre mice are a useful transgenic tool to mark and genetically manipulate quiescent and activated HSC in murine models of liver injury and fibrosis
Breitkopf K et al (2005) Expression patterns of PDGF-A, -B, -C and -D and the PDGF-receptors alpha and beta in activated rat hepatic stellate cells (HSC). Cytokine 31(5):349–357
Kinnman N et al (2001) Hepatic stellate cell proliferation is an early platelet-derived growth factor-mediated cellular event in rat cholestatic liver injury. Lab Invest 81(12):1709–1716
Ikeda K et al (1999) In vitro migratory potential of rat quiescent hepatic stellate cells and its augmentation by cell activation. Hepatology 29(6):1760–1767
Pinzani M et al (1996) Expression of platelet-derived growth factor and its receptors in normal human liver and during active hepatic fibrogenesis. Am J Pathol 148(3):785–800
Ikura Y et al (1997) Expression of platelet-derived growth factor and its receptor in livers of patients with chronic liver disease. J Gastroenterol 32(4):496–501
• Hayes BJ et al (2014) Activation of platelet-derived growth factor receptor alpha contributes to liver fibrosis. PLoS One 9(3):e92925. This study highlights an important role for PDGFRα in the pathogenesis of hepatic fibrosis
Carloni V et al (1997) Focal adhesion kinase and phospholipase C gamma involvement in adhesion and migration of human hepatic stellate cells. Gastroenterology 112(2):522–531
Yoshiji H et al (2005) Imatinib mesylate (STI-571) attenuates liver fibrosis development in rats. Am J Physiol Gastrointest Liver Physiol 288(5):G907–G913
Czochra P et al (2006) Liver fibrosis induced by hepatic overexpression of PDGF-B in transgenic mice. J Hepatol 45(3):419–428
Hudkins KL et al (2004) Exogenous PDGF-D is a potent mesangial cell mitogen and causes a severe mesangial proliferative glomerulopathy. J Am Soc Nephrol 15(2):286–298
Pinzani M et al (1991) Mitogenic signals for platelet-derived growth factor isoforms in liver fat-storing cells. Am J Physiol 260(3 Pt 1):C485–C491
Campbell JS et al (2005) Platelet-derived growth factor C induces liver fibrosis, steatosis, and hepatocellular carcinoma. Proc Natl Acad Sci U S A 102(9):3389–3394
Martin IV et al (2013) Platelet-derived growth factor (PDGF)-C neutralization reveals differential roles of PDGF receptors in liver and kidney fibrosis. Am J Pathol 182(1):107–117
Grappone C et al (1991) Expression of platelet-derived growth factor in newly formed cholangiocytes during experimental biliary fibrosis in rats. J Hepatol 31(1):100–109
Marra F et al (1994) Regulation of platelet-derived growth factor secretion and gene expression in human liver fat-storing cells. Gastroenterology 107(4):1110–1117
Kinnman N et al (2003) The myofibroblastic conversion of peribiliary fibrogenic cells distinct from hepatic stellate cells is stimulated by platelet-derived growth factor during liver fibrogenesis. Lab Invest 83(2):163–173
Li Z et al (2007) Transforming growth factor-beta and substrate stiffness regulate portal fibroblast activation in culture. Hepatology 46(4):1246–1256
Wells RG, Kruglov E, Dranoff JA (2004) Autocrine release of TGF-beta by portal fibroblasts regulates cell growth. FEBS Lett 559(1–3):107–110
Milani S et al (1991) Transforming growth factors beta 1 and beta 2 are differentially expressed in fibrotic liver disease. Am J Pathol 139(6):1221–1229
Sedlaczek N et al (2001) Proliferating bile duct epithelial cells are a major source of connective tissue growth factor in rat biliary fibrosis. Am J Pathol 158(4):1239–1244
Kinnman N et al (2000) PDGF-mediated chemoattraction of hepatic stellate cells by bile duct segments in cholestatic liver injury. Lab Invest 80(5):697–707
Malizia G et al (1995) Growth factor and procollagen type I gene expression in human liver disease. Gastroenterology 108(1):145–156
Bonner JC et al (1991) Differential proliferation of rat lung fibroblasts induced by the platelet-derived growth factor-AA, -AB, and -BB isoforms secreted by rat alveolar macrophages. Am J Respir Cell Mol Biol 5(6):539–547
Gay S et al (1989) Immunohistologic demonstration of platelet-derived growth factor (PDGF) and sis-oncogene expression in scleroderma. J Invest Dermatol 92(2):301–303
Friedman SL, Arthur MJ (1989) Activation of cultured rat hepatic lipocytes by Kupffer cell conditioned medium. Direct enhancement of matrix synthesis and stimulation of cell proliferation via induction of platelet-derived growth factor receptors. J Clin Invest 84(6):1780–1785
• Yoshida S et al (2014) Extrahepatic platelet-derived growth factor-β, delivered by platelets, promotes activation of hepatic stellate cells and biliary fibrosis in mice. Gastroenterology. 147(6):1378–1392. This study suggests that platelets are the primary source of PDGF during liver fibrosis
Hart C et al (1990) Purification of PDGF-AB and PDGF-BB from human platelet extracts and identification of all three PDGF dimers in human platelets. Biochemistry 29(1):166–172
Cohen MH et al (2002) Approval summary for imatinib mesylate capsules in the treatment of chronic myelogenous leukemia. Clin Cancer Res 8(5):935–942
Manley PW et al (2010) Structural resemblances and comparisons of the relative pharmacological properties of imatinib and nilotinib. Bioorg Med Chem 18(19):6977–6986
Neef M et al (2006) Oral imatinib treatment reduces early fibrogenesis but does not prevent progression in the long term. J Hepatol 44(1):167–175
Shaker ME et al (2011) Comparison of imatinib, nilotinib and silymarin in the treatment of carbon tetrachloride-induced hepatic oxidative stress, injury and fibrosis. Toxicol Appl Pharmacol 252(2):165–175
Shaker ME et al (2011) Nilotinib counteracts thioacetamide-induced hepatic oxidative stress and attenuates liver fibrosis progression. Fundam Clin Pharmacol 25(2):248–257
Teo YL, Ho HK, Chan A (2013) Risk of tyrosine kinase inhibitors-induced hepatotoxicity in cancer patients: a meta-analysis. Cancer Treat Rev 39(2):199–206
Hasinoff BB, Patel D (2010) The lack of target specificity of small molecule anticancer kinase inhibitors is correlated with their ability to damage myocytes in vitro. Toxicol Appl Pharmacol 249(2):132–139
Mughal T, Schrieber A (2010) Principal long-term adverse effects of imatinib in patients with chronic myeloid leukemia in chronic phase. Biologics 4:315–323
Goldman J, Marin D (2012) Is imatinib still an acceptable first-line treatment for CML in chronic phase? Oncology (Williston Park) 26(10):901–907
Liu Y et al (2011) Inhibition of PDGF, TGF-beta, and Abl signaling and reduction of liver fibrosis by the small molecule Bcr-Abl tyrosine kinase antagonist Nilotinib. J Hepatol 55(3):612–625
Medscape (2013) Nexavar (sorafenib) dosing, indications, interactions, adverse effects, and more. http://reference.medscape.com/drug/nexavar-sorafenib-342260 Accessed 26 Dec 2013
Wilhelm SM et al (2008) Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling. Mol Cancer Ther 7(10):3129–3140
Wang Y et al (2010) New insights into the antifibrotic effects of sorafenib on hepatic stellate cells and liver fibrosis. J Hepatol 53(1):132–144
Gonzalo T et al (2007) Local inhibition of liver fibrosis by specific delivery of a platelet-derived growth factor kinase inhibitor to hepatic stellate cells. J Pharmacol Exp Ther 321(3):856–865
Beljaars L et al (2003) The preferential homing of a platelet derived growth factor receptor-recognizing macromolecule to fibroblast-like cells in fibrotic tissue. Biochem Pharmacol 66(7):1307–1317
Borkham-Kamphorst E et al (2004) Dominant-negative soluble PDGF-beta receptor inhibits hepatic stellate cell activation and attenuates liver fibrosis. Lab Invest 84(6):766–777
Borkham-Kamphorst E et al (2004) Inhibitory effect of soluble PDGF-beta receptor in culture-activated hepatic stellate cells. Biochem Biophys Res Commun 317(2):451–462
Hao Z et al (2012) Vaccination with platelet-derived growth factor B kinoids inhibits CCl4-induced hepatic fibrosis in mice. J Pharmacol Exp Ther 342(3):835–842
Acknowledgments
The authors acknowledge the support of CORE/Children’s Liver Disease Foundation and the Wellcome Trust.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Cytokines That Affect Liver Fibrosis and Activation of Hepatic Myofibroblasts.