Abstract
Severe combined immunodeficiencies (SCIDs) are a group of rare genetic diseases characterized by profound abnormalities of cellular and humoral immunity. They have been identified as ideal candidates for treatment by gene therapy and have led the development of gene therapy for other bone marrow disorders. SCID-X1 and adenosine deaminase SCID have been successfully treated with hematopoietic stem cell gene therapy (HSC-GT) and patients show impressive levels of immune reconstitution. Initial clinical trials employed γ-retroviral vectors, but leukaemia arose in five SCID-X1 patients due to insertional mutagenesis. Clinical trials using self-inactivating γ-retroviral and lentiviral vectors incorporating improved safety features are currently being conducted in Europe and North America. With safer approaches, we anticipate other SCIDs such as recombinase activating gene and Artemis deficiency will be treated with HSC-GT. Looking to the future, gene editing strategies hold enormous promise, and SCID will continue to be the paradigm condition for the development of these applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Severe combined immunodeficiencies (SCIDs) are a diverse group of rare genetic diseases phenotypically characterized by the profound abnormalities of cellular and humoral immunity. They are the most severe of the primary immunodeficiencies (PID) [1] and clinically present with severe recurrent infections with the first year of life. The immunophenotype varies but SCID is defined by very low numbers or the absence of autologous naïve T cells. The presence or absence of B and NK cells is dependent on the underlying genetic defect.
All the known forms of SCID are monogenic conditions which manifest Mendelian patterns of recessive inheritance, either autosomal or X-linked depending on the SCID form (Table 1). As a result, autosomal recessive forms of SCID are reported at a higher frequency within consanguineous pedigrees and populations carrying pathogenic mutations due to a founder effect [2, 3] (for list of genetic defects causing SCID, see Picard et al., review in this series).
Treatment Options for SCID
Until very recently, the treatment of choice for most SCIDs was a hematopoietic stem cell transplant (HSCT), since the introduction of normal hematopoietic stem cells can result in functional immune cell development. Patients with SCID are usually diagnosed after opportunistic or recurrent infections or related complications, and after these acute infections are resolved, patients are placed on prophylactic antibiotics and immunoglobulin replacement until a suitable HSCT donor is found [4]. The nature of the donor is a major factor in the outcome of HSCT in SCID, and a number of studies highlight that the best survival outcomes are seen following the matched sibling donors (MSD) [4]. Results from matched unrelated donor transplants are good, but multiple mismatched grafts or mismatched haplo-identical transplants from a parental donor have the poorest outcome [4, 5] (see Wahlstrom et al., review in this series). Gene therapy protocols were therefore developed to offer a therapeutic option for those patients without a suitable donor.
Conditioning is commonly used before HSCT but must be carefully considered in the paediatric context of SCID due to the increased risk of chronic health conditions associated with conditioning children [6] and also because unconditioned transplants have excellent results in specific SCID forms such as SCID-XL and ADA SCID forms [4]. Even more careful deliberation must be undertaken in the case of patients with radiosensitive SCID because these SCID types are more susceptible to alkylating agents used in chemotherapy regimens [7].
Due to the metabolic nature of ADA deficiency, enzyme replacement therapy (ERT) exists for this SCID form by means of weekly intramuscular injection of pegademase bovine (PEG-ADA; a bovine ADA form conjugated to polyethylene glycol). Doses are relative to the weight of the patients, and the costs of the treatment are large. Moreover, the immune recovery is variable between patients, with approximately 20 % of non-responders and the rest showing a short-term full recovery followed by long-term decreasing T-cell numbers [8].
Gene Therapy for SCID
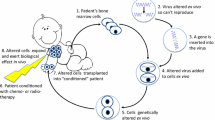
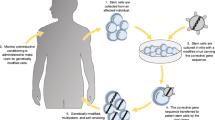
Gene therapy (GT) can be described as the delivery of nucleic acids into cells for the treatment, amelioration or cure of a disease. This delivery can be performed in vivo or ex vivo using either viral or non-viral vectors and can result in integrating or episomal gene delivery depending on the platform selected. SCID forms are ideal candidates for GT protocols with integrating vectors. First, the monogenic nature of these diseases allows the addition of a single gene to correct or ameliorate the SCID phenotype. Second, the technology to isolate and culture hematopoietic stem cells (HSC) from patients is well established, together with the protocols for successful ex vivo viral delivery to HSCs. Third, the use of integrating vectors is ideal to correct not only the HSCs but, through the passing of transgene to HSC cell progeny, also any downstream hematopoietic lineages affected in SCID. Finally, the genetic correction of SCID HSCs can provide cells with a strong positive selective advantage to restore the immune system and facilitate a positive outcome.
Human GT for SCID started in 1990 with a clinical trial for ADA SCID using ADA gene-corrected T cells. Cells were isolated from two patients who had both received ERT and were transduced with a γ-retroviral vector (GV) containing the human ADA under the control of the γ-retroviral long terminal repeat (LTR) enhancer/promoter. These gene modified cells were then repeatedly infused back into the patients over a period of 2 years [9]. A 12-year follow-up demonstrated the presence of gene-corrected T cells in the two patients treated, and although ERT was never stopped, the dosage of PEG-ADA administered was reduced [10]. Further pilot studies on ADA SCID again using GVs were performed with corrected bone marrow or umbilical cord CD34+ cells and proved to be safe, but all these initial studies did not show effective immune reconstitution and patients remained on PEG-ADA ERT [11–13].
These early studies demonstrated the safety and feasibility of GT for SCID conditions and the ability to gene modify primary haematopoietic cells. Following the research focused on achieving curative hematopoietic stem cell gene therapy (HSC-GT), several aspects of GT protocols were revised, including the development of cytokine cocktails to enhance activation and maintenance of stemness in HSCs [14] and the use of fibronectin to improve the co-localization of the GV with the HSCs to boost transduction [15]. So far, X-linked and ADA SCID forms have been successfully treated in humans using HSC-GT approaches (Table 2) [16–18, 19∙∙, 20∙∙]. However, the unexpected appearance of GT-related leukaemia in some X-linked SCID patients [21–23] has emphasized the importance of developing safer viral vectors and methods to assess genotoxicity of vector design.
Apart from SCIDs, HSC-GT has been employed to treat PIDs such as Wiskott–Aldrich syndrome (WAS) [24] and chronic granulomatous diseases (CGD) [25] with variable results, including unexpected transgene silencing and myelodysplasia in CGD HSC-GT [25].
Gene Therapy for SCID-XL
γ-Retroviral Trials and Insertional Mutagenesis
X-linked SCID (SCID-XL) is the most frequent form of SCID (accounting for 40–50 % of cases [20∙∙]), and it is caused by mutations in the common γ chain (γc) gene, IL2RG. The γc works as a pivotal subunit that is an integral part of the cytokine receptor complexes for IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21 [20∙∙]. γc is therefore critically important in multiple cytokine signalling pathways, and the absence of the functional gene results in a block of T-cell and NK-cell development. SCID-X1 was selected as a good candidate for HSC-GT because significant correction of the T-cell defect was documented in cases of revertant somatic mosaicism, which demonstrated the powerful selective advantage conferred upon wild-type corrected cells [26].
The first clinical trials for X-linked SCID (SCID-XL) were performed using a GV driving the γc expression via the viral endogenous LTR enhancer/promoter [27, 28]. Overall, ten patients were treated at the Necker–Enfants Malades Hospital in Paris from 1999 to 2002 [18], and ten were treated at the Great Ormond Street Hospital in London from 2001 to 2006 (Table 2) [20∙∙]. All the recruited patients, between 1 and 46 months of age, did not have optimal HSCT donor options and underwent ex vivo GT protocols without the use of any conditioning regimes [18, 20∙∙].
Patients discontinued prophylactic measures after immune restoration was observed and have since been naturally exposed to pathogens and resolved infections [18, 20∙∙]. Recovery of T-cell and NK-cell compartments could be observed during the first month after therapy, however, in the long-term patients maintained normal or near-normal T-cell counts, but NK-cell counts first improved and then decreased after a number of months [18, 20∙∙]. A possible explanation for this NK drop is that γc chain expression requirements seem to be higher for long-term maintenance than for development of NK cells [29], or that there was insufficient transduction of progenitor cells to allow long-term NK-cell recovery. The B cell compartment showed very little transduction, as corrected B cells do not present a selective advantage over SCID-X1 B cells, and as a consequence humoral immunity was only improved in a minority of treated patients [18, 20∙∙]. GT-related T-cell acute lymphoblastic leukaemia (T-ALL) arose in five patients between 2.5 and 5.5 years after treatment [21–23], one of whom died, and the rest have since been in remission after anti-leukaemia chemotherapy. Patients showed normal development with respect to weight and height [18, 20∙∙].
When the same γ-retroviral GT approach was conducted after failure of HSCT to treat three pre-adolescent cases, ages from 6 to 11, only one of the patients showed improvement in immunological parameters [30], and when used to treat two young adults, ages 15 and 20, results were unsuccessful in both cases (Table 2) [31]. The failure of HSC-GT in older patients suggests age plays a role in the outcome of HSC-GT, most probably due to the lack of thymic reserve [32].
These trials proved for the first time in any human study that HSC-GT is capable of correcting a genetic disease. However, important lessons were learnt from the unexpected insertional mutagenesis observed. Gene addition by means of retroviral integration is not independent of its genetic context, as GV show a preference for integration near transcription start sites (TSSs) [33] and gene-dense regions [34]. The leukaemia adverse events were found to be related to the integration pattern and LTR enhancer/promoter trans-activation of nearby proto-oncogenes LMO2, BMI1 and CCND2, together with other genetic abnormalities, which conferred HSCs with augmented proliferation capacities and elicited clonal expansion followed by leukemic transformation [22, 23]. It was suggested the γc chain transgene may also have had an oncogenic role by itself [35]. However, evidence from different groups argues against this possibility [36–39].
For future GT trials, new viral vectors were developed to confer a safer insertional mutagenic profile and incorporated important changes such as the deletion of the viral LTRs leading to self-inactivating (SIN) vectors and the switch from GV to lentiviral vectors (LV). Second, appropriate tests were developed to detect any potential genotoxic effects including in vitro immortalisation assays.
SIN retroviral vectors are vectors which carry a deletion of the enhancer/promoter sequence in the viral LTR and transgene expression is driven by internal endogenous promoters [40–42]. The internal promoters are typically less powerful and more specific than the wild-type LTR sequences, and the lack of enhancer sequences potentially abolishes unwanted trans-activation of neighbouring genes thereby eliminating the major initiating cause of leukaemia in previous GT trials [43, 44].
Lentiviruses are a different viral genus than γ-retroviruses within the same Retroviridae family, and show differences in their integration mechanism that are important for the purposes of GT. LV are capable of transducing both dividing and quiescent cells and facilitate high efficiency ex vivo transduction of HSC, with a reduction of culture time and better maintenance of cell stemness. More importantly, in terms of safety, they present a less genotoxic integration profile with fewer tendencies for hot spots and regions highly enriched in proto-oncogenes [34].
Both GV and LV have been engineered into their respective SIN versions. Comparison of these SIN vectors has confirmed the LV integration profile is inherently safer than that of GV, but the strength of the internal promoter is likely to play the bigger role in oncogenesis [43, 45]. Evidence of SCID-X1 correction with SIN γ-retroviral vectors was published in a murine model in 2008 [46], and similar successful results with SIN lentiviral vectors were reported in 2010 [44].
The insertional mutagenesis derived from the fact that the integration machinery in retroviruses has evolved to ensure productive viral infection by targeting accessible chromatin and sites of active transcription [33, 47–51]. Finding models for insertional mutagenesis is therefore of paramount interest to assess the safety of integrating GT approaches. In vivo, the first murine model to present leukaemia due to insertional mutagenesis was achieved after secondary transplant of cells transduced with GV [52], and a tumour prone mouse model was later introduced to compare vectors carrying full or self-inactivating LTR sequences [43, 53]. In vitro, insertional mutagenesis assays have been developed to detect enhancer-mediated up-regulation [45, 54], promoter driven insertional mutagenesis [55] and fusion mRNA and aberrant splicing [56–59]. Other long-range insertional mutagenesis mechanisms have been recently described in the γ-retroviral context [60] that may also be relevant in the GT setting. Overall, these results exemplify the intricacy of the retroviral integration and the importance of a better understanding of this phenomenon to ensure safer GT protocols in the clinic.
Self-inactivating (SIN) γ-Retroviral Trials
The second generation of GT trails for SCID-XL has been initiated. Patients have been recruited for SIN γ-retroviral GT at a number of sites in Europe and the USA. Alternatively, a SIN lentiviral vector pilot study for SCID-X1 has been registered as a multicentre clinical trial in the USA.
Interim data on the SIN γ-retroviral GT trial has recently become available with data from nine patients treated in Europe and the USA (Table 2) [61∙∙]. Corrected HSC was delivered without conditioning to patients. Unfortunately, one patient died before full reconstitution of the immune system due to adenoviral infection. After a median follow-up of almost 30 months, six patients had shown an early rise in NK-cell numbers, as well as achieved and maintained T-cell recovery with polyclonal diversity in most of the cases [61∙∙]. In regards to humoral immunity, two patients presented normal IgG and IgA levels, whereas the rest were still under immunoglobulin replacement therapy [61∙∙]. Peripheral blood cells from these patients were analysed and compared to those from the γ-retroviral trial; although a similar integration distribution was observed in both settings, there was a significant enrichment of cells with insertions near proto-oncogenes in the samples from the GV trial over the SIN GV trial [61∙∙]. No leukemic transformation or clonal expansion has been observed so far, suggesting that the SIN GV has a safer profile than the previous vector design. However, the follow-up is still below the 3 years of leukaemia latency reported in the γ-retroviral trial [22, 23], and continued follow-up is required.
Gene Therapy for ADA SCID
γ-Retroviral Trials
ADA SCID is a severe combined immunodeficiency that arises from the lack of adenosine deaminase (ADA, EC3.5.4.4), a cytosolic enzyme responsible for the degradation of adenosine and deoxyadenosine into inosine and deoxyinosine, respectively. As a result of the accumulation of adenosine and deoxyadenosine-related metabolites, several multi-organ pathologies emerge in patients with SCID being the most life threatening. However, non-immunological consequences including neuronal, skeletal, pulmonary and hepatic dysfunctions [62–66] have been observed. The only curative option for ADA SCID is bone marrow HSCT from a suitable donor, although GT protocols have now been developed with successful results. In addition, ERT is available for patients.
ERT for ADA deficiency consists of the systemic delivery of PEGylated bovine adenosine deaminase (PEG-ADA). PEG-ADA can be administered to patients by means of weekly injections [65], which is significantly expensive (about £400,000 per patients per year), and there is a great variability in the response to the treatment [8, 65]. PEG-ADA treatment may also be compromised by the development of immunity against the bovine ADA epitope [67]. PEG-ADA is often used in newly diagnosed patients until a suitable HSCT donor is found, or patients are enrolled in HSC-GT protocols [65]. As a substitute, long-term PEG-ADA has an estimated probability of 78 % of surviving 20 years, with half of these deaths happening in the first 6 months of treatment [65]. Nonetheless, it is important to note patients under long-term PEG-ADA ERT do not show effective immunological recovery as that seen in patients treated with HSCT and HSC-GT: immune function parameters are decreased, thymic function is lessened and bone marrow output is compromised [68–70].
Immune regulation and tolerance are also affected in ADA SCID patients, and autoimmune co-morbidities such as diabetes mellitus, autoimmune thyroiditis or haemolytic anaemia have been described in patients treated with HSCT, HSC-GT or PEG-ADA [71]. However, patients under PEG-ADA present with reduced Treg function and cell numbers compared to those treated with bone marrow HSCT or HSC-GT [72], and B cell tolerance appears to be corrected by bone marrow HSCT and HSC-GT but not with PEG-ADA ERT [73]. Overall, it seems the ADA protein must have intracellular roles necessary for the correct development and function of the immune system that cannot be cross-corrected via systemic (extracellular) PEG-ADA detoxification.
After several only partially successful GT pilot studies for ADA SCID [11–13], HSC-GT trials started in 2000 in HSR-TIGET, Milan (Table 2). Similar to the SCID-X1 GT trials, the Milan trial employed a GV with ADA transgene expression driven by the endogenous viral LTR enhancer/promoter sequence [17, 74]. Data from nine participants, 0.6- to 5.6-year old, were published in 2009 [17]. Patients were pre-conditioned with nonmyeloablative busulfan regimes in order to achieve therapeutic engraftment of corrected haematopoietic progenitors [74]. PEG-ADA administration was ceased 3 weeks before the transfusion to grant selective advantage to gene-corrected cells and enhance engraftment and immune recovery [75]. Only two out of ten patients restarted PEG-ADA during the follow-up [17]. After 1 year, peripheral blood cells showed significant transduction in T (88.0 %), B (52.5 %) and NK cells (59.2 %), and purine metabolites were significantly reduced in red blood cells, demonstrating HSC-GT resulted in multi-lineage reconstitution, and metabolic correction was enough to allow for the development of ADA deficient cells [17]. At the last follow-up, five patients showed normal T-cell counts and all presented with a polyclonal T-cell-receptor repertoire [17]. NK-cell numbers were within the normal range in three patients [17]. B cell counts and Ig levels increased progressively after therapy, and four patients achieved normal B cell counts, five reached normal serum IgG levels and the majority had normal IgA and IgM levels [17].
A similar trial was started at the Great Ormond Street Hospital (London) in 2003, recruiting up to eight patients for whom a HSCT donor was not found and who had responded poorly to ERT (Table 2) [19∙∙, 76]. Patients were pre-conditioned with a nonmyeloablative dose of melphalan or a busulfan regime, and PEG-ADA injections were stopped some weeks before transfusion of corrected HSCs [19∙∙, 76]. After long-term follow-up, three patients did not require PEG-ADA, and four showed stable CD3+ counts 5 years after HSC-GT [19∙∙]. These same four patients presented an early rise in NK cells, but only two have maintained normal NK-cell levels over time [19∙∙]. All four patients showed increased B cell counts, and all four could stop immunoglobulin replacement therapy [19∙∙].
Finally, a third γ-retroviral GT trial was performed between 2001 and 2009 at the Children’s Hospital Los Angeles (CHLA) and the Clinical Center of the National Institutes of Health (NIH) in Bethesda, Maryland (Table 2). The first ten patients recruited in the trial were simultaneously administered cells transduced with two different versions of a GV coding for the human ADA gene [77]. In the original protocol, the first four patients did not undergo pre-conditioning and did not stop PEG-ADA injections. However, after the positive results from the Milan trial, the GT protocol was amended to include busulfan conditioning and cessation of PEG-ADA for the next six patients. Notably, only the six last patients showed immune recovery. However, the simultaneous introduction of conditioning and ERT cessation made the relative contributions of each variable difficult to assess [77].
Further research with the mouse model for ADA SCID has tried to elucidate the individual impacts of conditioning and ERT cessation. These studies showed that conditioning is essential in ADA SCID to create “space” for engraftment and expansion of corrected HSC with higher degrees of myeloablation showing higher levels of gene-marked cells [78]. With regard to ERT, mice in whom ERT was continued after GT demonstrated that the vector copy number (VCN, the number of transgene copies in respect to the number of cells) in lymphocytes was maintained despite total lymphocyte numbers increasing [78]. This result suggests that ERT continuation does not blunt the selective advantage of the corrected cells but could instead provide these cells with a detoxified environment supportive for thymopoiesis [78].
Similar to the SCID-X1 trials, the integration analysis from the Milan trial showed a preference of the vectors to integrate near transcription start sites (TSSs) and gene-dense regions, including integrations near the LMO2 proto-oncogene [79]. However, unlike the SCID-X1 trials, leukaemia has not yet been described in any ADA SCID patient treated with γ-retroviral GT although the reasons for this remain unclear.
Lentiviral Vectors Development and Trials
Currently, a new generation of GT trials for ADA SCID is being conducted with LV (Table 2). Preclinical evidence has been recently published in the murine model for ADA SCID showing lentiviral HSC-GT can promote the recovery of the immune system and the detoxification of adenosine and deoxyadenosine metabolites while reducing the potential for insertional mutagenesis [83∙].
In Europe, the Great Ormond Street Hospital in London is conducting a lentiviral GT trial with ten patients recruited up to date. Preliminary data on the first five patients were disclosed at the American Society of Gene & Cell Therapy 17th Annual Meeting. All the patients (aged 1.2- to 4.5-year old) tolerated the procedure well which consisted of conditioning with a single dose of busulfan and reinfusion of transduced autologous HSCs [82∙]. After a mean follow-up of 361 days, ADA activity was detectable in red blood cells, and significant immunological recovery was evidenced by a rise in total T, CD4+ T and naïve CD4+ T-cell counts as well as in T-cell receptor excision circles (TREC) numbers [82∙]. Clonal expansions could be observed by integration analysis, but these clones were not persistent in time [82∙].
In the USA, lentiviral GT trials are recruiting patients at the Mattel Children’s Hospital—University of California, Los Angeles, and the National Institutes of Health Clinical Center in Bethesda, Maryland.
Other Gene Therapy Developments
Artemis and RAG 1/2 Deficiency
Artemis is a DNA repair protein also implicated in V(D)J recombination. Artemis deficiency, which results in T− B− NK+ SCID with sensitivity to ionizing radiation, has been identified as a suitable next candidate for HSC-GT. Current treatment presents various difficulties and post-HSCT morbidities include chronic GvHD, auto-inflammatory complications and recurrent and severe infections [26, 84]. In 2008, preclinical data were published on an Artemis deficiency murine model showing functional T and B cell restoration upon lentiviral HSC-GT [85], and functional restoration of human B cells was reported in a mouse recipient after lentiviral transduction of CD34+ cells isolated from Artemis and RAG1-deficient patients [86].
RAG1/2 are gene-encoding proteins differentially expressed in lymphocytes during cell development in order to elicit the V(D)J recombination necessary for TCR and immunoglobulin generation and variability. Without RAG-1 or RAG-2, lymphocytes fail to thrive and patients present with T− B− NK+ SCID.
Retroviral HSC-GT on a murine model for RAG1 deficiency was first reported in 2006 showing near-normal T- and B-lymphocyte reconstitution, albeit only when high VCN was achieved [87]. Successful RAG1 SIN lentiviral HSC-GT in mice followed in 2011, showing phenotype restoration without the need for high VCNs due to a more efficient vector design [88∙]. However, dissimilar results were recently published with an analogous lentiviral HSC-GT approach for the same RAG1 deficient mice. In the latter publication, only high VCN showed some degree of immune recovery and lower copy numbers resulted in autoimmune manifestations resembling those of Omenn’s syndrome [89]. Overall, RAG1 gene therapy will require further refinement before translation into humans.
In the case of RAG2 deficiency, a murine model exists that was first corrected with retroviral HSC-GT in 2002 [90], and favourable results with lentiviral HSC-GT were reported in 2012 [91]. No complications could be observed in any of these RAG2 HSC-GTs in mice.
Gene Editing Strategies
Gene or genome editing is the modification of the genome in a sequence-specific fashion, allowing for deletion, insertion or modification of the genome in a given site of interest. Gene editing relies on inducing a homology-directed repair (HDR) at the target site, a process in which a donor sequence containing the desired modifications is incorporated into the genome due to the presence of flanking homology arms. It has been described that inducing DNA damage such as double-strand breaks (DSBs) is the most significant way to elicit HDR. Therefore, gene editing relies on simultaneously supplying the cells with nucleases engineered to cut the site of interest and an appropriate donor molecule carrying the sequence changes required (Fig. 1).
Gene editing strategies. Several engineered nucleases are currently available for gene editing purposes, mainly zinc-finger nucleases (ZFN), transcription activator-like effector nucleases (TALEN) and the clustered regularly interspaced palindromic repeats/Cas9 (CRISPR/Cas9) system. All of these platforms enable the targeting and cutting of specific genomic sequences and the resulting double-strand break (DSB) induces the cell to swiftly repair the DNA damage or trigger apoptosis. Without a reference sequence, a set of proteins (which include artemis, DNA-PK, XLF and ligase IV causative of SCID) can repair the damage by joining the two ends in a random manner that results in arbitrary insertions or deletions at the target site, namely non-homologous end joining (NHEJ). In the presence of a donor DNA sequence (a sequence carrying the target modification flanked by homology arms of the target site), a different group of proteins can elicit homology-directed repair (HDR) using the donor DNA as a template while incorporating the target modification into the genome. HDR would allow for the correction of mutations causing SCID, thus avoiding the need for integrating vectors and potential genotoxicity, as well as allowing physiological expression of the corrected gene
Gene editing has received a lot of attention in the last years for its game-changing implications in eukaryotic genetic engineering, as illustrated by its election as method of the year in 2011 [92]. There are very clear advantages for using gene editing in SCID forms and GT in general. Edition of mutations causing SCID would completely remove the possibility of insertional mutagenesis and gene expression would be driven by its endogenous regulatory element, thereby allowing for cells to respond physiologically during engraftment, reconstitution and immune response. However, this technology presents some potential pitfalls: off-target effects (unwanted disruption of secondary target sites) have been reported in all gene editing platforms and editing efficiencies in the literature are still modest. The SCID setting, nonetheless, is favourable for proof-of-concept studies due to the selective advantage of the corrected cells. Indeed, gene editing of HSC with zinc-finger nucleases (ZFNs) has been demonstrated by the first time by Genovese et al. earlier this year, both in healthy and SCID-X1 human HSCs [93∙∙].
Conclusions
Gene therapy has come a long way since conceptualized in the early 70s [94], and the need for therapeutic options for rare, untreatable diseases has relentlessly driven research in the field during the last decades. In the context of SCID, GT has successfully treated X-linked and ADA SCID and is showing success in other blood disorders and specific metabolic conditions [24, 95–97]. Not surprisingly, a lot has been learnt in the process of modifying the genome with integrating vectors as illustrated by the genotoxicity reported in the initial GV mediated trials of GT for SCID-XL.
As highlighted in this review, the leukaemia cases in the SCID-X1 trials flagged safety as a priority for any following HSC-GT trials. Research showed the powerful LTR enhancer/promoter regions together with the tendency of these vectors to integrate upstream of active genes were the most likely forces driving the leukemic transformation [22, 23, 33, 48]. Currently, a new generation of trials is being conducted with novel SIN GV and LV with the main purpose of decreasing the potential insertional mutagenesis, and initial results appear to be very promising. If proven safe, we anticipate these new approaches will be translated into other SCID forms such as RAG1/2 and Artemis deficiency, with preclinical evidence of successful HSC-GT already demonstrated [85].
With regard to GT for SCID, there is still more room for improvement. More physiologically regulated transgene expression and even safer platforms can and will be developed in the years to come. Particularly, we believe gene editing strategies offer the greatest promise for SCID forms, as correction at the DNA sequence level would result in fully corrected autologous HSCs with normally regulated gene sequences [93∙∙]. Long-term HSC-GT could then become the standard of treatment for SCID if proven safe, as patients would not need to wait for compatible donors and HLA disparity and GvHD complications could be avoided.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Zhang L, Thrasher A, Gaspar H. Current progress on gene therapy for primary immunodeficiencies. Gene Ther. 2013;20(10):963–9.
Li L, et al. A founder mutation in Artemis, an SNM1-like protein, causes SCID in Athabascan-speaking Native Americans. J Immunol. 2002;168(12):6323–9.
Rozmus J, et al. Severe combined immunodeficiency (SCID) in Canadian children: a national surveillance study. J Clin Immunol. 2013;33(8):1310–6.
Gaspar H, et al. How I treat severe combined immunodeficiency. Blood. 2013;122(23):3749–58.
Gennery A, et al. Transplantation of hematopoietic stem cells and long-term survival for primary immunodeficiencies in Europe: entering a new century, do we do better? J Allergy Clin Immunol. 2010;126(3):602.
Armenian S, et al. Long-term health-related outcomes in survivors of childhood cancer treated with HSCT versus conventional therapy: a report from the Bone Marrow Transplant Survivor Study (BMTSS) and Childhood Cancer Survivor Study (CCSS). Blood. 2011;118(5):1413–20.
Dvorak C, Cowan M. Radiosensitive severe combined immunodeficiency disease. Immunol Allergy Clin North Am. 2010;30(1):125–42.
Booth C, Gaspar H. Pegademase bovine (PEG-ADA) for the treatment of infants and children with severe combined immunodeficiency (SCID). Biol Targets Ther. 2009;3:349–58.
Blaese R, et al. T lymphocyte-directed gene therapy for ADA-SCID: initial trial results after 4 years. Science. 1995;270(5235):475–80.
Muul L, et al. Persistence and expression of the adenosine deaminase gene for 12 years and immune reaction to gene transfer components: long-term results of the first clinical gene therapy trial. Blood. 2003;101(7):2563–9.
Bordignon C, et al. Gene therapy in peripheral blood lymphocytes and bone marrow for ADA-immunodeficient patients. Science. 1995;270(5235):470–5.
Hoogerbrugge P, et al. Bone marrow gene transfer in three patients with adenosine deaminase deficiency. Gene Ther. 1996;3(2):179–83.
Kohn D, et al. T lymphocytes with a normal ADA gene accumulate after transplantation of transduced autologous umbilical cord blood CD34+ cells in ADA-deficient SCID neonates. Nat Med. 1998;4(7):775–80.
Luens K, et al. Thrombopoietin, kit ligand, and flk2/flt3 ligand together induce increased numbers of primitive hematopoietic progenitors from human CD34+ Thy-1+ Lin− cells with preserved ability to engraft SCID-hu bone. Blood. 1998;91(4):1206–15.
Hanenberg H, et al. Optimization of fibronectin-assisted retroviral gene transfer into human CD34+ hematopoietic cells. Hum Gene Ther. 1997;8(18):2193–206.
Aiuti A, et al. Hematopoietic stem cell gene therapy for adenosine deaminase deficient-SCID. Immunol Res. 2009;44(1–3):150–9.
Aiuti A, et al. Gene therapy for immunodeficiency due to adenosine deaminase deficiency. N Engl J Med. 2009;360(5):447–58.
Hacein-Bey-Abina S, et al. Efficacy of gene therapy for X-linked severe combined immunodeficiency. N Engl J Med. 2010;363(4):355–64.
∙∙ Gaspar HB, et al. Hematopoietic stem cell gene therapy for adenosine deaminase-deficient severe combined immunodeficiency leads to long-term immunological recovery and metabolic correction. Sci Transl Med. 2011;3(97):97ra80. This work reports the follow-up of the second cohort of ADA patients treated with GV HSC-GT after mild conditioning and ERT cessation. After a mean follow-up of 43 months 4 of 6 patients are off ERT, supporting HSC-GT is a therapeutic option for ADA SCID.
∙∙ Gaspar H, et al. Long-term persistence of a polyclonal T cell repertoire after gene therapy for X-linked severe combined immunodeficiency. Sci Transl Med. 2011;3(97). Successful long-term outcome of the 10 SCID-X1 patient cohort in the UK treated with GV HSC-GT. After a mean follow-up of 80 months all patients showed a polyclonal T-cell repertoire and one patient developed T-ALL.
Hacein-Bey-Abina S, et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science. 2003;302(5644):415–9.
Hacein-Bey-Abina S, et al. Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J Clin Invest. 2008;118(9):3132–42.
Howe S, et al. Insertional mutagenesis combined with acquired somatic mutations causes leukemogenesis following gene therapy of SCID-X1 patients. J Clin Invest. 2008;118(9):3143–50.
Boztug K, et al. Stem-cell gene therapy for the Wiskott–Aldrich syndrome. N Engl J Med. 2010;363(20):1918–27.
Stein S, et al. Genomic instability and myelodysplasia with monosomy 7 consequent to EVI1 activation after gene therapy for chronic granulomatous disease. Nat Med. 2010;16(2):198–204.
Cavazzana-Calvo M, et al. Gene therapy for primary immunodeficiencies: part 1. Curr Opin Immunol. 2012;24(5):580–4.
Hacein-Bey-Abina S, et al. Sustained correction of X-linked severe combined immunodeficiency by ex vivo gene therapy. N Engl J Med. 2002;346(16):1185–93.
Gaspar H, et al. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet. 2004;364(9452):2181–7.
Orr S, et al. Implications for gene therapy-limiting expression of IL-2R gamma c delineate differences in signaling thresholds required for lymphocyte development and maintenance. J Immunol. 2010;185(3):1393–403.
Chinen J, et al. Gene therapy improves immune function in preadolescents with X-linked severe combined immunodeficiency. Blood. 2007;110(1):67–73.
Thrasher A, et al. Failure of SCID-X1 gene therapy in older patients. Blood. 2005;105(11):4255–7.
Antoine C, et al. Long-term survival and transplantation of haemopoietic stem cells for immunodeficiencies: report of the European experience 1968-99. Lancet. 2003;361(9357):553–60.
Wu X, et al. Transcription start regions in the human genome are favored targets for MLV integration. Science. 2003;300(5626):1749–51.
Cattoglio C, et al. Hot spots of retroviral integration in human CD34+ hematopoietic cells. Blood. 2007;110(6):1770–8.
Woods N-B, et al. Gene therapy: therapeutic gene causing lymphoma. Nature. 2006;440(7088):1123.
Pike-Overzet K, et al. Gene therapy: is IL2RG oncogenic in T-cell development? Nature. 2006;443(7109):7.
Thrasher A, et al. Gene therapy: X-SCID transgene leukaemogenicity. Nature. 2006;443(7109):E5–6.
Scobie L, et al. A novel model of SCID-X1 reconstitution reveals predisposition to retrovirus-induced lymphoma but no evidence of gammaC gene oncogenicity. Mol Ther. 2009;17(6):1031–8.
Ginn S, et al. Lymphomagenesis in SCID-X1 mice following lentivirus-mediated phenotype correction independent of insertional mutagenesis and gammac overexpression. Mol Ther. 2010;18(5):965–76.
Yu S, et al. Self-inactivating retroviral vectors designed for transfer of whole genes into mammalian cells. Proc Natl Acad Sci USA. 1986;83(10):3194–8.
Kraunus J, et al. Self-inactivating retroviral vectors with improved RNA processing. Gene Ther. 2004;11(21):1568–78.
Schambach A, et al. Overcoming promoter competition in packaging cells improves production of self-inactivating retroviral vectors. Gene Ther. 2006;13(21):1524–33.
Montini E, et al. The genotoxic potential of retroviral vectors is strongly modulated by vector design and integration site selection in a mouse model of HSC gene therapy. J Clin Invest. 2009;119(4):964–75.
Zhou S, et al. A self-inactivating lentiviral vector for SCID-X1 gene therapy that does not activate LMO2 expression in human T cells. Blood. 2010;116(6):900–8.
Modlich U, et al. Insertional transformation of hematopoietic cells by self-inactivating lentiviral and gammaretroviral vectors. Mol Ther. 2009;17(11):1919–28.
Thornhill S, et al. Self-inactivating gammaretroviral vectors for gene therapy of X-linked severe combined immunodeficiency. Mol Ther. 2008;16(3):590–8.
Schröder A, et al. HIV-1 integration in the human genome favors active genes and local hotspots. Cell. 2002;110(4):521–9.
Biasco L, et al. Integration profile of retroviral vector in gene therapy treated patients is cell-specific according to gene expression and chromatin conformation of target cell. EMBO Mol Med. 2011;3(2):89–101.
De Rijck J, et al. The BET family of proteins targets moloney murine leukemia virus integration near transcription start sites. Cell Rep. 2013;5(4):886–94.
Sharma A, et al. BET proteins promote efficient murine leukemia virus integration at transcription start sites. Proc Natl Acad Sci USA. 2013;110(29):12036–41.
De Ravin S, et al. Enhancers are major targets for murine leukemia virus vector integration. J Virol. 2014;88(8):4504–13.
Li Z, et al. Murine leukemia induced by retroviral gene marking. Science. 2002;296(5567):497.
Montini E, et al. Hematopoietic stem cell gene transfer in a tumor-prone mouse model uncovers low genotoxicity of lentiviral vector integration. Nat Biotechnol. 2006;24(6):687–96.
Modlich U, et al. Cell-culture assays reveal the importance of retroviral vector design for insertional genotoxicity. Blood. 2006;108(8):2545–53.
Bokhoven M, et al. Insertional gene activation by lentiviral and gammaretroviral vectors. J Virol. 2009;83(1):283–94.
Recchia A, et al. Retroviral vector integration deregulates gene expression but has no consequence on the biology and function of transplanted T cells. Proc Natl Acad Sci USA. 2006;103(5):1457–62.
Almarza D, et al. Risk assessment in skin gene therapy: viral-cellular fusion transcripts generated by proviral transcriptional read-through in keratinocytes transduced with self-inactivating lentiviral vectors. Gene Ther. 2011;18(7):674–81.
Moiani A, et al. Lentiviral vector integration in the human genome induces alternative splicing and generates aberrant transcripts. J Clin Invest. 2012;122(5):1653–66.
Cesana D, et al. Whole transcriptome characterization of aberrant splicing events induced by lentiviral vector integrations. J Clin Invest. 2012;122(5):1667–76.
Sokol M, et al. Novel principles of gamma-retroviral insertional transcription activation in murine leukemia virus-induced end-stage tumors. Retrovirology. 2014;11:36.
∙∙ Hacein-Bey-Abina S, et al. Gene therapy with a modified vector for X-linked severe combined immunodeficiency. N Engl J Med. in press. This publication reports the outcome of the first 9 patients treated with a novel SIN GV approach after a 12–38 months follow up. 7 of 8 HSC-GT survivors showed successful recovery of peripheral T cells and no T-ALL appearance was observed.
Bollinger M, et al. Brief report: hepatic dysfunction as a complication of adenosine deaminase deficiency. N Engl J Med. 1996;334(21):1367–71.
Rogers M, et al. Cognitive and behavioral abnormalities in adenosine deaminase deficient severe combined immunodeficiency. J Pediatr. 2001;139(1):44–50.
Titman P, et al. Cognitive and behavioral abnormalities in children after hematopoietic stem cell transplantation for severe congenital immunodeficiencies. Blood. 2008;112(9):3907–13.
Gaspar H, et al. How I treat ADA deficiency. Blood. 2009;114(17):3524–32.
Montiel-Equihua C, Thrasher A, Gaspar H. Gene therapy for severe combined immunodeficiency due to adenosine deaminase deficiency. Curr Gene Ther. 2012;12(1):57–65.
Chaffee S, et al. IgG antibody response to polyethylene glycol-modified adenosine deaminase in patients with adenosine deaminase deficiency. J Clin Invest. 1992;89(5):1643–51.
Malacarne F, et al. Reduced thymic output, increased spontaneous apoptosis and oligoclonal B cells in polyethylene glycol-adenosine deaminase-treated patients. Eur J Immunol. 2005;35(11):3376–86.
Chan B, et al. Long-term efficacy of enzyme replacement therapy for adenosine deaminase (ADA)-deficient severe combined immunodeficiency (SCID). Clin Immunol. 2005;117(2):133–43.
Serana F, et al. The different extent of B and T cell immune reconstitution after hematopoietic stem cell transplantation and enzyme replacement therapies in SCID patients with adenosine deaminase deficiency. J Immunol. 2010;185(12):7713–22.
Sauer AV, et al. Autoimmune dysregulation and purine metabolism in adenosine deaminase deficiency. Front Immunol. 2012;3:265.
Sauer A, et al. Alterations in the adenosine metabolism and CD39/CD73 adenosinergic machinery cause loss of Treg cell function and autoimmunity in ADA-deficient SCID. Blood. 2012;119(6):1428–39.
Sauer A, et al. Defective B cell tolerance in adenosine deaminase deficiency is corrected by gene therapy. J Clin Invest. 2012;122(6):2141–52.
Aiuti A, et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science. 2002;296(5577):2410–3.
Aiuti A, et al. Immune reconstitution in ADA-SCID after PBL gene therapy and discontinuation of enzyme replacement. Nat Med. 2002;8(5):423–5.
Gaspar HB, et al. Successful reconstitution of immunity in ADA-SCID by stem cell gene therapy following cessation of PEG-ADA and use of mild preconditioning. Mol Ther. 2006;14(4):505–13.
Candotti F, et al. Gene therapy for adenosine deaminase-deficient severe combined immune deficiency: clinical comparison of retroviral vectors and treatment plans. Blood. 2012;120(18):3635–46.
Carbonaro DA, et al. Gene therapy/bone marrow transplantation in ADA-deficient mice: roles of enzyme-replacement therapy and cytoreduction. Blood. 2012;120(18):3677–87.
Aiuti A, et al. Multilineage hematopoietic reconstitution without clonal selection in ADA-SCID patients treated with stem cell gene therapy. J Clin Invest. 2007;117(8):2233–40.
Kohn DB, et al. Engraftment of gene–modified umbilical cord blood cells in neonates with adenosine deaminase deficiency. Nat Med. 1995;1(10):1017–23.
Onodera M, et al. Successful peripheral T-lymphocyte-directed gene transfer for a patient with severe combined immune deficiency caused by adenosine deaminase deficiency. Blood. 1998;91(1):30–6.
∙ Gaspar HB, et al. Immunological and metabolic correction after lentiviral vector mediated haematopoietic stem cell gene therapy for ADA deficiency [abstract]. Mol Ther. 2014;22(S1):S106. This abstract summarizes the disclosed preliminary information from the current LV HSC-GT for ADA SCID clinical trial in the UK.
∙ Carbonaro DA, et al. Preclinical demonstration of lentiviral vector-mediated correction of immunological and metabolic abnormalities in models of adenosine deaminase deficiency. Mol Ther. 2014;22(3):607–22. This paper shows preclinical evidence of ADA SCID correction in mice using the LV that were translated into the clinics.
Neven B, et al. Long-term outcome after hematopoietic stem cell transplantation of a single-center cohort of 90 patients with severe combined immunodeficiency. Blood. 2009;113(17):4114–24.
Benjelloun F, et al. Stable and functional lymphoid reconstitution in artemis-deficient mice following lentiviral artemis gene transfer into hematopoietic stem cells. Mol Ther. 2008;16(8):1490–9.
Lagresle-Peyrou C, et al. Restoration of human B-cell differentiation into NOD-SCID mice engrafted with gene-corrected CD34+ cells isolated from Artemis or RAG1-deficient patients. Mol Ther. 2008;16(2):396–403.
Lagresle-Peyrou C, et al. Long-term immune reconstitution in RAG-1-deficient mice treated by retroviral gene therapy: a balance between efficiency and toxicity. Blood. 2006;107(1):63–72.
∙ Pike-Overzet K, et al. Correction of murine Rag1 deficiency by self-inactivating lentiviral vector-mediated gene transfer. Leukemia. 2011;25(9):1471–83. Preclinical correction of Rag1 deficiency using LV, known to be safer than GV.
van Til NP, et al. Recombination-activating gene 1 (Rag1)-deficient mice with severe combined immunodeficiency treated with lentiviral gene therapy demonstrate autoimmune Omenn-like syndrome. J Allergy Clin Immunol. 2014;133(4):1116–23.
Yates F, et al. Gene therapy of RAG-2−/− mice: sustained correction of the immunodeficiency. Blood. 2002;100(12):3942–9.
van Til NP, et al. Correction of murine Rag2 severe combined immunodeficiency by lentiviral gene therapy using a codon-optimized RAG2 therapeutic transgene. Mol Ther. 2012;20(10):1968–80.
Method of the Year 2011. Nat. Methods. 2011;9(1):1–1.
∙∙ Genovese P, et al. Targeted genome editing in human repopulating haematopoietic stem cells. Nature. 2014;510(7504):235–40. This work is the first proof of principle for gene editing in the context of human HSC and SCID, demonstrating engraftment of IL2RG gene edited HSC from both healthy and SCID-X1 human donors by means of ZFN technology.
Friedmann T, Roblin R. Gene therapy for human genetic disease? Science. 1972;175(4025):949–55.
Cartier N, et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science. 2009;326(5954):818–23.
Cavazzana-Calvo M, et al. Transfusion independence and HMGA2 activation after gene therapy of human β-thalassaemia. Nature. 2010;467(7313):318–22.
Biffi A, et al. Lentiviral hematopoietic stem cell gene therapy benefits metachromatic leukodystrophy. Science. 2013;341(6148):1233158.
Disclosure
Miguel Calero-Garcia and H. Bobby Gaspar declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Immunology.
Rights and permissions
About this article
Cite this article
Calero-Garcia, M., Gaspar, H.B. Gene Therapy for SCID. Curr Pediatr Rep 3, 11–21 (2015). https://doi.org/10.1007/s40124-014-0069-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40124-014-0069-1