Abstract
Kufri Bahar, a leading potato variety is facing a problem of deshaped elongated tubers. Deshaping of tubers in form of elongation with jelly end formation leads to spoilage during transportation and storage. This elongation may be due to the stress conditions and/or enhanced gibberellic acid (GA) formation during micropropagation. Photoperiod and EDTA salts are reported to influence the GA concentration. Therefore, the effect of photoperiod and EDTA salts was investigated on the endogenous GA concentration of tissue culture microplants of potato vars Kufri Bahar and Kufri Lauvkar. The GA concentration in microplants grown in tissue culture media with Na-EDTA, Fe-EDTA and without EDTA ranged from 0.84 to 1.95 nmol/ml FW (fresh weight). In the present study, no correlation was observed between EDTA and GA concentration. Shoot length, internodal distance and number of nodes were more in microplants grown under a long photoperiod (16 h). Root length, number of roots and the number of leaves were high in short photoperiod (12 h) grown microplants. Gibberellic acid content showed similar trend as that of shoot length and ranged from 0.65 to 1.49 nmol/ml. Concentration of GA was more in var. Kufri Bahar compared to var. Kufri Lauvkar under the long photoperiod, which showed that Kufri Bahar is more responsive to photoperiod. High GA content in var. Kufri Bahar microplants grown under the long photoperiod could be the possible reason for tuber elongation. Therefore, to avoid the deshaping of tubers, the photoperiod during in vitro propagation may be shortened, particularly for var. Kufri Bahar.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In vitro micropropagation is used for producing diseases and virus-free potato seed. In India, deshaping/elongation of potato seed in a particular potato variety Kufri Bahar has emerged when the first generation in vitro material is further multiplied in the field (Fig. 1). Kufri Bahar is an important cultivar since it occupied almost 14–16% potato area in the country, and in Uttar Pradesh this variety occupied almost 67% of potato area [16]. Deshaping/elongation in potato tubers of this cultivar has economic implications for farmers.
Light regulates plant development. Photoperiod effects morphology, source sink relationship and finally the yield of crop plants. Response of plant to light is affected by numerous factors such as plant species, type of explants (leaf, root, stem), type of development of explants (embryo, callus, meristem), etc. Similar light conditions affect the growth of different varieties of the same plant to varying extent. Generally, growth of plant is proportional to the day length. The photoperiod also influences plant hormone levels in tissue culture which are used for producing seedlings. The standard photoperiod used in tissue culture is 16 h light and 8 h dark.
Gibberellins are diterpenoids, and their function as plant growth hormone is known since 1950s. Gibberellins play role in various plant growth and developmental processes such as seed germination, stem elongation, leaf expansion, floral initiation and fruit development [3]. Some compounds are reported to enhance the activity of gibberellic acid. These compounds are succinic acid 2,2 dimethylhydrazide, sodium-ethylenediaminetetraacetic acid (Na-EDTA) and 2 chloroethylphosphoric acid [18]. Two experiments were conducted to study the potato microplant morphology and its correlation with GA concentration in the tissue culture that was used to produce seedlings. First experiment was conducted to investigate the effect of different EDTA salts on endogenous GA concentration and its correlation with morphological parameters. Second experiment was conducted to study the effect of photoperiod on endogenous GA concentration and its correlation with morphological parameters of seedlings.
Materials and Methods
The experiment was conducted at ICAR-Central Potato Research Institute, Shimla. The explants used in the study were double-node cuttings derived from the middle portion of microplantlets of potato variety Kufri Lauvkar (single clone: P2B) and Kufri Bahar (two clones: P7A and P2A). The standard culture MS medium was used for the experiment (without hormones/growth regulators) [15]. For the first experiment, three types of culture media were prepared that varied in the type of EDTA. In the first set, sodium-EDTA (Na-EDTA) was incorporated in the culture media, in the second set iron EDTA (Fe-EDTA) was incorporated, whereas the culture media of third set were devoid of EDTA. In these three sets, three single-node pieces of explants were vertically inserted in MS culture medium per tube at equilateral distance. Culture tubes were kept in culture room at 12 h light (irradiance of 60 µmol/m2/s) and 12 h dark photoperiod and at 22 ± 1 °C temperature for 21 days (i.e. one cycle). For the second experiment, culture media of uniform composition were used for both the photoperiodic treatments. After vertically placing the three single-node pieces of explants in MS culture medium, culture tubes were kept for 21 days in culture room at two photoperiods, i.e. 16 and 12 h light (irradiance of 60 µmol/m2/s) and 8 and 12 h dark, respectively, at 22 ± 1 °C temperature. Various growth parameters, namely microplant height, root length, number of nodes, leaves and roots and fresh weight, were recorded after 21 days of incubation for two cycles in photoperiod experiment and for one cycle in EDTA experiment. In case of roots, only primary roots were counted. Root length was recorded for the longest root in each microplant. Fresh weight of shoot and root was observed separately. Along with morphological parameters, GA analysis was also carried out from the same samples.
Morphological Analysis
Shoot and root length was measured with the help of scale. Minimum three plants were used. Internodal distance, i.e. distance between two nodes, was estimated with the help of scale. Number of nodes, leaves and roots was counted from minimum three microplants. Shoot was separated from root and weighed directly. Media were removed from roots with washing. Roots were surface dried on filter paper and weighed.
GA Analysis
Extraction
Fresh tissue (microplant) was crushed in liquid nitrogen. The powder was extracted with 1 ml of 10% trichloroacetic acid and incubated overnight at − 20 °C (Deep Freezer, WEIBER make, Germany). Samples were centrifuged at 8000 rpm for 1 h at 4 °C. Supernatant was discarded, and 1 ml chilled acetone was added to the residue and vortexed. Samples were centrifuged at 8000 rpm for 15 min at 4 °C. Supernatant was lyophilized, and 5 ml of lysis buffer (2.7 g urea and 0.2 g of 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulfonate in 5 ml of distilled water) was used for reconstitution.
DAS-ELISA
GA was analysed using ‘plant gibberellic acid (GA) ELISA Kit’ (TSZ ELISA, Framingham, MA). Samples were analysed using the assay procedure provided with the kit. Optical density was read at 450 nm in ELISA plate reader (SUNRISE Absorbance Reader, Tecan Austria GmbH, Austria-Europe).
Statistical Analysis
A completely randomized design was followed with each treatment having three replications. The data were analysed using MSTAT 4.0C software using the procedure of Gomez and Gomez [7].
Results and Discussion
Effect of EDTA Salts on GA Content and Its Correlation with Morphological Parameters
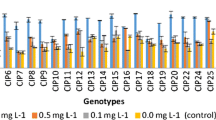
To study the effect of EDTA salts on endogenous GA concentration and microplant morphology, nodal cuttings of variety viz. Kufri Lauvkar and Kufri Bahar (P7A and P2A clone) were grown in three types of culture media. The three sets varied in the composition of culture media, wherein the culture media of one set contained Na-EDTA, second contained Fe-EDTA, and third set was devoid of EDTA. Microplants of both the varieties as well as clone of same variety grown on these different types of culture media behaved differently in terms of GA concentration and hence morphological parameters. Microplants of Kufri Lauvkar showed the maximum shoot (8.99 cm) and root length (8.14 cm) when grown in culture media containing Fe-EDTA and the minimum shoot (7 cm) and root length (6.63 cm) observed in the absence of EDTA. Shoot length of P7A (14.1 cm) and P2A clone (11.3 cm) of Kufri Bahar was the maximum in microplants grown on culture media devoid of EDTA, whereas culture media containing Na-EDTA resulted in least shoot length (9.61 cm and 10.66 cm in P7A and P2A clone, respectively). Kufri Bahar clone P7A responded significantly to the three types of culture media, where the shoot length of microplants grown in culture media containing Fe-EDTA is significantly higher than those grown on Na-EDTA containing culture media and shoot length of microplants grown in the absence of EDTA is significantly higher than that of Fe-EDTA containing culture media (Fig. 2). Internodal distance showed similar trend as that of shoot length, wherein Kufri Lauvkar microplants grown on Fe-EDTA containing culture media showed non-significantly higher intermodal distance, number of nodes and number of leaves. In P7A clone of Kufri Bahar, internodal distance was the maximum in microplants grown in the absence of EDTA, number of nodes remained unaffected in all the treatments and number of roots was the maximum in Na-EDTA containing culture media. In case of P2A clone of Kufri Bahar, internodal distance remained unaffected, number of nodes was more in microplants grown on Na-EDTA containing culture media, and number of leaves and number of roots were more in microplants grown in the absence of EDTA (Fig. 2). Shoot fresh weight showed the similar trend as that of shoot length, wherein shoot fresh weight was the maximum in Fe-EDTA containing media in case of Kufri Lauvkar and in the absence of EDTA in case of Kufri Bahar clones (Fig. 3). Gibberellic acid content ranged from 0.65 to 1.49 nmol/ml FW (fresh weight) including all clones and treatments (Fig. 4). The role of GA in cell elongation and stem elongation is well established [3], and our results are in accordance with this. GA concentration showed similar trend as that of shoot length of microplants and is positively correlated to shoot length (r = 0.798). Kufri Lauvkar microplants contained maximum concentration of endogenous GA (0.93 nmol/ml FW) when grown in culture media containing Fe-EDTA and minimum when grown in the absence of EDTA (0.65 nmol/ml FW). Whereas in case of Kufri Bahar clones, GA concentration was the maximum in microplants grown in the absence of EDTA and the endogenous GA concentration was 1.49 nmol/ml FW in P7A clone and 1.45 nmol/ml FW in P2A clone. Reports are available on the role of endogenous/exogenous GA in controlling the root growth and shoot growth by regulating the cell elongation [2, 3, 9, 10, 21,22,23]. In the present study also shoot length and root length showed positive correlation with the endogenous GA concentration (Table 1). Palevitch and Thomas reported that Na-EDTA enhances the activity of GA [18]. However, in the present results no such correlation was reported between Na-EDTA treatment and endogenous GA concentration. The results revealed that EDTA as such did not have much effect on endogenous GA concentration and hence on morphological parameters of tissue culture microplants. Our results are in accordance with the previous reports, and gibberellic acid showed high positive correlation with shoot length, root length and internodal distance [3, 9, 10, 21,22,23].
Effect of Photoperiod on GA Content and Its Correlation with Morphological Parameters
Plant growth is regulated by various factors including environmental stimuli such as light, temperature and touch as well as endogenous phytohormone level. Plant growth and development is regulated by the interaction of these stimuli. Gibberellic acid biosynthesis as well as responsiveness is regulated by phytochrome. In the present study, the effect of photoperiod on endogenous GA concentration and growth parameters was studied. Nodal cuttings of variety, namely Kufri Lauvkar and Kufri Bahar (P7A and P2A clone), were grown in two sets, wherein one set was kept under 11–12 h of light, i.e. short day (SD) and another under 16 h of light, i.e. long day (LD). Shoot length was more in microplants grown under long photoperiod in both the varieties. Gibberellins are reported to interact positively with auxin and ethylene to promote the elongation of the light grown seedlings [20]. Response of Kufri Lauvkar was non-significant in first cycle, whereas long day increased the shoot length (8.46 cm) significantly in second tissue culture cycle. Shoot length increased significantly in both clones of Kufri Bahar during both the cycles (Fig. 5). The results of shoot length showed that Kufri Bahar is more responsive to photoperiod compared to Kufri Lauvkar. In contrast to shoot length, root length was significantly high in SD microplants. Exogenous GA application is reported to delay the rooting and reduce the number of roots in long day conditions, but increases the number of roots in short day conditions and our results are in accordance to the earlier reported results [6, 17]. Internodal distance as well as number of nodes was non-significantly high in LD microplants. Number of leaves was non-significantly high in LD microplants of only P2A clone of Kufri Bahar (Fig. 6). Different photoperiodic conditions are known to affect the number of leaves. Extension of day length generally stimulates leaf development. Cook noticed an increase in the number of leaves of Chenopodium rubrum plants grown under 15 h days compared to ones grown under 12 h days [4]. Mitrovic et al. also observed more stem elongation and number of leaves in long day grown C. rubrum plants under in vitro conditions. Our results of shoot fresh weight were in accordance with the earlier reported results, where shoot fresh weight was more in SD microplants during both the cycles [13]. More fresh weight was recorded in Kufri Lauvkar and Kufri Bahar P7A clone during second cycle (Fig. 7). Trend in morphological parameters remained almost same during both the cycles. Overall, GA content ranged from 0.93 to 2.43 nmol/ml FW (Fig. 8). In the second tissue culture cycle of Kufri Lauvkar, the endogenous GA concentration increased significantly under both photoperiodic conditions, whereas in case of Kufri Bahar clones, GA concentration was high in first tissue culture cycle which reduced in the second cycle. In both the cycles, concentration of GA was high in LD microplants. Kufri Lauvkar microplants showed non-significant increase in GA concentration when grown under LD conditions. LD grown microplants of P7A clone of Kufri Bahar showed significant increase in GA concentration during second tissue culture cycle, whereas in LD grown microplants of P2A clone of Kufri Bahar, the increase was significant in first cycle. The results of GA concentration were well correlated with the morphological data, where the morphological parameters such as shoot length and internodal distance showed positive correlation with GA concentration in both the tissue culture cycles (Table 2). Both cell division and cell enlargement are affected by GA. Studies have shown that GA-mediated growth is regulated partially by modulation of cellular GA concentration as well as by alteration in the ability of cells to respond to this hormone [19]. The effect of light quality, photoperiod, light duration and light intensity has been evaluated by various researchers [1, 5, 8, 11, 14]. Although GA showed positive correlation with some morphological parameters in both the experiments, GA interacts with other phytohormones also during growth and development of plants. GA interacts synergistically with brassinosteroids which are also known to enhance cell division, cell elongation, vascular differentiation, reproductive development and modulation of gene expression [12]. GA interacts antagonistically with abscisic acid and positively with auxin. The type of interaction between GA and ethylene depends on the developmental and environmental circumstances. Cytokinin and GA interact negatively during various developmental processes such as root elongation, cell differentiation, shoot regeneration in culture and meristem activity. However, for normal shoot apical meristem function high cytokinin and low GA signals are required [24]. Therefore, the overall morphological data in both the experiments could be result of either GA alone or of its interaction with other phytohormones.
Conclusions
No correlation was observed between the different forms of EDTA with the endogenous GA concentration and hence the morphology of tissue culture microplants. Present study highlights the effect of photoperiod on the endogenous concentration of GA in tissue culture microplants positively. The effect of photoperiod was cultivar dependent, where the cultivar Kufri Bahar was found to be more responsive to long photoperiod compared to Kufri Lauvkar. Endogenous GA concentration as well as the length of microplants was more in Kufri Bahar grown under long photoperiod. High concentration of GA in microplants could be the possible reason for tuber deshaping/elongation in case of Kufri Bahar. The results hint towards the need to shorten the photoperiod during in vitro propagation from 16 h, particularly for Kufri Bahar to avoid the deshaping of tubers. However, due to interaction of GA with other phytohormones there is still a need to further investigate the role of other phytohormones and other associated reasons of tuber deshaping in cultivar Kufri Bahar.
References
Appelgren M (1991) Effects of light quality on stem elongation of Pelargonium in vitro. Sci Hortic 45:345–351
Bidadi H, Yamaguchi S, Asahina M, Satoh S (2009) Effects of shoot-applied gibberellin/gibberellin-biosynthesis inhibitors on root growth and expression of gibberellin biosynthesis genes in Arabidopsis thaliana. Plant Root 4:4–11
Chudasama RS, Thaker VS (2007) Relationship between gibberellic acid and growth parameters in developing seed and pod of pigeon pea. Braz J Plant Physiol 19(1):43–51
Cook RE (1975) The photoinductive control of seed weight in Chenopodium rubrum L. Am J Bot 62:427–431
Donneley D, Vidaver WE, Lee KY (1985) The anatomy of tissue cultured red raspberry prior to and after transfer to soil. Plant Cell Tissue Organ Cult 4:43–50
Ernstsen A, Hansen J (1986) Influence of gibberellic acid and stock plant irradiance on carbohydrate content and rooting in cuttings of Scots pine seedlings (Pinus sylvestris L.). Tree Physiol 1(1):115–125
Gomez KA, Gomez AA (1984) Statistical procedures for agriculture research, 2nd edn. Wiley, New York
Hayashi M, Fujita N, Kitaya Y, Kozai T (1992) Effect of sideward lighting on the growth of potato plantlets in vitro. Acta Hortic 319:163–166
Inada S, Shimmen T (2000) Regulation of elongation growth of gibberellins in root segments of Lemna minor. Plant Cell Physiol 41(8):932–939
Inada S, Tominaga M, Shimmen J (2000) Regulation of root growth by gibberellins in Lemna minor. Plant Cell Physiol 41(6):657–665
Iwanami Y, Kimura T, Kozai T, Kitaya Y, Kino S (1992) Effects of supplemental red and far-red lighting using light emitting diodes on stem elongation and growth of potato plantlets in vitro. In: Abstracts, international symposium on transplant production systems, July 1992, Yokohama, Japan, p 183
Megbo BC (2010) Brassinosteroids and gibberellic acid act synergistically to influence plant growth and development. Int J Sci Eng Res 1(1):68–72
Mitrovic A, Giba Z, Culafic A (2007) The photoperiodic control of growth and development of Chenopodium rubrum L. plants in vitro. Arch Biol Sci 59(3):203–208
Moe R (1983) Temperature and day length responses in Dianthus carthusianorum cv. Napoleon III. Acta Hortic 141:165–171
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Naik PS, Lal SS (2009) Region specific technologies for potato production in India. All India Coordinated Research Project on Potato. ICAR-Central Potato Research Institute, Shimla, pp 142–149
Nanda KK, Purohit AN, Bala A (1967) Effect of photoperiod, auxins and gibberellic acid on rooting of stem cuttings of Bryophyllum tubiflorum. Physiol Plant 20(4):1096–1102
Palevitch D, Thomas TH (1976) Enhancement by low pH of gibberellins effects on dormant celery seeds and embryoless half-seeds of barley. Physiol Plant 37:247–252
Richards DE, King KE, Ait-Ali T, Harberd NP (2001) How gibberellins regulates plant growth and development: a molecular genetic analysis of gibberellins signaling. Annu Rev Plant Mol Biol 52:67–88
Saibo NJM, Vriezen WH, Beemster GTS, Van der Straeten D (2003) Growth and stomata development of Arabidopsis hypocotyls are controlled by gibberellins and modulated by ethylene and auxins. Plant J 33:989–1000
Tanimoto E (2005) Regulation of root growth by plant hormones—roles for auxin and gibberellin. Crit Rev Plant Sci 24:249–265
Tanimoto E (2012) Tall or short? Slender or thick? A plant strategy for regulating elongation growth of roots by low concentration of gibberellins. Ann Bot 110:373–381
Wang G-L, Que F, Xu Z-S, Wang F, Xiong A-S (2015) Exogenous gibberellin altered morphology, anatomic and transcriptional regulatory networks of hormones in carrot root and shoot. BMC Plant Biol. https://doi.org/10.1186/s12870-015-0679-y
Weiss D, Ori N (2007) Mechanisms of cross talk between gibberellins and other hormones. Plant Physiol 144:1240–1246
Author information
Authors and Affiliations
Contributions
PR was involved in GA analysis, TB conducted in vitro experiments, BS, RKS, and BPS were involved in planning and conduct of experiments, BK contributed to morphological analysis.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Raigond, P., Buckseth, T., Singh, B. et al. Influence of Photoperiod and EDTA Salts on Endogenous Gibberellic Acid Concentration of Tissue Culture Grown Potato Microplants. Agric Res 8, 176–183 (2019). https://doi.org/10.1007/s40003-018-0364-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40003-018-0364-0