Abstract
Pediatric abdominal tumors have many different etiologies; most of them are generally benign. Malignant tumors are rare, but early diagnosis and work-up are essential to ensure patient’s cure. Diffusion-Weighted Images (DWI) helps providing functional information at a microscopic level of the movement of water. DWI has being increasingly utilized in body oncology imaging for both tumor detection and therapy evaluation, and is particularly promising in children due to the large proportion of small round cell tumors which demonstrate restricted water diffusion. This technique has been applied specially as a noninvasive radiation-free cancer staging and follow up. This article reviews the diffusion magnetic resonance sequence in childhood Neuroblastoma, Wilm´s tumor, Lymphoma, and Hepatoblastoma, helping providing additional information to differentiate high cellularity tumors from others, as well as reducing ionizing radiation exposure, adding another tool for diagnostic imaging, especially in this age group patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pediatric abdominal tumors have many different etiologies; most of them are generally benign such as hydronephrosis, multicystic kidney, infectious hepatosplenomegaly, bezoar, bowel malformation or cysts.
Malignant tumors are rare, but early diagnosis and work-up are essential to ensure patient’s cure [1••].
Most diagnosed abdominal malignancies in the pediatric population commonly include neuroblastoma, Wilms’ tumor, hepatoblastoma, lymphoma, and germ cell tumors [2•].
Magnetic resonance imaging (MRI) use in pediatric oncology continues to grow with the development of faster scanning techniques, coil improvements, moving table platforms, and advanced post-processing techniques. MRI provides high soft tissue contrast and spatial resolution, lack of ionizing radiation, multiplanar capability, and excellent soft tissue characterization; unfortunately MRI studies sometimes have limited availability, relatively long examination time, physiologic motion artifact, high cost, and suboptimal evaluation of the lungs [3•].
Diffusion-weighted images (DWI) helps providing functional information at a microscopic level of the movement of water. Images from the motion of water molecules are obtained by this sequence, as the more cellular density associated with numerous intact cell membranes is present (e.g., high cellular tumor tissue) the more the water motion is restricted. In areas of low cellularity or where the cellular membrane has been broken, the motion of water molecules is less restricted (e.g., a cyst or necrotic tissue). Using these principles, DWI can be used on the detection and further characterization of pathologic processes, including malignant tumors and treatment response.
Initially, it was thought that patient movement (e.g., respiration) limited the successful application of diffusion-weighted techniques to studies performed with breath holding or respiratory triggering, and therefore, decreasing its usefulness in whole-body (WB) examinations due to its increased scanning time. However, with the advent of DWI under free-breathing, diffusion-weighted WB imaging with background body signal suppression (DWIBS), has further increased the interest in WB DWI. [4•].
DWI has being increasingly utilized in body oncology imaging for both tumor detection and therapy evaluation, and is particularly promising in children due to the large proportion of small round cell tumors which demonstrate restricted water diffusion. This technique has been applied specially as a noninvasive radiation-free cancer staging.
The use of WB-MRI for pediatric oncology is well established, with a 96 % of malignancy detection sensitivity when compared to PET-CT and an 100 % bone lesion detection sensitivity [5••, 6•], detecting more metastasis on 17 % of the cases reported by Schlemmer et al. [5••, 6•, 7], when compared to computed tomography (CT).
In 2010 Muller et al. performed DWIBS on a series of 48 healthy children and demonstrated that the restricted diffusion signal within the pelvis and lumbar spine was present in 48 % of patients as a normal finding, advising caution with its interpretation for cancer staging [4•].
This article will review the diffusion magnetic resonance sequence in most common abdominal oncologic tumors of childhood.
Neuroblastoma
Neuroblastic tumors include all tumors that originate from the sympathetic nervous system derived from primordial neural crest cells: ganglineuroma, ganglioreuroblastoma, and neuroblastoma. [8]
Two-thirds of neuroblastomas are primary from the adrenal gland, and the remaining occurs along the paravertebral sympathetic chain. On both CT and MR imaging, neuroblastoma is irregular, lobulated, and heterogeneous, demonstrating coarse amorphous calcifications and variable contrast enhancement, as well as possible invasion of adjacent organs and encasement of vessels, with luminal compression at as many as 70 % of patients with metastatic disease at the time of the diagnosis.
Neuroblastoma is the most common congenital neoplasm representing the second most common congenital tumor (30 % of tumors). It is the third most common tumor of childhood after leukemia and tumors of the central nervous system, accounting for 8–10 % of all pediatric tumors and 15 % of deaths from cancer in pediatrics. [9••, 10••]
Routine protocols traditionally use a combination of CT, metaiodobenzylguanidine scintigraphy (MIBG), and bone marrow aspirate to evaluate local and distant disease spread, however, regional MRI is playing an increasing role, often replacing CT, for assessment of the local disease. It should be remembered that MIBG also has its limitations including the failure to detect bone marrow disease and the lack of MIBG avidity in up to 6 to 10 % of neuroblastoma patients, the loss of MIBG avidity may also occur at relapse, and suboptimal visualization of small lesions in the liver. PET and PET-CT also play a role imaging neuroblastoma, despite its radiation exposure; it is often used in cases of non-MIBG avidity. Further studies are needed to compare WB DWI MRI with MIBG and PET-CT, and potentially also PET-MRI [4•].
Studies with WB MRI with short-time inversion recovery (STIR) and diffusion, although has not yet had completely proven its clinical impact, have shown good results in the initial assessment of distant metastases, even thought are limited detecting calcifications and differentiating viable and necrotic tissue in residual disease after treatment [8].
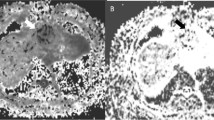
Neuroblastic tumors are usually composed of high cellularity small round cells promoting high signal of restricted water diffusion over its projection. The level of diffusion restriction can be compared and measured over the ADC map, without yet clinical correlation described. Diffusion images can also to be reconstructed from multiple axial 3D data with background suppression (DWIBS) creating a maximum intensity projection 3D WB image that has shown good correlation with disease location (Figs. 1, 2) [5••].
Wilm’s Tumor
Wilm’s tumor, also known as nephroblastoma, is the most common renal malignancy of childhood (representing 87 % of renal tumors). Wilm’s tumor manifests as a solid intrarenal mass with a pseudocapsule associated to distortion of the renal parenchyma and collecting system. The tumor typically spreads by direct extension and displaces adjacent structures, but does not typically encase or elevate the aorta; such encasement or elevation is a distinguishing characteristic of neuroblastoma. There might be vascular invasion of the renal vein and inferior vena cava with occasional extension into the right atrium [11].
MRI shows heterogeneous mass with low signal intensity on T1 and high signal on T2 that may contain hemoglobin degradation products. As ultrasound, MRI has good accuracy to evaluate its extension to intravascular sites from the renal vein to the inferior vena cava [8].
Metastases are most commonly found in the lungs (85 % of cases), liver, and regional lymph nodes, and vascular invasion [11, 12].
As well as neuroblastoma, Ewing’s sarcoma (EWS), peripheral neuroectodermal tumor, rhabdomyosarcoma, synovial sarcoma, non-Hodgkin’s lymphoma, retinoblastoma, and hepatoblastoma [13], nephroblastoma tumors are characterized by high cellularity, small, round, relatively undifferentiated cells, determining restriction on DW images that can be useful for both staging, treatment evaluation, and follow up.
This Brownian motion restriction is represented on DW images as high signal on diffusion sensitive sequences that can be helpful on the search of distant disease as well as on the determination of the tumor viable area.
Humphries et al. made initial examination over three children with nephroblastoma with DW techniques on clinical 1.5-T MR units confirming that DWI can help guiding biopsy away from necrotic areas and suggesting poor response to chemotherapy before tumor shrinkage is expected. However, up to now, DWI was unable to differentiate between benign and malignant lesions based only on ADC values [14, 15], even though the majority of DWI studies are accompanied of some other MR sequences that may improve its sensibility.
Lymphoma
Lymphoma is the third most common malignancy in pediatrics accounting for more than 10 % of cancer on childhood [5••, 16].
The evaluation of the disease extension (staging) is important for the appropriate treatment planning and prognosis determination. Imaging methods play an essential role staging lymphomas, monitoring therapy response and detecting recurrence [17]. As recent studies reported, diffusion-weighted MRI is highly sensitive in detection of malignant diseases, and diffusion-weighted WB imaging has been proposed as a powerful screening tool for these high cellularity tumors [17].
Staging both Hodgkin and non-Hodgkin lymphoma involves the search for bone marrow involvement, present at up to 16 %, which that can be missed at plain radiography [5••, 18]. WB-MRI has proved to be more sensitive than scintigraphy detecting both marrow and non-marrow involvement [5••, 19] and can be a very good alternative to patients who cannot undergo a PET-CT [5••, 20] during its important staging phase (Fig. 3).
DWI studies have been carried out to prove the preliminary results of its importance due to the relationship between cellularity, water diffusion restriction, and disease activity on lymphoma patients [5••, 21], which might help to improve WB-MRI acceptance by clinicians as an alternative to PET-CT during the diagnostic workflow [22••].
Hepatoblastoma
This is mainly a tumor of childhood that often is diagnosed under the age of 3 years [23]. There is a slight male predominance [23, 24] and association with Beckworth Wiedemann syndrome and hemihypertrophy [23, 25].
Children tend to present with an abdominal mass associated with almost always elevated serum alpha-fetoprotein (AFP) to extreme levels (>105 ng/mL) and thrombocytosis. Metastasis occurs almost exclusively in the lungs [26]. The elevation of AFP may also be seen in infants with other tumors like yolk-sac tumors, sarcomas, and hamartomas [27].
Congenital hepatoblastoma is a malignant embryonic tumor composed of only epithelial cells or a mixture of epithelial and mesenchymal cells. They present as lobulated, sometimes multifocal, large solid masses, most often in the right hepatic lobe (60 %). The association with hemihypertrophy (2 %), Beckwith-Wiedemann and intestinal polyposis is well established. The α-fetoprotein may be negative in a considerable percentage of fetal and neonatal tumors.
CT and MRI show a heterogeneous solid hepatic lesion with non-uniform enhancement. The differential diagnosis of congenital hepatoblastoma is the malignant hepatic rhabdoid tumor.
Imaging studies demonstrate a large solitary hypervascular mass, often located in the right lobe of the liver. Cystic change or calcification may be present [23].
Hepatoblastoma is hypointense at MRI in comparison to normal liver in T1-weighted sequences and hyperintense in T2-weighted sequences, while dynamic imaging with gadolinium shows early enhancement with rapid washout [28].
As well as the other tumors described in this article, DW sequences also present restriction for hepatoblastoma, helping on the determination of its multifocal characteristic or in the differentiation to other liver tumors.
Conclusion
Summarizing, diffusion-weighted MRI sequences can provide important information for initial diagnosis of abdominal tumors in childhood, helping providing additional information to differentiate high cellularity tumors from others, as well as reducing ionizing radiation exposure and as an extra tool on early treatment follow up, especially in this age group patients. Several different diffusion aimed clinical trials still needed to be carried, even with the increasing use of this sequence for abdominal diseases, collaborating to increase the reliability of these useful findings.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•• Gunderman RB, Transue DL. Radiology’s role in the battle against pediatric cancers. Acad Radiol. 2012;19(10): 1300–3. This article reviews Radiology's role in pediatric oncology.
• Golden CB, Feusner JH. Malignant abdominal masses in children: quick guide to evaluation and diagnosis. Pediatr Clin North Am. 2002;49(6):1369–92, viii. In this article the authors present their single institution experience of how abdominal malignancies masses present and their distribution by age and diagnosis.
• States LJ, Meyer JS. Imaging modalities in pediatric oncology. Radiol Clin North Am. 2011;49:579–588. This article reviews the roles of specific imaging modalities in the diagnosis and management of noncentral nervous system childhood cancer.
• Atkin KL, Ditchfield MR. The role of whole-body MRI in pediatric oncology. J Pediatr Hematol Oncol. 2014;36(5):332. This article describes the role of whole-body MRI in pediatric oncology.
•• Schiavon JLO, Lederman HM. Whole body MRI and diffusion weighed images in pediatric oncology: lymphomas and several others tumors. Curr Radiol Rep. 2014;2:54. doi:10.1007/s40134-014-0054-z. This article reviews most use of WB-MRI in pediatric oncology.
• Krohmer S, Sorge I, Krausse A, Kluge R, Bierbach U,Marwede D, Kahn T, Hirsch W. Whole-body MRI for primary evaluation of malignant disease in children. Eur J Radiol. 2010;74(1):256–61. doi:10.1016/j.ejrad.2009.01.037. This study compared whole-body (WB) MRI to a combined reference standard of conventional cross-sectional imaging methods and FDG-PET in the detection of malignant disease spread in children.
Schlemmer HP, Schafer J, Pfannenberg C, et al. Fast whole-body assessment of metastatic disease using a novel magnetic resonance imaging system: initial experiences. Investig Radiol. 2005;40:64–71.
Lederman HM, Schiavon JLO, Regacini R, Tostes VS. Tumores abdominais na infância. In: Colégio Brasileiro de Radiologia e Diagnóstico por Imagem; Athaide ACM, Caserta NG, organizadores. PRORAD Programa de Atualização em Radiologia e Diagnóstico por Imagem: Ciclo 3. Porto Alegre:Artmed Panamericana; 2014.p.41-81(Sistema de Educação Continuada a Distância, v.4).
•• Rajiah P, Sinha R, Cuevas C, Dubinscky TJ, Bush WH Jr, Kolokythas O. Imaging Uncommon retroperitoneal masses. Radiographics. 2011; 31(4):949–76. This article reviews CT and MR imaging features of various retroperitoneal masses.
•• Sheth RA, Bittencourt LK, Guimaraes AR. Diffusion-weighted imaging of the male pelvis. Magn Reson Imaging Clin N Am. 2014;22:145–163. In this article, the authors detail the role of DW imaging for pathologic processes involving the male pelvis.
Lowe LH, Isuani BH, Heller RM, Stein SM, Johnson JE, Navarro OM, Hernanz-Schulman M. Pediatric renal masses: wilms tumor and beyond. RadioGraphics. 2000;20:1585–603.
Lonergan GJ, Martinez-Leon MI, Agrons GA, Montemarano H, Suarez ES. Nephrogenic rests, nephroblastomatosis, and associated lesions of the kidney. RadioGraphics. 1998;18:947–68.
Akhtar M, Iqbal MA, Mourad W, Ali MA. Fine-needle aspiration biopsy diagnosis of small round cell tumors of childhood: a comprehensive approach. Diagn Cytopathol. 1999;21:81–91.
Humphries PD, Sebire NJ, Siegel MJ, et al. Tumors in pediatric patients at diffusion-weighted MR imaging: apparent diffusion coefficient and tumor cellularity. Radiology. 2007;245:848–54.
Anne M. Smets & Jan de Kraker Malignant tumours of the kidney: imaging strategy. Pediatr Radiol. 2010;40:1010–8. doi:10.1007/s00247-010-1584-z.
Ley S, Ley-Zaporozhan J, Schenk JP. Whole-body MRI in the pediatric patient. Eur J Radiol. 2009;70:442–51.
Daniel Nava, Heverton Cesar de Oliveira, Flavio Augusto Luisi, Andrea Regina da Silveira Ximenes, Henrique Manoel Lederman Whole-body magnetic resonance imaging for staging and follow-up of pediatric patients with Hodgkin’s lymphoma:comparison of different sequences. Radiol Bras [online]. 2011, vol.44, n.1, pp. 29-34. ISSN 0100-3984.
Krishnan A, Shirkhoda A, Tehranzadeh J, Armin AR, Irwin R, Les K. Primary bone lymphoma: radiographic-MR imaging correlation. Radiographics. 2003;23(6):1371–83;discussion 1384–7.
Kellenberger CJ, Miller SF, Khan M, et al. Initial experience with FSE STIR whole-body MR imaging for staging lymphoma in children. Eur Radiol. 2004;14:1829–41.
Schmidt GP, Haug AR, Schoenberg SO, et al. Whole-body MRI and PETCT in the management of cancer patients. Eur Radiol. 2006;16:1216–25.
Lin C, Itti E, Luciani A, Haioun C, Meignan M, Rahmouni A. Whole-body diffusion-weighted imaging in lymphoma. Cancer Imaging. 2010;10 Spec no A:S172–8. doi:10.1102/1470-7330. 2010.9029. The use of DWI on lymphoma patients was reviewed in this article.
•• Vilanova JC, Barcelo´ J. Diffusion-weighted whole-body MR screening. Eur J Radiol. 2008;67(3):440–7. doi:10.1016/j.ejrad.2008.02.040. The possibilities, limitations and the preliminary clinical results of the whole-body MR imaging using a DWI of the entire body are reviewed in this article.
Linda F. Current imaging strategies of primary and secondary neoplasms of the liver. Seminars in Interventional Radiology. volume 23, number 1 2006.
Emre S, McKenna GJ. Liver tumors in children. Pediatr Transplant. 2004;8:632–8.
Fukuzawa R, Hata J, Hayashi Y, Ikeda H, Reeve AE. Beckwith-Wiedemann syndrome–associated hepatoblastoma. Pediatr Dev Pathol. 2003;6:299–306.
Roebuck DJ. Assessment of malignant liver tumors in children. Cancer Imaging. 2009; 9(Special issue A): S98–S103. Published online Oct 2, 2009. doi: 10.1102/1470-7330.2009.9041 PMCID: PMC2797472.
Fernandez-Pineda I, Cabello-Laureano R. Differential diagnosis and management of liver tumors in infants. World J Hepatol. 2014; 6(7): 486–495. Published online Jul 27, 2014. doi: 10.4254/wjh.v6.i7.486 PMCID: PMC4110540.
Alamo L, Beck-Popovic M, Gudinchet F, Meuli R Congenital tumors: imaging when life just begins. Insights Imaging. 2011; 2(3): 297–308. Published online Feb 14, 2011. doi: 10.1007/s13244-011-00738 PMCID: PMC3259397.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on MRI for Pediatric Oncology.
Rights and permissions
About this article
Cite this article
de Oliveira Schiavon, J.L., Pioner, G.G. & Lederman, H.M. MRI in Pediatric Oncology, Abdominal Tumors: Diffusion. Curr Radiol Rep 2, 75 (2014). https://doi.org/10.1007/s40134-014-0075-7
Published:
DOI: https://doi.org/10.1007/s40134-014-0075-7