Abstract
Spinal epidural angiolipomas are rare lesions composed of mature lipomatous and angiomatous elements. In this paper, the authors review a surgical series of ten patients with epidural angiolipomas. All patients had performed preoperative and postoperative magnetic resonance imaging. The diagnosis of angiolipoma was based on pathology. All the follow-up data were obtained during office visits. There were 5 males and 5 females with a mean age of 53.6 years. One tumor was located in the cervicothoracic, six in the thoracic, and three in the lumbar spine. The most common symptom was progressive motor deficit. Gross total resection of the tumor was achieved in nine cases, and subtotal resection was obtained in one case. No recurrence or regrowth of the residual tumor was observed during a follow-up period of 50.8 months. At the last follow-up, 90% of patients experienced improvement in the neurological function. Epidural angiolipomas are benign but clinically progressive lesions. Early surgery should be performed to prevent irreversible neurological deficits. Postoperative radiotherapy is not recommended. The risk of long-term recurrence/regrowth of the lesions is low, and a good clinical outcome after total or subtotal removal can be expected.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Angiolipomas are specific vascular variants of lipoma, consisting of mature fatty tissue and abnormal blood vessels [1, 2]. Most of angiolipomas are observed subcutaneously on the trunk and limbs [3]. Spinal epidural angiolipomas are extremely rare, accounting for about 1% of all spinal axis tumors [4, 5].
With the aid of magnetic resonance imaging (MRI), spinal epidural angiolipomas are being discovered in increasing numbers; nevertheless, most studies are case reports and large series studies are very limited in the literature [1, 2, 4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32]. In this article, we present the clinical presentation, radiological features, and surgical outcomes of ten patients surgically treated with in a single institution and review the pertinent literature.
Methods
After obtaining approval from Institutional Review Board of The First Affiliated Hospital of USTC, we retrospectively reviewed the data of ten patients with epidural angiolipomas between 2011 and 2017. Preoperatively, all patients underwent MRI as the standard radiological investigation. Surgery was performed in all patients through posterior median approach with intraoperative monitoring of somatosensory and motor-evoked potentials. Histological specimens were sent to the Department of Pathology for histological confirmation. Modified McCormick Classification (MMC) (Table 1) was applied to assess neurological function [33]. All patients were monitored post-operatively by outpatient examination.
Results
Clinical presentation
There were five male and five female patients aged from 25 to 69 years (mean 53.6 years). The presentations included motor deficit, sensory disturbance, pain, and sphincter dysfunction. The mean duration of symptoms was 16.8 months (range 2–48 months). Four patients had Grade II status according to the MMC, three patients had Grade III, two patients had Grade Ib, and one patient who had Grade IV.
Preoperative imaging diagnosis
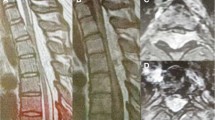
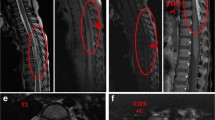
The lesions were located in cervicothoracic (1 case, 10%), thoracic (6 cases, 60%), and lumbar (3 cases, 30%) spine. Based on T1-weighted images (WI), the tumors had slight hypointensity in 1 case, isointensity in 2 cases, hyperintensity in 6 cases, and mixed signal intensity in 1 case. T2-WI revealed hyperintensity in 9 cases and mixed signal intensity in 1 case. Contrast-enhanced T1-WI revealed homogeneous enhancement in 6 cases and heterogeneous enhancement in 4 cases. Eight lesions were located in the dorsal canal and 2 were in the lateral canal. In 1 case, the lesion extended into the intervertebral foramen, and extended to the paravertebral space. An illustrative example of case 4 is illustrated in Figs. 1, 2, and 3.
Magnetic resonance imaging showed a spindle-shaped epidural tumor at the T8–T11 levels. The tumor was iso–hyperintense on T1-weighted image (WI) (a) and hyperintense on T2-WI (b). Contrast-enhanced T1-WI revealed homogeneous markedly enhancement and severe spinal cord compression (c). Coronary contrast-enhanced T1-WI exhibited dorsal location (d)
Surgical outcomes and pathological examination
Intraoperatively, the lesions were yellowish or red-grayish, soft or rubbery hard, and with moderate vascularity. Most tumors were well demarcated from the dura, which facilitated their exposure and dissection. Gross total resection (GTR) was achieved in 9 cases (90%) and subtotal resection (STR) was achieved in 1 case (10%). Histological examination showed mature adipose tissue interlaced with numerous blood vessels ranging from capillary to venular in size (Fig. 4).
Follow-up
No patient received postoperative radiotherapy. During a mean follow-up period of 50.8 months, no recurrence or regrowth of the residual tumor was observed on MRI. Neurological status had markedly improved in most patients and remained stable in one patient. The detailed clinical profiles are summarized in Table 2.
Discussion
Epidural angiolipomas are rare entities, accounting for about 2–3% of epidural tumors [4, 24, 29]. They can be traditionally categorized into two subtypes: non-infiltrating and infiltrating, the latter extending into the vertebral body [34]. Based on the literature, patients in their 4th–6th decades of life are the most affected with a slight female predominance [1, 31]. In our group, the age ranged from 25 to 69 years, with a mean age of 53.6 years, which accorded with the literature. In addition, female/male ratio of 1:1 was noted. Some authors have reported a female/male ratio of 1.5:1, which might suggest that clinical symptoms are more frequent in female patients because of possible hormonal influences [14, 18]. However, this hypothesis has not yet been proven. The most common location is the thoracic spine in about 78.8% of cases, followed by lumbar in fewer than 10.2%; a cervical location is extremely rare [1, 4, 12, 15, 16, 23,24,25, 29]. In the study, 60% of the angiolipomas were in the thoracic region, 30% were in the lumbar region, and 10% were in the cervicothoracic region. It was proposed that an insufficiency of blood supply in thoracic spine contributes to the thoracic predominance [5].
Patients in our study commonly present with spinal cord or nerve root compression. The relatively slow clinical course (mean 14.2 months) reflects the benign nature of angiolipomas. Motor deficit or pain is always the most common initial symptom. Despite vascularization of angiolipomas, accelerated onset of symptoms secondary to spontaneous bleeding is relatively unusual. To our knowledge, only a few cases presenting with acute paraplegia caused by a hemorrhagic epidural angiolipoma have been described [6, 8, 13, 18, 20, 27, 28, 30]. Trauma, pregnancy, and hormonal changes were assumed to be aggravating factors [28, 30]. Lack of well-developed arterioles and low vascular characteristics inside the tumor may correlate with low hemorrhagic risk [21].
Some theories of etiology suggested that angiolipomas may originate from primitive, pluripotential mesenchyme tissue [10, 16], and congenital malformations [11]. Pagni and Canavero et al. support the theory of abnormal developmental origin, based on their findings of spinal angiolipomas in patients with birth defects outside the central nervous system [29]. Barodosi et al. postulated that they arise from pluripotential stem cells with secretory activity [9]. Thus, various pathogenic mechanisms may cause spinal angiolipomas.
MRI is the modality of choice for epidural angiolipomas [35]. Non-infiltrating lesions are usually spindle shaped with taper ends and locate in the dorsal space of the epidural space [1]. Infiltrating lesions usually infiltrate the surrounding tissues, especially the bone with extending into the paraspinal region [21]. Owing to fatty content, they are usually hyperintense on T1-WI and T2-WI, hypointense on fat-suppressed images [2, 14, 18]; nevertheless, mixed intensity or hypointensity can also be noticed on T1-WI due to increased vascularity within the tumor that is likely to be encountered at surgery [5, 23, 32]. Contrast-enhanced T1-WI shows significant enhancement because of vascular component [18]. Thus, the differential diagnosis including epidural contrast-enhancing lesions, such as arteriovenous malformations, cavernous angiomas, schwannomas, and metastases [32]. High-flow arteriovenous malformations always have vascular flow voids on MRI [1, 25]; nevertheless, angiolipomas do not contain vascular flow void due to the presence of capillaries and venous channels [14, 21]. Cavernous angiomas are generally isointense on T1-WI and hyperintense on T2-WI [36]. Schwannomas always have necrosis, extending into the paraspinal region with the enlargement of intervertebral foramen [36, 37]. However, paravertebral component with foraminal extension can also be noticed in infiltrating epidural angiolipomas, which make angiolipomas difficult to differentiate from schwannomas. Most of epidural metastases are associated with bone marrow and paravertebral involvement [38]. Because of its rare occurrence, we believe that definitive preoperative diagnosis may be challenging based only on MRI. An accurate diagnosis still depends on pathological examinations. Histologically, angiolipomas are composed of mature adipose tissue and blood vessels, features of which are described as being either normal or mimicking capillary angiomas, cavernous angiomas, or arteriovenous malformations [17]. The ratio of fat to vessels is variable, ranging from 1:3 to 2:3 [15]. Atypia, pleomorphism, and mitotic figures of both adipose and angiomatous component were never encountered [29]. In the study, all histological characteristics were consistent with angiolipomas.
Surgical resection is the first choice for the treatment of epidural angiolipomas. According to the literature, the GTR rate of epidural angiolipomas is 72.4–74.1% [4, 12]. In our series, 90% of tumors showed well-demarcated dissection plane with no adhesions to the dura, and can be easily removed by a full or partial laminectomy. However, for infiltrating tumors which involve the vertebral body with huge extra-spinal extension, GTR can be more difficult. Labram et al. recommended combined microneurosurgical posterior approach, stabilization of the affected vertebrae, and thoracoscopic approach in two-stage surgery [5]. In our case, we opted for a less aggressive treatment by performing GTR of the intraspinal component and STR of paravertebral component. The tumor-invaded vertebral body which was considered to be analogous to vertebral hemangiomas may not enlarge [7], and thus, it was preserved to maintain spinal stabilization.
Tumor recurrence of epidural angiolipomas following surgery is rare and only two cases were reported [10, 19]. In our series, all patients did not receive postoperative radiotherapy, and had no tumor recurrence or regrowth of the residual tumor. We believe that postoperative radiotherapy may be unnecessary because of slow growth and low recurrence risk of the tumors. If a residual tumor showed obvious regrowth with clinical deterioration, an early reoperation is recommended as the primary treatment [10].
In our series, during a mean follow-up period of 50.8 months, 90% of patients experienced an improvement in the MMC. However, the neurological function did not get improvement in one GTR case with duration of symptoms for 4 years. Long-term spinal cord compression could cause irreversible neurological dysfunction. Therefore, we recommend that if neurological deficits are progressive, early surgery should be performed. Because these tumors grow slowly, we will continue to perform periodic neurological examination and MRI to obtain longer follow-up data of the patients.
In conclusion, angiolipomas should be considered in the differential diagnosis of epidural contrast-enhancing lesions, especially if a thoracic lesion has dorsal localization and hyperintense on T1-WI and T2-WI. Chronically progressive spinal cord compression is main clinical symptoms. Early surgery should be performed to prevent irreversible neurological deficits. Postoperative radiotherapy is not recommended. A good clinical outcome after GTR or STR can be expected, and the risk of long-term recurrence is low.
References
Garg A, Gupta V, Gaikwad S, Deol P, Mishra NK, Sharma MC, Vaish S (2002) Spinal angiolipoma: report of three cases and review of MRI features. Australas Radiol 46:84–90
Nadi MM, Nadi AM, Zabara MY, Ahmad TM (2015) Management of infiltrating spinal epidural angiolipoma. Neurosciences (Riyadh) 20:159–163
Kitagawa Y, Miyamoto M, Konno S, Makino A, Maruyama G, Takai S, Higashi N (2014) Subcutaneous angiolipoma: magnetic resonance imaging features with histological correlation. J Nippon Med Sch 281:313–319
Gelabert-González M, García-Allut A (2009) Spinal extradural angiolipoma: report of two cases and review of the literature. Eur Spine J 18:324–335
Labram EK, el-Shunnar K, Hilton DA, Robertson NJ (1999) Revisited: spinal angiolipoma–three additional cases. Br J Neurosurg 13:25–29
Akhaddar A, Albouzidi A, Elmostarchid B, Gazzaz M, Boucetta M (2008) Sudden onset of paraplegia caused by hemorrhagic spinal epidural angiolipoma. A case report. Eur Spine J 17(Suppl 2):S296–S298
Andaluz N, Balko G, Bui H, Zuccarello M (2000) Angiolipomas of the central nervous system. J Neurooncol 49:219–230
Anson JA, Cybulski GR, Reyes M (1990) Spinal extradural angiolipoma: a report of two cases and review of the literature. Surg Neurol 34:173–178
Bardosi A, Schaake T, Friede RL, Roessmann U (1985) Extradural spinal angiolipoma with secretory activity. An ultrastructural, clinico-pathological study. Virchows Arch A Pathol Anat Histopathol 406:253–259
Bender JL, Van Landingham JH, Manno NJ (1974) Epidural lipoma producing spinal cord compression. Report of two cases. J Neurosurg 41:100–103
Bucy PC, Ritchey H (1947) Klippel-Feil’s syndrome associated with compression of the spinal cord by an extradural hemangiolipoma. J Neurosurg 4:476–481
Chotai S, Hur JS, Moon HJ, Kwon TH, Park YK, Kim JH (2011) Spinal angiolipoma–case report. Neurol Med Chir (Tokyo) 51:539–542
da Costa MD, Paz Dde A, Rodrigues TP, Gandolfi AC, Lamis FC, Stavale JN, Suriano IC, Cetl LD, Cavalheiro S (2014) Hemorrhagic onset of spinal angiolipoma. J Neurosurg Spine 21:913–915
Diyora B, Nayak N, Kukreja S, Kamble H, Sharma A (2011) Thoracic epidural angiolipoma with bilateral multilevel extraspinal extensions: a rare entity. Neurol India 59:134–136
Fujiwara H, Kaito T, Takenaka S, Makino T, Yonenobu K (2013) Thoracic spinal epidural angiolipoma: report of two cases and review of the literature. Turk Neurosurg 23:271–277
Griebel RW, Khan M, Rozdilsky B (1986) Spinal extradural angiolipoma. A case report and literature review. Spine (Phila Pa 1976) 11:47–48
Haddad FS, Abla A, Allam CK (1986) Extradural spinal angiolipoma. Surg Neurol 26:473–486
Hu S, Hu CH, Hu XY, Wang XM, Dai H, Fang XM, Cui L (2013) MRI features of spinal epidural angiolipomas. Korean J Radiol 14:810–817
Kujas M, Lopes M, Lalam TF, Fohanno D, Poirier J (1999) Infiltrating extradural spinal angiolipoma. Clin Neuropathol 18:93–98
Lacour M, Gilard V, Marguet F, Curey S, Perez A, Derrey S (2018) Sudden paraplegia due to spontaneous bleeding in a thoracic epidural angiolipoma and literature review. Neurochirurgie 64:73–75
Leu NH, Chen CY, Shy CG, Lu CY, Wu CS, Chen DC, Guo TH (2003) MR imaging of an infiltrating spinal epidural angiolipoma. Am J Neuroradiol 24:1008–1011
Pagni CA, Canavero S (1992) Spinal epidural angiolipoma: rare or unreported? Neurosurgery 31:758–764
Park JH, Jeon SR, Rhim SC, Roh SW (2008) Lumbar spinal extradural angiolipoma: case report and review of the literature. J Korean Neurosurg Soc 44:265–267
Preul MC, Leblanc R, Tampieri D, Robitaille Y, Pokrupa R (1993) Spinal angiolipomas. Report of three cases. J Neurosurg 78:280–286
Provenzale JM, McLendon RE (1996) Spinal angiolipomas: MR features. Am J Neuroradiol 17:713–719
Rabin D, Hon BA, Pelz DM, Ang LC, Lee DH, Duggal N (2004) Infiltrating spinal angiolipoma: a case report and review of the literature. J Spinal Disord Tech 17:456–461
Sandvik U, Svensdotter E, Gustavsson B (2015) Spinal cavernous extradural angiolipoma manifesting as a spontaneous spinal epidural hematoma in a child. Childs Nerv Syst 31:1223–1226
Sankaran V, Carey M, Shad A (2010) Traumatic bleeding of spinal angiolipoma presenting with subacute paraparesis–a case report and histopathological aspects. Br J Neurosurg 24:714–715
Si Y, Wang Z, Pan Y, Lin G, Yu T (2014) Spinal angiolipoma: etiology, imaging findings, classification, treatment, and prognosis. Eur Spine J 23:417–425
Tsutsumi S, Nonaka Y, Abe Y, Yasumoto Y, Ito M (2011) Spinal angiolipoma in a pregnant woman presenting with acute epidural hemorrhage. J Clin Neurosci 18:849–851
Turgut M (1999) Spinal angiolipomas: report of a case and review of the cases published since the discovery of the tumour in 1890. Br J Neurosurg 13:30–40
Wang B, Yang Z, Yang J, Wang G, Xu Y, Liu P (2014) Spinal angiolipoma: experience of twelve patients and literature. Neurol India 62:367–370
Aghakhani N, David P, Parker F, Lacroix C, Benoudiba F, Tadie M (2008) Intramedullary spinal ependymomas: analysis of a consecutive series of 82 adult cases with particular attention to patients with no preoperative neurological deficit. Neurosurgery 62:1279–1285
Lin JJ, Lin F (1974) Two entities in angiolipoma. A study of 459 cases of lipoma with review of literature on infiltrating angiolipoma. Cancer 34:720–727
Yamashita K, Fuji T, Nakai T, Hamada H, Kotoh K (1993) Extradural spinal angiolipoma: report of a case studied with MRI. Surg Neurol 39:49–52
Feng J, Xu YK, Li L, Yang RM, Ye XH, Zhang N, Yu T, Lin BQ (2009) MRI diagnosis and preoperative evaluation for pure epidural cavernous hemangiomas. Neuroradiology 51:741–747
Demachi H, Takashima T, Kadoya M, Suzuki M, Konishi H, Tomita K, Yonezawa K, Ubukata A (1990) MR imaging of spinal neurinomas with pathological correlation. J Comput Assist Tomogr 4:250–254
Boukobza M, Mazel C, Touboul E (1996) Primary vertebral and spinal epidural non-Hodgkin’s lymphoma with spinal cord compression. Neuroradiology 38:333–337
Acknowledgements
We would like to thank all the patients who trusted us and all the physicians and staff who helped this study.
Funding
The authors declare that no funding was involved.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Ethical approval
This study was approved by Institutional Review Board of The First Affiliated Hospital of USTC.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, T., Niu, C. Microsurgical treatment for spinal epidural angiolipomas. Acta Neurol Belg 121, 421–427 (2021). https://doi.org/10.1007/s13760-019-01192-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-019-01192-6