Abstract
Characterizing biological richness at the landscape level is conveniently done using the plant as an indicator of biota. The congruence between plant and animal biological richness (BR) was studied by extending the scope of methodology from past studies in order to understand the question; does higher plant richness encourages higher animal richness? Using satellite images, 16 forest vegetation classes to integrate BR attributes for 85 plants and 271 animal species were derived. Plant BR analysis linked six biodiversity attributes (i.e., spatial, phytosociological, social, physical, economic and ecological) together based on their relative importance. The information of four terrestrial animal taxa (i.e., mammal, bird, reptile and amphibian) from various resources was utilized over five surrogates of biological richness (habitat suitability, spatial heterogeneity, eco-climatic stability, plant structural properties, and forage). A methodological basis of spatial enumeration of animal richness was provided in one of the most biologically rich landscapes of India, forming part of Indian Gangetic plains. It was observed that positive congruence between BR of plant and animal with a spatial overlap of 82.23% for the inclusive BR. Significant positive correlation (R 2: 0.7) was observed for high BR values (7–9) of animal and plant. Authors believe the strengths of our study are (i) translation of animal characterization onto a spatial map, (ii) collection and utilization of scattered data of animals from varied resources for Indian region where proper documentation is lacking (iii) generation of an inclusive BR map having higher conservation potential, and (iv) creation of a database having retrieval and future modification capability. This methodology has the potential for inclusive plant and animal biological richness for effective conservation implications with more site-specific data of a wider range of animals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Biological richness (BR) is a cumulative property of an ecological habitat and its surrounding environment [1]. Characterizing BR offers useful insights for conservation prescription and policy investments. The extraordinary richness (as a component of diversity) of the terrestrial fauna, is clearly due largely to the diversity provided by terrestrial plants and remains poorly documented [2]. Central questions among conservation biologists and ecologists include whether plant diversity influences animal diversity. Documentation for ecological status of the terrestrial species either animal or plant has long been practiced, but inclusive studies (integrating both plant and animal) for spatial BR distribution are limited [3] and theoretical [4].
Animals are known to influence and sometimes help maintain plant species richness in terrestrial systems and vice versa [5]. Ehrlich and Raven [6] advocated how plant–animal interactions have played a very important role in the generation of Earth’s biodiversity. So, it is important to understand the sociological relationships that living organisms have with each other to understand their ecology. Studies on BR considering plants [1, 7, 8] or animal [9,10,11,12,13] has increased in the recent past, but very less [3] has been talked about the inclusive BR analysis combining earth observation data and field inputs. Our understanding to access inclusive BR analysis has increased with technology [14], but there is still a gap in knowledge to understand the ecological interactions between sessile plants and moving animals.
A variety of ecological applications require data from broad spatial extents that cannot be collected using field-based methods [15]. With the onset of geoinformatics techniques comprising remote sensing, global positioning system (GPS), integrative tools, such as GIS, is realized as a complementary system for ground-based ecological studies, and a strong tool to understand the species spatial distribution. Vegetation distribution map, inventory, and in situ community analysis are important means to study the interaction between forest and their wildlife [16,17,18,19,20]. Such maps can provide baseline information against which changes in species richness can be monitored at different levels and scales. Many studies based on remote sensing techniques have been carried out for plant BR analysis [7, 8, 21,22,23,24], but only a few [3, 25, 26] for animals. Remote sensing techniques used to study plants are based on characteristic spectral reflectance features of species or communities, and for this purpose, objects need to be sessile for accurate assessment. Techniques used in plant BR analysis cannot be applied to the animals in a similar fashion. A major issue complicating the assessment of animal species occurrence is its mobility, especially migrants with longer mobility occupying a wide range of natural and anthropogenic habitats. The idea of mapping plant BR as a proxy to map high animal BR area is based on the accurate plant cover data and availability of animal data for the same region. The abundance of plant species in an area might influence animal richness, and at a small spatial scales, such positive associations have been tested in observational studies [27]. Much evidence has also been produced to validate the relation that ecosystem functions provided by diverse plant communities support rich animal communities. At some very gross level plant and animal richness patterns must be congruent, since both increases from the poles to the tropics [28].
The multi-faceted interactions among living organism make BR calculation difficult to capture in one description and thereby, its measurement with a single factor [29,30,31]. Many researchers suggested to use species diversity of certain taxa as a surrogate to understand the same of other taxa thereby it helps identifying areas to be protected [32,33,34,35]. Studies conducted by Hutchinson [2], Lamoreux et al. [36] and Qian [37] have used non-spatial regression methods to calculate congruence between plant and animal richness. Duro et al. [38] used a combination of direct and indirect approaches to access spatial diversity that can be derived from satellite data by considering surrogates like productivity; disturbance, topography and land cover to monitor potential biodiversity change. Rocchini et al. [39] reviewed spectral heterogeneity of existing sensors and their ability for estimating species diversity. Here authors argued that spatial variability extracted from remotely sensed images can be used as a proxy of species diversity, as these data provide an inexpensive means of deriving environmental information for large areas in a consistent and regular manner. Kuenzer et al. [40] in their review article about the use remote sensing for biodiversity monitoring, suggested that mapping vegetation attributes for diversity are easier and many more studies assess vegetation biodiversity from space than animal biodiversity. Leyequien et al. [14] in their review study of remote sensing based animal biological richness analysis suggested five vegetation based surrogates. They proposed that habitat suitability, spatial heterogeneity, eco-climatic stability, structural properties of habitat, and forage quality can act as surrogates to evaluate the biological richness for terrestrial animals (like mammals, birds, reptiles, amphibians, and many invertebrates). The use of spatial heterogeneity as a proxy to understand the pattern of biological richness is limited to a small number of studies [26, 41]. It has long been accepted that spatially heterogeneous ecosystem may support richer species assemblages compared to homogenous ecosystems [42,43,44,45] because of the creation of niche differentiation [46, 47]. The most commonly used remote sensing product for quantifying productivity and above ground biomass of ecosystems is the normalized difference vegetation index (NDVI) [48,49,50,51].
In this article authors analyzed the congruence between plant and animal species richness by extending the scope of Matin et al. [3] methodological basis from past study in order to understand the question; does higher plant richness encourages higher animal richness? We extended the possibility of congruence between plant and animal richness on a moderate spatial scale (1:50,000)by incorporating four terrestrial animal taxa. This study might be able to nullify the methodological bias for calculating BR of animal characterization onto a spatial map. This study may also provide a baseline data in GIS format, collected from scattered sources on animal distribution from varied resources, having retrieval and future modification capability.
2 Materials and Methods
The entire work is an integration of data collected from various sources and means (Table 1). Remote sensing data to prepare vegetation type map was acquired from National Remote Sensing Agency, India [52]. Authors collected plant data from the field to calculate inputs like species richness (SR), ecosystem uniqueness (EU) and economic value (EV; Table 2). Data like NDVI(max), temperature and rainfall were downloaded from their respective websites. Literature-based information to calculate spectral properties of vegetation, forage type as attractant and deterrent were gathered from various published articles (see Table 1). Road, rail and point settlement data were digitized from toposheets and updated with the Google Earth and IRS P6-L-III satellite images.
2.1 Study Area
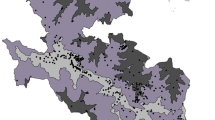
This study was conducted to the extent of 730 km from Himalayan foothills to the Vindhyan range of mountains. The study area (Fig. 1a) is located between the parallels of 25°18′ N and 30° 25′ N latitude and 77° 04′ E and 84°38′ E longitude with a geographical extent of 187150 km2 in part of Indian Gangetic plains. The entire study area has been biogeographically divided into three parts, i.e., Northern Terai, Central Gangetic plains and Southern Semi-arid land (divisions can be seen in Fig. 1b). Vegetation cover is distinct in all three zones with respect to phenological adaptation, composition, structure, and diversity [52]. The total forest area mapped in the region is 5300 km2, occupying 2.93% of the total geographical area. Most of the forests in the region, especially in the Central Gangetic plains are known to be influenced by biotic interferences and the original vegetation community no longer exists in these areas [53]. The Terai zone is listed among the most productive eco-regions of the world, well known for its vast biodiversity and high productivity [54]. Terai possesses marshy grasslands, savannas, and forests located south of the outer foothills of the Himalaya, the Siwalik hills, and north of the Indo-Gangetic plain of the Ganges. The Southern Semi-arid zone is mosaic of dry gullied land, with scrub as the dominant vegetation cover. The main motivation for selecting this study site is the wealth of wildlife animals, especially in the Northern Terai. Several species of wildlife have become extinct in the last few decades, even though its avifauna is among the richest in the country. The entire study area is the home of some of many endangered species like Panthera tigris, Gavialis gangeticus, Gazella gazelle, Grus antigone, Haliaeetus leucoryphus and Platanista gangetica.
a Showing part of Indian Gangetic Palins (Uttar Pradesh state) as study area with winter season IRS P6 LISS-III satellite data, and locations of 466 nested quadrate samples as black dots, b vegetation and land cover map derived from two seasons satellite images, showing 16 vegetation and 5 non-vegetation classes. Three bio-geographical zones (i.e., upper north Terai, central Gangetic plains and Semi-arid, in lower left side) are delineated and shown as thick black lines
2.2 Vegetation Type Mapping
The Indian remote sensing, linear imaging self-scanning (IRS-P6, LISS-III) satellite product of 23.5 m spatial resolution were used to prepare the vegetation type map. A total 26 scenes of LISS-III sensor with bands ranging from 0.52 to 1.70 µm wavelength were acquired. Vegetation classification scheme (Table S1) adopted in this study was based on physiognomy, the structural floristic influence of climate, topography and biotic factors.
We used two season satellite data for optimal discrimination of various vegetation types considering deciduous phenological stage during April to May in association with October and November (2005–2006) mostly in fair weather. Rectified satellite data were used to classify vegetation and land cover map at 1:50,000 scale. On-screen visual interpretation technique, considering visual keys, such as tone, texture, shape, pattern, and relationship to other objects were applied to classify the entire land use and land cover (LULC)onto a false color composite image (NIR, red and green band combination). Automated mapping gives rapid results but with relatively lower accuracy when compared to the manual on-screen classification system. A total of 21 LULC units (sixteen vegetation and five non-vegetation) were mapped in this study based on a predefined classification scheme (Table S1). A minimum of 10 GPS based ground control points were collected (from the core zone of each class) during the field visits, which later utilized to validate the classified map.
2.3 Plant Data Collection
Field tours were carried out during 2008–2011 in the different season by a team of five members. Sixteen vegetation types were determined prior to the sampling according to vegetation classification developed by expert and based on Champion and Seth classification scheme [55]. In order to carry out the ground level phytosociological study; a sampling intensity of 0.002% of the forest area was envisaged. Accordingly, the entire region required 555 sample points, distributed proportionally among all the major forest types. However, based on accessibility and administrative difficulties in the region, a total of 466 sampling points were laid. We enumerated 54937 numbers of plants (density of tree species) including 284 unique species in 16 vegetation types (based on Champion and Seth classification scheme 1968). Stratified random sampling with probability proportional to size method [56] was adopted for field inventory using nested quadrats to record various life-forms. Based on the species-area curve, an optimized field plots size of 0.0004 km2 was adopted uniformly for all the vegetation types. While the sample size for tree, shrub, and herbs were considered 20 m × 20 m, 5 m × 5 m and 1 m × 1 m, respectively. In each quadrate CBH (circumference at breast height at 1.37 m from ground level) and height of trees were measured. Collected data were enumerated for community structure analyses like Shannon diversity [57] and Margalef richness index [58] to calculate diversity and abundance. All the species were also evaluated for their endemism status and total economic value [59] (Table S3).
2.4 Animal Data Collection
For animal species information, we used data documented by the Uttar Pradesh state forest department in their working plan book series for the year 2005–2015 [60]. A total of 271 animal species from four classes of terrestrial taxa were evaluated for the IUCN red list of threatened species. Species information then used to calculate the EU status, which is nothing but the average value calculated from a number of endemic species observed in different vegetation types according to Red data book citations (www.iucnredlist.org). Besides, ground knowledge, species composition, the extent of the area, contiguity, importance in the landscape and critical habitat value of the patches were also considered for EU calculation [1].
2.5 Surrogates of Animal Biological Richness
2.5.1 Habitat Suitability Mapping
Habitat suitability is widely used as a remotely sensed proxy for species distribution and richness [3]. It mainly considers vegetation type map as the primary input for suitability calculation. We have prepared habitat suitability map using raster layers of primary habitat, species richness, biodiversity index, vegetation fragmentation and disturbance index (DI), in SPLAM software [52]. DI, as biotic disturbance proximity buffer zones were created around roads and human settlements for 200, 500, and 1000 m to understand a different level of disturbance with respect to distance from the source. Out of five parameters used, fragmentation and DI possess negative influences to species richness, and hence have negative participation in the weight assignment. Habitat usage or characteristics of nesting, breeding, or burrowing sites were identified on the satellite image and validated from inputs provided by forest department (animal sighting) and similar pixel was classified and mapped as primary habitat. Similarly, spectral properties of the pixel corresponding to a known location were used as training data to classify the imagery for a larger area. Species richness and ecosystem uniqueness maps were generated using field inputs (Table 2).
2.5.2 Spatial Heterogeneity in Living Environment
Spatial heterogeneity with respect to primary productivity was calculated using 1 km global product of Aqua MODIS normalized difference vegetation index (NDVImax) composite of 8 days (15–22 April, 2005), along with vegetation interspersion index and juxtaposition index (proximity of the vegetation types) map. This is based on the assumption that high NDVI value corresponds to higher above ground primary productivity [61], and can be correlated with high animal occurrence and diversity [14]. Interspersion, which is a count of dissimilar neighboring classes with respect to central class, calculated using 3 × 3 pixel convolution window, along with juxtaposition to evaluate the proximity of habitat types and relative importance of adjacency [62]. Areas with high-level of juxtaposition could possess a high degree of the biological interface, and therefore given highest priority value for conservation [1].
2.5.3 Eco-climatic Stability
Accessing species richness for animals may further increase by adding climate dependent variables to the analysis. Seasonal variations control differences in plant species growth and establishment patterns, leading to changes in species composition and productivity [63]. Consequently, annual variations in vegetation could induce a change in the spatial distribution of plant phenology and growth [49]. Hence, based on the assumption that climatically more stable region may support high animal richness [41]; we have calculated climate anomaly for the region. We used past 50 years rainfall and temperature data provided by climate research unit (www.cru.uea.ac.uk), to generate the eco-climatic stability index map. Rainfall and temperature data of 50 years were divided into ten layers of 5 year duration, and compared for the anomaly. Areas with the highest deviation from the average value for both rainfall and temperature were marked as least stable and vice versa.
2.5.4 Structural Properties of Habitat
A vertically diverse forest can possess a rich biota [64], similar case observed in the current study region. Here we have prepared map based on the structural properties of habitat (height of the tree, its density, and biomass) using structural information from published literature (Table 1; 60,65). We identified representative vegetation species for each class with similar age group and latitudinal location and analyzed for above mentioned structural parameters.
2.5.5 Forage Types as Attractant or Deterrent
It was evident that the spatial distribution of many wildlife species is passively influenced by the variation in plant type and its chemical constituents [66]. Accordingly, representative vegetation species were searched for major chemical constituent and classified as attractant or deterrent based on the existing literature [67]. Plants based on chemical compounds were classified as deterrents (e.g., diformyl phloroglucinols) [68] and attractants (e.g., Sodium and Calcium) [69].
2.6 Modeling Approach
2.6.1 Plant BR Calculation
Spatial modeling techniques provide the means to model spatial relationships in data and fill the gap where no data are available [70]. We used the existing methodology [1, 7, 8] to calculate plant BR. Six parameters (i.e., spatial, phytosociological, social, physical, economic, and field observations) were considered to compute plant BR using a customized SPLAM software. Higher plant BR was assigned to the habitats with (i) high species diversity, (ii) high degree of ecosystem uniqueness (EU), (iii) high economic value (Table S3), and (v) low disturbance level. This simple idea of integrated three-tier modeling approach for (i) utilization of geospatial tools, (ii) field survey and (iii) landscape analysis; formed the basis of rapid assessment of plant biological richness.
2.6.2 Animal BR Calculation
In the lineup with the existing methodology adopted by Matin et al. [3], we have integrated four groups of terrestrial animal taxa i.e., mammal, bird, reptile, and amphibian, and evaluated for five surrogates of animal BR (habitat suitability, spatial heterogeneity, eco-climatic stability, structural properties, and forage). In addition, direct and indirect measurements of species diversity, uniqueness, and distribution were also illustrated. Finally, all the data were spatially harmonized with respect to highest pixel size (1 km) among all the raster data and re-projected to fit into single GIS platform in ArcGIS software.
2.6.3 Weight Assignment for Modeling
Rescaling of the values by applying a transformation function to convert the resulting values onto a specified continuous evaluation scale helps to retain the data consistency and to standardize the range of independent variables or features of data. All raster-based spatial layers were rescaled to 1 and 9 [71]. Raster layers were prepared using field input values of relative importance. Animal habitat suitability was calculated for individual species with defined criteria. Here, we have evaluated individual species from each taxon for their habitat preference and finally overlaid to create a single map for one taxon. Four maps for all the four taxa were created and merged with relative weight to prepare a final animal habitat suitability map. Species richness and ecosystem uniqueness coined with positive weights (Table 3), while disturbance and fragmentation index map with negative weights [72]. Weights assigned to structural properties of vegetation, i.e., height, density and biomass as surrogates of richness were based on published literature (Table 4) [65] while the final map was created using mean values of all the three layers. All four classes of the animal were individually related with representative vegetation species mapped for forage attractant or deterrent and assigned weights accordingly. Finally, the average weights were converted to integer values ranging from 1 to 9 [71]. We used known conservation status [73] to derive relative weights for EU using species endemism status and SR. We calculated number of individuals for richness [58], and BV through Shannon’s index [57] for plants and animals. EU, SR, and BV were enumerated quantitatively for BR weights computation (Table 3). Higher BR values were assigned to the layers with (i) high species diversity (SD), (ii) high degree of EU, (iii) high economic value (EV) and (v) low disturbance [1].
2.6.4 Inclusive BR Calculation
Plant and animal data collected in the field with other spatial and ancillary data were utilized to prepare the map of plant and animal BR and finally merged together to prepare an inclusive BR map (Fig. 2). Before final overlaying, both layers were checked for similar pixel size and projection parameters. To analyses how animal BR value varies along all the gradient of plant BR, we calculated a correlation using all the data points from both the BR maps. A total 29035 pixels corresponds to nine gradients of BR were arranged to see a correlation at different levels of the gradients.
3 Results
3.1 Vegetation Type Distribution and Diversity Analysis
The vegetation classes reported in this study were mapped into 16 different units (9 forest and 7 non-forest classes Fig. 1b), covering 2.93% and 2.34% of the total geographical area, respectively. Dense forests were found only in the Northern Terai region, dominated by species of moist and dry sal (Shorea robusta). Moist lands are occupied with tall grass of Eulaliopsis binata, Saccharum spontaneum and Imperata cylindrica, which possesses suitable habitat for many ruminant species and hence its predators. Scrubland was found dominating in the southern Semi-arid zone, where the temperature is relatively high with scanty rainfall. Such areas are good habitat for shrubby species like Prosopis juliflora, Acacia nilotica, Calotropis procera and for reptiles. The Central Gangetic plains were found to be almost treeless, with only 0.73% area under forest cover with planted patches of Eucalyptus hybrid, Populus deltoids, and Prosopis juliflora. Diversity and richness indices for 16 vegetation types were calculated and final values used as an input for BR calculation. Highest richness was observed in moist deciduous forest followed by the mixed formation of sal and teak (Tectona grandis), and least for orchards and forest plantation. Among forest types, maximum diversity was observed in Aegle marmelos, followed by lowland swamp forest and other formations of dry and moist sal (Fig. 3). Acacia catechu (Khair) was found showing surprisingly high diversity value may be due to the high number of associated regenerated species. Scrubs possess higher diversity (Anogeissus pendula and Salvadora oleoides) than of forest. We have observed six threatened plant species as per international IUCN status(Table S2) in the region, and calculated total importance value (TIV) as the economic value [59] (Table S3) for 179 species.
Comparing Shannon diversity and Margalef richness index for 16 vegetation types where APS, Anogeissus pendula scrub; SOS, Salvadora oleoides; KHR, Acacia catechu; AMF, Aegle marmelos; RGL, riverine grass land; FPL, forest plantation; LSF, lowland swamp forest; DDF, dry deciduous forest; TMD, Teak mixed moist deciduous forest; SMD, sal mixed moist deciduous forest; THF, thorn forest; OSC, open scrub; PJS, Prosopis juliflora scrub; GLD, grass land; MDF, moist deciduous forest; ORC, orchards
3.2 Animal Data Collection and Analysis
A total of 271 animal species has been observed, falling geographically in 16 vegetation types. Panthera tigris, Elephas maximus, and Gazella gazelle were found in the dense forest of the Northern Terai zone, while many species of birds (like Sarus crane) and mammals (like Macaca mulatta) were reported in the Central Gangetic plains. The Semi-arid northern zone was found to be rich in reptile population, especially Gavialis gangeticus in areas of Chambal crocodile sanctuary. Out of 271 animal species in the region, 50 were found internationally threatened, 3 critically endangered, 18 endangered, 16 vulnerable and 13 near threatened as per the IUCN red list 2012 (Table S2),and used to calculate the EU. Protected forests and wildlife sanctuaries were observed with maximum richness for both plant and animal.
3.3 BR Calculation and Congruence
All the three BR maps were scaled between 1 and 9 (1: low and 9: high) for the quantitative representation of the result, and assigned with different colors. Plant BR map was showing high richness values in Southern Terai region, and least in Central Gangetic plans (Fig. 4a). High plant BR was observed in sal mixed moist deciduous forest, followed by moist deciduous forest and lowland swamp forest. On the other hand, least plant BR was observed in orchards and scrubs of Semi-arid zone. Animal BR found highest in the protected areas and wildlife sanctuaries. Maximum animal BR was observed in the core protected zones of lowland swamp forest, followed by sal mixed moist deciduous forest, and deciduous forest in the Terai zone (southern side). Lowest animal BR was reported in all the scrublands areas followed by dry grassland and orchards (Fig. 4b). The inclusive BR map (Fig. 4c) exhibits highest BR values in the area of lowland swamp forest, followed by and moist deciduous forest formation, while minimum in grass and orchards. Inclusive BR map found holding more area under high BR category while compared to plant BR map (Table 5).
Biological richness (BR) map of a plants, b animals, c inclusive BR for both plants and animals (82.23% overlapping). Correlation matrix of plant BR and animal for d BR values between 1 and 3, e BR values between 4 and 6, f BR values between 7 and 9 and g considering all values. Color represents green as high and brown as low BR values, and rest in yellow color
Areas with high plant richness encourage greater animal richness was witnessed in the regions of high plant BR. Although high animal richness was also observed in some of the moderately rich vegetation units, and least in grassland and scrubs, where plant BR reported very low. Though, the maximum spatial overlapping between plant and animal BR maps is 82.23%, which don’t ponder the quantitative variations between richness, but it shows only total overlapping areas. A total 29035 pixel values correspond to nine gradients of BR shown that BR congruence is decreasing with a decrease in plant BR value. Highest congruence was observed in high BR values (7–9) with 17439 pixels which are 60% of the total matching pixels with a correlation coefficient of R 2 : 0.75 (Fig. 4f). The BR value for the range 1–6 was observed with minimum positive correlation (Fig. 4d, e). Moderate correlation of R 2 : 0.43 was observed for all the nine gradients of BR (Fig. 4g).
4 Discussion
Our analysis indicated that plant richness drives animal richness and such observations are possible only using integrated approach by field-based information with geoinformatics technique. Furthermore, we also believe that our study has generated a database on animal distribution for Indian Gangetic plains which have retrieval and future modification capabilities. We analyzed the spatial extent of the vegetation types and wildlife within it, as a proxy to understand the entire ecosystem and explored the possibilities using satellite-based observations. Assessing species information by either direct method by field sampling [74] or an indirect method using remotely sensed information [75] have their own advantages but latter is more in practice [40]. Such tool provides quantifiable information that can be repeatedly obtained and updated [38]. Remote sensing techniques that have aided the studies on the plant cannot be applied directly to animals; hence surrogacy concept is obligatory [76]. Among five surrogates of animal BR used in this study, mapping habitat suitability is most straightforward approach and has a strong positive correlation with the animal species richness [77]. Furthermore, habitat use patterns can also be derived from the movements of radio or satellite collared individuals [78] using the method of area of occupancy and extent of occurrence [79]. Though, other surrogates like spatial heterogeneity, eco-climatic stability and structural properties of habitat are dynamic in nature and have an indirect influence on animal BR. These surrogates may change with prevailing climatic conditions and cannot stand same for a longer span of time. Most studies relating forest structural properties to animal richness relied on height variability, percent canopy cover as density, and aboveground biomass [14]. Chemical constituents as attractant or deterrent are more consistent surrogate for animal BR but require extensive laboratory-based analysis. Although some authors like Grant et al. [66] recommended, that studies should focus on monitoring seasonal changes in foliar nutrient concentration as well as extending the method to predict other macro nutrients (P, K, Na, Mg and Ca) and secondary compounds in both grass and tree canopies.
In the current study, we considered 4 taxa of animals, which include 271 animals in 16 vegetation units. In this way, we extended the method proposed by Matin et al. [3], which enabled us to cover more vulnerable species for wider perspective for conservation. This paper validates the assumption that high plant richness areas support high animal richness, by superimposing plant and animal BR maps and observed adequate results to approve the hypothesis. We observed a positive correlation between plant and animal BR while compared for congruence between high BR values. The highest correlation was observed in high plant BR, mostly in the swampy and dense forest. BR was observed decreasing with decrees in BR values, this shows that there is no or least congruence between low BR areas and high in high BR areas, which supports our hypothesis. Currie [80] also reported similar observation with a very strong correlation between amphibians and plant biological richness across North America. Andrews and O’Brien [81] also established a positive relationship between plant and mammal species richness for southern Africa and found that variation in plant species richness was responsible for up to 75% of the variation in mammal species richness. Zhao et al. [82] in their study on the relationship between species richness of vascular plants and terrestrial vertebrates on a broad scale across 186 nature reserves in China also observed positive correlation. Their analysis based on multiple regression methods indicated that plant richness was a significant predictor of richness patterns for terrestrial vertebrates (except birds). Qian [37] examined the relationship between the biological richness of vascular plants with four classes of terrestrial vertebrates at a regional scale (3.5 × 106 km2), in 28 provinces of China. The author observed that plant richness was correlated with animal richness more strongly than the richness of different animal groups correlated with each other. Contrary, Hawkins and Pausas [28] in their study on the relationship between mammals and vascular plants species richness in Catalonia, Spain reported insignificant results. They suggested that mammal richness pattern, as well as those of herbivores and carnivores if considered separately, only weakly corresponded to the pattern of plants and depend mostly upon other climatic factors. Contrary, there are very few studies available to validate the reverse scenario where high animal diversity considered for high plant richness. High animal diversity intensifies the count of herbivores and increases feeding rates within the region. Depending on the balance of these counteracting features, species-rich animal groups may put plants under top-down control or may release them from grazing pressure [83].
Such observational correlations for BR, reported in the previous studies are contradictory. Most of the studies resulted in strong and positive correlations, whereas others showed weak or negative relationships. These inconsistencies may result from limitations like sample size, spatial extent, and scale of studies [36]. Positive congruence of BR is more frequent in large-scale analyses, whereas weak or negative at small scales observations. We prepared different layers to analyze BR at moderate scale (1: 50,000), which reduces the effect of scale for correlation analysis. We understand that these divergences in results may be influenced by factors like different methods in evaluating surrogacy [84], different spatial scales, sample size [85], and diverse underlying bio-geographical patterns [86]. Though, all such relationship is more theoretical and lacks ground validation. Unlike any other study, we emphasized animal BR analysis on vulnerable species information by giving higher weight to species with critically endangered or endangered status (Table S2). Understanding correlation between vegetation types and vulnerable animals is more important for conservation prospective than delineating areas of high animal abundance. Hence, weight assignment is very crucial especially in animal BR calculation and have a vast effect on final output.
5 Conclusions
This study highlights the capability of remote sensing techniques to map ecological attributes and probably the only tool to generate seamless landscape maps. Vegetation is static, and hence the data collection is point-based, but due to the fugitive behavior of the animal, its data collection is either extent or surrogate based. Plant-based information are consistent, easy to sample and map hence, more reliable and could be considered primary information for any such analysis. Diverse plant habitats require considerably less attention than of degraded animal habitats because such areas may possess less species abundance, but with rare or endangered fauna. So, data on endangered and vulnerable species are crucial and must be used to prepare a priority map for animal conservation.
In process of extending the methodological basis for inclusive BR calculation; resulted in the positive congruence between plant and animal BR, especially in the areas of high plant richness. High plant BR area possesses a wide range of plant species which serve as food and shelter for animals and results in a diverse assemblage of animal species and customs a healthy ecosystem with producer and predator living in the same place. We believe that conservation actions would be more efficient if there is a strong congruence among taxa in the distribution of species. Positive BR congruence at higher values and insignificant in moderate and lower values may help the researcher to classify areas based on the priority for conservation and policy makers to propose different strategies. A rich BR area may be considered for conservation and mark as “protected land” while the rest for establishing a different level of biodiversity based on their BR gradients. Besides, understanding such correlations, between plant and animal may reduce effort to quantify both, and conserving one may work for other.
In contrast, it is intricate to prepare a map of BR comprising all forms of living organism and also to establish correlation between plant and animal species for their preferred habitat by using satellite data. Most promising development in terms of methodology observed in the assimilation of remote sensing data is to predict future scenarios for species using species distribution models [3, 14]. For generating future scenarios for inclusive BR maps, the inputs from the current study may serve as baseline information in the same study area, and the proposed methodology for any similar analysis.
6 Supporting Information
Table S3 list of TIV values of species found in the region, the value assigned as per field knowledge, existing literature and expert consultation.
References
Behera MD, Kushwaha SPS, Roy PS (2005) Geo-spatial modeling for rapid biodiversity assessment in Eastern Himalayan region. For Ecol Manag 207:363–384
Hutchinson GE (1959) Homage to Santa Rosalia or why are there so many kinds of animals? Am Nat 93:145–159
Matin S, Chitale VS, Behera MD, Mishra B, Roy PS (2012) Fauna data integration and species distribution modelling as two major advantages of geoinformatics-based phytobiodiversity study in today’s fast changing climate. Biol Cons 21:1229–1250
Wagner HH, Fortin J (2005) Spatial analysis of landscapes: concepts and statistics. Ecology 86:1975–1987
Hewitt N, Miyanishi K (1997) The role of mammals in maintaining plant species richness in a floating Typha Marsh in Southern Ontario. Biodivers Conserv 6:1085–1102
Ehrlich P, Raven P (1964) Butterflies and plants: a study in co-evolution. Evolution 18:586–608
Roy PS, Tomar S (2000) Biodiversity characterization at landscape level using geospatial modeling technique. Biol Cons 95:95–109
Behera MD, Roy PS (2010) Assessment and validation of biological richness at landscape level in part of the Himalayas and Indo-Burma hotspots using geospatial modeling approach. J Indian Soc Remote Sens 38:415–429
Löffler E, Margules C (1980) Wombats detected from space. Remote Sens Environ 9:47–56
Nellis MD, Bussing CE (1990) Spatial Variation in Elephant Impact on the Zambezi Teak Forest in the Chobe National Park, Botswana. Geocarto Int 5:55–57
Saveraid EH, Debinski DM, Kindscher K, Jakubauskas ME (2001) A comparison of satellite data and landscape variables in predicting bird species occurrences in the Greater Yellowstone Ecosystem, USA. Landscape Ecol 16:71–83
Kerr JT, Southwood TRE, Cihlar J (2001) Remotely sensed habitat diversity predicts butterfly species richness and community similarity in Canada. Proc Natl Acad Sci 98:11365–11370
Sibanda M, Murwira A (2012) Cotton fields drive elephant habitat fragmentation in the Mid Zambezi Valley, Zimbabwe. Int J Appl Earth Obs Geoinf 19:286–297
Leyequien E, Verrelst J, Slot M, Schaepman-Strub G, Heitkonig IMA, Skidmore A (2007) Capturing the fugitive: applying remote sensing to terrestrial animal distribution and diversity. Int J Appl Earth Obs Geoinf 9:1–20
Kerr JT, Ostrovsky M (2003) From space to species: ecological applications of remote sensing. Trends Ecol Evol 18:299–305
Sudhakar S, Rao PHVV, Krishnamurthy YVN, Rao RVR (1986) Application of remote sensing for vegetation mapping: a case study along the northern coastal districts of Andhra Pradesh. Photonirvachak J Indian Soc Remote Sens 14:1–8
Trisurat Y, Eiumnoh A, Murai S, Husain MZ, Shrestha RP (2000) Improvements of tropical vegetation mapping using a remote sensing technique: a case study of Khao National Park, Thailand. Int J Remote Sens 21:2031–2042
Franco-Lopez H, Ek AR, Baue ME (2001) Estimation and mapping of forest stand density, volume, and cover type using the k-nearest neighbors method. Remote Sens Environ 77:251–274
Thompson ID, Maher SC, Rouillard DP, Fryxell JM, Baker JA (2007) Accuracy of forest inventory mapping: some implications for boreal forest management. For Ecol Manag 252:208–221
Yakovleva AN (2010) A model of vegetation pattern at the Verkhneussuriysky biogeocenotic station. Russ J Ecolog 41:307–315
Gould W (2000) Remote sensing of vegetation, plant species richness, and regional biodiversity hotspots. Ecol Appl 10:1861–1870
Griffiths GH, Lee J, Eversham BC (2000) Landscape pattern and species richness; regional scale analysis from remote sensing. Int J Remote Sens 21:13–14
Foody GM, Cutler MEJ (2006) Mapping the species richness and composition of tropical forests from remotely sensed data with neural networks. Ecol Model 195:37–42
Engler R, Waser LT, Zimmermann NE, Schaub M, Berdos S, Ginzler C, Psomas A (2013) Combining ensemble modeling and remote sensing for mapping individual tree species at high spatial resolution. For Ecol Manag 310:64–73
Bohning-Gaese K (1997) Determinants of avian species richness at different spatial scales. J Biogeogr 24:49–60
Oindo BO (2002) Patterns of herbivore species richness in Kenya and current ecoclimatic stability. Biodivers Conserv 11:1205–1221
Haddad NM, Tilman D, Haarstad J, Ritchie M, Knops JMH (2001) Contrasting Effects of Plant Richness and Composition on Insect Communities: A Field Experiment. Am Nat 158(1):17–35
Hawkins BA, Pausas JG (2004) Does plant richness influence animal richness?: The mammals of Catalonia (NE Spain). Divers Distrib 10:247–252
Noss RF (1990) Indicators for monitoring biodiversity: a hierarchical approach. Conserv Biol 4:355–364
Wolfgang B (2003) Biodiversity and agri-environmental indicators—general scopes and skills with special reference to the habitat level. Agr Ecosyst Environ 98:35–78
Scholes RJ, Biggs R (2005) A biodiversity intactness index. Nature 434:45–49
Pearson DL, Carroll SS (1999) The influence of spatial scale on cross-taxon congruence patterns and prediction accuracy of species richness. J Biogeogr 26:1079–1090
Myers N, Mittermeier RA, Mittermeier CG, Da Fonseca GAF, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Moore JL, Balmford A, Brooks T, Burgess ND, Hansen LA, Rahbek C, Williams PH (2003) Performance of sub-Saharan vertebrates as indicator groups for identifying priority areas for conservation. Conserv Biol 17:207–218
Kemp JE, Ellis AG (2017) Significant local-scale plant-insect species richness relationship independent of abiotic effects in the temperate cape floristic region biodiversity hotspot. PLoS ONE 12(1):1–16
Lamoreux JF, Morrison JC, Ricketts TH, Olson DM, Dinerstein E, McKnight MW, Shugart HH (2006) Global tests of biodiversity concordance and the importance of endemism. Nature 440:212–214
Qian H (2007) Relationships between plant and animal species richness at a regional scale in China. Conserv Biol 21:937–944
Duro DC, Coops NC, Wulder MA, Han T (2007) Development of a large area biodiversity monitoring system driven by remote sensing. Prog Phys Geogr 31:1–26
Rocchini D, Balkenhol N, Carter GA, Foody GM, Gillespie TW, He KS, Kark S, Levin N, Lucas K, Luoto M, Nagendra H, Oldeland J, Ricotta C, Southworth J, Neteler M (2010) Remotely sensed spectral heterogeneity as a proxy of species diversity: recent advances and open challenges. Ecol Inf 5:318–329
Kuenzer C, Ottinger M, Wegmann M, Guo H, Wan C, Zhang GJ, Dech S, Wikelski M (2014) Earth observation satellite sensors for biodiversity monitoring: potentials and bottlenecks. Int J Remote Sens 35:6599–6647
Fjeldsa J, Lovett JC (1997) Biodiversity and environmental stability. Biodivers Conserv 6:315–323
Simpson EH (1949) Measurement of diversity. Nature 163:688
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
Lack D (1969) The numbers of bird species on islands. Bird Study 16:193–209
Huston MA (1994) Biological diversity: the coexistence of species on changing landscapes. Cambridge University Press, Cambridge
Tilman D, Lehman CL, Thomson KT (1997) Plant diversity and ecosystem productivity: theoretical considerations. Proc Natl Acad Sci 94:1857–1861
Loreau M (1998) Separating sampling and others effects in biodiversity experiments. Oikos 82:600–602
Tucker CJ (1979) Red and photographic infrared linear combinations for monitoring vegetation. Remote Sens Environ 8:127–150
Tucker CJ, Sellers PJ (1986) Satellite remote sensing of primary production. Int J Remote Sens 7:1395–1416
Box EO, Holben BN, Kalb V (1989) Accuracy of the AVHRRVegetation Index as a predictor of biomass, primary productivity and net CO2 flux. Vegetation 80:71–89
Devagiri GM, Money S, Singh S, Dadhwal VK, Patil P, Khaple AK, Devakumar AS, Hubballi S (2013) Assessment of above ground biomass and carbon pool in different vegetation types of south western part of Karnataka, India using spectral modelling. Trop Ecol 54:149–165
Anonymous (2011) Biodiversity characterization at landscape level in northern plains using remote sensing and GIS, Bishen Singh and Mahendra Pal Singh, Dehradun India. ISBN: 978-81-211-806-5
Singh S, Roy A, Upadhyay D, Matin S, Debnath B (2011) Biodiversity characterization at landscape level in Northern Plains using satellite remote sensing and geographic information system, regional analysis and synthesis of report, xxiv, 302. Bishen Singh and Mahendra Pal Singh, Dehradun
Chitale VS, Behera MD, Matin S, Roy PS, Sinha VK (2014) Characterizing Shorearobusta communities in the part of Indian Terai landscape. J For Res 25:121–128
Champion HG, Seth SK (1968) A revised survey of forest types of India. Manager of Publications, Government of India, Delhi
Anonymous (1998) Biodiversity Characterization at landscape level using geographic information system and geographic information system: project manual. Indian Institute of Remote Sensing, Dehradun
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Margalef R (1958) Information theory in ecology. Gen Syst 3:36–71
Belal A, Springuel BA (1996) Economic value of plant diversity in arid environments. Nat Resour J 31:33–39
UPFD-Lucknow (2008) Uttar Pradesh state forest department—working plan book series for the year 2005–2015
An N, Price KP, Blair JM (2013) Estimating above-ground net primary productivity of the tall grass prairie ecosystem of the Central Great Plains using AVHRR NDVI. Int J Remote Sens 34:3717–3735
Lyon JG (1983) Landsat derived landcover classifications for locating potential kestrel nesting habitat. Photogram Eng Remote Sens 49:245–250
Hobbs RJ (1990) Remote sensing of spatial and temporal dynamics of vegetation. In: Hobbs RJ, Mooney HA (eds) Ecological studies. Remote sensing of biosphere functions, vol 79. Springer, Berlin, pp 203–219
Brokaw N, Lent R (1999) Vertical structure. In: Hunter ML (ed) Maintaining biodiversity in forest ecosystems. Cambridge University Press, Cambridge, pp 373–399
Pandey U, Kushwaha SPS, Kachhwaha TS, Kunwar P, Dadhwal VK (2010) Potential of Envisat ASAR data for woody biomass assessment. Trop Ecol 51(117):124
Grant CC, Davidson T, Funston PJ, Pienaar DJ (2002) Challenge faced in the conservation of rare antelope: a case study on the northern basalt plains of the Kruger National Park. Koedoe 45:1–22
Kattge J, Ogle K, Bönisch G, Díaz S, Lavorel S, Madin J, Nadrowski K, Nöllert S, Sartor K, Wirth C (2010) A generic structure for plant trait databases. Methods Ecol Evol 2:202–213
Dury SJ, Turner B, Foley WJ (2001) The use of high spectral resolution remote sensing to determine leaf palatability of eucalypt trees for folivorous marsupials. Int J Appl Earth Observ Geoinf 3:328–336
Mutanga O, Skidmore AK (2004) Narrow band vegetation indices overcome the saturation problem in biomass estimation. Int J Remote Sens 25:3999–4014
Pearson DL, Carroll SS (1998) Global patterns of species richness: spatial models for conservation planning using bioindicator and precipitation data. Conserv Biol 12:809–821
Saaty T (1977) A scaling method for priorities in hierarchical structures. J Math Psychol 15:234–281
Bond WJ, Midgley J, Volk J (1988) When is an Island not an Island? Insular effects and their causes in fynbosshrublands. Acta Oecol 77:515–521
IUCN (2011) IUCN Red list of threatened species. IUCN, Gland, Switzerland. http://www.redlist.org. Accessed November 15, 2011
Lavorel S, Grigulis K, McIntyre S, Williams NSG, Garden D, Dorrough J, Berman S, Thébault FQA, Bonis A (2008) Assessing functional diversity in the field—methodology matters! Funct Ecol 22:134–147
Turner W, Spector S, Gardiner N, Fladeland M, Sterling E, Steininger M (2003) Remote sensing for biodiversity science and conservation. Trends Ecol Evol 18:306–314
Sætersdal M, Gjerde I, Blom HH, Ihlen PG, Myrseth EW, Pommeresche R, Skartveit J, Solhøy T, Aaas O (2003) Vascular plants as a surrogate species group in complementary site selection for bryophytes, macrolichens, spiders, carabids, staphylinids, snails, and wood living polypore fungi in a northern forest. Biol Cons 115:21–23
Tews J, Brose U, Grimm V, Tielborger K, Wichmann MC, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31:79–92
Bechtel R, Sánchez-Azofeifa A, Rivard B, Hamilton G, Martin J, Dzus E (2004) Associations between Woodland Caribou telemetry data and Landsat TM spectral reflectance. Int J Remote Sens. 25:4813–4821
Boitani L, Sinibaldi I, Corsi F, Biase A, Carranza ID, Ravagli M, Reggiani G, Rondinini C, Trapanese P (2007) Distribution of medium- to large-sized African mammals based on habitat suitability models. Biodivers Conserv 17:605–621
Currie DJ (1991) Energy and large-scale patterns of animal-species and plant-species richness. Am Nat 137:27–49
Andrews P, O’Brien EM (2000) Climate, vegetation, and predictable gradients in mammal species richness in southern Africa. J Zool 251:205–231
Zhao S, Fang J, Peng C, Tang Z (2006) Relationships between species richness of vascular plants and terrestrial vertebrates in China: analyses based on data of nature reserves. Divers Distrib 12:189–194
Schneider FD, Brose U, Rall BC, Guill C (2016) Animal diversity and ecosystem functioning in dynamic food webs. Nat Commun 7(12718):1–8. https://doi.org/10.1038/ncomms12718
Lund MP, Rahbek C (2002) Cross taxon congruence in complementarity and conservation of temperate biodiversity. Anim Conserv 5:163–171
Warman LD, Sinclair ARE, Scudder GGE, Klinkenberg B, Pressey RL (2004) Sensitivity of systematic reserve selection to decisions about scale, biological data and targets: case study from southern British Columbia. Conserv Biol 18:655–666
Howard PC, Viskanic P, Davenport TRB, Kiyenyi FW, Baltzer M, Dickinson CJ, Lwanga JS, Matthews RA, Balmford A (1998) Complementarity and the use of indicator groups for reserve selection in Uganda. Nature 394:472–475
Acknowledgements
The study was taken under joint DOS-DBT program entitled ‘Biodiversity characterization at landscape level using remote sensing and GIS’ with financial assistance from NRSC, Hyderabad, their co-operation is thankfully acknowledged. Authors are also thankful to Additional PCCF, Dr. S.K. Dutta, and entire team of department of the forest, Uttar Pradesh, Lucknow, for providing data and their kind co-operation during various field tours. We also acknowledge climate research unit (CRU) for providing climate data.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Matin, S., Behera, M.D. & Roy, P.S. Demonstrating Surrogacy of Animal Diversity with Plant Diversity and Their Integration to Assess Inclusive Biodiversity: A Geoinformatics Basis. Proc. Natl. Acad. Sci., India, Sect. A Phys. Sci. 87, 911–925 (2017). https://doi.org/10.1007/s40010-017-0459-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40010-017-0459-1