Abstract
Background:
Glucosamine hydrochloride (GlcN·HCl) has been shown to inhibit cell growth and matrix synthesis, but not with N-acetyl-glucosamine (GlcNAc) supplementation. This effect might be related to an inhibition of critical growth factors (GF), or to a different metabolization of the two glucosamine derivatives. The aim of the present study was to evaluate the synergy between GlcN·HCl, GlcNAc, and GF on proliferation and cartilage matrix synthesis.
Method:
Bovine chondrocytes were cultivated in monolayers for 48 h and in three-dimensional (3D) chitosan scaffolds for 30 days in perfusion bioreactors. Serum-free (SF) medium was supplemented with either growth factors (GF) TGF-β (5 ng mL−1) and IGF-I (10 ng mL−1), GlcN·HCl or GlcNAc at 1mM each or both. Six groups were compared according to medium supplementation: (a) SF control; (b) SF + GlcN·HCl; (c) SF + GlcNAc; (d) SF + GF; (e) SF + GF + GlcN·HCl; and (f) SF + GF + GlcNAc. Cell proliferation, proteoglycan, collagen I (COL1), and collagen II (COL2) synthesis were evaluated.
Results:
The two glucosamines showed opposite effects in monolayer culture: GlcN·HCl significantly reduced proliferation and GlcNAc significantly augmented cellular metabolism. In the 30 days 3D culture, the GlcN·HCl added to GF stimulated cell proliferation more than when compared to GF only, but the proteoglycan synthesis was smaller than GF. However, GlcNAc added to GF improved the cell proliferation and proteoglycan synthesis more than when compared to GF and GF/GlcN·HCl. The synthesis of COL1 and COL2 was observed in all groups containing GF.
Conclusion:
GlcN·HCl and GlcNAc increased cell growth and stimulated COL2 synthesis in long-time 3D culture. However, only GlcNAc added to GF improved proteoglycan synthesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Glucosamine (GlcN—abbreviation for general formula) is a hexosamine monosaccharide with potential effects to act as a chondrogenic and chondroprotector substance when prescribed as a supplement for osteoarthritis. GlcN assists in the healing process by inhibiting pro-inflammatory cytokine agents [1,2,3], it suppresses the degradation of the matrix by blocking proteases [4] and aggrecanases [5, 6], it stimulates chondrocyte proliferation and differentiation [7], and it might also be used as a substrate for glycosaminoglycan synthesis [8,9,10,11]. Nevertheless, the effect on cell proliferation and biosynthesis is controversial [12, 13].
GlcN enters cells via glucose transporter proteins (GLUTs) [14, 15] and it is metabolized in uridine-diphosphate-N-acetylglucosamine (UDP-N-GlcNAc) [16] through the hexosamine biosynthesis pathway (HBP) [17]. UDP-N-GlcNAc is the substrate for O-linked-N-acetylglucosamine transferase enzyme (OGT), which promotes the O-linked GlcNAcylation of serine/threonine residues of many nucleocytoplasmic proteins, kinases, and transcription factors [18,19,20]. This post-translational modification modulates the function and fate of these proteins, regulating many cellular processes such as signaling dynamics and gene expression [19]. The beneficial effect of exogenous GlcN might be related to an increase of O-GlcNAcylation. Low doses of GlcN increase O-GlcNAcylation of the hyaluronan synthase enzyme (HAS) [21]. It protects HAS from proteases by improving the stability and stimulates the synthesis of hyaluronan, which is an important proteoglycan of articular cartilages. Additionally, GlcN increases the O-GlcNAcylation of Sp1 transcription factor upregulating the expression of the transforming growth factor beta (TGF-β) type I [18].
The stimulation of TGF-β expression has a positive effect on chondrogenesis. TGF-β binds to type II receptor (TGF-rII) and recruits type I receptor (TGF-rI), also called activin-like kinase (ALK) 1–7 [22]. The activation of ALK5 downstream smad2/3, MEK-ERK1/2-ELK1/2, and PI3K-AKT-mTOR pathways, These pathways are responsible for cell proliferation, and collagen II [13, 23,24,25] and aggrecan [22, 23, 25, 26] expressions. The Insulin-like growth factors (IGF) are also critical for cartilage homeostasis. Their anabolic effect is additive to the effects of TGF-βs but independent [27]. IGF-1 phosphorylates the IGF-1 receptor (IGF-Ir). This phosphorylation and conformational change of IGF-Ir permits the phosphorylation of the insulin receptor-1 (IRS-1) [28], which downstream MEK-ERK1/2 and Akt-mTOR pathways. The first pathway stimulates cell proliferation [28] and proteoglycan synthesis [29], while the second stimulates collagen II expression [30].
Glucose is considered the main precursor and a critical energy source for chondrogenic differentiation [31] and matrix synthesis in cartilage [15, 32]. However, the imbalance of nutrition such as hyperglycemia has been linked to the hypertrophy of chondrocytes and development of osteoarthritis through the increase of O-GlcNAcylation. High concentrations of glucose (20–30 mM) stimulate the O-GlcNAcylation of several kinases, including PKC [33]. This modification inhibits the phosphorylation and activity of PKC, down-regulating the TGF-rII expression [33]. This pathological mechanism may explain the decreased TGF-rII expression and TGF-β activity in osteoarthritic chondrocytes [34], despite the enhancement of TGF-β expression found in osteoarthritis [35]. On the other hand, contradicting results have been shown in smooth muscle cells [36] and dental pulp stem cells [17], which however might have a completely different glucose metabolism. The proliferation induced by high glucose was related to an overexpression of TGF-rII stimulated by PKC [36], and GlcN and GlcNAc stimulated the expression of TGF-rI [17]. The IGF-I signaling also appears to be tightly modulated by post-translational modification of IGF-Ir and IRS-1, since exogenous GlcN increases the O-GlcNAcylation of serine/threonine residues of IRS-1 [37] and IGF-Ir [38]. This modification inhibited the phosphorylation of IRS-1 and the Akt downstream [39], resulting in insulin resistance, decreased glucose uptake [39], and inhibition of proliferation [38].
The association of the nutritional state and inhibition of TGF-rII and IGF-Ir expressions give rise to the hypothesis that GlcN reduces the chondrogenic effect of these two growth factors, which could explain the inhibition of cell proliferation and matrix synthesis after GlcN addition to medium containing native growth factors included in fetal bovine serum (FBS) [40,41,42]. Therefore, the present study evaluated the effect of two GlcNs—glucosamine hydrochloride (GlcN·HCl) and N-acetylglucosamine (GlcNAc), which have hitherto shown differential biological activities [43], solely or in combination with TGF-β and IGF-I, on the biosynthesis and proliferation of chondrocytes seeded in chitosan scaffolds for cartilage engineering. The use of serum-free medium is a more reliable design due to the absence of other molecules present in FBS that could interfere in the results. However, first the expression of the glucose transporter type II (GLUT2), which has higher affinity for GlcN than for glucose [44] was confirmed, and then the effect of GlcN·HCl and GlcNAc doses on proliferation in monolayer cultures was evaluated, individually and in crosstalk with TGF-β and IGF-I.
2 Materials and methods
2.1 Materials
The composition of the medium was: DMEM/F12 high glucose medium, dexamethasone, ascorbate 2-phosphate, fetal bovine serum (FBS), GlcN·HCl, and GlcNAc (Sigma Aldrich, Darmstadt, Germany), AlbuMax (Thermo Scientific, Schwerte, Germany), penicillin/streptomycin (GE Healthcare, Freiburg, Germany), transferrin-selenite (Fisher Scientific, Germany), collagenase IV (Invitrogen, Darmstadt, Germany), TGF-β3 (R&D Systems, Wiesbaden, Germany), and IGF-I (PeproTech, Hamburg, Germany). The 3D scaffold was prepared with chitosan (99% purity and deacetylation degree; Heppe Medical Chitosan GmbH (HMC+), Halle, Germany) and glutaraldehyde solution (Sigma Aldrich, Darmstadt, Germany). Immunohistochemistry was evaluated with anti-GLUT2 antibody (Abcam 111117, Darmstadt, Germany), rabbit polyclonal anti-collagen I (Abcam AB34710, Darmstadt, Germany), secondary polyclonal antibody Goat Anti-rabbit IgG H&L (Biotin) (Thermo Scientific, Schwerte, Germany), mouse polyclonal anti-collagen 2, secondary polyclonal antibody anti-Goat/Sheep IgG (R&D Systems, Wiesbaden, Germany), pierce peroxidase immunohistochemistry detection kit (Thermo Scientific, Schwerte, Germany). The evaluation of the proliferation was performed with the Quanti-iT Pico Green dsDNA assay kit (Thermo Scientific, Schwerte, Germany), and the resazurin Kit (Sigma Aldrich, Darmstadt, Germany). The absorbance and fluorescence were analyzed in a Polar Star Optima spectrophotometer (BMG LabTech GmbH, Ortemberg, Germany). The lactate production was measured in a Labtrend (BST, Berlin, Germany).
2.2 Primary cell isolation and cultivation
Primary chondrocytes were isolated from bovine carpal joints. Briefly, the articular cartilage tissue obtained was cultivated in DMEM/F12 high glucose medium supplemented with 1% penicillin/streptomycin, and 1 mg mL−1 collagenase IV (160 U L−1). The suspension was centrifuged at 200 g for 5 min and resuspended in DMEM/F12 high glucose medium supplemented with 10% FBS, 0.1% AlbuMax, 1% v/v penicillin/streptomycin, and 1% v/v transferrin-selenite. Chondrocytes from passage 4–9 were used in the two-dimensional (2D) and 3D experiments. All cells were cultivated at 37 °C, 5% CO2, and under normoxic conditions.
2.3 GLUT2 expression
For the evaluation of the GLUT2 expression, 10 × 103 chondrocytes were cultivated in chamber slides for 24 h at 37 °C, 5% CO2 in high glucose medium. The cells were fixed with 4% formaldehyde for 2 h, rinsed 3 times with TBS + Tween 20. Blocking was achieved by incubating the samples for 30 min with 1% bovine albumin serum in PBS, incubated with anti-GLUT2 antibody and anti-goat/sheep IgG antibody for 1 h at room temperature. Peroxidase labeled streptavidin–biotin was used as the detection system counterstained with Harris modified hematoxylin according to the manufacturer’s protocol.
2.4 Chondrocyte monolayer culture
Bovine chondrocytes were seeded in 96 well plates at a concentration of 5 × 103 in 100 µL medium per well, and cultivated for 3 h for cell adhesion. After cell adhesion, the medium was changed to serum-free medium containing DMEM/F12 high glucose medium, 0.1% AlbuMax, 1% penicillin/streptomycin, 170 µL dexamethasone at a concentration of 2 mg mL−1, 1% transferrin-selenite, 0.01% ascorbate 2-phosphate, supplemented with GlcN·HCl or GlcNAc at concentrations of 1 µM, 0.5 mM, 1 mM, 5 mM, and 10 mM. The best concentration of GlcN·HCl and GlcNAc was used to study the interaction with TGF-β3 + IGF-I. Three groups were formed. The serum-free medium was supplemented with GF, GF/GlcN·HCl, and GF/GlcNAc. For both experiments the same medium without GlcN·HCl or GlcNAc was used as control. The cells were cultivated for 72 h, at 37 °C, 5% CO2, and under normoxic conditions. All experiments were carried out in duplicate.
2.5 Cell proliferation quantification
Cell proliferation was evaluated using three methods according to the manufacturer’s protocol: (1) measurement of the dsDNA concentration with 5 µL mL−1 Quanti-iT Picogreen assay (excitation 480 nm/emission 520 nm), (2) measurement of the redox reaction of 2% resazurin in resorufin (excitation 560 nm/emission 590 nm) by NADPH/NADH with incubation for 2 h, and (3) measurement of the lactate production. The production of lactate was measured aliquoting 20 µL of sample medium and diluting it in 1 mL of measuring system solution.
2.6 3D scaffold synthesis
Porous chitosan sponge scaffolds were produced as described before [45]. Chitosan powder was solubilized in a 0.1 N HCl/deionized water solution at a concentration of 2%. The pH of this solution was adjusted to 5 by adding 1 N NaOH. Chitosan was crosslinked by adding 1% glutaraldehyde in a 1:1 proportion. This solution was poured into a mold with a height of 4 mm and a diameter of 14 mm, frozen for 12 h, lyophilized for 12 h, and sterilized with gamma irradiation. The scaffold and internal structure can be seen in Fig. 1.
2.7 Cell cultivation in 3D scaffold
The scaffolds were washed with medium to balance the pH for 14 h changing the medium 3 times. One million bovine chondrocytes resuspended in 100 µL medium were seeded on top of the scaffold and incubated for 20 min for cell adhesion at 37 °C, 5% CO2, to allow cell adhesion. After adhesion, the samples were cultivated for 24 h in DMEM/F12 high glucose supplemented with 10% FBS, 0.1% AlbuMax, 1% penicillin/streptomycin, and 1% transferrin-selenite. The scaffolds were transferred into perfusion bioreactors and cultivated for 30 days in serum-free medium as described above, supplemented with GlcN·HCl, GlcNAc, GF, GF + GlcN·HCl, and GF + GlcNAc; pure SF medium served as control. The concentrations of GF were 5 ng mL−1 TGF-β3 and 10 ng mL−1 IGF-I. The GlcN·HCl and GlcNAc concentrations were defined in the monolayer experiment described above. A total of 200 mL of medium per bioreactor was used and perfused with a flow velocity of 1 mL min−1. The sample size of each treatment group was 3.
2.8 Histology, immunohistochemistry and scanning electron microscopy
The 3D matrix samples were fixed in 4% formaldehyde for 12 h, dehydrated in a series of graded ethanol with Roticlear, embedded in paraffin, and cut into 6 µm sections that were stained with hematoxylin & eosin to evaluate cell proliferation, and Alcian Blue pH 2.5 for proteoglycans. The peroxidase labeled streptavidin–biotin immunohistochemistry detection system was performed to evaluate the synthesis of COL1 and COL2. The primary antibodies were applied and allowed to react for 12 h, the secondary antibody reacted for 30 min, and finally the samples were counterstained with Harris modified hematoxylin for 1 min. For scanning electron microscopy (SEM), the samples were fixed in aqueous 0.2 M sodium cacodylate trihydrate containing 1% glutaraldehyde, 2% formaldehyde and 5% sucrose solution for 4 h, followed by an equal fixation period in 0.2 M sodium cacodylate trihydrate, 3% glutaraldehyde, and 3% formaldehyde solution, dehydrated in an ascending acetone series, dried in an overcritical process of CO2, and sputter-coated with carbon and gold.
2.9 Statistical analysis
The one-way single factor analysis of variance (ANOVA) with the Tukey post hoc test or the Kruskal–Wallis rank sum test with the Nemeniy post hoc test were used at a significance level of 0.05.
3 Results
3.1 Evaluation of GLUT-2 expression
The IHC of plated chondrocytes clearly showed positive staining of GLUT-2, and no background was observed in control without the primary antibody (Fig. 2). Thus, the GLUT-2 transporter is present in the bovine chondrocytes used in the experiment.
3.2 Effect of GlcN·HCl and GlcNAc concentration on chondrocyte proliferation
In order to establish a possible dose–response relationship for the effect of the glucosamines on cell proliferation, we tested a range from 1 µM to 10 mM of both substances. The concentration of 1 µM estimated the viable concentration in synovia fluid after an oral dose of 1500 mg of GlcN [46, 47]. The results are shown in the supplementary section. A high concentration (10 mM) of GlcN·HCl showed the lowest cell proliferation and lactate production, while there was no difference between the concentrations below 1 mM. The concentrations of 1 and 10 mM resulted in the highest cell proliferation, whereas 1 mM GlcNAc resulted in the highest lactate production. Neither GlcN·HCl nor GlcNAc supplementations altered the resazurin metabolization. Therefore, the concentration of GlcN·HCl and GlcNAc for the next experiments was set at 1 mM due to the GlcNAc effect.
3.3 Effect of GlcN·HCl, GlcNAc and growth factors synergy on cell monolayer proliferation
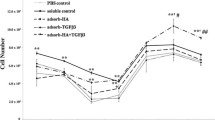
The three parameters evaluated showed a striking difference between the two glucosamines on cell proliferation (Fig. 3 and Table 1). The addition of GlcN·HCl alone did not change the proliferation compared to control for all parameters measured (p > 0.91), and had a strong inhibitory effect in combination with GF, producing the lowest values for all parameters (p < 0.001 compared to GF). On the other hand, although GlcNAc alone only increased the DNA concentration slightly (p > 0.378), the lactate values increased significantly compared to control and GlcN·HCl (p < 0.01). The resazurin redox was similar between them (p > 0.75). Combined to GF, GlcNAc produced similar proliferation (p > 0.127), but slightly higher lactate production compared to GF (p < 0.05). Therefore, the addition of GlcNAc to GF led to higher metabolic activity, since the number of cells was similar to GF, while the reduced values of lactate and resazurin redox obtained for GlcN·HCl + GF correlated with the low dsDNA values and thus might be a consequence of a lower number of cells.
Effect of GlcN·HCl, GlcNAc and GF on bovine chondrocyte proliferation. A dsDNA measurement (F statistic, p < 0.001). * and ** = p < 0.001 compared to GF/GlcN·HCl. # = p < 0.05 compared to control and GlcN·HCl. § = p < 0.01 compared to GF/GlcN·HCl. B NADPH/NADH synthesis (Kruskal–Wallis Chi square statistic, p < 0.001). * and ** = p < 0.001 compared to control, GlcN·HCl, GlcNAc, and GF/GlcN·HCl. C Lactate synthesis (F statistic, p < 0.001). * = p < 0.001, compared to control, GlcN·HCl, GlcNAc, GF, GF/GlcNAc. # = p < 0.001, compared to control, GlcN·HCl, and GlcNAc. $ = p < 0.05, compared to GF. § = p < 0.001, compared to control and GlcN·HCl. ** = p < 0.01, compared to control and GlcN·HCl
3.4 Effect of GlcN·HCl, GlcNAc and GF synergy on cell proliferation and biosynthesis in 3D culture
Cell distribution and proteoglycan synthesis are shown in Figs. 4 and 5, respectively. The data are summarized in Table 2. GlcN·HCl and GlcNAc without growth factors did not alter the cell proliferation compared to control. This low cell density was responsible for a very low proteoglycan synthesis. On the other hand, higher cell proliferation and proteoglycan synthesis were observed in groups with GF supplementation. Among these groups, the addition of GlcN·HCl (p < 0.001) and GlcNAc (p < 0.001) to GF clearly improved the cell proliferation compared to GF. However, the proliferation was higher in GF/GlcNAc than GF/GlcN·HCl (p < 0.001) in concordance with the 2D culture result t. Although the addition of GlcN·HCl to GF stimulated cell proliferation, the proteoglycan synthesis was slightly lower (min: 1.95, max: 8.27% of total area) compared to GF (min: 4.36, max: 8.88% of total area), but not significantly different (p = 0.663). the addition of GlcNAc to GF (min: 8.27, max: 18.69% of total area) showed higher proteoglycan synthesis compared to GF/GlcN·HCl and GF, but again not statistically significant (p = 0.159 and 0.467, respectively). All groups with GF showed a synthesis of COL1 and COL2 (Fig. 6). No synthesis of either collagen was observed in samples without GF and therefore the images were omitted. The total matrix production observed in SEM was similar between the GF and GF/GlcN·HCl groups (Fig. 7). Nevertheless, the highest biosynthesis was observed in the GF/GlcNAc group. The superficial porosity on top of the chitosan scaffold was completely closed by cells and matrix, while uncovered chitosan parts and pores can be seen in GF and GF/GlcN·HCl, respectively.
A hematoxylin & eosin staining, purple dots show cell growth, and pink shows chitosan fibers. B cell density quantification (F statistic, p < 0.001), *—p < 0.001, compared to all groups. **—p < 0.001, compared to GF, GlcNAc, GlcN·HCl, and control. #—p < 0.01, compared to control. §—p < 0.05, compared to GlcN·HCl and GlcNAc. (n = 3; magnification 100 ×; scale bar: 300 µm). (Color figure online)
4 Discussion
The supplementation of GlcN·HCl in low and high doses has shown a deleterious effect on cell proliferation and proteoglycan synthesis. The nutritional imbalance created by GlcN·HCl supplementation could promote suppression of the chondrogenic effect of GF through the inhibition of the TGF-βr expression [33], and inhibition of IRS-1 [37] and IGF [38] phosphorylation. To test this hypothesis, a group with TGF-β3 + IGF-I, termed “GF”, and GF/GlcN·HCl were compared in two experimental settings: 48 h monolayer culture and 30 days in 3D culture. The proliferation was inhibited by the addition of GlcN·HCl to GF, as observed in DNA, resazurin redox, and lactate quantifications. Conversely, a proliferative effect was observed in the long time 3D culture.
Interestingly, this higher cell concentration was not traduced in higher proteoglycan synthesis, since there was no difference between GF and GF/GlcN·HCl. This result suggests that GlcN·HClGlcN·HCl was not used as a substrate for proteoglycan synthesis. The concentration of GlcN is very low in cartilage, and glucose is the main substrate for biosynthesis via HBP. Nevertheless, studies that have reported biosynthesis impairment with GlcN·HCl supplementation have suggested that the glucose metabolism is modified. GlcN increases the post-translational inhibition of IRS-1 [46] and IGF-Ir [38], which results in insulin resistance, decreased glucose uptake [39], and downregulates the expression of GLUT types with higher affinity for glucose via depletion of the ATP and hexosamine pathways [43, 47, 48]. Furthermore, upon entering the cell, GlcN undergoes phosphorylation by glucokinase and competes with glucose for binding to glucokinase [42], which inhibits the glucose metabolism.
An even stronger proliferative effect was observed with GlcNAc supplementation. The addition of GlcNAc to GF did not inhibit the cell proliferation in the monolayer culture; however, it increased the cell metabolism compared to GF. In the long-term culture, the addition of GlcNAc greatly improved the cell proliferation and proteoglycan synthesis compared to GF/GlcN·HCl and GF alone. These results suggest that GlcN·HCl and GlcNAc have a differential metabolic effect, as shown in a previous study [43]. First, GlcNAc (1 and 5 mM) does not compete for GLUT, thus perming the normal glucose uptake [42, 43]. Second, GlcNAc does not interfere in glucose metabolism. GlcNAc undergoes phosphorylation by GlcNAc kinase and does not compete with glucose for glucokinase [49, 50]. Moreover, this product of GlcNAc phosphorylation (GlcNAc-6 phosphate) enters the hexosamine pathway more distally than GlcN-6 phosphate and does not possess any known negative allosteric effects toward glucokinase [49]. Beyond GlcNAc does not change the glucose metabolism, its supplementation might directly be used as a substrate for proteoglycan synthesis. Therefore, these findings may account, at least in part, for the different results between GlcN·HCl and GlcNAc observed in the present study.
The natural cartilage matrix is mainly composed of COL2 with very small amounts or absence of COL1 [51]. The synthesis of COL2 is mediated by GF. The cells under the influence of GF, and GF supplemented with GlcN·HCl or GlcNAc synthesized both COL1 and COL2. The presence of COL2 confirmed the maintenance of the chondrocytic phenotype and reinforced the non-inhibitory effect of GlcN·HCl and GlcNAc on GF. However, the synthesis of COL1 indicated the existence of dedifferentiated cells.
The difference between GlcN·HCl and GlcNAc on cell proliferation and matrix synthesis has been reported in previous monolayer and 3D culture studies. Whereas small concentrations of GlcN·HCl (0.5 and 5 mM) inhibited cell proliferation [40, 52] and decreased lactate and glycosaminoglycan synthesis [40,41,42], GlcNAc did not alter the proliferation [41, 52] and proteoglycan synthesis [12, 42, 52]. However, contradicting results with GlcN·HCl supplementation were also recorded. An improvement of COL2 and proteoglycan synthesis was observed with a supplementation of 2 mM GlcN·HCl [13] and might be related to the upregulation of the TGF-β expression [13, 18]. Conversely, the COL2 expression [41, 53] and synthesis [53] were not changed in other studies.
Due to the suppressive effect of GlcN when added to GF on cell proliferation in the monolayer culture compared to GlcNAc, the GlcN and GlcNAc were tested without growth factors in 3D culture. However, neither altered the cell proliferation compared to control. The low cell concentration resulted in non-significant synthesis of the matrix. Therefore, GlcN and GlcNAc have no effect when added without growth factors in a long-time culture.
In summary, GlcN·HCl and GlcNAc without GF have no effect on cell proliferation and matrix synthesis. When added to GF, the inhibitory effect of GlcN·HCl, observed in the monolayer short-term culture, was not observed in the 3D long-term culture. GlcN·HCl improved cell proliferation but did not benefit proteoglycan synthesis. However, the addition of GlcNAc to GF resulted in higher cell proliferation and synthesis of proteoglycan compared to GF and GF + GlcN·HCl. The presence of collagen I and II was observed in all groups containing GF. Therefore, although GlcN·HCl and GlcNAc do not inhibit the effect of GF, GlcNAc at the concentration used is a better option for the proliferation and synthesis of PG in cartilage engineering.
References
Scotto d’Abusco A, Politi L, Giordano C, Scandurra R. A peptidyl-glucosamine derivative affects IKKalpha kinase activity in human chondrocytes. Arthritis Res Ther. 2010;12:R18.
Imagawa K, de Andrés MC, Hashimoto K, Pitt D, Itoi E, Goldring MB, et al. The epigenetic effect of glucosamine and a nuclear factor-kappa B (NF-kB) inhibitor on primary human chondrocytes—implications for osteoarthritis. Biochem Biophys Res Commun. 2011;405:362–7.
Chan PS, Caron JP, Rosa GJ, Orth MW. Glucosamine and chondroitin sulfate regulate gene expression and synthesis of nitric oxide and prostaglandin E2 in articular cartilage explants. Osteoarthritis Cartilage. 2005;13:387–94.
Ratcliffe A, Hardingham T. Cartilage proteoglycan binding region and link protein. Radioimmunoassays and the detection of masked determinants in aggregates. Biochem J. 1983;213:371–8.
Fenton JI, Chlebek-Brown KA, Peters TL, Caron JP, Orth MW. The effects of glucosamine derivatives on equine articular cartilage degradation in explant culture. Osteoarthritis Cartilage. 2000;8:444–51.
Sandy JD, Gamett D, Thompson V, Verscharen C. Chondrocyte-mediated catabolism of aggrecan: aggrecanase-dependent cleavage induced by interleukin-1 or retinoic acid can be inhibited by glucosamine. Biochem J. 1998;335:59–66.
Jeon JH, Suh HN, Kim MO, Han HJ. Glucosamine-induced reduction of integrin β4 and plectin complex stimulates migration and proliferation in mouse embryonic stem cells. Stem Cells Dev. 2013;22:2975–89.
Bassleer C, Rovati L, Franchimont P. Stimulation of proteoglycan production by glucosamine sulfate in chondrocytes isolated from human osteoarthritic articular cartilage in vitro. Osteoarthritis Cartilage. 1998;6:427–34.
Wang L, Detamore MS. Effects of growth factors and glucosamine on porcine mandibular condylar cartilage cells and hyaline cartilage cells for tissue engineering applications. Arch Oral Biol. 2009;54:1–5.
Dodge GR, Jimenez SA. Glucosamine sulfate modulates the levels of aggrecan and matrix metalloproteinase-3 synthesized by cultured human osteoarthritis articular chondrocytes. Osteoarthritis Cartilage. 2003;11:424–32.
Igarashi M, Kaga I, Takamori Y, Sakamoto K, Miyazawa K, Nagaoka I. Effects of glucosamine derivatives and uronic acids on the production of glycosaminoglycans by human synovial cells and chondrocytes. Int J Mol Med. 2011;27:821–7.
Uitterlinden EJ, Jahr H, Koevoet JL, Jenniskens YM, Bierma-Zeinstra SM, Degroot J, et al. Glucosamine decreases expression of anabolic and catabolic genes in human osteoarthritic cartilage explants. Osteoarthritis Cartilage. 2006;14:250–7.
Varghese S, Theprungsirikul P, Sahani S, Hwang N, Yarema KJ, Elisseeff JH. Glucosamine modulates chondrocyte proliferation, matrix synthesis, and gene expression. Osteoarthritis Cartilage. 2007;15:59–68.
Windhaber RA, Wilkins RJ, Meredith D. Functional characterisation of glucose transport in bovine articular chondrocytes. Pflugers Arch. 2003;446:572–7.
Mobasheri A, Vannucci SJ, Bondy CA, Carter SD, Innes JF, Arteaga MF, et al. Glucose transport and metabolism in chondrocytes: a key to understanding chondrogenesis, skeletal development and cartilage degradation in osteoarthritis. Histol Histopathol. 2002;17:1239–67.
Huang JB, Clark AJ, Petty HR. The hexosamine biosynthesis pathway negatively regulates IL-2 production by Jurkat T cells. Cell Immunol. 2007;245:1–6.
Chen YJ, Yao CC, Huang CH, Chang HH, Young TH. Hexosamine-induced TGF-β signaling and osteogenic differentiation of dental pulp stem cells are dependent on N-acetylglucosaminyltransferase V. Biomed Res Int. 2015;2015:924397.
Ali AA, Lewis SM, Badgley HL, Allaben WT, Leakey JE. Oral glucosamine increases expression of transforming growth factor β1 (TGFβ1) and connective tissue growth factor (CTGF) mRNA in rat cartilage and kidney: implications for human efficacy and toxicity. Arch Biochem Biophys. 2011;510:11–8.
Hart GW, Housley MP, Slawson C. Cycling of O-linked β-N-acetylglucosamine on nucleocytoplasmic proteins. Nature. 2007;446:1017–22.
Slawson C, Copeland RJ, Hart GW. O-GlcNAc signaling: a metabolic link between diabetes and cancer? Trends Biochem Sci. 2010;35:547–55.
Vigetti D, Deleonibus S, Moretto P, Karousou E, Viola M, Bartolini B, et al. Role of UDP-N-acetylglucosamine (GlcNAc) and O-GlcNAcylation of hyaluronan synthase 2 in the control of chondroitin sulfate and hyaluronan synthesis. J Biol Chem. 2012;287:35544–55.
Grimaud E, Heymann D, Rédini F. Recent advances in TGF-beta effects on chondrocyte metabolism. Potential therapeutic roles of TGF-beta in cartilage disorders. Cytokine Growth Factor Rev. 2002;13:241–57.
Mariani E, Pulsatelli L, Facchini A. Signaling pathways in cartilage repair. Int J Mol Sci. 2014;15:8667–98.
Miyazaki Y, Tsukazaki T, Hirota Y, Yonekura A, Osaki M, Shindo H, et al. Dexamethasone inhibition of TGF beta-induced cell growth and type II collagen mRNA expression through ERK-integrated AP-1 activity in cultured rat articular chondrocytes. Osteoarthritis Cartilage. 2000;8:378–85.
Li C, Wang Q, Wang JF. Transforming growth factor-β (TGF-β) induces the expression of chondrogenesis-related genes through TGF-β receptor II (TGFRII)-AKT-mTOR signaling in primary cultured mouse precartilaginous stem cells. Biochem Biophys Res Commun. 2014;450:646–51.
Li J, Zhao Z, Liu J, Huang N, Long D, Wang J, et al. MEK/ERK and p38 MAPK regulate chondrogenesis of rat bone marrow mesenchymal stem cells through delicate interaction with TGF-β1/Smads pathway. Cell Prolif. 2010;43:333–43.
Longobardi L, O’Rear L, Aakula S, Johnstone B, Shimer K, Chytil A, et al. Effect of IGF-I in the chondrogenesis of bone marrow mesenchymal stem cells in the presence or absence of TGF-β signaling. J Bone Miner Res. 2006;21:626–36.
Tahimic CG, Wang Y, Bikle DD. Anabolic effects of IGF-1 signaling on the skeleton. Front Endocrinol (Lausanne). 2013;4:6.
Starkman BG, Cravero JD, Delcarlo M, Loeser RF. IGF-I stimulation of proteoglycan synthesis by chondrocytes requires activation of the PI 3-kinase pathway but not ERK MAPK. Biochem J. 2005;389:723–9.
Zhang M, Zhou Q, Liang QQ, Li CG, Holz JD, Tang D, et al. IGF-1 regulation of type II collagen and MMP-13 expression in rat endplate chondrocytes via distinct signaling pathways. Osteoarthritis Cartilage. 2009;17:100–6.
Sun C, Shang J, Yao Y, Yin X, Liu M, Liu H, et al. O-Glc NA cylation: a bridge between glucose and cell differentiation. J Cell Mol Med. 2016;20:769–81.
Lee GM, Tioran ME, Jansen M, Graff RD, Kelley SS, Lin P. Development of selective tolerance to interleukin-1β by human chondrocytes in vitro. J Cell Physiol. 2002;192:113–24.
Tsai TL, Manner PA, Li WJ. Regulation of mesenchymal stem cell chondrogenesis by glucose through protein kinase C/transforming growth factor signaling. Osteoarthritis Cartilage. 2013;21:368–76.
Dehne T, Karlsson C, Ringe J, Sittinger M, Lindahl A. Chondrogenic differentiation potential of osteoarthritic chondrocytes and their possible use in matrix-associated autologous chondrocyte transplantation. Arthritis Res Ther. 2009;11:R133.
Pombo-Suarez M, Castaño-Oreja MT, Calaza M, Gomez-Reino J, Gonzalez A. Differential upregulation of the three transforming growth factor beta isoforms in human osteoarthritic cartilage. Ann Rheum Dis. 2009;68:568–71.
Yasuda Y, Nakamura J, Hamada Y, Nakayama M, Chaya S, Naruse K, et al. Role of PKC and TGF-β receptor in glucose-induced proliferation of smooth muscle cells. Biochem Biophys Res Commun. 2001;281:71–7.
Klein AL, Berkaw MN, Buse MG, Ball LE. O-Linked N-acetylglucosamine modification of insulin receptor substrate-1 occurs in close proximity to multiple SH2 domain binding motifs. Mol Cell Proteomics. 2009;8:2733–45.
Song KH, Kang JH, Woo JK, Nam JS, Min HY, Lee HY, et al. The novel IGF-IR/Akt-dependent anticancer activities of glucosamine. BMC Cancer. 2014;14:31.
Vosseller K, Wells L, Lane MD, Hart GW. Elevated nucleocytoplasmic glycosylation by O-GlcNAc results in insulin resistance associated with defects in Akt activation in 3T3-L1 adipocytes. Proc Natl Acad Sci U S A. 2002;99:5313–8.
de Mattei M, Pellati A, Pasello M, de Terlizzi F, Massari L, Gemmati D, et al. High doses of glucosamine-HCl have detrimental effects on bovine articular cartilage explants cultured in vitro. Osteoarthritis Cartilage. 2002;10:816–25.
Nakatani S, Mano H, Im R, Shimizu J, Wada M. Glucosamine regulates differentiation of a chondrogenic cell line, ATDC5. Biol Pharm Bull. 2007;30:433–8.
Uitterlinden EJ, Jahr H, Koevoet JL, Bierma-Zeinstra SM, Verhaar JA, Weinans H, et al. Glucosamine reduces anabolic as well as catabolic processes in bovine chondrocytes cultured in alginate. Osteoarthritis Cartilage. 2007;15:1267–74.
Shikhman AR, Brinson DC, Valbracht J, Lotz MK. Differential metabolic effects of glucosamine and N-acetylglucosamine in human articular chondrocytes. Osteoarthritis Cartilage. 2009;17:1022–8.
Uldry M, Ibberson M, Hosokawa M, Thorens B. GLUT2 is a high affinity glucosamine transporter. FEBS Lett. 2002;524:199–203.
Hoffmann B, Seitz D, Mencke A, Kokott A, Ziegler G. Glutaraldehyde and oxidised dextran as crosslinker reagents for chitosan-based scaffolds for cartilage tissue engineering. J Mater Sci Mater Med. 2009;20:1495–503.
Persiani S, Rotini R, Trisolino G, Rovati LC, Locatelli M, Paganini D, et al. Synovial and plasma glucosamine concentrations in osteoarthritic patients following oral crystalline glucosamine sulphate at therapeutic dose. Osteoarthritis Cartilage. 2007;15:764–72.
Mroz PJ, Silbert JE. Use of 3H-glucosamine and 35S-sulfate with cultured human chondrocytes to determine the effect of glucosamine concentration on formation of chondroitin sulfate. Arthritis Rheum. 2004;50:3574–9.
Henrotin Y, Chevalier X, Herrero-Beaumont G, McAlindon T, Mobasheri A, Pavelka K, et al. Physiological effects of oral glucosamine on joint health: current status and consensus on future research priorities. BMC Res Notes. 2013;6:115.
Miwa I, Mita Y, Murata T, Okuda J, Sugiura M, Hamada Y, et al. Utility of 3-O-methyl-N-acetyl-D-glucosamine, an N-acetylglucosamine kinase inhibitor, for accurate assay of glucokinase in pancreatic islets and liver. Enzyme Protein. 1994–1995;48:135–42.
Hinderlich S, Berger M, Schwarzkopf M, Effertz K, Reutter W. Molecular cloning and characterization of murine and human N-acetylglucosamine kinase. Eur J Biochem. 2000;267:3301–8.
Gelse K, Pöschl E, Aigner T. Collagens—structure, function, and biosynthesis. Adv Drug Deliv Rev. 2003;55:1531–46.
Terry DE, Rees-Milton K, Pruss C, Hopwood J, Carran J, Anastassiades TP. Modulation of articular chondrocyte proliferation and anionic glycoconjugate synthesis by glucosamine (GlcN), N-acetyl GlcN (GlcNAc) GlcN sulfate salt (GlcN.S) and covalent glucosamine sulfates (GlcN-SO4). Osteoarthritis Cartilage. 2007;15:946–56.
Stoppoloni D, Politi L, Leopizzi M, Gaetani S, Guazzo R, Basciani S, et al. Effect of glucosamine and its peptidyl-derivative on the production of extracellular matrix components by human primary chondrocytes. Osteoarthritis Cartilage. 2015;23:103–13.
Acknowledgement
This study was supported by grants from CAPES Foundation within the Ministry of Education of Brazil, University of Bayreuth, and the Friedrich-Baur Biomed Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest that would prejudice the impartiality of this scientific work.
Ethical statement
There are no animal experiments carried out for this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1.
The effect of GlcN·HCl and GlcNAc doses on cell proliferation and viability. A dsDNA quantification (F statistic p < 0.001). * = p < 0.05, compared to 0.5 mM. ** = p < 0.001, compared to 1 mM and 10 mM. # = p < 0.05, compared to 0 mM. B Effect of GlcN·HCl (F statistic, p = 0.24) and GlcNAc (F statistic, p = 0.69) on NADPH/NADH2 synthesis. C Lactate synthesis, (F statistic p < 0.001). * = p < 0.01 compared to 0, 1 µM, and 0.5 mM. # = p < 0.05 compared to 5 mM. § = p < 0.05 compared to control, 1 µM, and 0.5 mM. ƚ = p < 0.001 compared to 5 mM and 10 mM. (PPTX 57 kb)
Rights and permissions
About this article
Cite this article
Pizzolatti, A.L.A., Gaudig, F., Seitz, D. et al. Glucosamine Hydrochloride and N-Acetylglucosamine Influence the Response of Bovine Chondrocytes to TGF-β3 and IGF in Monolayer and Three-Dimensional Tissue Culture. Tissue Eng Regen Med 15, 781–791 (2018). https://doi.org/10.1007/s13770-018-0150-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-018-0150-x