Abstract
Termites provide ecosystem services and have been used as a biological index of soil quality and fertility because of their key role in soil nutrient cycling that enables trees associated with their activities to remain evergreen throughout the year. To gain further insight into this situation, we compared the soil physico-chemical properties of termitaria and their surrounding soils in three Nigerian ecological zones including savannah, derived savannah and rainforest. Parameters evaluated were the pH, cation exchange capacity (CEC), organic carbon (OC), available phosphorus (Av.P) and total nitrogen (TN); base saturation (BS) and soil texture were also determined alongside the elemental (K, Na, Ca, Mn, H, Al) composition. The pH, OC, Av.P and TN in the termitaria were significantly higher than those in the surrounding soils. There were significant differences in the composition of the elements among sites in the termitaria and surrounding soils. The predominant analyte in the termitaria was Al, being in significantly higher concentration than in the surrounding soil. Na and Ca were also much higher in the termitaria than the surrounding soils in the guinea savannah zone. Soil pH had strong positive correlation with OC and CEC in the termitaria soil, but negative correlation with the surrounding soils outside the termitaria. On the other hand, both the termitaria and surrounding soils, base saturation, cation and soil texture were highly correlated between the ecozones. The findings from this study might promote nature conservation and enrichment of biodiversity in the different ecological zones, and could also support the local and poor farmers’ decisions in growing specific crops closer to the termitaria, especially the crops that require higher uptake of OC, N, Na and Ca for their growth and yields.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Termites are insects belonging to the order, Isoptera, and phylum, Arthropoda. They exist in the six families of Mastotermitidae, Kalotermitidae, Termopsidae, Hodotermitidae, Rhinotermitidae and Termitidae [38, 39]. In some agroecological zones, mound-building termites are known to have adverse effects on a number of arable and tree crops and human structures [9, 48, 49]. Mound-building termites are termite species, such as fungus-growing termites, [17] which cause significant damage to forest and arable crops. Many studies and research have been concentrated on how to control the pest, termite species [4, 48, 52, 54, 59]. However, out of over 2250 well-known species of termites, between 10 and 20% have a negative impact on humans, socially and economically [23]. The remaining 80% is an indication that termites are of more benefit to man and the environment than harm [58, 62]. For example, termites have been used as a biological tool to assess soil quality and fertility [21, 37, 47]. Some termite societies such as Macrotermes and Coptotermes are found in mounds (termitaria) which they build by excavating soil particles from depths using resources from the surrounding soil [32]. Kalotermitidae, Termopsidae and Rhinotermitidae are dominantly found in wood species while others are scattered in soil matrix and nest [25]. All species of termites in colonies are composed of different castes, namely the workers, soldiers and reproductive which are made up of the king and the queen, and each of the castes has defined role that contributes to the survival of the colony [31, 59]. Subterranean termites are associated with underground and vertical tunnels of buildings that look like mud tubes [8]. The tunnels provide protection to the termites from the external air, allowing for free movement of food by the workers within the nest. Generally, termites are influenced by the environment where they live when inflicting physical and chemical changes in the plant–litter soil system.
The mounds depict how termites are well adapted to the environment; construction of termitaria from the soil or mixture of soil and other materials or within soil horizons affects the physical and chemical composition of both the soil used for construction and soil of surrounding areas from which the materials are derived [54, 65]. Microhabitats are produced by termites which favor the development and sustenance of symbiotic microbial associations, providing it with optimum security from attacks and interference by predators. In the tropics, Kaschuk et al. [36] and Turner [69] found that trees associated with termite activities remain ever green throughout the year due to their activities due to nutrient cycling that led to improved soil fertility.
Termites by exercising their bioturbation activity influences the dynamics of soil and water at different spatial and temporal scales [11, 33, 43]. At a medium scale, termites have been identified as important drivers of weathering because they regulate the mineralogy of clay and the chemical compositions and properties of soil including the soil aggregates, which consequently controls the soil organic matter, soil carbon, and soil nutrient dynamics and cycling as well as soil water-holding capacity [43, 68]. At the soil profile level, termites explore and exploit soils from different soil profiles or generally select soil particles of small sizes (i.e., clays) for the construction of their termitaria [34]. The potential of termites in the enrichment of the soil in clay particles is very significant as their impact on soil organic matter is very remarkable. For example, many studies found higher carbon content in termitaria than in the surrounding soil in different tropical regions of the world [19, 23, 70], while Cheik et al. [18] found a negative or neutral effect of termite bioturbation activity. Finally, termites act as heterogeneity drivers and shape the distribution of natural resources such as water and nutrients at the landscape scale through the edification of mounds that are comparable to nutrient patches or soil fertility spots [7, 19, 40, 45, 46].
Generally, soil biochemical and physical properties of termitaria have been intensively discussed, especially in tropical Africa, where they exercise crucial roles in the dynamics and structure of vegetation zones and plant species richness [35, 67]. Other sect of authors in the region have described elaborately the roles of termites and their termitaria in promoting the soil ecosystem services by increasing the robustness and resilience of African arid ecosystems against water constraints and desertification [10]. Soils of the termitaria have high clay and organic matter contents, which contribute to the cation exchange capacity, buffering of the soil pH and aeration of the soil due to the burrows associated with their movement and transportation of materials. Termites of the genus macrotermes in Africa are generally referred to as the mound-building termites. The mounds can be about 12–18 m in diameter and up to 7 m high [28]. This study is aimed at determining and comparing the physiochemical properties of termitaria and their surrounding soils in three Nigerian ecozones to gain further insights into the effects of termite's activities, especially on a tropical soil ecosystem. Therefore, we hypothesized that termitaria can be identified as biogeochemical hot spots with enriched soil properties (e.g., clay, organic matter and other nutrients).
Materials and Methods
Study Area
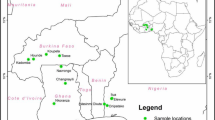
This study was conducted in three agroecological zones (guinea savanna, derived savanna and the mangrove zone) of Cross River State, Nigeria. Cross River State is a coastal state located in the Niger Delta region and occupies 20,156 square kilometers. It shares boundaries with Benue state to the north, Ebonyi and Abia states to the west, to the east by Sud-Ouest Province in Cameroon, and to the south by Akwa-Ibom and the Atlantic Ocean [5]. Samples from large termitariun (Supplementary Fig. 1) (LT) (with 15–20 entry points), small termitarium (ST) (5–10 entry points) [61] and surrounding soil (SS) were collected from 3 agroecological zones of Cross River State, Nigeria (Fig. 1). The locations are Obudu in the guinea savanna zone of the state, Ikom in the derived savanna zone and Calabar in the mangrove.
Guinea Savanna
Obudu (6°40′ 5.48″ N; 9° 09′ 52.31″ E) lies in the tropical Guinea savanna zone of Cross River State, Nigeria, with the broadest vegetation type, and it consists of a combination of perennial trees and grasses. It has an annual rainfall of 1000–1500 mm and distributed within a period of 6–8 months. The vegetation is pruned to wildfires during the drier periods of the year, which leads to an abundance of tree species that are resistant to wildfire. The major occupation in the area is farming and the soil is an Oxisol (ferrasol) [47] with pH range of 4.7–5.7 and organic carbon of 4.9–74.8 g kg−1 [4].
Derived Savanna
Ikom (5° 58′ 0.08″ N; 8° 42′ 22.75″ E) has the appearance and composition of the derived savanna, but with vestiges of lowland rainforest. In some areas, especially where human impacts on the vegetation has been high, there is a considerable reduction of plant species. The derived savanna zone has a mean annual rainfall of 2000 mm and minimum and maximum temperature of 25 and 37 °C, respectively [56]. The primary soil type is Alfisols with loamy sand, pH value ranging from 4.36 to 6.98, organic carbon ranging from 1.29 to 1.9% and nitrogen ranging from 0.2 to 0.27% [63].
Mangrove Forest Vegetation
Calabar (4°57′ 32.15″ N; 8° 19′37.02″ E) is located in the tropical Mangrove vegetation region with dense plants capable of trapping rainwater [26]. The area has a mean annual rainfall of between 2000 and 3500 mm, and it distributed within a period of 8–10 months [3]. It is characterized by dense vegetation comprising of trees, shrubs, herbs and tall grasses with a pH value of 5.43–5.54 and organic carbon (2.51–3.05%) [2].
At each of the 3 sites, samples were collected from the crest of the termitaria and surfaces (15 cm deep) of the soils that surround the termitaria, 50 m away from the base of the mound and outside the zone with noticeable termite activity. The distance of 50 m was applied to avoid any encroachment or runoff from the termitarium. At each site, 3 bulk samples for physico-chemical analyses were taken from every mound and from the surrounding soil [43]. The various soil samples were labelled appropriately, air-dried and sent to the Department of Soil Science, University of Calabar, for the analysis of organic carbon, phosphorus, potassium, aluminum, exchangeable bases, cation exchange capacity, pH and soil texture.
Soil Analysis
The pH of the soil was measured electrometrically using a pH meter in a 1:2.5 soil:water suspension. Available phosphorus (Av.P) was extracted with acidic fluoride using the Bray P 1 method [14], and phosphorus in the extract was determined colorimetrically by the molybdenum blue method of Murphy and Riley [44]. Exchangeable bases (calcium, potassium, magnesium and sodium) were extracted using 1:10 soil solution ratio. Soil textural analysis and classification were determined using Bouyoucos hydrometer method [12]. The content of soil organic carbon (OC) was determined according to the Walkley and Black method [29, 60], while the soil total nitrogen (TN) content was determined using the Kjeldahl method [15, 51].
Statistical Analysis
Prior to the statistical analysis, all soil variables were log-transformed and standardized to meet the assumptions of normality and linearity [28]. All soil data were expressed as mean standard error (± SE). One-way analysis of variance (ANOVA) and t-test were used to determine the differences and the significance of the differences in soil chemical properties, base saturation and cation concentrations, and in the soil texture particulates between termitarium and surrounding soils across the vegetation zones. Significant means were separated using least significant difference at 5% significance level to assess the mean differences in soil physiochemical properties under the termitarium and surrounding soils in different vegetation types. The multivariate analysis such as correlation and the redundancy analysis (RDA) were used to compare the relationships between the contents of the soil physiochemical properties in different vegetation types/ecozones. The STATISTICA 13.0 software (Statsoft, Tulsa, OK, USA) and the redundancy analysis (RDA) followed by a Monte Carlo Permutation test with 999 permutations in the CANOCO 5.0 software [66] were used for the analyses. Ordination diagram was produced by applying the CanoDraw program which prompted the presentation and visualization of the RDA result.
Results
Data in Table 1 show the variations in pH, organic carbon, total nitrogen, available phosphorus and cation exchange capacity of termitaria and surrounding soils from the various ecozones. Generally, the termitaria from the three zones were acidic to slightly acidic with pH of between 4.0 and 6.2. The pH was much higher in the guinea savanna and derived savanna than the mangrove forest zone. The pH of termitaria in guinea savannah and derived savannah was significantly higher than values in the surrounding soils, while the pH of soils surrounding the termitaria in the mangrove forest vegetation zone was similar to that of the termitaria. Organic carbon in derived savannah was higher in termitaria (2.40%) compared with the surrounding soils (1.46%). There was no significant difference in OC between termitaria and surrounding soils in guinea savannah (p = 0.06) and mangrove ecozones. Table 1 shows that the termitaria total nitrogen in all the locations were not different from that of the surrounding soils. Total nitrogen was lower in the mangrove forest zones, whether in surrounding soils (0.09%) or termitaria (0.07%). On average, total nitrogen was higher in surrounding soils (0.90%) from the three ecozones than termitaria (0.11%); however, there was no statistical difference between the amount in termitaria and surrounding soils across the ecozones. Available phosphorus (Av.P) in the savannah ecozone was higher in termitarium than surrounding soils. The Av.P in the termitaria had an average of 12.55 mg kg−1 compared with surrounding soils with an average of 11.86 mg kg−1. Termitaria across the various ecozones had significantly higher cation exchange capacity compared with surrounding soils averaging 10 cmol kg−1 in termitaria and 5 cmol kg−1 in surrounding soils.
There were not much significant differences between the termitaria and the surrounding soils, as indicated in the selected soil chemical properties, especially base saturation (BS) and cation concentrations (Table 2). Mean exchangeable potassium was about same in termitaria and surrounding soils across the ecozones, but exchangeable sodium was slightly higher in termitaria (0.1 cmol kg−1) than surrounding soils (0.10 cmol kg−1); however, there was no significant difference between exchangeable potassium and exchangeable sodium in termitaria in surrounding soils across ecozones. Exchangeable calcium was higher in termitaria than the surrounding soils in guinea savannah and Mangrove ecozones. In the savannah zone, there was no significant difference between exchangeable calcium in termitarium and surrounding soil. Concentrations of magnesium were higher in termitarium (3.56 cmol kg−1) than in surrounding soils (1.80 cmol kg−1) in the guinea savannah ecozone, 1.80 and 2.50 cmol kg−1 in the derived savannah and 0.97 and 1.80 cmol kg−1 in the mangrove forest ecozone, respectively. The base saturation was higher in termitaria than in surrounding soils across the three ecozones, with an average of 80% compared with surrounding soils with an average of 71%.
There was variability in soil texture across the three ecological zones with clay being significant in all (Table 3). The content of sand was higher in surrounding soils than in the termitaria that of silt was similar while the clay content was higher in termitaria than surrounding soils.
The chemical properties of the termitaria soils across the vegetation types showed that the pH had strong correlations with OC (r = 0.571), TN (r = − 0.988) and CEC (r = 0.740) (Supplementary Table 1). On the other hand, in the surrounding soils CEC showed negative correlations with pH (r = − 0.630), OC (r = − 0.757), TN (r = − 0. 148) and Av.P (r = − 0.220). Similarly, a positively strong correlation (r = 0.900) was revealed between CEC and Av.P. The correlations between Av.P and other soil chemical properties such as pH (r = 0.896), OC (r = 0.803) and TN (r = − 0.931) were high. The base saturation (BS) and cation contents for termitaria soils revealed that Mg had high correlation with K (r = − 0.979) and Na (r = 0.907) (Supplementary Table 3). Calcium showed a high correlation with H (r = 0.968), BS (r = 0.823) and Al (r = 0.567) while H showed a negatively high correlation with Al (r = − 0.771). Strong correlations were recorded between BS and Na (r = 0.897) and H (r = 0.629). Further, the surrounding soils revealed strong correlations between Ca and Mg (r = − 0.987), H (r = 0.990), Al (r = 0.847) and BS (r = 0.994). Sodium indicated negatively high correlations with Ca (r = − 0.879), Mg (r = − 0.792), H (r = − 0.805), Al (r = − 0.997) and BS (r = − 0.824). In both the termitaria and surrounding soils, the base saturation and cation contents revealed very high correlation (r = 0.999) between the ecozones (Supplementary Table 2). In terms of soil texture, sand had strong correlation with clay (r = 0.687) and silt (r = − 0.998) under the termitaria soils (Supplementary Table 3). In the surrounding soils, sand was negatively correlated with silt (r = − 0.978) and clay (r = − 0.937). In general, the soil chemical properties were highly correlated (r = 0.997) between guinea savannah (GS) and derived savannah (DS). Similarly, high correlation of the soil chemical properties was recorded between mangrove forest (MF) and GS (r = 0.993) and DS (r = 0.986). On the contrary, negatively weak or low correlations were recorded between MF and GS (r = − 0.364), and DS (r = − 0.134), while DS and GS were also negatively weak (r = − 0.133) (Supplementary Table 4). A further comparative correlation analysis between the ecozones revealed that the soil texture, the cation and base saturation were highly correlated in both termitaria and surrounding soils (Supplementary Tables 5 and 6).
Discussion
The results presented above confirmed earlier reports that physicochemical parameters of termitaria soil and those of the surrounding soil are relatively different [68]. The difference in pH, for instance, may be because of the organic materials used or added to the clay during the building of the nest as this could buffer the soil pH. It may also arise from the amount of precipitation and infiltration rates between the termitaria soil and those 50 m apart. Soil farther (50 m) away would tend to have higher infiltration and leaching rates than soils of the termitaria. This could also explain the variations in other chemical properties of the soil.
This is evidenced by the lower pH of samples from the mangrove vegetation receiving between 2500 and 3000 mm rainfall, which favors the leaching of exchangeable bases. Brady and Weil [13] reported that soils from humid climates are commonly acidic with a low soil pH, mostly because of precipitation. Precipitation and potential evapotranspiration control soil pH variations scales [20]. Termitaria across the various ecozones showed significantly higher cation exchange capacity than surrounding soils. This may be caused by the high clay content of the termitaria compared with the surrounding soils in all the locations. Clay is always negatively charged and easily attracts anions that are readily exchangeable and clay soil acts as a magnet or attractant [39]. In this sense, termitaria has a sponge effect, sucking up anions from adjacent soil catena. The OC was higher in the termitaria than the surrounding soils, especially in the derived savannah, but TN was lower in the mangrove forest zones whether in surrounding soils or termitaria while the available phosphorus across zones was higher in the termitaria. Sarcinelli et al. [60] also found that pH and the contents of organic C and N, P, Ca and Mg were significantly higher in mounds than adjacent soil. In particular, Ca and perhaps Mg may be acting as useful elements in binding or cementing organic materials for the building of termitaria. Termite dependence on dead wood, litter and grass as sources of food also facilitates the cycling and recycling of mineral elements and the formation of humus [65]. This is in consonance with Dhembare [24] report that termite ingestion of soil organic matter and returning it as fecal matter, protected organic matter physically and chemically, forming stable aggregates in soil. These unfailing but continuous activities of the termites, as well as their fecal pellets mixed with saliva, could be responsible for the higher pH and the other soil mineral contents (organic C, N, P, Ca and Mg) in the termitaria when compared with the surrounding soils [60].
Mean levels of potassium, sodium, magnesium, hydrogen and aluminum were the same in termitaria and surrounding soils across the ecozones, but base saturation was significantly higher in surrounding soils. For soils experiencing such relatively high precipitation, the base saturation which exceeded 80% was considered satisfactory. Rowland [57] reported that termite excrement and fecal residues though rich in N showed widely fluctuating nitrate levels according to season or rainfall. Phosphorus (P) level in soil can affect the bioavailability of P, especially if it has been built over the years and the P fixing capacity is satisfied (at soil pH of < 7.0). However, desorption of phosphates in the soil causes rapid increase in availability of P in the soil. The content of sand was significantly lower in the termitaria than the surrounding soils, while the clay contents were significantly higher in the termitaria than the surrounding soils across the three zones. The high clay content in the termitaria than surrounding soils is of importance as clay particles are negatively charged and can attract and retain more water and nutrients than the surrounding soils. Meanwhile, this is not just ordinary clay, but a clay rich with organic matter, OC, N and Ca contents formed through the decompositions of the organic plants’ residues by termites. This could describe why the termitaria soil is richer in nutrients. Clays are often found in deeper layers of soils in the tropics, but since the termites are scavengers, they dig into the deeper layers and bring up the clay used in building the termitaria. The activities of the termites also help in creating the tubular pores in the soil. Tubular pores help in creating a well aerated environment around and within the termitaria. The termites themselves help in recycling some nutrients as they break down some organic used as their food and they excrete or respire. They add some organic matter to the micro-soil environment. Mujinya et al. [41, 42] reported somewhat higher clay contents in termite mounds than in subsurface parts of surrounding soils. Similarly, Dhembare [24] and Deke et al. [23] found that termite mound soils were richer in clay, organic carbon, copper, iron, potassium, zinc, magnesium and phosphorus than surrounding soils.
The negative correlations between CEC, pH, OC, TN and Av.P in the surrounding soils might be attributed to the fact that the surrounding soils had high contents of sandy particles. Thus, the CEC of the sand fraction tended to encourage dispersion rather than aggregation [27, 30]. This could probably be explained by the reason that the content of clay was high in the termitaria soils; thus, the role of clay in uniting OC, TN and CEC has been reported in previous studies [1, 3, 30]. In agreement with our findings, a recent study in a grassland vegetation focusing on the interactions between soil base cations and micronutrients reported that pH significantly correlated with CEC [50]. The soil chemical properties across the ecozones showed negative correlations under the surrounding soils while the relationships under the termitaria soils were positive which might be explained by high CEC in the termitaria soils due to substantial amount of clay and high microbial (and termites) activities [39, 64].
A close association found in the result between Ca and BS has been previously reported by Bunt [16]. The author in his finding concluded that Ca does not only has high correlation with BS but accounted for the largest portion of BS and that the high CEC revealed the potential for greater adsorption of divalent cation. Similarly, in Alberta, base cations including Ca, Mg, K and Na were found to have positive correlations with CEC and BS [57]. This could be defined by the nutrients biochemical processes which affirm that increase in Ca, Mg and/or K in most cases would lead to a corresponding increase in CEC if prevailing environmental and anthropogenic covariates are favorable. In the surrounding soils, sand was negatively correlated with silt and clay. This result was consistent with the findings by Owoade et al. [53], who reported a high negative correlation between sand and clay in Southern Nigeria. There have been other studies focusing on the relationships between soil properties in different ecozones in Nigeria and West Africa [6, 53, 55]. Significant relationships were found between soil pH, OC, Av.P and N under the mangrove (or coastal) vegetation and the transitional vegetation zones in Ghana [22, 53]. In the Loess Plateau of Northern China, correlations between soil properties such as soil OC content and soil texture particles were reported to be high and significant across different vegetation types [55]. The strong relationships between soil OC and soil texture might be explained by the reason that OC is derived from a substantial enrichment of the soil by organic matter which consequently has a large influence of the soil texture. Owing to the uniqueness of termite mound soils, many researchers have recommended their use as a soil amendment in low-input cropping systems [25].
Conclusions
The study reveals that there are variations in the physicochemical composition of the termitaria soils and those farther (50 m) away from the termitaria across the ecozones. Clay content was equally higher in termitaria soils than those of the surrounding soils. Exchangeable cations were also higher in termitaria soils than in the surrounding soils, especially in the guinea savanna zone. Surprisingly, exchangeable Al was also higher in termitaria soils that tend to have more organic carbon and clay than the soils farther from the termitaria. It can, therefore, be concluded that the level of interactions between the colony and the environment determine to a large extent the variation that exists between termitaria soil and those of soil farther from termitaria. Sodium, calcium, organic carbon and potassium were also much higher in the termitaria than the surrounding soils in the guinea savannah zone. This study will promote nature conservation and enrichment of biodiversity in the different ecological zones and might also support the local and poor farmers’ decisions in growing specific crops closer to the termitaria, especially the crops that the require higher uptake of sodium and calcium for their growth and yields.
References
Afu SM, Asady CLA, Ofem KI, Olim DM (2016) Contribution of organic matter fractions to cation exchange capacity of organic matter under four land uses in Southeastern Nigeria. Afr J Agric Sci Technol 4:871–876
Afu SM, Isong IA, Awaogu CE (2019) Agricultural potentials of floodplain soils with contrasting parent material in Cross River State, Nigeria. Glob J Pure Appl Sci 25:13–22. https://doi.org/10.4314/gjpas.v25i1.3
Afu MS, Asadu CLA, Nsor ME (2019) Characterization and classification of selected soils under different land use in Northern Cross River State, Nigeria. J Sustain Agric Environ 16(2):390–403
Ahmad F, Fouad H, Liang SY, Hu Y, Mo JC (2021) Termites and Chinese agricultural system: applications and advances in integrated termite management and chemical control. Insect Sci 28:2–20. https://doi.org/10.1111/1744-7917.12726
Akpan JF, Aki EE, Isong IA (2017) Comparative assessment of wetland and coastal plain soils in Calabar, Cross River State. Glob J Agric Sci 16:17–30. https://doi.org/10.4314/gjass.v16i1.3
Akpan-Idiok A, Ofem K (2014) Physicochemical characteristics, degradation rate and vulnerability potential of Obudu Cattle Ranch Soils in Southeast Nigeria. Open J Soil Sci 4:57–63. https://doi.org/10.4236/ojss.2014.42008
Bachha B, Sahoo S, Mishra SS, Kusum A (2022) Physicochemical properties and biochemical activities of Termitaria soil of Odontotermes spp. and surrounding soil in Sambalpur district, Odisha, India. J Entomol Zool Stud 10(2):124–128
Bama PS, Ravindran AD (2018) Influence of combined termite mound materials and inorganic fertilizers on growth parameters of maize under non sterilized pot culture study. Elixir Appl Zool 125:52303–52305
Bhardwaj N, Tomar S (2018) Indigenous knowledge of termite (Odontotermes obesus) control. A review. Int J Adv Sci Res 1:124–127
Bonachela JA, Pringle RM et al (2015) Termite mounds can increase the robustness of dryland ecosystems to climatic change. Science 347:651–655. https://doi.org/10.1126/science.1261487
Bottinelli N, Jouquet P, Capowiez Y, Podwojewski P et al (2015) Why is the influence of soil macrofauna on soil structure only considered by soil ecologists? Soil Tillage Res 146:118–124. https://doi.org/10.1016/j.still.2014.01.007
Bouyoucos GJ (1962) Hydrometer method improved for making particle size analysis of soils. Agron J 54:464–465
Brady NC, Weil RR (2016) The nature and properties of soils, 15th edn. Pearson Publishers, pp 375–419
Bray RH, Kurtz LT (1945) Determination of total organic and available forms of phosphorus in soils. Soil Sci 59:39–45. https://doi.org/10.1097/00010694-194501000-00006
Bremner JM, Mulvaney CS (1982) Nitrogen-total. In: Page et al (eds) Methods of soil analysis. Part 2, chemical and microbiological properties. AME Soc Agron, Madison, pp 595–624
Bunt AC (1988) Media and mixes for container grown plants. Unwin Hyman, London
Calovi DS, Bardunias P, Carey N, Turner J, Nagpal R, Werfel J (2019) Surface curvature guides early construction activity in mound-building termites. Philos Trans R Soc B 374:2018 2023
Cheik S, Bottinelli N, Sukumar R, Jouquet P (2018) Fungus-growing termite foraging activity increases water infiltration but only slightly and temporally impacts soil physical properties in southern Indian woodlands. Eur J Soil Biol 89:20–24. https://doi.org/10.1016/j.ejsobi.2018.09.001
Chen C, Zou X et al (2021) Accumulation and spatial homogeneity of nutrients within termite (Odontotermes yunnanensis) mounds in the Xishuangbanna region, SW China. CATENA 198:105057. https://doi.org/10.1016/j.catena.2020.105057
Cheng-Jim JI, Yuan-He YA, Wen-Xuan NG, Yan-Fang HAN, Smith J, Smith P (2014) Climatic and edaphic controls on soil pH in alpine grasslands on the Tibetan Plateau, China: a quantitative analysis. Pedosphere 24(1):39–44
Dawes TZ (2010) Re-establishment of ecological functioning by mulching and termite invasion in a degraded soil in an Australian Savanna. Soil Biol Biochem 42:1825–1834
De Souza HJ, Delabie JHC et al (2020) Termite participation in the soil-forming processes of “murundus” structures in the semi-arid region of Brazil. Rev Bras Ciência do Solo 44:1–18. https://doi.org/10.36783/18069657rbcs20190133
Deke AL, Adugna WT, Fite AT (2016) Soil physic-chemical properties in termite mounds and adjacent control soil in Miyo and Yabello Districts of Borana Zone, Southern Ethiopia. Am J Agric For 4:69–74
Dhembare A (2013) Physico-chemical properties of termite mound soil. Arch Appl Sci Res 5:123–126
Eneji IS, Sha’Ato R, Ejembi SE (2016) Comparative analysis of termitaria and surrounding soil properties in the University of Agriculture, Makurdi, Nigeria. Chem Search J 6(1):57–61
Eze BE, Abua AM (2002) Environmental hydrology. Ushie Printer, Calabar, pp 6–59
Garba M, Cornelis WM, Steppe K (2011) Effects of termite mound material on the physical properties of sandy soil and on the growth characteristics of tomato (Solanum lycopersicum L.) in semi-arid Niger. Plant Soil 338:451–466
Grace JB, Anderson TM, Seabloom EW, Borer ET, Adler PB, Harpole WS, Bakker JD, Buckley YM, Crawley MJ, Damschen IE et al (2016) Integrative modelling reveals mechanisms linking productivity and plant species richness. Nature 529:390–393
Husein H, Mousa M, Sahwan W, Bäumler R, Lucke B (2019) Spatial distribution of soil organic matter and soil organic carbon stocks in semi-arid area of Northeastern Syria. Nat Res 10:415–432. https://doi.org/10.4236/nr.2019.1012028
Igwe CA, Nkemakosi JT (2007) Nutrient element contents and cation exchange capacity in fine fractions of Southeastern Nigerian soils in relation to their stability. Commun Soil Sci Plant Anal 38(9–10):1221–1242. https://doi.org/10.1080/00103620701328347
Jouquet P, Bottinelli N, Lata J-C, Philippe M, Sandrine C (2007) Role of the fungus-growing termite. Psaudacanthotermes Spiniger (Isoptera. macromitinae) in the dynamic of clay and soil organic matter content. Exp Anal 307
Jouquet P, Guilleux N, Caner L, Chintakunta S, Ameline M, Shanbhag R (2016) Influence of soil pedological properties on termite mound stability. Geoderma 262:45–51
Jouquet P, Bottinelli N et al (2016) Termites: the neglected soil engineers of tropical soils. Soil Sci 181:157–165. https://doi.org/10.1097/SS.0000000000000119
Jouquet P, Airola E et al (2017) Abundance and impact on soil properties of cathedral and lenticular termite mounds in Southern Indian woodlands. Ecosystems 20:769–780. https://doi.org/10.1007/s10021-14016-0060-5
Jouquet P, Traoré S et al (2020) Moving beyond the distinction between the bright and dark sides of termites to achieve sustainable development goals. Curr Opin Insect Sci 40:71–76
Kaschuk G, Santos JCP, Almeida JA (2006) Termite’s Activity in Relation to Natural Grassland Soil Attributes. J Sci Agric (Piracicaba, Braz) 63(6):583
Khucharoenphaisan K, Sripairoj N, Sinma K (2012) Isolation and identification of actinomycetes from termite’s gut against human pathogen. Asian J Anim Vet Adv 7:68–73
Korb J, Kasseney BD, Cakpo YT, Robin H, Daza D, Norbert J, Gbenyedji KB, Lboudo ME, Josens G, Koné NA, Meusemann K, Abdoulaye B, Ndiaye AB, Okweche SI, Poulsen M, Roisin Y, Sankara F (2019) Termite taxonomy, challenges and prospects: West Africa, a case example. Insects 32:1–8
Krohmer J (2004) Umwetwahmehmung und - Klassifikation bei Fulbegruppen in Verschiedenen Naturraumen Burkina Faso’s und Benin’s. Ph.D. Thesis. Johann- Wolfgang-Goe the –universitat, Frankfurt
López-Hernández D (2023) Termite mound as nutrient hot-spots in savannah with emphasis in P cycling and the potential use of mounds as soil amendment. Pedobiologia. https://doi.org/10.1016/j.pedobi.2023.150888
Mujinya BB, Mees F, Boeckx P, Bodé S, Baert G, Erens H, Delefortrie S, Verdoodt A, Ngongo M, Van Ranst E (2011) The origin of carbonates in termite mounds of the Lubumbashi area, D.R. Congo. Geoderma 165:95–105
Mujinya B, Mees F, Erens H, Dumon M, Baert G, Boeckx P, Ngongo M, Van Ranst E (2013) Clay composition and properties in termite mounds of the Lubumbashi area, D.R. Congo. Geoderma 192:304–315
Muon R, Ket P et al (2023) Termite constructions as patches of soil fertility in Cambodian paddy fields. Geoderma Reg 33:e00640
Murphy J, Riley JP (1962) A modified single solution method for the determination of phosphorus in natural waters. Anal Chim Acta 2:27–35
Muvengwi J, Mbiba M et al (2017) Termite diversity along a land use intensification gradient in a semi-arid savanna. J Insect Conserv 21:801–812. https://doi.org/10.1007/s10841-017-0019-7
Muvengwi J, Witkowski ETF (2020) Cascading effects of termite mounds in African savannas. N Z J Bot 58:167–193. https://doi.org/10.1080/0028825X.2020.1767162
National Forest Reference Emission Level (NFREL) for the Federal Republic of Nigeria (2019) Federal Department of Forestry Federal Ministry of Environment Federal Republic of Nigeria, pp 1–54
Negassa W, Sileshi GW (2018) Integrated soil fertility management reduces termite damage to crops on degraded soils in western Ethiopia. Agric Ecosyst Environ 251:124–131
Nithyatharani R, Kavitha US (2018) Termite soil as bio-Indicator of soil fertility. Int J Res Appl Sci Eng Technol 6:659–661
Niu G, Wang R, Hasi M, Wang Y, Geng Q, Wang C, Jiang Y, Huang J (2021) Availability of soil base cations and micronutrients along soil profile after 13-year nitrogen and water addition in a semi-arid grassland. Biogeochemistry 152:223–236. https://doi.org/10.1007/s10533-020-00749-5
Nwaogu C, Okeke OJ, Fashae O, Nwankwoala H (2018) Soil organic carbon and total nitrogen stocks as affected by different land use in an Ultisol inImo Watershed, southern Nigeria. Chem Ecol 34(9):854–870. https://doi.org/10.1080/02757540.2018.1508461
Oludairo OO, Aiyedun JO, Adeyi AJ, Ayeni AMJ, Aungbias AA (2016) Control and prevention of biodeterioration caused by termites (isoptera). J Res For Wildl Environ 8(3):44–56
Owoade FM, Adiku SGK, Atkinson CJ, MacCarthy DS (2021) Differential impact of land use types on soil productivity components in two agro-ecological zones of Southern Ghana. In: Leal Filho W, Oguge N, Ayal D, Adeleke L, da Silva I (eds) African handbook of climate change adaptation. Springer, Cham. https://doi.org/10.1007/978-3-030-45106-6144
Paul B, Khan MA, Paul S, Shankarganesh K, Chakravorty S (2018) Termites and Indian agriculture. In: Khan MA, Ahmad W (eds) Termites and sustainable management. Springer, Cham, pp. 51–96
Pei Y, Huang L, Li D, Shao M (2021) Characteristics and controls of solute transport under different conditions of soil texture and vegetation type in the water-wind erosion crisscross region of China’s Loess Plateau. Chemosphere 273:129651. https://doi.org/10.1016/j.chemosphere.2021.1296510045-6535
Philip fon (Jr), Akintoye OA, Olorundami T, Nkpena CO, Ukata SU, Harrison EU (2014) Forest resources of cross river state: their potentials, threats and mitigation measures. IOSR Environ Sci Toxicol Food Technol 8(6):64–71
Rippy JFM, Nelson PV (2007) Cation exchange capacity and base saturation variation among Alberta, Canada, Moss Peats. HortScience 42(2):349–352
Rowland C, Brauman A, Labatt M, Lepage M (1993) Nutritional factors affecting methane emission from termite. Chemosphere 26(1–4):617–622
Santos MN (2020) Research on termites in urban areas: approaches and gaps. Urban Ecosyst 23:587–601. https://doi.org/10.1007/s11252-020-00944-0
Sarcinelli T, Schaefer C, Lynch L, Arato H, Viana J, Filho M, Telles GMT (2009) Chemical, physical and micromorphological properties of termite mounds and adjacent soils along a toposequence in Zona da Mata, Minas Gerais State, Brazil. CATENA 76:107–113. https://doi.org/10.1016/j.catena.2008.10.001
Schnitzer M (1982) Total carbon, organic matter, and carbon. In: Page et al (eds) Methods of soilanalysis. Part 2. 2nd ed. Agronomy monograph, vol 9. American Society of Agronomy, Madison, pp 539–577
Sekhar V (2018) Termites and food security. AkiNik Publications, 169, C-11, Sector - 3, Rohini, Delhi-110085, India
Shittu AK, Nwoke OC et al (2022) Effects of contrasting agricultural land-use systems on selected soil properties in South-West Nigeria. Tanzan J Agric Sci 21(2):204–213
Silesshi G, Mafongoya PI (2010) Agroforestry systems, traditional fallows and monoculture on Nitrogen-limited soils in eastern Zambia. Agric For Entomol 7:61–69
Slame AJ (2005) Termite mounds as fertilizer: low external input sustainable agriculture 21 Pp
Ter Braak, CJF, Smilauer P (2012) CANOCO reference manual and canodraw for windows user’s guide: software for Canonical Community Ordination (version 5.0). www.canoco.com. Accessed 15 Jan 2021
Traoré S, Tigabu M et al (2015) Long-term effects of Macrotermes termites, herbivores and annual early fire on woody undergrowth community in Sudanian woodland, Burkina Faso. Flora Morphol Distrib Funct Ecol 211:40–50
Traoré S, Bottinelli N, Aroui H, Harit A, Jouquet P (2019) Termite mounds impact soil hydro-structural properties in southern Indian tropical forests. Pedobiologia 74:1–6
Turner JS (2006) Termites as the mediators of water economy of arid savanna ecosystems. In: Porporato A, D’Odorico P (eds) Dry land ecohydrology. Springer, Berlin, pp 303–313
Wakbulcho G, Kenea O (2021) Impact of termite activities on soil physicochemical characteristics in different land use types in Lalo Asabi District, Western Ethiopia 7:8–16
Acknowledgements
We are grateful to Prof. Egrinya Eneji of the Department of Soil Science, University of Calabar, Nigeria, for his technical/scientific reviews of this manuscript and to the Department of Soil Science, University of Calabar, for the soil analysis.
Funding
The work did not receive external funding.
Author information
Authors and Affiliations
Contributions
SIO, HPM, EBE and CN contributed to the conception, experimental work, interpretation of the analyzed data, and writing and reviewing the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Okweche, S.I., Matthew, H.P., Effa, E.B. et al. Physico-Chemical Properties of Termitaria and their Surrounding Soils in Some Nigerian Ecozones. Agric Res 13, 509–518 (2024). https://doi.org/10.1007/s40003-024-00707-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40003-024-00707-9