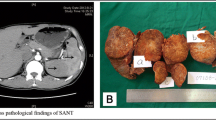
Abstract
Purpose of review
Sclerosing angiomatoid nodular transformation of the spleen (SANT) is a rare benign tumor. Its epidemiological and morphological characteristics remain unknown. The main objective of this work is to reveal the epidemiological, biological and morphological characteristics of a SANT.
Recent findings
Seventy-three publications were included in this review describing 188 cases of SANT. The average age of the patients was 45.9 years and the gender ratio was 0.66. SANT was found incidentally in 60% of the cases. In most cases (85.6%), there was no anomaly in biological tests. Abdominal ultrasound showed a hypoechoic mass which is heterogeneous. Abdominal CT scan was performed in 49.5% of the cases. It showed a hypodense (92%), heterogeneous (92%) mass. After contrast agent injection, the spoke wheel aspect was observed (100%). In all cases, the density of the mass was lower than that of the non-tumoral splenic parenchyma. Abdominal MRI was performed in 23.9% of the cases. After dynamic injection of Gadolinium, it showed the same aspect as in the CT scan. All patients were operated either immediately (88.2%) or after clinical and/or radiological changes during monitoring (9.6%).
Summary
The diagnosis of SANT remains a real challenge for the clinician and the radiologist. Establishing the differential diagnosis with other rare tumors is important in order to develop a standardized management protocol.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Sclerosing angiomatoid nodular transformation of the spleen or SANT is a rare benign vascular tumor, first described in 2004 by Martel et al. [33••] Its discovery is most often incidental. Its epidemiological profile and morphological characteristics have remained poorly known. Due to a lack of knowledge of this entity and fear of possible degeneration, splenectomy for diagnostic purposes was unavoidable in the majority of cases.
The main objective of this work is to reveal through a systematic review of the literature the epidemiological, biological and morphological characteristics of a SANT. At the end of this review, a diagnostic approach is proposed.
Methods
This review followed the PRISMA recommendations. Articles were selected through electronic databases of Medline, EMBASE, and Cochrane Library, and only articles published between January 2004 and January 2020 were included. The search was performed using the following keywords: “sclerosing,” “angiomatoid,” “nodular,” “transformation,” and “spleen”.
All articles included in our search were reviewed by two independent readers.
Inclusion Criteria
All articles related to SANT cases, published between 2004 and January 2020, in French or English language.
Exclusion Criteria
-
1.
Articles reporting the same cases or results of the same series
-
2.
Articles in languages other than English and French.
-
3.
Unpublished data and isolated abstracts.
Judging Criteria
From each article we collected demographic, clinical, biological, radiological and therapeutic data as well as postoperative and pathological examination data.
The Statistical Study
For data entry and exploration of the results, we used SPSS software in its standard version 20.0. Quantitative variables were described using means, standard deviation, limits, median and extremes. The study of the normality of quantitative variables distribution was done by the skewness and kurtosis coefficients and by the Kolmogorov–Smirnov test. The qualitative variables were described using the calculation of the observed numbers and relative frequencies (percentages).
Results
The study selection process is summarized in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart (Fig. 1).
At the end of this literature search, we retained 73/103 publications, describing 188 cases of SANT (Fig. 1) [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73]. Between 2004 and January 2020, the average publication was 4.56 articles per year.
Descriptive Study
Patients and Characteristics
In our study, we collected 188 cases of SANT, described in 73 articles. Between 2004 and January 2020, the average publication was 4.56 articles per year.
The average age of the patients was 45.9 years with a standard deviation of 15 years. This series included 75 men and 113 women with a male-to-female ratio of 0.66.
In this review, 68 patients had at least one medical or surgical antecedent. The majority of these histories included tumor pathology which was noted in 39 patients (57%).
In the majority of cases, SANT was found incidentally. It was symptomatic in 40% of the cases. Abdominal pain was the most frequent symptom. The main clinical features have been summarized in Table 1.
Biology
In most cases (85.6%), there was no anomaly in biological tests. Biological abnormalities were non-specific in 27 cases (14.4%). The various biological abnormalities are summarized in Table 2.
Radiological Examinations
Morphological features were described in 106 cases (56.4%). The splenic mass was unique in 99 cases (93.4%) The average size of the splenic mass was 50 mm, with a range from 8 to 130 mm, with clear boundaries, without capsule (n = 103, 97.1%), lobulated (n = 33; 56%) or rounded (n = 26; 44%). Its topography was respectively upper polar (58%), lower polar (21%) and medio-splenic (21%). Splenomegaly was not constant (n = 15, 14%). Other associated extra-splenic masses were reported in 17 cases (9%), most often in the liver and the mesentery.
Abdominal ultrasound, performed in 50 cases (26.6%), had described an hypoechoic mass (n = 44; 88%), heterogeneous (n = 50; 100%) with intra-tumoral calcification (n = 6; 12%).
Abdominal CT scan was performed in 93 cases (49.5%), including 66 cases with injection of contrast media (71%).
The SANT was spontaneously hypodense (n = 86; 92%), heterogeneous (n = 48; 92%), without calcification (95%) and with necrotic-hemorrhagic areas (100%). Contrast media uptake from the arterial phase in the periphery sparing the center (n = 45; 94%) with centripetal and progressive uptake at the portal phase realizing the spoke wheel appearance (n = 66; 100%). In all cases, the density of the mass was lower than that of the non-tumoral splenic parenchyma at arterial and portal phases.
The late vascular phase, performed in 48 cases, showed preservation of the spoke wheel appearance with central scar in 40 cases (83%), and homogenization with the non-tumoral splenic parenchyma in 8 cases (17%).
Abdominal MRI was performed in 45 cases (23.9%), with T1-weighted image (n = 40), T2-weighted image (n = 43), in-phase and out-of-phase sequences (n = 8), diffusion imaging (n = 12) with b values ranging from 700 to 900 and dynamic Gadolinium injection (n = 34).
The lesion appearance was on T1, hypointense (n = 18; 45%), isointense (n = 18; 45%) and hyperintense (n = 4; 10%). The mass was heterogeneous (n = 6; 75%) and homogeneous (n = 2; 25%). On T2, the signal was hypointense (n = 30; 70%), hyperintense (n = 9; 21%) and isointense (n = 4; 9%) (2,3,6,10). The mass was heterogeneous in all cases, by the presence of a central hypointense scar and hyperintense areas of peripheral and septal disposition. A signal drop on in-phase was described in 7 cases (n = 7/8), testifying to the presence of iron overload. Diffusion imaging did not show diffusion restriction with an average ADC value (9 cases/12) estimated at approximately 1.20 × 10–3 mm2/s.
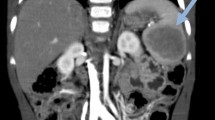
The lesion characteristics after dynamic injection of Gadolinium being detailed in 28 cases, is similar to that described previously on the CT scan. Figure 2 shows the different characteristics of SANT on CT scan and MRI.
Imaging characteristics of SANT. a Axial CT image shows predominantly hypodense mass with lobulated contours with rim-style contrast enhancement of external borders of lesion (arrow). b Fat-saturated T1-weighted image shows central hyperintensity that is consistent with hemorrhage. c T2-weighted MR image shows spoke wheel pattern of lesion that is predominantly hyperintense with central hypointense areas with hyperintense radiations towards center of lesion. d and e Postcontrast arterial (d) and delayed venous phase (e) T1-weighted MR images clearly show progressive enhancement from periphery to center of lesion, which is similar to spoke wheel pattern. [11•] Reproduced courtesy of M Karçaaltıncaba
Other Explorations
Positron Emission Tomography (PET Scan)
FDG uptake was present in 17 cases (81%) of the 21 cases performed.
Technetium-99 m Scintigraphy
The scintigraphy was negative in the 3 cases performed.
Fine Needle Biopsy (FNA)
After microscopic and immunohistochemical study, FNA confirmed the diagnosis in four cases (66.66%) among the six cases performed.
Therapeutic Management
The therapeutic strategy was detailed in all patients. The majority of patients were operated immediately and only 22 patients, or 12%, were monitored regularly.
Monitoring
Twenty-two patients (12%) were followed up regularly with a median duration of monitoring estimated at 16 months (Extremes:3–60 months).
Surveillance was clinical, radiological in all cases by PET scan in two cases (9%) and anatomopathological by radio-guided needle biopsy in six cases.
Clinically, none of the cases presented a complication.
On imaging, no change in the appearance of enhancement or morphological characteristics of the mass was reported except for a discrete progression in size found in 19 cases (86%).
On PET scan, fixation was increased in two cases.
Given the appearance of clinical and/or radiological modifications (the progression in size) or of fixation in the PET scan, 18 cases (82%) were operated. There were no cases of tumor degeneration.
The 4 cases of SANT confirmed by FNA (18.2%), had benefited from exclusive surveillance.
Surgery
Surgery was decided either immediately in 88.2% of cases or after clinical and/or radiological changes during monitoring in 9.6% of cases. The approach was specified in only 41 cases. Laparoscopy was performed in 68.3% of cases, followed by laparotomy in 31.7%. The type of surgery was total splenectomy in 180 cases. The average of hospitalization was 8 days (extremes: 2–6 days) with a simple postoperative course in 98.9% of cases and no recurrence.
Anatomopathological Examination of the Surgical Specimen
Anatomopathological examination of the surgical specimen (184 patients) revealed a single lesion in 169 cases (93.8%) with a size of approximately 51 mm (between 10 and 170 mm), circumscribed in 143 cases (79%) and not.
encapsulated in 109 cases (98.2%). It was nodular in 153 cases (84%) with central fibrotic scarring in 151 cases (98.7%).
Microscopic examination described angiomatoid nodules within a fibrosclerotic stroma in 181 cases (99%) without evidence of intra-tumor necrosis. Immunohistochemical study by CD34, CD31 and CD8 had confirmed the diagnosis of SANT. The tumor showed 3 types of blood vessels: capillary (CD34 + /CD31 + /CD8-) in 166 cases (91.2%), sinusoid (CD34-/CD31 + /CD8 +) in 164 cases (90.1%) and venule (CD34-/CD31 + /CD8-) in 165 cases (90.6%). No cases of degeneration were observed.
In addition, extra-splenic masses were identified which were associated with the splenic masses already described. They were usually located in the liver (35%), mesentery (25%), kidney (15%) and stomach (10%). The different histological types described are cystic type (35%), fibro-calcified pseudo tumoral (30%) and hemangioma (10%).
Discussion
At the end of this study, SANT is a rare benign tumor, poorly elucidated, without risk of degeneration or complication. It affects mostly women with an average age of 45 years.
It is usually asymptomatic and is discovered incidentally (60%). Splenomegaly is rare (14%). On imaging, it appears as a single, well-limited mass without capsule.
On ultrasound, it was generally hypoechoic (88%) and heterogeneous (100%).
On CT scan, it was spontaneously hypodense (n = 86; 92%), heterogeneous (n = 48; 92%), without necrotic-hemorrhagic areas (100%) and calcification (95%).
It enhances from the arterial phase (n = 45, 94%) in the periphery in a progressive and centripetal way presenting septa realizing the aspect of a spoke wheel with a central scar which persists unenhanced at the portal and late vascular phases. Apart from the scar, the density of the tumor mass remained lower than that of the non-tumor splenic parenchyma at portal time (100%) with possible homogenization at late vascular phase (77%).
On MRI, the tumor was on T1-weighted hyposignal (n = 18; 45%), T2 weighted hyposignal (n = 30; 70%), heterogeneous with septal and peripheral areas in hypersignal, with no evidence of restriction on diffusion sequences. A signal drop in-phase could be suggestive. The vascular attitude after dynamic injection of Gadolinium is superimposable to that described on the CT scan.
Nevertheless, this radiological presentation is sometimes misleading and may mimic other benign, borderline or malignant conditions (primary or secondary). Knowledge of alternative diagnoses can help avoid unnecessary splenectomy.
Regarding malignant tumors or tumors with malignant potential, in addition to lymphomas and metastases, three main tumors may pose diagnostic difficulties in the absence of clinical and/or biological guidance: hemangiopericytoma, hemangioendothelioma and primary angiosarcoma of the spleen (Table 3).
Splenic lymphoma, in its unique form, may be difficult to diagnose. But, unlike a SANT, it is usually poorly limited, poorly enhancing after contrast media injection and is responsible for diffusion restriction [74, 75].
Metastasis can occur as an isolated mass, synchronous or metachronous to a primary tumor. It is most often poorly enhanced after injection, except for metastases of renal tumors and chorioepitheliomas. Targeted contrast uptake and a halo appearance with peripheral T2 weighted hypersignal are suggestive [76•].
Hemangiopericytoma is a mesenchymal tumor that may be revealed by fever and/or weight loss [77•]. It affects males preferentially. Unlike SANT, it is most often partially calcified with a T2 weighted hypersignal [77, 78].
Hemangioendothelioma is a rare borderline tumor of vascular origin, observed in middle-aged adults, with a gender ratio of 1:1. The clinical and biological signs are not specific. Unlike SANT, it is characterized by a T2 weighted hypersignal with the presence of necrotic-hemorrhagic areas. The lesion center is enhanced at late phase [79, 80].
Primary angiosarcoma of the spleen is a tumor of poor prognosis, discovered at the metastatic stage. It affects elderly subjects with a gender ratio of 1:1. Abdominal pain is present in more than 80% of cases associated with splenomegaly. Spontaneous rupture of the spleen is a common complication. Unlike SANT, the tumor mass is poorly limited with cystic and necrotic-hemorrhagic areas with massive calcifications in stars, causing a diffuse and heterogeneous enhancement [79] (Fig. 3).
In addition, other benign tumor conditions can sometimes be confused with SANT, such as cavernous hemangioma, inflammatory pseudo tumor, or littoral cell angiomas in this case (Table 3).
Cavernous hemangioma is the most frequent benign tumor of the spleen occurring in middle-aged adults without gender predominance. Unlike SANT, this lesion is on T1 and T2 weighted hyper signal, containing cysto-hemorrhagic areas, centripetal enhancement, and peripheral clumps with homogenization at late phase, without real scar [79, 81].
Littoral cell angioma is a rare tumor specific to the spleen. Unlike SANT, it is symptomatic, often associated with splenomegaly with biological hypersplenism. It is in frank T2 weighted hyposignal, with a multi and micronodular appearance, taking the contrast homogeneously, without central scar at the late phase [79].
Inflammatory pseudotumor or inflammatory myofibroblastic proliferation of the spleen is of exceptional splenic localization. It is often symptomatic, revealed by pain in the left hypochondrium, splenomegaly, fever and/or an inflammatory state. On imaging, the T2 weighted hypersignal is an additional evocative argument [75, 82
In order to be able to orientate oneself towards one pathology or another, when a hypoechoic mass of the spleen is found, enhanced after injection of contrast media, certain semiological criteria must be checked, in this case: the association with a splenomegaly, the appearance of the lesion’s boundaries, the detection of calcifications, areas of necrosis and/or hemorrhage and the study of the attitude of the lesion after dynamic injection of contrast media (Fig. 4).
Indeed, a diagnostic approach could be proposed to facilitate the diagnosis of SANT in its typical form. We, thus, require the existence of these three conditions together (Fig. 5):
-
(1)
Check for the absence of signs predictive of malignancy although they are not specific, such as a non-painful splenomegaly, ill-defined boundaries (lymphoma, angiosarcoma + +), lesion heterogeneity (necrotic and/or hemorrhagic areas) (angiosarcoma + + /hemangioendothelioma), presence of massive calcification (hemangiopericytoma or angiosarcoma), intense enhancement (angiosarcoma), central enhancement (hemangioendothelioma) or target enhancement (metastasis).
-
(2)
Gather the following radiological signs:
-
Single, hypoechoic mass
-
Centripetal enhancement,” spoke wheel” appearance
-
Non-enhanced central scar
-
Intensity of contrast uptake “non-tumor splenic parenchyma”
-
Heterogeneous T2 weighted hyposignal due to the presence of hyperintense septa
-
No broadcast restrictions
-
Absence of calcifications
-
-
(3)
Check for the absence of clinical and/or biological inflammatory signs
However, in all cases, radiological signs should not be interpreted independently of the clinical and/or biological context.
The interpretation of radiological signs requires a dynamic acquisition after injection of contrast medium with a late phase.
Limits
Our study was based on the analysis of 73 articles, including only case reports.
Although our sample included 188 cases, the diagnostic approach to this condition remains poorly elucidated and non-coded (debatable on a case-by-case basis).
In addition, the use of imaging was not systematic, and radiological data were not consistently reported in all publications.
Indeed, only 35% of the cases had undergone a CT scan with dynamic injection of contrast medium and 24% of the cases had had an abdominal MRI according to a non-standardized protocol in all patients.
Conclusion
Despite the continuous progress of imaging techniques, the diagnosis of SANT remains a real challenge for the clinician and the radiologist. Due to the rarity of this lesion, the radiological exploration and management protocol is still not standardized. The differential diagnosis is essentially with other rare tumors without specific diagnostic criteria.
Highlights
-
SANT in its typical form is asymptomatic without splenomegaly
-
Imaging often shows, in the absence of malignancy criteria, a unique, well limited lesion which is hypoechoic on ultrasound and has an early centripetal and progressive enhancement resulting in a spoke wheel appearance (CT-MRI) with a non-enhanced central scar on the different injection phase. On T2-weighted imaging, it exhibits a heterogeneous hyposignal lesion and shows no restricted diffusion on diffusion-weighted imaging.
-
In the case of a typical form of SANT, monitoring could be proposed.
-
Splenectomy might be indicated if symptomatic or contains calcifications or contains heterogeneous zones or has a rapid evolution.
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Matsubara K, Oshita A, Nishisaka T, Sasaki T, Matsugu Y, Nakahara H, et al. A case of sclerosing angiomatoid nodular transformation of the spleen with increased accumulation of fluorodeoxyglucose after 5-year follow-up. Int J Surg Case Rep. 2017;39:9–13.
Watanabe M, Shiozawa K, Ikehara T, Kanayama M, Kikuchi Y, Ishii K, et al. A case of sclerosing angiomatoid nodular transformation of the spleen: correlations between contrast-enhanced ultrasonography and histopathologic findings. Journal of Clinical Ultrasound févr. 2014;42(2):103–7.
Eusébio M, Sousa AL, Vaz AM, da Silva SG, Milheiro MA, Peixe B, et al. A case of sclerosing angiomatoid nodular transformation of the spleen: Imaging and histopathological findings. Gastroenterología Hepatología. 2016;39(9):600–3.
Koyama R, Minagawa N, Maeda Y, Shinohara T, Hamada T. A sclerosing angiomatoid nodular transformation (SANT) mimicking a metachronous splenic metastasis from endometrioid cancer and ovarian cancer. Int J Surg Case Rep. 2019;9(65):292–5.
Gaeta R, Donati F, Kauffmann EF, Campani D. A Splenic IgG4+ sclerosing angiomatoid nodular transformation (SANT) treated by hemisplenectomy: a radiologic, histochemical, and immunohistochemical study. Appl Immunohistochem Mol Morphol. 2020;28(5):e40–6.
Chikkappa MG, Morrison C, Lowe A, Antrim R, Swirsky DM, Gokhale J. Case report and magnetic resonance images of sclerosing angiomatoid nodular transformation (SANT) of the spleen. Case Reports. 2009;2009:bcr0720092131.
Weinreb I, Bailey D, Battaglia D, Kennedy M, Perez-Ordoñez B. CD30 and Epstein-Barr virus RNA expression in sclerosing angiomatoid nodular transformation of spleen. Virchows Archiv Juill. 2007;451(1):73–9.
Lee J-C, Lien H-C, Hsiao C-H. Coexisting sclerosing angiomatoid nodular transformation of the spleen with multiple calcifying fibrous pseudotumors in a patient. J Formosan Med Assoc. 2007;106(3):234–9.
Menozzi G, Maccabruni V, Ferrari A, Tagliavini E. Contrast sonographic appearance of sclerosing angiomatoid nodular transformation of the spleen. J Ultrasound. 2015;18(3):305–7.
•Karaosmanoglu DA, Karcaaltincaba M, Akata D. CT and MRI Findings of Sclerosing Angiomatoid Nodular Transformation of the Spleen: Spoke Wheel Pattern. Korean J Radiol. 2008;9(Suppl):S52. This article gives a typical image of SANT on MRI and CT scan.
Kashiwagi S, Kumasaka T, Bunsei N, Fukumura Y, Yamasaki S, Abe K, et al. Detection of Epstein-Barr virus-encoded small RNA-expressed myofibroblasts and IgG4-producing plasma cells in sclerosing angiomatoid nodular transformation of the spleen. Virchows Arch. 2008;453(3):275–82.
Lapa C, Steger U, Ritter CO, Wild V, Herrmann K. Differentiation of an unclear splenic lesion in a patient with cholangiocarcinoma. Clin Nuclear Med. 2014;39(5):470–1.
Feng Y-M, Huang YC, Tu C-W, Kao W-S, Tu D-G. Distinctive PET/CT features of splenic SANT. Clin Nuclear Med. 2013;38(12):e465.
Lee D, Wood B, Formby M, Cho T. F-18 FDG-avid sclerosing angiomatoid nodular transformation (SANT) of the spleen: case study and literature review. Pathology. 2007;39(1):181–3.
Kakisaka T, Kamiyama T, Yokoo H, Orimo T, Wakayama K, Tsuruga Y, et al. Hand-assisted laparoscopic splenectomy for sclerosing angiomatoid nodular transformation of the spleen complicated by chronic disseminated intravascular coagulation: a case report: SANT complicated by DIC. Asian J Endosc Surg. 2014;7(3):275–8.
Raman SR, Parithivel VS, Niazi M. Image of the month sclerosing angiomatoid nodular transformation of the spleen. Arch Surg. 2010;145(2):205–6.
Kornprat P, Beham-Schmid C, Parvizi M, Portugaller H, Bernhardt G, Mischinger HJ. Incidental finding of sclerosing angiomatoid nodular transformation of the spleen. Wien Klin Wochenschr. 2012;124(3–4):100–3.
Diebold J, Le Tourneau A, Marmey B, Prevot S, Müller-Hermelink HK, Sevestre H, et al. Is sclerosing angiomatoid nodular transformation (SANT) of the splenic red pulp identical to inflammatory pseudotumour? Report of 16 cases. Histopathology. 2008;53(3):299–310.
Chouchane Mlik O, Lardenois S, Salmon R, Servois V, Sastre-Garau X. La transformation angiomatoïde nodulaire sclérosante de la rate. Ann Pathol. 2008;28(4):317–20.
Chen Y-C, Huang J-W, Su W-L, Chang W-T, Kuo K-K. Laparoscopic approach is the treatment of choice for sclerosing angiomatoid nodular transformation of the spleen. Kaohsiung J Med Sci. 2015;31(9):496–7.
Bushati M, Sommariva A, Montesco M, Rossi C. Laparoscopic splenectomy for sclerosing angiomatoid nodular transformation of the spleen. J Min Access Surg. 2017;13(4):309.
Kim K-H, Lee S, Youn SH, Lee MR, Kim MC, Rha S-H, et al. Laparoscopic splenectomy for sclerosing angiomatoid nodular transformation of the spleen. J Korean Surg Soc. 2011;80(Suppl 1):S59–62.
Cao Z, Wang Q, Li J, Xu J, Li J. Multifocal sclerosing angiomatoid nodular transformation of the spleen: a case report and review of literature. Diagn Pathol. 2015;10:1–5.
Lim H, Tan C, Teo L, Ho C. Multimodality imaging of splenic sclerosing angiomatoid nodular transformation. Singapore Med J. 2015;56(06):e96–9.
Chang K-C, Lee J-C, Wang Y-C, Hung L-Y, Huang Y, Huang W-T, et al. Polyclonality in sclerosing angiomatoid nodular transformation of the spleen. Am J Surg Pathol. 2016;40(10):1343–51.
Nagai Y, Hayama N, Kishimoto T, Furuya M, Takahashi Y, Otsuka M, et al. Predominance of IgG4+ plasma cells and CD68 positivity in sclerosing angiomatoid nodular transformation (SANT). Histopathology. 2008;53(4):495–8.
Zeeb LM, Johnson JM, Madsen MS, Keating DP. Sclerosing angiomatoid nodular transformation. Am J Roentgenol. 2009;192(5):W236–8.
Önder S, Kosemehmetoglu K, Himmetoglu Ç, Firat P, Uner A. Sclerosing angiomatoid nodular transformation (SANT) of spleen: a case report describing cytology, histology, immunoprofile and differential diagnosis: sclerosing angiomatoid nodular transformation of spleen. Cytopathology. 2012;23(2):129–32.
Imamura Y, Nakajima R, Hatta K, Seshimo A, Sawada T, Abe K, et al. Sclerosing angiomatoid nodular transformation (SANT) of the spleen: a case report with FDG-PET findings and literature review. Acta Radiologica Open. 2016;5(8):205846011664979.
Koreishi AF, Saenz AJ, Fleming SE, Teruya-Feldstein J. Sclerosing angiomatoid nodular transformation (SANT) of the spleen: a report of 3 cases. Int J Surg Pathol. 2009;17(5):384–9.
Falk GA, Nooli NP, Morris-Stiff G, Plesec TP, Rosenblatt S. Sclerosing angiomatoid nodular transformation (SANT) of the spleen: case report and review of the literature. Int J Surg Case Rep. 2012;3(10):492–500.
Gutzeit A, Stuckmann G, Dommann-Scherrer C. Sclerosing angiomatoid nodular transformation (SANT) of the spleen: sonographic finding. J Clin Ultrasound. 2009;37(5):308–11.
••Martel M, Cheuk W, Lombardi L, Lifschitz-Mercer B, Chan JKC, Rosai J. Sclerosing Angiomatoid Nodular Transformation (SANT): Report of 25 cases of a distinctive benign splenic lesion. Am J Surg Pathol. 2004;28(10):1268–1279. This article is the first article describe SANT as a separate and new entity.
Zhang S, Yang W, Xu H, Wu Z. Sclerosing angiomatoid nodular transformation of spleen in a 3-year-old child. Indian Pediatr. 2015;52(12):1081–3.
Dutta D, Sharma M, Mahajan V, Chopra P. Sclerosing angiomatoid nodular transformation of spleen masquerading as carcinoma breast metastasis: importance of splenic biopsy in obviating splenectomy. Indian J Pathol Microbiol. 2016;59(2):223.
Niu M, Liu A, Wu J, Zhang Q, Liu J. Sclerosing angiomatoid nodular transformation of the accessory spleen: a case report and review of literature. Medicine. 2018;97(26):e11099.
Murthy V, Miller B, Nikolousis EM, Pratt G, Rudzki Z. Sclerosing angiomatoid nodular transformation of the spleen. Clin Case Rep. 2015;3(10):888–90.
Kim H-H, Hur Y-H, Koh Y-S, Kim J-C, Kim H-J, Kim J-W, et al. Sclerosing angiomatoid nodular transformation of the spleen related to IgG4-associated disease: report of a case. Surg Today. 2013;43(8):930–6.
Lee M, Caserta M, Tchelepi H. Sclerosing angiomatoid nodular transformation of the spleen. Ultrasound Quart. 2014;30(3):241–3.
Efared B, Sidibé IS, Erregad F, Hammas N, Chbani L, El Fatemi H. Sclerosing angiomatoid nodular transformation of the spleen (SANT) in a patient with clear cell carcinoma of the uterus: a case report. J Med Case Rep. 2018;12:15.
Kuo T, Chen T-C, Lee L. Sclerosing angiomatoid nodular transformation of the spleen (SANT): Clinicopathological study of 10 cases with or without abdominal disseminated calcifying fibrous tumors, and the presence of a significant number of IgG4+ plasma cells. Pathol Int. 2009;59(12):844–50.
Raman SP, Singhi A, Horton KM, Hruban RH, Fishman EK. Sclerosing angiomatoid nodular transformation of the spleen (SANT): multimodality imaging appearance of five cases with radiology–pathology correlation. Abdom Imaging. 2013;38(4):827–34.
Corrado G, Tabanelli V, Biffi R, Petralia G, Tinelli A, Peccatori FA. Sclerosing angiomatoid nodular transformation of the spleen during pregnancy: diagnostic challenges and clinical management: SANT of the spleen during pregnancy. J Obstetr Gynaecol Res. 2016;42(8):1021–5.
Delgado MA, Fleming A, El-Gohary Y, Abdelhafeez A, Santiago T, McCarville ME, et al. Sclerosing angiomatoid nodular transformation of the spleen in a four-year-old with anemia. J Pediatr Surg Case Rep. 2018;38:53–6.
Huang XD, Jiao HS, Yang Z, Chen CQ, He YL, Zhang XH. Sclerosing angiomatoid nodular transformation of the spleen in a patient with Maffucci syndrome: a case report and review of literature. Diagn Pathol. 2017;12:1–6.
Bamboat ZM, Masiakos PT. Sclerosing angiomatoid nodular transformation of the spleen in an adolescent with chronic abdominal pain. J Pediatr Surg. 2010;45(7):e13–6.
Sitaraman L, Linn JG, Matkowskyj KA, Wayne JD. Sclerosing angiomatoid nodular transformation of the spleen masquerading as a sarcoma metastasis. Rare Tumors. 2010;2(4):45.
Demirci I, Kinkel H, Antoine D, Szynaka M, Klosterhalfen B, Herold S, Janßen H. Sclerosing angiomatoid nodular transformation of the spleen mimicking metastasis of melanoma: a case report and review of the literature. J Med Case Rep. 2017;11:1–6.
Langer R, Dinges J, Dobritz M, Brauer RB, Perren A, Becker K, et al. Sclerosing angiomatoid nodular transformation of the spleen presenting as a rapidly growing tumour in a patient with rectal cancer. Case Rep. 2009;2009:bcr1120081191.
Nagai Y, Satoh D, Matsukawa H, Shiozaki S. Sclerosing angiomatoid nodular transformation of the spleen presenting rapid growth after adrenalectomy: report of a case. Int J Surg Case Rep. 2017;30:108–11.
Capaldi M, Fransvea P, Ricci G, Stella F, Trombetta S, Cerasari S, Cataldi C, Casale S, Marini P. Sclerosing angiomatoid nodular transformation (SANT) of spleen mimicking a splenic abscess: Case report and review of the literature. Int J Surg Case Rep. 2019;56:1-4.
Budzyński A, Demczuk S, Kumiega B, Migaczewski M, Matłok M, Zub-Pokrowiecka A. Sclerosing angiomatoid nodular transformation of the spleen treated by laparoscopic partial splenectomy. Videosurg Other Miniinvasive Techniq. 2011;4:249–55.
Cafferata B, Pizzi M, D’Amico F, Mescoli C, Alaggio R. Sclerosing angiomatoid nodular transformation of the spleen, focal nodular hyperplasia and hemangioma of the liver: a tale of three lesions. Pathol—Res Pract. 2016;212(9):855–8.
Wang T-B, Hu B-G, Liu D-W, Gao Z-H, Shi H-P, Dong W-G. Sclerosing angiomatoid nodular transformation of the spleen: a case report and literature review. Oncol Lett. 2016;12(2):928–32.
Gooch C, Gaballah AH, Nelson J, Wade M, Chen R. Sclerosing angiomatoid nodular transformation of the spleen: a case report of thrombocytopenia and a hypervascular splenic mass. Radiol Case Reports avr. 2019;14(4):521–5.
Agrawal M, Uppin SG, Bh S, Uppin MS, Bheerappa N, Challa S. Sclerosing angiomatoid nodular transformation of the spleen: a new entity or a new name? Turk J Pathol. 2016;32(3).
Atas H, Bulus H, Akkurt G. Sclerosing angiomatoid nodular transformation of the spleen: an uncommon cause of abdominal pain. Euroasian J of Hepato-Gastroenterol. 2017;7(1):89–91.
El Demellawy D, Nasr A, Alowami S. Sclerosing angiomatoid nodular transformation of the spleen: case report. Pathol—Res Practice. 2009;205(4):289–93.
Pinheiro JL, Catarino S, Duarte L, Ferreira M, Simão R, Pinheiro LF, Casimiro C. Sclerosing angiomatoid nodular transformation of the spleen: case report of a metastatic carcinoma-simulating disorder. J Surg Case Rep. 2019;2019(9):rjz249.
Kim HJ, Kim KW, Yu ES, Byun JH, Lee SS, Kim JH, et al. Sclerosing angiomatoid nodular transformation of the spleen: clinical and radiologic characteristics. Acta Radiol. 2012;53(7):701–6.
Lewis RB, Lattin GE, Nandedkar M, Aguilera NS. Sclerosing angiomatoid nodular transformation of the spleen: CT and MRI features with pathologic correlation. Am J Roentgenol. 2013;200(4):W353–60.
Thacker C, Korn R, Millstine J, Harvin H, Van Lier Ribbink JA, Gotway MB. Sclerosing angiomatoid nodular transformation of the spleen: CT, MR, PET, and 99mTc-sulfur colloid SPECT CT findings with gross and histopathological correlation. Abdom Imaging. 2010;35(6):683–9.
Nomura R, Tokumura H, Katayose Y, Nakayama F, Iwama N, Furihata M. Sclerosing angiomatoid nodular transformation of the spleen: lessons from a rare case and review of the literature. Intern Med. 2019;58(10):1433–41.
Subhawong TK, Subhawong AP, Kamel I. Sclerosing angiomatoid nodular transformation of the spleen: multimodality imaging findings and pathologic correlate. J Comput Assist Tomogr. 2010;34(2):206–9.
Wang HL, Li KW, Jian WA. Sclerosing angiomatoid nodular transformation of the spleen: report of five cases and review of literature. Chin Med J. 2012;125(13):2386–9.
Martínez Martínez PJ, Solbes Vila R, Bosquet Úbeda CJ, Roig JM. Sclerosing angiomatoid nodular transformation of the spleen: a case report. Rev Esp Enferm Dig. 2017;109(3):214–5.
Pelizzo G, Villanacci V, Lorenzi L, Doria O, Caruso AM, Girgenti V, Unti E, Putignano L, Bassotti G, Calcaterra V. Sclerosing angiomatoid nodular transformation presend nodulartransformation presenting witha bdominal hemorrhage: first report in infancy. Pediatr Rep. 2019;11(2):7848.
Dazé Y, Gosselin J, Bernier V. Transformation angiomatoïde nodulaire sclérosante de la rate (TANS): présentation d’un cas. Ann Pathol. 2008;28(4):321–3.
Frampas E, Regenet N, Moreau A, Léauté F, Dupas B. Transformation nodulaire angiomatoïde sclérosante de la rate. J Radiol. 2009;90(11):1755–8.
Yoshimura N, Saito K, Shirota N, Suzuki K, Akata S, Oshiro H, et al. Two cases of sclerosing angiomatoid nodular transformation of the spleen with gradual growth: usefulness of diffusion-weighted imaging. Clin Imaging. 2015;39(2):315–7.
Cao J-Y. Ultrasonography of sclerosing angiomatoid nodular transformation in the spleen. World J Gastroenterol. 2010;16(29):3727.
Opletalová K, Michels J-J, Chasle J. Une tumeur de la rate. Ann Pathol. 2009;29(6):512–4.
Zhou J, Zhang D, Hu G, Zheng X, Shen Q, Li W, et al. Upregulated expression of CD30 protein in sclerosing angiomatoid nodular transformation (SANT): studies of additional 4 cases and analyses of 6 cases previously published cases. Int J Clin Exp Pathol. 2015;8(6):6064–9.
•Bowerson M, Menias CO, Lee K, Fowler KJ, Luna A, Yano M, et al. Hot spleen: hypervascular lesions of the spleen. Abdom Imaging. 2015;40(7):2796–2813. This article gives details about splenic lymphoma.
Vilgrain R. Imagerie de l’abdomen. Lavoisier; 2010. p. 1082.
•Compérat E, Bardier-Dupas A, Camparo P, Capron F, Charlotte F. Splenic metastases: clinicopathologic presentation, differential diagnosis, and pathogenesis. Arch Pathol Lab Med. 2007;131(6):965–969. This articles relates to splenic metastases and how they compare to SANT.
•Illuminati G, Pizzardi G, Calio F, Pacilè MA, Carboni F, Palumbo P, et al. Hemangiopericytoma of the spleen. Int J Surg. 2015;15:6–10. Characteristics of splenic hemangiopericytomas.
Yang W, Lai J-Y, Cheng C-H, Hsueh C. Splenic hemangiopericytoma in a one-month-old infant. J Pediatr Surg. 2013;48(3):e21–4.
•Abbott RM, Levy AD, Aguilera NS, Gorospe L, Thompson WM. From the archives of the AFIP: primary vascular neoplasms of the spleen: radiologic-pathologic correlation. RadioGraphics 2004;24(4):1137‑63. This article describes the correlation between pathology and radiology of different vascular tumors and how do they compare with each other.
Ferrozzi F, Bova D, De Chiara F. Hemangioendothelioma of the spleen. Clin Imaging. 1999;23(2):111–4.
Giovagnoni A. Tumours of the spleen. Cancer Imaging. 2005;5(1):73–7.
•Hamdi I, Marzouk I, Toulali L, Amara H, Bellara I, Ridene I, et al. Pseudotumeur inflammatoire de la rate et corrélation radio-anatomopathologique. J Radiol. 2006;87(12):1894‑6. This article details Characteristics of Inflammatory pseudotumors of the spleen.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
We confirm that all the authors have had material input into the submission. To our knowledge, all the claims, statements and conclusions are true and are our jointly held opinions.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare. All co-authors have seen and agree with the contents of the manuscript and there is no financial interest to report. We certify that the submission is original work and is not under review at any other publication.
Human and Animal Rights and Informed Consent
No animal or human subjects by the authors were used in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Trigui, A., Toumi, N., Bouzid, A. et al. Sclerosing Angiomatoid Nodular Transformation of the Spleen: A Systematic Review. Curr Surg Rep 12, 161–172 (2024). https://doi.org/10.1007/s40137-024-00402-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40137-024-00402-2