Abstract
Introduction
Many of the activities in daily living require different walking skills such as straight walking (SW), walking with turning (WwT), curved walking (CW) or backward walking (BW) in a dual-task condition. However, there is a lack of evidence regarding the dual task cost (DTC) during different types of walking. Therefore, this study was planned to compare the DTC during different types of walking in people with Parkinson’s disease (PwPD) and healthy controls.
Methods
Thirty-one PwPD and 31 healthy controls were included. Different types of walking were assessed using the 10-Meter Walk Test for SW, the Timed Up and Go Test for WwT, the Figure-of-Eight Walk Test for CW, and the Three-Meter Backward Walk Test for BW. Walking assessments were performed in a single-task and a dual-task condition.
Results
The DTC on SW was the lowest in both groups. The DTC on WwT, BW, and CW were similar in healthy controls, whereas the order of the DTC on remaining walking types from lowest to highest was; WwT, BW, and CW in PwPD. Also, the DTC on WwT, BW, and CW were higher in PwPD than healthy controls. However, the DTC on SW was similar in PwPD and healthy controls.
Conclusion
The DTC is different during SW, WwT, BW, and CW in PwPD. Therefore, walking type should be considered in studies investigating dual task walking in PwPD. Also, dual task exercises consisting of different types of walking should be included in rehabilitation programs of PwPD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many of the activities in daily living require ability to walk in a safe and independent manner [1]. Usual straight walking and more complex walking skills such as turning, curved walking or backward walking are essentials for this ability [2,3,4]. However, walking impairments are one of the most common symptoms in people with Parkinson’s disease (PwPD). Therefore, PwPD have difficulties in performing their activities of daily living [5]. Moreover, most activities of daily living require the ability to perform two tasks at the same time such as walking and talking [6]. The ability to perform two tasks concurrently called as the dual-task ability and relies on automatic motor control and cognitive functions such as executive function. However, PwPD also have difficulties to perform two tasks at the same time due to impairments in these functions [7].
The dual task cost (DTC) refers to deterioration in performance of one or both tasks while performing two tasks concurrently [8]. The DTC on walking is a widely investigated topic in PwPD. However, most of the studies investigating the DTC on walking in PwPD used a single walking task and a single cognitive task [7]. The majority of these studies examined the DTC while performing usual straight walking task [9]. However, in the study of McIsaac et al., which created a framework for dual-task studies, it was reported that the type of motor tasks such as walking during dual-task can affect the DTC and therefore, this situation should be taken into account in dual-task studies [8]. Also, as mentioned earlier, walking activities performed in daily life not only include straight walking, but also require many different walking types such as turning, curved walking or backward walking [2,3,4]. Furthermore, it was reported that curved walking alone constitutes 30% of daily walking activities [10]. In addition, more than half of the steps taken in daily life consist of turning steps [11]. Considering that most of the daily living activities consist of dual-task activities, the DTC during different types of walking in PwPD seems to be an issue that needs to be investigated. However, we have not come across any study investigating this issue.
McIsaac et al. also stated that the type of cognitive task may affect the DTC during dual-tasking [8]. Indeed, there are studies in the literature comparing the DTC of different cognitive tasks during walking in neurological diseases such as multiple sclerosis and Parkinson's disease [6, 12]. However, there is no study comparing the DTC during different walking tasks. Therefore, this study was planned to compare the DTC during different types of walking in PwPD and healthy controls.
Methods
Study design
This cross-sectional study was carried out at the Neurology Clinic at the University of Health Sciences, Ankara Diskapi Yildirim Beyazit Training and Research Hospital between April and May 2022. The study was approved by the Clinical Research Ethics Committee of the University of Health Sciences, Ankara Diskapi Yildirim Beyazit Training and Research Hospital with decision number 134/06. All participants included in the study provided informed written consent according to the Declaration of Helsinki.
Participants
A sample size calculation was performed based on previous data available [13], which estimated that minimum 31 participants were required for each of the patient and control groups to achieve a significance level of 0.05 at a power of 0.90. 38 patients were screened for eligibility. 2 patients refused to participate in the study and 5 patients did not meet the inclusion criteria. Therefore, a convenience sample of 31 PwPD and a control group of 31 age matched healthy participants included in the study.
The inclusion criteria were: (1) being diagnosed with idiopathic PD by an experienced neurologist, (2) ability to walk independently with or without walking aids (3) being 40 years or older. Patients with a neurologic condition other than PD, deep brain stimulation history, Mini Mental State Examination score < 24 [14], visual or hearing disorder, disartria or similar speech disorder were not included in the study. Also, healthy controls with Mini Mental State Examination score < 24, visual or hearing disorder, and any vestibular, orthopedic or rheumatological disorder that hinder walking assessments were not included in the study.
Experimental procedures
All assessments were completed in a single assessment session by an experienced physiotherapist. Assessments of PwPD were conducted in “on” medication condition, 45 to 60 min after taking their usual morning dose of anti-Parkinson medication. All patients were receiving the most appropriate treatment determined by an experienced neurologist. The neurologist adjusted the dosages, frequency, and timing of medication to achieve best symptom control and optimal functioning of PwPD in order to give the most appropriate treatment. Assessments were performed in a quiet, well-lit room. Three-minute resting periods were given between each walking assessments to minimize fatigue effects. Each of the walking assessments was completed in a single task and a dual-task condition. In order to minimize practice effects, single and dual-task walking assessments were counter-balanced in each walking test. The dual-task walking assessments were performed using the same walking task as the single-task condition, but in combination with a serial sevens subtraction task from a randomly chosen number between 300 and 999 as a cognitive task. A different number was used for each dual-task walking assessment in order to avoid the learning effect. The participants were instructed to give equal attention to the walking task and the cognitive task. Prior to the assessment session, demographic data of the participants (e.g., age, gender, body mass index, educational status) were recorded. Disease duration and levodopa equivalent daily dose for PwPD were also recorded.
Outcome measures
Severity of Parkinson’s disease was determined according to the Hoehn & Yahr (H&Y) scale [15]. Severity of motor symptoms was assessed using the Unified Parkinson Disease Rating Scale motor section (UPDRS-III) [16].
Four different walking tests were used for assessing four different types of walking. The first test was the 10 Meter Walk Test (10MWT). The 10MWT is a widely used measure to assess usual straight walking speed. The 10MWT requires the participants to walk at a comfortable pace over a 10-m distance. The total time taken to compete the test is recorded with a stopwatch [17].
The second test was the Timed Up and Go Test (TUG). The TUG is used to assess functional mobility and the ability to incorporate walking with turning. It requires the participants to rise from a chair, walk three meters distance, turn around 180°, walk back three meters distance to the chair, and sit down. The total time taken to compete the test is recorded with a stopwatch [18].
The third test was the Figure of Eight Walk Test (F8WT). The F8WT is used to assess curved walking performance. The test requires the participants to walk in a figure of eight between two cones which are placed 5 ft. apart. The total time taken to compete the test is recorded with a stopwatch [19].
The fourth test was the Three Meter Backward Walk Test (3MBWT). The is used to assess backward walking performance. The test requires the participants to stand straight facing backward on a three meters distance and to walk that distance backwards. The total time taken to compete the test is recorded with a stopwatch [20].
The DTC was calculated separately for each walking test using following formula: (dual-task walking time–single-task walking time)/(single-task walking time) X 100 [21].
Statistical analysis
Data analysis was performed by using IBM SPSS Statistics 21.0 (IBM Corp. Armonk, NY). Demographic data were expressed as mean ± standard deviation for continuous variables and number (percentage) for categorical variables. The Shapiro–Wilk test was used to assess the variables’ distribution. Independent t test for normally distributed variables, the Mann–Whitney U test for non-normally distributed variables, and the chi-square test for categorical variables were used to compare demographic variables of PwPD and controls. A mixed model repeated measures ANOVA with group (PwPD and control) as between factor and the DTC on different walking tests (the 10MWT, the TUG, the F8WT, and the 3MBWT) as within factor was applied to compare the DTC during different types of walking. Main effects and interactions were examined with the repeated measures ANOVA. If a main effect or interaction was found, post-hoc pairwise comparisons with Bonferroni adjustment were performed. Significance value was set as p < 0.05. Effect sizes were expressed as partial eta-squared (ηp2).
Results
Demographic and clinical characteristics of participants were given in Table 1. Comparisons of the DTC during different types of walking in PwPD and controls were given in Table 2.
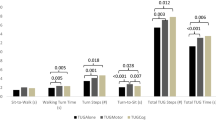
There was a significant main effect of group regarding of the DTC (F (1,60) = 20.436, p < 0.001, ηp2 = 0.254). This indicated that the overall DTC was higher in PwPD than healthy controls. Also, there was a significant main effect of walking type (F (2.569,154.170) = 52.540, p < 0.001, ηp2 = 0.467). Post-hoc analyses revealed that the overall DTC on the 10MWT was lowest than other types of walking (p < 0.001 for all). Also, the overall DTC on the F8WT was highest than other types of walking (p < 0.001 for all). In addition, the overall DTC on the TUG and the 3MBWT were similar (p = 0.307) (Fig. 1).
There was a significant group X walking type interaction effect in terms of DTC (F (2.569,154.170) = 10.247, p < 0.001, ηp2 = 0.146). Post-hoc analyses revealed that DTC was higher in PwPD than healthy controls in the TUG (p < 0.040), the F8WT (p < 0.001), and the 3MBWT (p < 0.001). However, the DTC on 10MWT was similar in PwPD and healthy controls (p = 0.372). Also, the DTC on the 10MWT was significantly lower than other types of walking in PwPD (p < 0.001 for all). In addition, the DTC on the F8WT was significantly higher than other types of walking in PwPD (p < 0.001 for the TUG and p = 0.001 for the 3MBWT). The DTC on 3MBWT was higher than the DTC on the TUG in PwPD (p = 0.019). Similar with PwPD, the DTC on the 10MWT was significantly lower than other types of walking in healthy controls (p < 0.001 for the TUG and the F8WT, p = 0.034 for 3MBWT). On the other hand, the DTC on the TUG, the F8WT, and the 3MBWT were similar in healthy controls (p = 0.229 between the TUG and the F8WT, p > 0.05 between the TUG and the 3MBWT, p = 0.219 between the F8WT and the 3MBWT) (Fig. 1).
Discussion
This study aimed to compare the DTC during different types of walking in PwPD and healthy controls. Ov erall, the results showed that the DTC on straight walking was the lowest in both groups. Interestingly, the DTC on walking with turning, backward walking, and curved walking were similar in healthy controls, whereas the order of the DTC on remaining walking types from lowest to highest was; walking with turning, backward walking, and curved walking in PwPD. Also, the DTC on walking with turning, backward walking, and curved walking were higher in PwPD than healthy controls. However, the DTC on straight walking was similar in PwPD and healthy controls.
As mentioned earlier, we have not come across any study comparing the DTC during different walking tasks. Therefore, interpreting our results with previous findings is not possible. However, it is known that more cognitive resources are used in complex walking tasks than straight walking tasks in both PwPD and healthy controls [22]. It is also known that individuals have a flexible but limited cognitive capacity and exceeding this limit may result in DTC [9]. In addition, complex walking tasks require more balance ability and postural control than straight walking [19, 23]. Therefore, complex walking tasks in this study such as walking with turning, backward walking, and curved walking may have required more cognitive resources. Also, these tasks may have required more balance ability and postural control. These requirements of complex walking tasks may have ultimately resulted in more DTC than straight walking in both groups.
When the DTC on complex walking tasks in PwPD examined, the order of the DTC types from lowest to highest was; walking with turning, backward walking, and curved walking. Evidence from previous studies in PwPD shows that walking with turning, backward walking, and curved walking are all more associated with cognitive functions than straight walking [19, 24, 25]. However, there is a lack of evidence regarding the cognitive load of these types of walking. Previous studies in healthy adults have shown that cognitive functions are particularly associated with the turning phase of the TUG test [26]. Therefore, it can be concluded that turning causes more cognitive load than straight walking. This load can be expected to be even greater due to the loss of automatic motor control ability in PwPD [27]. Furthermore, deteriorated cognitive functions of patients as a result of the disease may reduce the limit of the cognitive capacity [28], and thus concurrently performing a cognitive task with walking and turning may have resulted in more DTC than straight walking PwPD. Similar with turning, backward walking also associated with cognitive functions in PwPD [25]. Previous studies have shown that backward walking increases the cognitive load of postural control [29]. In addition to the increased cognitive load, backward walking requires more proprioception than other types of walking [4]. These evidences may explain the reason behind that backward walking causes more DTC than straight walking and walking with turning in PwPD. Similar with turning and backward walking, curved walking is also associated with cognitive functions [19]. In addition, walking along curved trajectories has different spatiotemporal walking patterns than walking along straight trajectories [3]. Therefore, it requires more coordination, more complex control of posture, and more brain activation [19]. Since both walking with turning and walking backwards require walking along straight trajectories, we think that curved walking causes more cognitive load than these walking types. Thus, given the decreased cognitive capacity in PwPD as well, these reasons may help explain why curved walking resulted in highest DTC in PwPD in this study.
Apart from the cognitive load, the motor requirements of walking with turning, backward walking, and curved walking are different in PwPD. Turning requires segmental coordination of rotation [11]. Backward walking relies on proprioception [4]. Curved walking requires complex control of the trunk, asymmetric action of the legs, mediolateral stability and ability to shift the center of pressure [19]. We used the TUG to assess walking with turning. However, the TUG requires participants to perform only one 180-degree turn [18]. However, the F8WT requires performing a constant turn in two different directions [19]. Therefore, this may contribute to the difference between the DTC on walking with turning and curved walking seen in the results of PwPD. Proprioception also plays a major role in curved walking [30]. However, as mentioned earlier, it also requires complex control of the trunk, asymmetric action of the legs, mediolateral stability and ability to shift the center of pressure [19]. Therefore, it can be speculated that curved walking has more requirements than backward walking. Consequently, this may be the reason behind the difference between the DTC on backward walking and curved walking in PwPD.
In contrast to PwPD, the DTC on walking with turning, backward walking, and curved walking were similar in healthy controls. These walking types have similar requirements and similar walking patters in healthy controls with PwPD [31, 32]. However, it is known that healthy controls have more automatic motor control ability than PwPD [27]. Therefore, these walking types may result in lesser cognitive load in healthy controls. In addition, healthy controls have more cognitive capacity than PwPD [28]. We think that less cognitive load and higher cognitive capacity in healthy controls allow individuals to not exceed their cognitive capacity limit in these three walking types. Thus, similar DTCs can be observed between walking with turning, backward walking, and curved walking in healthy controls.
The DTC on walking with turning, backward walking, and curved walking were higher in PwPD than healthy controls. However, the DTC on straight walking was similar in PwPD and healthy controls. Previous studies have shown that the DTC on complex walking tasks is greater in PwPD than healthy controls [6]. It was reported that gait disturbances became more pronounced in complex gait tasks of PwPD due to impairments in automatic motor control required for balance adjustments and muscle tone regulation [33]. Therefore, this may account for the difference in DTC regarding complex walking tasks between PwPD and healthy controls. In addition, it is also shown that that the DTC on straight walking is similar in PwPD and healthy controls in the early stages of the disease [34]. Considering that the patients in our study had relatively low disease severity, it can be said that our results are in line with these results.
There are several limitations in this study. First, all the PwPD included in our study were in H&Y stage 1, 2 and 3. Therefore, generalization of current results to patients in more severe stages can be misleading. Secondly, all the PwPD included in this study were cognitively intact. Thus, these results could be different in patients with cognitive problems such as mild cognitive impairment or dementia. Third, although the functional performance tests we used to evaluate different types of walking are valid and reliable, spatiotemporal gait parameters were not evaluated in this study. Therefore, results from studies using computer-assisted gait analysis may differ from the results of this study.
In conclusion, order of the DTC during different types of walking in PwPD is; straight walking < walking with turning < backward walking < curved walking. However, this order is different in healthy controls: straight walking < walking with turning = backward walking = curved walking. Moreover, the DTC during different types of walking is higher in PwPD than in healthy controls except for straight walking. The DTC during straight walking is similar in PwPD and healthy controls. These results indicate that walking type should be considered in studies investigating dual task walking in PwPD. Also, dual-task exercises consisting of different types of walking should be included in rehabilitation programs of PwPD.
Data Availability
The data relative to the study are available from the corresponding author upon reasonable request.
References
Shumway-Cook A, Patla AE, Stewart A, Ferrucci L, Ciol MA, Guralnik JM (2002) Environmental demands associated with community mobility in older adults with and without mobility disabilities. Phys Ther 82(7):670–681. https://doi.org/10.1093/PTJ/82.7.670
Glaister BC, Bernatz GC, Klute GK, Orendurff MS (2007) Video task analysis of turning during activities of daily living. Gait Posture 25(2):289–294. https://doi.org/10.1016/J.GAITPOST.2006.04.003
Lowry KA, Brach JS, Nebes RD, Studenski SA, Vanswearingen JM (2012) Contributions of cognitive function to straight- and curved-path walking in older adults. Arch Phys Med Rehabil 93(5):802–807. https://doi.org/10.1016/J.APMR.2011.12.007
Hackney ME, Earhart GM (2010) The effects of a secondary task on forward and backward walking in Parkinson disease. Neurorehabil Neural Repair 24(1):97. https://doi.org/10.1177/1545968309341061
Mirelman A, Bonato P, Camicioli R et al (2019) Gait impairments in Parkinson’s disease. Lancet Neurol 18(7):697–708. https://doi.org/10.1016/S1474-4422(19)30044-4
Zirek E, Ersoz Huseyinsinoglu B, Tufekcioglu Z, Bilgic B, Hanagasi H (2018) Which cognitive dual-task walking causes most interference on the timed up and go test in Parkinson’s disease: a controlled study. Neurol Sci 39(12):2151–2157. https://doi.org/10.1007/S10072-018-3564-2
Raffegeau TE, Krehbiel LM, Kang N et al (2019) A meta-analysis: Parkinson’s disease and dual-task walking. Parkinsonism Relat Disord 62:28. https://doi.org/10.1016/J.PARKRELDIS.2018.12.012
McIsaac TL, Lamberg EM, Muratori LM (2015) Building a framework for a dual task taxonomy. Biomed Res Int. https://doi.org/10.1155/2015/591475
Kelly VE, Eusterbrock AJ, Shumway-Cook A (2012) A review of dual-task walking deficits in people with Parkinson’s disease: motor and cognitive contributions mechanisms and clinical implications. Parkinsons Dis. https://doi.org/10.1155/2012/918719
Segal AD, Orendurff MS, Czerniecki JM, Shofer JB, Klute GK (2008) Local dynamic stability in turning and straight-line gait. J Biomech 41(7):1486–1493. https://doi.org/10.1016/J.JBIOMECH.2008.02.012
Mancini M, Weiss A, Herman T, Hausdorff JM (2018) Turn around freezing: Community-living turning behavior in people with Parkinson’s disease. Front Neurol. https://doi.org/10.3389/FNEUR.2018.00018
Kirkland MC, Wallack EM, Rancourt SN, Ploughman M (2015) Comparing three dual-task methods and the relationship to physical and cognitive ımpairment in people with multiple sclerosis and controls. Mult Scler Int 2015:1–7. https://doi.org/10.1155/2015/650645
Baker K, Rochester L, Nieuwboer A (2007) The immediate effect of attentional, auditory, and a combined cue strategy on gait during single and dual tasks in Parkinson’s disease. Arch Phys Med Rehabil 88(12):1593–1600. https://doi.org/10.1016/J.APMR.2007.07.026
Güngen C, Ertan T, Yaşar R, Eker E (2002) Standardize mini mental test’in türk toplumunda hafif demans tanısında geçerlik ve güvenilirliği. Türk Psikiyatr Derg 13(4):273–281
Hoehn MM, Yahr MD (1967) Parkinsonism: onset, progression and mortality. Neurology 17(5):427–442. https://doi.org/10.1212/WNL.17.5.427
Ramaker C, Marinus J, Stiggelbout AM, van Hilten BJ (2002) Systematic evaluation of rating scales for impairment and disability in Parkinson’s disease. Mov Disord 17(5):867–876. https://doi.org/10.1002/MDS.10248
Lang JT, Kassan TO, Devaney LL, Colon-Semenza C, Joseph MF (2016) Test-retest reliability and minimal detectable change for the 10-Meter Walk Test in older adults with Parkinson’s disease. J Geriatr Phys Ther 39(4):165–170. https://doi.org/10.1519/JPT.0000000000000068
Morris S, Morris ME, Iansek R (2001) Reliability of measurements obtained with the Timed “Up & Go” Test in people with Parkinson disease. Phys Ther 81(2):810–818. https://doi.org/10.1093/PTJ/81.2.810
Soke F, Erkoc Ataoglu NE, Ozcan Gulsen E et al (2023) The psychometric properties of the figure-of-eight walk test in people with Parkinson’s disease. Disabil Rehabil 45(2):301–309. https://doi.org/10.1080/09638288.2022.2028020
Carter VA, Farley BG, Wing K, De Heer H, “dirk”, Jain TK, (2020) Diagnostic accuracy of the 3-Meter backward walk test in persons with Parkinson disease. Top Geriatr Rehabil 36(3):140–145. https://doi.org/10.1097/TGR.0000000000000272
Ozkul C, Eldemir K, Apaydin Y, Gulsen C, Irkec C, Guclu-Gunduz A (2023) Effects of multi-task training on motor and cognitive performances in multiple sclerosis patients without clinical disability: a single-blinded randomized controlled trial. Acta Neurol Belg. https://doi.org/10.1007/s13760-023-02172-7
Maidan I, Rosenberg-Katz K, Jacob Y et al (2016) Altered brain activation in complex walking conditions in patients with Parkinson’s disease. Parkinsonism Relat Disord 25:91–96. https://doi.org/10.1016/J.PARKRELDIS.2016.01.025
Söke F, Demirkaya Ş, Yavuz N et al (2022) The 3-m backward walk test: reliability and validity in ambulant people with multiple sclerosis. Int J Rehabil Res 45(3):209–214. https://doi.org/10.1097/MRR.0000000000000530
Mellone S, Mancini M, King LA, Horak FB, Chiari L (2016) The quality of turning in Parkinson’s disease: a compensatory strategy to prevent postural instability? J Neuroeng Rehabil. https://doi.org/10.1186/S12984-016-0147-4
Tseng IJ, Jeng C, Yuan RY (2012) Comparisons of forward and backward gait between poorer and better attention capabilities in early Parkinson’s disease. Gait Posture 36(3):367–371. https://doi.org/10.1016/J.GAITPOST.2012.03.028
Sunderaraman P, Maidan I, Kozlovski T et al (2019) Differential associations between distinct components of cognitive function and mobility: Implications for understanding aging, turning and dual-task walking. Front Aging Neurosci 11:166. https://doi.org/10.3389/FNAGI.2019.00166
Wu T, Hallett M (2005) A functional MRI study of automatic movements in patients with Parkinson’s disease. Brain 128(Pt 10):2250–2259. https://doi.org/10.1093/BRAIN/AWH569
Muslimović D, Post B, Speelman JD, Schmand B (2005) Cognitive profile of patients with newly diagnosed Parkinson disease. Neurology 65(8):1239–1245. https://doi.org/10.1212/01.WNL.0000180516.69442.95
Correno MB, Hansen C, Chardon M, Milane T, Bianchini E, Vuillerme N (2022) Association between backward walking and cognition in Parkinson disease: a Systematic Review. Int J Environ Res Public Health. https://doi.org/10.3390/IJERPH191912810
Godi M, Giardini M, Schieppati M (2019) Walking along curved trajectories Changes with age and Parkinson’s disease. Hints to rehabilitation. Front Neurol. https://doi.org/10.3389/fneur.2019.00532
Belur P, Hsiao D, Myers PS, Earhart GM, Rawson KS (2020) Dual-task costs of texting while walking forward and backward are greater for older adults than younger adults. Hum Mov Sci 71:102619. https://doi.org/10.1016/J.HUMOV.2020.102619
Orendurff MS, Segal AD, Berge JS, Flick KC, Spanier D, Klute GK (2006) The kinematics and kinetics of turning: limb asymmetries associated with walking a circular path. Gait Posture 23(1):106–111. https://doi.org/10.1016/J.GAITPOST.2004.12.008
Stuart S, Vitorio R, Morris R, Martini DN, Fino PC, Mancini M (2018) Cortical activity during walking and balance tasks in older adults and in people with Parkinson’s disease: a structured review. Maturitas 113:53–72. https://doi.org/10.1016/j.maturitas.2018.04.011
Ozcan Gulsen E, Tunca Yilmaz O (2022) Dual task effect on upper and lower extremity skills in different stages of Parkinson’s disease. Acta Neurol Belg. https://doi.org/10.1007/S13760-022-02007-X
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
Conception and study design (BK and ÇG), data collection (BK, ÇG, FS, and EÖG), statistical analysis (NE), interpretation of results (BK, ÇG, FS, EÖG, and NE), drafting the manuscript (BK, ÇG, FS, and EÖG), revising and editing the manuscript (ÖTY and SSÇ), supervision (ÖTY and SSÇ).
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study was approved by the Clinical Research Ethics Committee of the University of Health Sciences, Ankara Diskapi Yildirim Beyazit Training and Research Hospital with decision number 134/06.
Informed consent
Informed consent was obtained from all participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Koçer, B., Gülşen, Ç., Söke, F. et al. Comparison of dual-task costs during different types of walking in people with Parkinson’s disease: a controlled study. Acta Neurol Belg 123, 2221–2227 (2023). https://doi.org/10.1007/s13760-023-02271-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-023-02271-5