Abstract
The growth of international trade, coupled with an expansion of large-scale pine plantations in South America during the second half of the twentieth century, has significantly increased the opportunities for the invasion of forest insects. Bark beetles (Coleoptera: Curculionidae, Scolytinae) are a large and diverse group of insects, commonly recognized as one of the most important tree mortality agents in coniferous forests worldwide and an important group among invasive forest species. In this study, we combined data from field sampling with published records of established non-native pine bark beetles, to describe their distribution and invasion history in pine plantations across southern South America, reviewing the available information on their phenology and host range. We obtained records of established populations of six Eurasian species distributed in two major regions: the southwest region comprises plantations in Chile and the Argentine Patagonia, with four bark beetle species: Hylastes ater, Hylastes linearis, Hylurgus ligniperda, and Orthotomicus laricis; the northeastern zone includes northeastern Argentina, Uruguay, and southern Brazil, and includes three bark beetle species: Cyrtogenius luteus, H. ligniperda, and O. erosus. The establishment of non-native populations across the study area began in the 1950s, and from the 1980s onwards, there has been an exponential increase in introductions. We predict that several of these species will continue spreading across South America and that new species will continue arriving. We highlight the importance of international collaboration for early detection and management of non-native pine bark beetles.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Numerous forest insect species are being transported and introduced to regions beyond their native range due to the growing process of globalization (Brockerhoff and Liebhold 2017). In some cases, these species manage to establish and subsequently inflict ecological and economic impacts on forest ecosystems (Freer-Smith and Webber 2017; Kenis et al. 2017). The invasion of phytophagous insects is usually closely related to the prior introduction of their hosts, and it has been demonstrated that there can be a significant time lag between the introduction of the hosts and the arrival of associated insects (Bonnamour et al. 2023). Consequently, the introduction of forest insects is expected to continue increasing (Brockerhoff and Liebhold 2017; Seebens et al. 2017).
Bark beetles (Coleoptera: Curculionidae, Scolytinae) are a large and diverse group of forest insects that breed in and feed on the phloem of trees (Kirkendall et al. 2015; Gómez et al. 2023). Several species of this group are commonly recognized as important tree mortality agents in coniferous forests worldwide (Raffa et al. 2008; Biedermann et al. 2019). Furthermore, because of their cryptic nature and association with bark and wood, they can be transported accidentally in timber, wood products, and wood packaging materials. Many species have successfully established in various parts of the world, representing an important group of invasive forest insects (Brockerhoff et al. 2014; Lantschner et al. 2020; Pureswaran et al. 2022; Grégoire et al. 2023).
Among true bark beetles, there are at least 220 species that are specialized in feeding and reproducing on pine trees (Pinus spp.) (Wood and Bright 1992), and 25 of these species (11%) have established outside of their native range (Wood and Bright 1992; Lantschner et al. 2020). Currently, pine species exhibit a broad global distribution. While native pine forests dominate numerous areas within the Holarctic region, extensive plantations of non-native pines have been established across various regions in the Southern Hemisphere (Procheş et al. 2012; FAO 2020; Villacide et al. 2023). Furthermore, pines are frequently utilized for constructing wood packaging material used in international trade (Krishnankutty et al. 2020). Consequently, the considerable diversity of pine bark beetles, coupled with the global prevalence of pine trees and the widespread transportation of pine wood, renders species feeding on pines a significant threat in terms of invasion (Lantschner et al. 2017; Faccoli et al. 2020; Vilardo et al. 2022). In this context, through the global initiative to address the risk associated with trade, international standards for phytosanitary measures (ISPMs) were developed under the International Plant Protection Convention. The standard related to wood boring pests in wood packaging material, ISPM 15, was first adopted in 2002 and is now implemented by 100 countries, including those of southern South America (Greenwood et al. 2023).
In southern South America, plantation forestry with non-native fast-growing trees has substantially increased since the mid-twentieth century, to produce timber, pulp, and fuel. Pines represent one of the main planted genera, usually consisting of intensively managed, even aged, and regularly spaced stands of single species (FAO 2020). In forest plantations in South America, the detection of established non-native pests has increased exponentially in recent decades (Corley et al. 2020; Estay 2020; Villacide et al. 2023). Forty-five species of exotic Scolytinae have been documented as established in South America (Lantschner et al. 2020) and seven of these species are pine bark beetles: Cyrtogenius luteus (Blandford 1894), Dryocoetes autographus (Ratzeburg, 1837), Hylastes ater (Paykull 1800), Hylastes linearis Erichson 1836, Hylurgus ligniperda (Fabricius 1787), Orthotomicus erosus (Wollaston 1857) and Orthotomicus laricis (Fabricius 1792) (Wood 2007; Mausel et al. 2007; Tiranti 2010; Gómez et al. 2017; Kirkendall 2018).
The initial stage in developing an effective control strategy for non-native species entails promptly identifying them in recently invaded areas and understanding the biological characteristics they exhibit in the new environmental conditions. Although the occurrence of some of these species of non-native pine bark beetles and ecological characteristics such as the patterns in flight phenology have been studied in some locations (Mausel et al. 2007; Gómez et al. 2017), the distribution and invasion history of these species within pine plantations of South America remain poorly understood. The aim of this study was to describe the distribution and invasion history of the established bark beetles in pine plantations of southern South America and to review the available information on their phenology and host range.
Methods
Study area
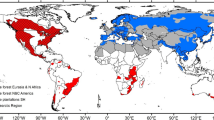
Our study focused on the main pine (Pinus spp.) growing regions in the southern part of South America (latitudes 10° to 50° S), encompassing Argentina, Bolivia, south of Brazil, Chile, Paraguay, and Uruguay (Fig. 1). Across this region, about 5.6 million ha of land are planted with pines (BRACELPA 2004; López 2004; Paraguay 2009; MAGyP 2011; FAO 2020; Álvarez González et al. 2022). Pine plantations are mainly composed of North American species such as P. contorta, Pinus elliottii, P. pinaster, P. ponderosa, P. radiata, and P. taeda (Fig. 1).
Bark beetle data
We carried out sampling of non-native true bark beetle known to feed on pines (Pinus spp.) across the different pine growing areas of our study area (see description of sampling sites in supplementary material, Table S1). Variable numbers of sampling sites were established in pine plantations across different administrative divisions of Argentina: provinces of Misiones (2 sites), Corrientes (6 sites), Entre Ríos (1 site), Córdoba (3 sites), Neuquén (2 sites), Río Negro (2 sites), and Chubut (3 sites); Chile: provinces of Biobío (3 sites), Los Lagos (1 site), and Los Ríos (2 sites); and Uruguay: departments of Tacuarembó (1 site) and Rivera (1 site). At each site, bark beetle species were captured by placing variable numbers (3 to 6) of traps (i.e., Lindgren funnel traps, Cross vane panel traps), during at least 2 months for at least one season from 2015 to 2023. Traps were baited with generic bark beetle attractants (i.e., ethanol, α-pinene, and/or turpentine). Baits were replaced at regular intervals as needed and captured beetles were collected regularly. Additionally, we also manually collected bark beetle species by inspecting cut pine logs available in some sites. In these cases, we removed the bark of the logs and searched for bark beetle adults. All the sampled insects were collected and conserved in 70% ethanol for future identification.
To complement the information obtained from field samplings, we compiled information on the history of introductions and distribution of non-native pine bark beetles across southern South America, from a variety of sources including both published scientific articles, regional reports, and National surveys (i.e., SAG 2022 for Chile) (see Table S1). Only records of species that have established self-sustaining populations in pine growing regions of southern South America were included in our analyses (i.e., species that have been found for consecutive years the new location). For each species, we compiled information about the locations where the species were recorded, and the year of the first report in each country and province. We also compiled information on the countries of their native range, other countries where the species have established outside of their native range and the year of the first detection in these countries, the recorded hosts, and other biological attributes of each species (i.e., body size, mating system, voltinism, phenology; Table S2).
Data processing and analysis
Collected bark beetles were identified to species level following identification keys based on morphological features (Wood 2007; Gómez 2014). We combined the information of locations where each species was registered from both sampled specimens and bibliography. To avoid overrepresented locations and to create a more spatially uniform dispersion of locations, we buffered collection point locations by 50 km distance, removing points within this distance.
In order to characterize the environmental conditions where each species is currently distributed in the study area, we obtained data on the planted pine species and climatic variables of each selected location. We used two climatic variables that have been found to be important in determining the distribution of insects: minimum temperature of the coldest month (bio6) and maximum temperature of the warmest month (bio5), both derived from the WorldClim database version 2.1, for de period 1970–2000, 5 arc minutes resolution (Fick and Hijmans 2017). For each locality where each species was recorded, we extracted the values of bio 5 and bio 6 using the software QGIS (QGIS.org 2023). Then, we estimated the average and range of these variables in which each species is distributed in South America.
Results
We sampled and collected information for six of the known non-native pine bark beetle species established in southern South America: Cyrtogenius luteus, Hylastes ater, H. linearis, Hylurgus ligniperda, Orthotomicus laricis, and O. erosus. We obtained information of occurrences of these species for most of the regions where pine plantations are distributed across the study area: Chile, Brazil, Argentina, and Uruguay (Fig. 2), while we did not obtain data for Bolivia and Paraguay. Additionally, one more pine bark beetle species has been described to be established in the study area: Dryocoetes autographus. This species was reported from the state of São Paulo, Brazil (Wood 2007). However, we did not find any new references confirming its establishment anywhere in South America. Moreover, because D. autographus is a spruce specialist (Picea spp.), this report is unlikely.
Hylurgus ligniperda (Fabricius, 1787)
Hylurgus ligniperda is the bark beetle species that was first detected in South America (1956), in Uruguay (Fig. 3); three decades later, it was detected in Chile (1985) and subsequently in Brazil (1991) and Argentina (2007). In this way, this species has the widest distribution in the region, ranging from southern Brazil to southern Argentina and Chile—from latitude 24 to 47°S (Table 1, Fig. 2). In the study area, the locations where the species was found range from a maximum temperature of 17 to 32 °C and minimum temperature of − 7 to 10 °C (Fig. 4). We describe, in this study, the occurrence of this species for the first time in the Argentine provinces of Entre Rios, Corrientes, Córdoba, and Río Negro (Table 1). This species is native to Europe, West Asia, and North Africa (Wood and Bright 1992), and has also invaded most parts of the world where pines are grown, during the last century. It was first detected outside of its native range in Japan (1935), then in Australia (1942), South Africa (1973), New Zealand (1974), Sri Lanka (unknown date), the USA (1995), South Korea (1999), and China (2019) (Bain 1977; Geertsema 1982; Wood and Bright 1992; Hoebeke 2001; Park et al. 2017; Lin et al. 2021) (Table S2).
Range of temperature conditions of the locations where each bark beetle species was registered. Black: maximum temperature of the warmest month, red: minimum temperature of the coldest month. Points indicate the mean value among all locations where each species was detected, and bars indicate the maximum and minimum value observed for each variable
Hylurgus ligniperda is monogamous and usually has one generation per year but may have up to three generations per year (CABI 2021a). The flight activity in Chile has been observed to have two peaks that are variables in magnitude, one in the spring and another one in the autumn (Lanfranco et al. 2002; Mausel et al. 2007, Ahumada personal observation), while in Uruguay the peak occurs earlier, during the winter, with a shorter peak in the summer (Gómez et al. 2017). Captures of this species in Uruguay were found to be significantly predicted by temperature anomaly and season since it was most abundant during unusually warm periods of winter months, while its abundance was not significantly related to precipitation during trapping periods (Gómez et al. 2020). This species develops in the lower part of trunk, root collar, and emerging roots of recently dead, weakened, or cut Pinus species (Wood and Bright 1992; Lieutier et al. 2016). In the study area, it has been found feeding on P. contorta, P. elliottii, P. pinaster, P. ponderosa, P. radiata, P. sylvestris, and P. taeda (Tables S1 and S2).
Hylastes ater (Paykull, 1800)
Hylastes ater was first detected in Chile in 1983, and it has spread across all the pine growing areas of this country. Later, in 2007, it was also detected in the neighboring pine plantations of southern Argentina, reaching also to the province of Córdoba in central Argentina. Here we present the first records for this species for the provinces of Córdoba and Río Negro in Argentina (Table 1, Fig. 2). The current distribution of H. ater in South America ranges from latitude 32 to 46°S, and it has not been registered in any of sampled locations of the northeastern part of the study area (north-east Argentina, Uruguay, and S Brazil, Figs. 2 and 3). In the study area, this species was found in locations with maximum temperatures ranging from 17 to 33 °C and minimum temperatures ranging from − 4 to 8 °C (Fig. 4). Hyastes ater is native from Europe and Asia and has also invaded New Zealand in 1929 and Australia in 1936 (Clark 1932; Boomsma and Adams 1943) (Table S2).
Hylastes ater is a monogamous species and has one or two generations per year (Mausel et al. 2007; Lieutier et al. 2016). In Chile, its flight period has been observed to exhibit either a bimodal pattern, with peaks of abundance in both spring and fall, or a unimodal pattern, with a peak of abundance in the fall (Mausel et al. 2007). This species infests the base of trunks, collars, and thick exposed root of weakened or felled trees. Its host range includes primary Pinus species, and rarely other genera of Pinaceae (Abies, Picea, Larix, Pseudotsuga), Cupressaceae (Thuja, Sequoia, Chamaecyparis), Taxaceae (Taxus), and Araucariaceae (Araucaria) (Wood and Bright 1992; Lieutier et al. 2016). In the study area, it was observed feeding on P. contorta, P. elliottii, P. ponderosa, Pinus radiata, and P. taeda (Tables S1 and S2).
Orthotomicus laricis (Fabricius, 1792)
Orthotomicus laricis was detected for the first time in Chile, in 1986 (where it was misidentified as O. erosus, see Kirkendal 2018 for more detail), and about two decades later it was also detected in southern Argentina (Río Negro, first report, Table 1), spreading across all pine plantation areas of these regions (Mausel et al. 2007; Tiranti 2010). More recently, it was also detected in the province of Tucuman, in the north-west of Argentina (Córdoba et al. 2021). Currently, the distribution of this species ranges from latitude 26 to 43°S, encompassing locations with maximum temperatures ranging from 20 to 32 °C and minimum temperatures ranging from − 3 to 7 °C (Fig. 4). Orthotomicus laricis is native to Europe, Asia, and North Africa, and there have been no reports of its establishment outside of its native range in other regions of the world.
Orthotomicus laricis is a polygamous species and has one to two generations per year (Chararas 1962). Its phenology and flight activity has not been investigated in the study area. The species breeds on recently cut or felled logs. The host range of this species includes trees from the genera Pinus, Picea, and Larix (Wood and Bright 1992; Lieutier et al. 2016), although in the study area it has been observed feeding only on P. contorta, P. ponderosa, and P. radiata (Tables S1 and S2).
Hylastes linearis Erichson, 1836
Hylastes linearis is the only species registered in just one country across the study area. The species was first found in South America in 2006 (Fig. 3), in the most northern areas with pine plantations of Chile (O’Higgins province), and during the last years it has spread further south up to the province of Osorno, ranging from latitudes 33 to 40.6°S (Figs. 2 and 3, Table 1) (SAG 2010). The maximum temperature of the locations where this species was registered range from 19 to 31 °C and the minimum temperature ranged from − 1 to 7 °C (Fig. 4). Unlike the other pine bark beetle species registered in Chile, H. linearis was not detected in the pine plantations of southern Argentina. This species is native to Europe, West Asia, and North Africa, and has also established in South Africa in 1931 (Schedl 1957; Wood and Bright 1992).
Hylastes linearis is a monogamous species and has one or two generation per year (Mendel et al. 1985; Pfeffer 1995). Its phenology and flight activity has not been studied in the study area. This species breeds in dead roots and stumps of Pinus species (Wood and Bright 1992), and in the study area, it has been recorded breeding on P. radiata (Tables S1 and S2).
Cyrtogenius luteus (Blandford, 1894)
Cyrtogenius luteus was recorded in the study area for the first time in southern Brazil (states of Paraná, Río Grande do Sul, and São Paulo) in 2006 (Flechtman and Atkinson 2018); 3 years later, it was detected in Uruguay (Gómez et al. 2012) and later in north-east Argentina (provinces of Entre Ríos, Corrientes, and Misiones). Here we present the first records for this species for the provinces of Entre Rios and Misiones in Argentina (Table 1, Fig. 2). Currently, the species ranges from latitude 23 to 34°S (Figs. 2 and 3, Table 1). The maximum temperatures of the locations where C. luteus was found range from 26 to 32 °C and the minimum temperature from 5 to 11 °C (Fig. 4). This species is native to parts of south-east Asia and has also invaded Italy in 2009 (Wood and Bright 1992; Beaver and Liu 2010; Faccoli et al. 2012).
Cyrtogenius luteus is a polygynous species and has at least two generations per year. The flight activity in Uruguay and Brazil occurs mostly during early summer (Gómez et al. 2017; Flechtman and Atkinson 2018), and the species abundance peaks during periods of high precipitation, while it shows no response to temperature (Gómez et al. 2020). The species usually infests stressed or dying Pinus species but is also recorded in other Pinaceae (Larix and Picea) (Wood and Bright 1992). In the study area, it has been observed feeding on P. elliottii, P. pinaster, P. taeda, and possibly Araucaria angustifolia (Gómez et al. 2012; Flechtman and Atkinson 2018) (Tables S1 and S2).
Orthotomicus erosus (Wollaston, 1857)
Orthotomicus erosus is the most recently detected species in the study area. It was registered for the first time in Uruguay, in 2009 (Gómez and Martínez 2013), and 4 years later in the province of Corrientes, northeast Argentina (Klasmer et al. 2014). Here, we report the first records for the provinces of Entre Ríos and Córdoba, in northeast and center of Argentina, respectively (Figs. 2 and 3, Table 1). Currently, the distribution of O. erosus in South America ranges from latitude 28 to 35°S, the maximum temperatures where this species was registered ranged from 26 to 32 °C and the minimum temperatures ranged from 0 to 9 °C (Fig. 4). This species is native to the Mediterranean region and southern Europe, Asia, and North Africa, and has also invaded other pine growing regions of the world: South Africa in 1968, Eswatini in 1983, Fiji in 1984 and the USA (California) in 2004 (Beaver 1987; Wood and Bright 1992; Lee et al. 2005). More recently, it was reported from additional states in the USA, including Arizona and Nevada (USDA FS 2019; G Phillips et al. 2020).
Orthotomicus erosus is a polygamous species and can have up to four generations per year (Pfeffer 1995; Lieutier et al. 2016). In Uruguay, its flight activity occurs during late winter, spring, and summer (Gómez et al. 2017). This species infests main branches and trunks of Pinus species. Maturing beetles can occasionally also feed in Abies, Cedrus, Picea, and Pseudotsuga, but it does not breed in trees other than pines (Lieutier et al. 2016; CABI 2021b). In the study area, it was observed feeding on P. caribea, P. elliottii, and P. taeda (Gómez and Martínez 2013; Klasmer et al. 2014) (Tables S1 and S2).
Discussion
Pine bark beetle invasion success in southern South America
Here we present detailed information on the distribution and invasion history of the six pine bark beetle species established across the largest pine plantation surface areas in South America. These six species have proven to be successful invaders, with a great capacity to spread throughout pine growing regions. The establishment of non-native populations across the study area began in the 1950s. From the 1980s onwards, there has been an exponential increase in introductions, and it is expected that the arrival of exotic species will continue to rise (Kirkendall 2018; Bonnamour et al. 2023).
The success of these species in establishing in South America, as well as in other regions of the world—i.e., all species, except O. laricis, have established populations outside of their native range in other parts of the world—can be attributed, on the one hand, to their propagule pressure. They are strongly associated to certain pathways facilitating their non-native long-distance movement, such as the global transport of timber and wood packaging material (Meurisse et al. 2019). All the studied species have been intercepted in wood packaging material in some parts of the world. Ortothomicus erosus and H. ligniperda have been found to be among the most frequently transported bark beetles around the world (Haack 2001; Brockerhoff et al. 2014; Wang et al. 2018); O. laricis and H. ater have also been frequently intercepted, while H. linearis and C. luteus show much lower interception frequencies (Haack 2001; Brockerhoff et al. 2014).
On the other hand, factors associated to the invasibility of the new region (i.e., southern South American pine plantations), such as the climatic similarities among their native and invaded geographic regions and host availability in the location of arrival may have determined their success in establishing in South America (Vilardo et al. 2022). In this regard, the expansion of large-scale pine plantations during the second half of the twentieth century in regions of South America with climatically similar conditions to areas of the Northern Hemisphere where these species are native to, has significantly increased their opportunities for invasion (Lantschner et al. 2017; Corley et al. 2020; Vilardo et al. 2022).
An interesting finding of our study is that, although the native pool of pine bark beetles from North America is similar to the one of Eurasia (Wood and Bright 1992), the six pine bark beetle species established in South America are native to Europe and/or Asia. This pattern of “invasion asymmetry,” where the Palearctic region is overrepresented and the Nearctic region under-represented as donors of non-native established species, has been commonly observed in forest insects (Mattson et al. 2007; Lantschner et al. 2020; Isitt et al. 2023). There is no clear explanation for this pattern in our case since the Nearctic and Palearctic regions have been historically strongly connected with the Neotropical region via trade (Lantschner et al. 2020), both regions have considerable areas with similar climatic conditions to those where pines are grown in South America (Lantschner et al. 2017; Vilardo et al. 2022), and additionally, most of the planted pine species in South America are native from North America (Lantschner et al. 2017). The invasion asymmetry could be related to various factors, including differences in biosecurity polices of each donor region, differences in the abilities of establishing (i.e., ability to survive underpopulation or Allee effects) between Nearctic and Palearctic bark beetle species, because of differences in the evolutionary pressures between regions (Mattson et al. 2007), or because of the small size of our sample. Further work is required to better understand these patterns.
It should also be noted, that although we know where the species are native from, it is unclear which was the route of invasion of the bark beetle species and whether there was a single or multiple introduction events for each species in South America. In this sense, invasions of new regions might originate from previously invaded regions—i.e., “bridgehead effect” (Bertelsmeier and Keller 2018). To better understand the invasion history and pathways of pine bark beetles in South America, more detailed studies are needed, analyzing, for example, interception history of the species in international wood packaging material and reconstructing the invasion histories of established populations based on genetic analysis (Lin et al. 2021; Turner et al. 2021).
Geographic patterns of pine bark beetle establishments and future predictions
We found that in southern South America, two major regions can be identified, both based on the distribution of pine trees and the composition of bark beetle species, except for the case of H. ligniperda, which is widely distributed throughout the study area. The southwest region comprises pine plantations in Chile and the Argentine Patagonia, and presents three unique bark beetle species: O. laricis, H. ater, and H. linearis (the last one only in Chile). On the other hand, the northeastern zone includes northeastern Argentina, Uruguay, and southern Brazil, where two other bark beetle species are found: C. luteus and O. erosus (the last species only in Argentina and Uruguay). This distribution pattern is likely the result of a combination of (1) the geographical distribution pattern of pine plantations, (2) the history of bark beetle species introduction and spread, and (3) environmental differences between regions.
The geographical distribution of pine plantations shows the existence of two large areas where most pine plantations of South America are concentrated, with 2.4 million ha in the northeastern region, and 2 million ha in the southwestern region. Although the patches of pine plantations inside of each of these regions are not continuous, they are close enough to each other to likely act as a continuous area for the spread of pine bark beetles. This situation underscores the significance of prioritizing the surveillance and management of invasive species at a regional level, rather than individual countries addressing the issue independently (Wingfield et al. 2015; Villacide et al. 2023).
The history of species introduction and spread across each region, of course, also determines current distribution of species. The occurrence of a species in one region and not in others can be a product of where it was first introduced and since how long. In this sense, the only species that has shown the ability to colonize all regions is Hylurgus ligniperda. This species established in South America several decades before the other species, and hence has had more time to spread across regions and countries. Given the current distribution of the studied species, it is expected that some of them will continue to spread across and between regions. For example, it could be expected that H. linearis, which has experienced significant expansion in Chile over the last decade, will also spread to neighboring Argentina. Additionally, in the northeast region, recently detected species such as C. luteus and O. erosus could continue to spread throughout Argentina and southern Brazil. Interestingly, pine plantations located between these two regions in central Argentina (province of Córdoba) show a mix of the species established in each region (H. ligniperda, H. ater, and O. erosus), which suggests that species might be spreading between regions using this area as a steppingstone.
Finally, the differences between the environmental characteristics of each region are probably also determining where each species can establish and spread. Climatic conditions, and particularly temperature, play a major role in the survival and development of bark beetles, and consequently in their abilities to establish (Lieutier et al. 2016; Bentz et al. 2022; Pureswaran et al. 2022). Moreover, because bark beetle species studied here have a different native range, ranging from tropical to temperate, we can expect to observe differences in their responses to the effect of temperature and in their emergence, as well as their seasonality (Gómez et al. 2020). The climate of the southwest region is predominantly temperate, while in the northeast region it is humid subtropical. Consequently, the species that establish in pine plantations of the southwestern region are exposed to lower minimum temperatures in winter (Fig. 4), and this could limit the survival and establishment of other species. Summer temperatures, on the other hand, can reach very high values, particularly in the northern regions, affecting eggs and larvae survival of some species. According to Vilardo et al. (2022), who studied the potential distribution of European bark beetles in pine plantations of the southern hemisphere based on bioclimatic conditions, Orthotomicus erosus and Hylastes linearis have the potential to establish in both major regions, consequently O. erosus might be able to establish in Chile and southern Argentina if introduced there, while H. linearis could be able to establish in north-east Argentina, Uruguay and southern Brazil. Orthotomicus laricis, on the other hand, is only predicted establish in the southwestern region, where it is already present, while Hylastes ater could find suitable climatic conditions in a very restricted areas of the northeastern region (Vilardo et al. 2022). On the other hand, C. luteus, as a tropical insect, depends primarily on rainfall volume to reach its optimal development conditions, and this kind of conditions is mainly found across the north-eastern region of our study area (NW Argentina, Uruguay, and S Brazil), which presents a subtropical climate (Gómez et al. 2020). Furthermore, other environmental variables such as the availability of suitable hosts may also influence the invasion pattern of pine bark beetles in South America. In this regard, it is worth noting that different species are cultivated in each region (Fig. 1), and both the type and intensity of silvicultural management, as well as the length of the rotation, can vary considerably. While the recorded species may seem to exhibit low specificity among planted pine species, regions with a higher abundance of pines and increased logging and thinning activities likely provide greater opportunities for species establishment. However, these characteristics do not appear to be significant in determining which species established where, as it would be expected that all species are similarly influenced by the same factors.
It should be noted that not all areas were studied with similar sampling efforts. There are areas with large surfaces of pine plantations, such as southern Brazil, for which very little information exists. It is possible that there are species present in some areas that have not yet been detected. We also lack information for countries like Bolivia and Paraguay, although it must be noted surface areas of pine plantations is scarce. It is also important to consider that there is a delay between when a species arrives in a new area and when it is detected, and this delay is likely to vary greatly, depending among other things, on sampling efforts. Therefore, the invasion history of each species in each country should be interpreted with caution, and it should be considered that several of the species may have been present in different areas for several years before being detected.
Finally, we conclude that Dryocoetes autographus has probably not established in the study area. Although we were not able to sample in the area where it was originally described (state of São Paulo), we did not find any information of other authors confirming its establishment in Brazil. Additionally, this species is specialized in feeding on spruce trees (Picea spp.), which are very uncommon in South America. Therefore, we assume that its report was probably a misidentification or the interception of a specimen from imported goods, rather than originating from an established population. This species presents a very wide distribution across the Holarctic, being native to Europe, Asia, North Africa, and North America, and has not been described to establish in other regions of the Southern Hemisphere (Wood and Bright 1992).
Economic impact of the established pine bark beetles
Understanding the distribution and invasion history of non-native pine bark beetles in southern South America is critical to predict their future spread and potential economic impact on pine plantations. The six bark beetle species described in this study are common colonizers of highly stressed and recently dead pine species, and consequently, they usually do not kill trees and are considered a secondary pest. However, under certain conditions, some of these species can kill large number of trees. In South America, outbreaks of C. luteus, H. ligniperda, and O. erosus have been documented in Uruguay in 2009 and in the province of Corrientes, Argentina in 2022. In both cases, the infestations resulted in significant mortality rates within Pinus taeda and P. elliottii plantations that were already experiencing severe drought conditions (Gómez and Martínez 2013; Escobar 2022). Among these bark beetle species, O. erosus has been observed to periodically reach epidemic levels in other parts of the world and to cause the death of living trees under stress caused by severe drought or the attack of other insects (Bevan 1984; Ferreira 1986; Tribe 1990). It should be noted that the most important factor of population increase of bark beetles is the quantity of suitable breeding material. Consequently, abiotic or biotic factors such as storms, severe droughts, extensive thinning operations, and fire, that provide large quantities of highly suitable breeding material, can trigger outbreaks of these species (Lieutier et al. 2016; Lantschner and Corley 2023).
Another cause of damage by some of the pine bark beetles is the decay or death of young pine stands and in nursery stock due to bark and crown damage resulted from the maturation feeding carried out by immature adults after they emerge. This kind of damage has been recorded occasionally by H. ligniperda and H. ater in Chile and can cause up to 5 to 10% of mortality of P. radiata seedlings in some stands (Ahumada, personal observation). Similar damage was also reported by these species in other parts of the world (Neumann 1979; Reay et al. 2002). Additionally, the pine bark beetle species registered in this study, as many other bark beetles, are vectors of ophiostomatoid fungi, that enter with the adults into the galleries (Wingfield et al. 2017). These fungi can favor the development of bark beetles in two ways: first, by increasing the nutritional value of the substrate (fungus-infected phloem enhances their nitrogen intake), and second, by altering the condition of trees (Lieutier et al. 2009; Six and Wingfield 2010; Kirkendall et al. 2015). Moreover, the dispersal of blue stain fungi by pine bark beetle species is very common in pine hosts, affecting the wood quality of economically valuable species (Lanfranco et al. 2004; de-Errasti et al. 2018).
Biosecurity implications
The pine bark beetle species established in southern South America are shared among the countries of the region, and in most cases also with other regions of the Southern Hemisphere in which pines are also planted. This situation reflects the high spread and dispersal of these forest insects, and it is evident that pine plantations in many regions provide a continuous habitat with permeable borders. In this sense, international collaboration to understand their distribution and spread of pine bark beetles to apply continent-wide management strategies is needed (see Villacide et al. 2023 for a recent initiative).
Moreover, as these bark beetles are attracted to freshly cut pine woody materials, they can become associated with exported wood commodities, posing a phytosanitary risk for the importing country. Consequently, several of these species are subject to quarantine concerns for wood exports to certain countries (Lanfranco et al. 2004; Mausel et al. 2007; Ruiz and Lanfranco 2008; Clarke and Hartshorn 2021). The spread of certain non-native bark beetle species to new regions, as well as the potential introduction of new species, could increase the cost of exports due to the necessity of log and timber phytosanitary treatments described in ISPM 15—i.e., debarking of logs, methyl bromide fumigation, pesticide treatment (Pawson et al. 2019; Meurisse et al. 2021).
In southern South America, the requirements outlined in ISPM 15 have been incorporated into the conditions for importing solid wood packaging materials between 2004 and 2006 (2004 in Brazil; 2005 in Argentina, Bolivia, Chile, Paraguay; and 2006 in Uruguay) (COSAVE 2016). Despite the expected reduced risk after the implementation of ISPM 15 in the region, several new introductions of bark beetles were registered. These introductions may still occur due to inadequate application of treatment, non-compliance, or pest survival (Greenwood et al. 2023), as well as because a delay between the introduction and first detection of new species.
As populations of non-native insects establish and expand into new areas, its management becomes progressively more challenging and costly (Liebhold and Tobin 2008). Therefore, detecting a non-native species at the early stage of its invasion is critical to promptly initiate mitigation measures and implement responses to prevent its permanent establishment and spread, thus reducing costs and minimizing damage (Epanchin-Niell and Liebhold 2015; Poland and Rassati 2019). Consequently, we highlight the importance of conducting biosecurity surveillance on pine bark beetles to early detect arriving individuals or newly established populations, and to further understand the pathways driving the introduction of forest insect species in South America (Faccoli et al. 2020).
Gaining insight into the factors driving invasion success is crucial for developing predictive tools that can identify species with a higher likelihood of becoming invaders and geographic regions more susceptible to invasions. By doing so, we can minimize the ecological and economic impacts associated with invasions and enhance the management of invasive insect populations.
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Álvarez González V, Poblete Hernández P, Soto Aguirre D et al (2022) ANUARIO FORESTAL 2022. In: Instituto Forestal, Boletín Estadístico N° 187, Santiago. https://doi.org/10.52904/20.500.12220/32501
Bain J (1977) Hylurgus ligniperda (Fabricius) (Coleoptera: Scolytidae). In: Forest and timber insects in New Zealand. New Zealand Forest Research Institute, Rotorua, pp 1–5
Beaver R (1987) Bark and ambrosia beetles (Coleoptera: Scolytidae) newly recorded from Fiji, and their potential economic importance. S Pac J Nat Sci 9:1–7
Beaver RA, Liu LY (2010) An annotated synopsis of Taiwanese bark and ambrosia beetles, with new synonymy, new combinations and new records (Coleoptera: Curculionidae: Scolytinae). Zootaxa 2602:1–47
Bentz BJ, Hansen EM, Davenport M, Soderberg D (2022) Chapter 2 - Complexities in predicting mountain pine beetle and spruce beetle response to climate change. In: KJK G, Hofstetter RW (eds) Bark Beetle management, ecology, and climate change. Academic Press, pp 31–54. https://doi.org/10.1016/B978-0-12-822145-7.00013-1
Bertelsmeier C, Keller L (2018) Bridgehead effects and role of adaptive evolution in invasive populations. Trends Ecol Evol 33:527–534. https://doi.org/10.1016/j.tree.2018.04.014
Bevan D (1984) Orthotomicus erosus (Wollaston) in Usutu pine plantations, Swaziland. For Res Rep Usutu Pulp Company Limited 64:1–34
Biedermann PHW, Müller J, Grégoire J-C et al (2019) Bark beetle population dynamics in the anthropocene: challenges and solutions. Trends Ecol Evol 34:914–924. https://doi.org/10.1016/j.tree.2019.06.002
Bonnamour A, Blake RE, Liebhold AM et al (2023) Historical plant introductions predict current insect invasions. Proc Natl Acad Sci 120:e2221826120. https://doi.org/10.1073/pnas.2221826120
Boomsma CD, Adams AJS (1943) The pine bark beetle (Hylastes ater) at Mount Burr, South Australia. Aust for 7:33–37. https://doi.org/10.1080/00049158.1943.10675211
BRACELPA (2004) Relatorio estadistico florestal 2004. In: Associação Brasileira de Celulose e Papel, Sao Paulo, Brazil, p 54. http://www.bibliotecaflorestal.ufv.br/handle/123456789/7740
Brockerhoff EG, Liebhold AM (2017) Ecology of forest insect invasions. Biol Invasions 19:3141–3159. https://doi.org/10.1007/s10530-017-1514-1
Brockerhoff EG, Kimberley M, Liebhold AM et al (2014) Predicting how altering propagule pressure changes establishment rates of biological invaders across species pools. Ecology 95:594–601. https://doi.org/10.1890/13-0465.1
CABI (2021a) Hylurgus ligniperda (red-haired pine bark beetle). CABI Compendium. https://doi.org/10.1079/pwkb.species.27364. Accessed 11 Jul 2023
CABI (2021b) Orthotomicus erosus (Mediterranean pine beetle). CABI Compendium. https://doi.org/10.1079/pwkb.species.37954 Accessed 11 Jul 2023
Chararas C (1962) Scolytides des Conifères. Lechevalier, Paris
Clark AF (1932) The pine bark beetle, Hylastes ater in New Zealand. N Z J Sci Technol 14:1–1
Clarke SR, Hartshorn J (2021) Contrasting competitor and predator responses to a short-lived southern pine beetle outbreak: a case study. For Sci 67:22–29. https://doi.org/10.1093/forsci/fxaa041
Córdoba SP, Mendoza EA, Atkinson TH, Manzo MV (2021) Diversidad y dinámica poblacional de Scolytinae (Coleoptera: Curculionidae) en plantaciones de palto y bosque natural circundante en el NW de Argentina. Dugesiana 28:115–130
Corley JC, Villacide JM, Lanstchner MV (2020) Chapter 8 - Invasive insects in forest plantations of Argentina: ecological patterns and implications for management. In: Estay S (ed) Forest pest and disease management in Latin America. Springer International Publishing, pp 107–122. https://doi.org/10.1007/978-3-030-35143-4_8
COSAVE (2016) Perspectiva general de la implementación de los reglamentos internacionales para el embalaje de madera para exportación e importación. In: Workshop on the Implementation of ISPM 15 with emphasis in the Americas. August 29 to September 2, 2016. IICA, Costa Rica
de-Errasti A, Pildain MB, Rajchenberg M (2018) Ophiostomatoid fungi isolated from three different pine species in Argentinian Patagonia. For Pathol 48:e12393. https://doi.org/10.1111/efp.12393
Epanchin-Niell RS, Liebhold AM (2015) Benefits of invasion prevention: effect of time lags, spread rates, and damage persistence. Ecol Econ 116:146–153. https://doi.org/10.1016/j.ecolecon.2015.04.014
Escobar P (2022) Sequía, plagas e incendios afectan el crecimiento de las plantaciones, generan pérdidas y desalientan la inversión forestal. Argentina Forestal https://www.argentinaforestal.com/2022/12/09/la-sequia-afecta-el-crecimiento-de-las-plantaciones-en-corrientes/. Accessed 11 Jul 2023
Estay SA (2020) Insect pests affecting exotic trees in Chile and their management. In: Estay SA (ed) Forest pest and disease management in Latin America: modern perspectives in natural forests and exotic plantations. Springer International Publishing, Cham, pp 185–195
Faccoli M, Simonato M, Petrucco Toffolo E (2012) First record of Cyrtogenius Strohmeyer in Europe, with a key to the European genera of the tribe Dryocoetini (Coleoptera: Curculionidae, Scolytinae). Zootaxa 3423:27–35
Faccoli M, Gallego D, Branco M et al (2020) A first worldwide multispecies survey of invasive Mediterranean pine bark beetles (Coleoptera: Curculionidae, Scolytinae). Biol Invasions 22:1785–1799. https://doi.org/10.1007/s10530-020-02219-3
FAO (2020) Global forest resources assessment 2020: Main report. Food & Agriculture Organization of the UN, Rome. https://doi.org/10.4060/ca9825en
Ferreira MCFG (1986) Pests of maritime pine in Portugal - scolytids. Bol Agric 36:1–4
Fick SE, Hijmans RJ (2017) WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. Int J Climatol 37:4302–4315. https://doi.org/10.1002/joc.5086
Flechtman CAH, Atkinson TH (2018) Oldest record of Cyrtogenius luteus (Blandford) (Coleoptera: Curculionidae: Scolytinae) from South America with notes on its distribution in Brazil. Insecta Mundi 0645:1–4
Freer-Smith PH, Webber JF (2017) Tree pests and diseases: the threat to biodiversity and the delivery of ecosystem services. Biodivers Conserv 26:3167–3181. https://doi.org/10.1007/s10531-015-1019-0
Geertsema H (1982) A historical review of forest entomology in South Africa. Saasveld 50:199–207
Gómez D, Martínez G (2013) Bark beetles in pine tree plantations in Uruguay: first record of Orthotomicus erosus Wollaston (Coleoptera: Curculionidae: Scolytinae). Coleopt Bull 67:470–472. https://doi.org/10.1649/0010-065X-67.4.470
Gómez D, Martinez G, Beaver RA (2012) First record of Cyrtogenius luteus (Blandford) (Coleoptera: Curculionidae: Scolytinae) in the Americas and its distribution in Uruguay. Coleopt Bull 66:362–364. https://doi.org/10.1649/072.066.0414
Gómez D, Hirigoyen A, Balmelli G et al (2017) Patterns in flight phenologies of bark beetles (Coleoptera: Scolytinae) in commercial pine tree plantations in Uruguay. Bosque 38:47–53
Gómez DF, Skelton J, De María M, Hulcr J (2020) Influence of temperature and precipitation anomaly on the seasonal emergence of invasive bark beetles in subtropical South America. Neotrop Entomol 49:347–352. https://doi.org/10.1007/s13744-019-00760-y
Gómez D (2014) Diversidad y Estacionalidad de Escarabajos de Corteza (Coleoptera: Scolytinae) en Pinus taeda L. en Uruguay. Master of Science Thesis, Universidad de la Republica, Montevideo
Gómez DF, Riggins JJ, Cognato AI (2023) Chapter 10 - Bark beetles. In: Allison DJ, Paine TD, Slippers B, Wingfield MJ (eds) Forest entomology and pathology. Springer International Publishing, Cham, pp 299–337. https://doi.org/10.1007/978-3-031-11553-0_10
Greenwood LF, Coyle DR, Guerrero ME et al (2023) Exploring pest mitigation research and management associated with the global wood packaging supply chain: What and where are the weak links? Biol Invasions 25:2395–2421. https://doi.org/10.1007/s10530-023-03058-8
Grégoire J-C, Jactel H, Hulcr J et al (2023) Cosmopolitan Scolytinae: strong common drivers, but too many singularities for accurate prediction. NeoBiota 84:81–105. https://doi.org/10.3897/neobiota.84.89826
Haack RA (2001) Intercepted Scolytidae (Coleoptera) at U.S. ports of Entry: 1985–2000. Integr Pest Manag Rev 6:253–282. https://doi.org/10.1023/A:1025715200538
Hoebeke ER (2001) Hylurgus ligniperda: a new exotic pine bark beetle in the United States. Newsl Mich Entomol Soc 46:1–2
Isitt R, Liebhold AM, Turner RM et al (2023) Drivers of asymmetrical insect invasions between three world regions. bioRxiv. https://doi.org/10.1101/2023.01.13.523858
Kenis M, Roques A, Santini A, Liebhold AM (2017) Impact of non-native invertebrates and pathogens on market forest tree resources. In: Vilà M, Hulme PE (eds) Impact of biological invasions on ecosystem services. Springer International Publishing, Cham, pp 103–117
Kirkendall LR (2018) Invasive bark beetles (Coleoptera, Curculionidae, Scolytinae) in Chile and Argentina, including two species new for South America, and the correct identity of the Orthotomicus species in Chile and Argentina. Diversity 10(2):40. https://doi.org/10.3390/d10020040
Kirkendall LR, Biedermann PHW, Jordal BH (2015) Chapter 3 - Evolution and diversity of bark and ambrosia beetles. Bark Beetles: Biol Ecol Native and Invasive Species 85–156
Klasmer P, Ramos S, Kirkendall L, Diez de Ulzurrun I (2014) Coleopteros escolitidos de importancia económica para plantaciones forestales: Orthotomicus erosus (Coleoptera: Scolytidae), primer registro para Argentina y Pissodes castaeus (Coleoptera: Curculionidae). II Jornadas Argentinas de Sanidad Forestal, September 24 - 26, Montecarlo, Misiones
Krishnankutty S, Nadel H, Taylor AM et al (2020) Identification of tree genera used in the construction of solid wood-packaging materials that arrived at U.S. ports infested with live wood-boring insects. J Econ Entomol 113:1183–1194. https://doi.org/10.1093/jee/toaa060
Lanfranco D, Ide S, Peredo H (2004) An analysis of health risk reduction in Chilean primary forest products for export. For: Int J For Res 77:193–203. https://doi.org/10.1093/forestry/77.3.193
Lanfranco D, Ide S, Ruiz C, Peredo H, Vives I (2002) Agentes entomopatógenos asociados a productos forestales primarios de exportación. Ediciones el Kultrún, Valdivia, p 87
Lantschner MV, Corley JC, Liebhold AM (2020) Drivers of global Scolytinae invasion patterns. Ecol Appl 30:eap.2103. https://doi.org/10.1002/eap.2103
Lantschner MV, Atkinson TH, Corley JC, Liebhold AM (2017) Predicting North American Scolytinae invasions in the Southern Hemisphere. Ecol Appl 27:66–77. https://doi.org/10.1002/eap.1451
Lantschner MV, Corley JC, Liebhold AM (2020) Drivers of global Scolytinae invasion patterns. Ecol Appl 30:eap.2103. https://doi.org/10.1002/eap.2103
Lee JC, Smith SL, Seybold SJ (2005) The Mediterranean pine engraver, pest alert R5-PR-016. U.S, Department of Agriculture, Forest Service, Pacific Southwest Research Station, Berkeley, CA
Liebhold AM, Tobin PC (2008) Population ecology of insect invasions and their management. Annu Rev Entomol 53:387–408
Lieutier F, Yart A, Salle A (2009) Stimulation of tree defenses by Ophiostomatoid fungi can explain attack success of bark beetles on conifers. Ann for Sci 66:801. https://doi.org/10.1051/forest/2009066
Lieutier F, Mendel Z, Faccoli M (2016) Bark beetles of Mediterranean conifers. In: Paine TD, Lieutier F (eds) Insects and diseases of Mediterranean forest systems. Springer International Publishing, Cham, pp 105–197
Lin W, Park S, Jiang Z-R et al (2021) Native or invasive? The red-haired pine bark beetle Hylurgus ligniperda (Fabricius) (Curculionidae: Scolytinae) in East Asia. Forests 12:950. https://doi.org/10.3390/f12070950
López J (2004) Estudio de Tendencias y Perspectivas del Sector Forestal en América Latina Documento de Trabajo: Informe Nacional Bolivia. Ministerio de Asuntos Campesinos y Agropecuarios, FAO, Rome
MAyG (2009) Censo Agropecuario Nacional 2008. In: Ministerio de Agricultura y Ganadería, Dirección de Censos y Estadísticas Agropecuarias. Republica del Paraguay, San Lorenzo
MAGyP (2011) Mapa de Plantaciones Forestales de la República Argentina. Ministrerio de Agricultura, Ganadería y Pesca, Buenos Aires, Argentina
Mattson W, Vanhanen H, Veteli T et al (2007) Few immigrant phytophagous insects on woody plants in Europe: legacy of the European crucible? Biol Invasions 9:957–974. https://doi.org/10.1007/s10530-007-9096-y
Mausel DL, Gara RI, Lanfranco D et al (2007) The introduced bark beetles Hylurgus ligniperda and Hylastes ater (Coleoptera: Scolytidae) in Chile: seasonal flight and effect of Pinus radiata log placement on colonization. Can J for Res 37:156–169. https://doi.org/10.1139/x06-215
Mendel Z, Madar Z, Golan Y (1985) Comparison of the seasonal occurrence and behavior of seven pine bark beetles (Coleoptera: scolytidae) in Israel. Phytoparasitica 13:21–32. https://doi.org/10.1007/BF02994434
Meurisse N, Rassati D, Hurley BP et al (2019) Common pathways by which non-native forest insects move internationally and domestically. J Pest Sci (2004) 92:13–27. https://doi.org/10.1007/s10340-018-0990-0
Meurisse N, Pawson SM, Somchit C (2021) Bark beetles on pine logs: forecasting winter colonisation dynamics based on trap catches and temperature records. J Pest Sci (2004) 94:1357–1373. https://doi.org/10.1007/s10340-021-01361-8
Neumann FG (1979) Beetle communities in eucalypt and pine forests in north-eastern Victoria. Aust For Res 9:277–293
Park S, Jung JC, Han T (2017) A new species and five newly recorded species of Scolytinae (Coleoptera: Curculionidae) from Korea. Entomol Res Bull 33:131–137
Pawson SM, Bader MK-F, Brockerhoff EG et al (2019) Quantifying the thermal tolerance of wood borers and bark beetles for the development of Joule heating as a novel phytosanitary treatment of pine logs. J Pest Sci (2004) 92:157–171. https://doi.org/10.1007/s10340-018-1015-8
Pfeffer A (1995) Zentral-und westpaläarktische Borken-und Kernkäfer:(Coloptera: Scolytidae, Platypodidae). Entomologica Basiliensia 17:5–310
Phillips G, Knight J, Williams J, Taveras A (2020) 2019 Forest pest conditions in Nevada. Nevada Division of Forestry, Department of Conservation and Natural Resources, Nevada https://forestry.nv.gov/uploads/missions/20190101_AMT_2019_Nevada_Forest_Pest_Conditions_Report.pdf
Poland TM, Rassati D (2019) Improved biosecurity surveillance of non-native forest insects: a review of current methods. J Pest Sci (2004) 92:37–49. https://doi.org/10.1007/s10340-018-1004-y
Procheş Ş, Wilson JRU, Richardson DM, Rejmánek M (2012) Native and naturalized range size in Pinus: relative importance of biogeography, introduction effort and species traits. Glob Ecol Biogeogr 21:513–523. https://doi.org/10.1111/j.1466-8238.2011.00703.x
Pureswaran DS, Meurisse N, Rassati D et al (2022) Chapter 1 - Climate change and invasions by nonnative bark and ambrosia beetles. In: Gandhi KJK, Hofstetter RW (eds) Bark beetle management, ecology, and climate change. Academic Press, pp 3–30. https://doi.org/10.1016/B978-0-12-822145-7.00002-7
QGIS.org (2023) QGIS Geographic Information System. QGIS Association. http://qgis.org
Raffa KF, Aukema BH, Bentz BJ et al (2008) Cross-scale drivers of natural disturbances prone to anthropogenic amplification: the dynamics of bark beetle eruptions. Bioscience 58:501–517. https://doi.org/10.1641/B580607
Reay SD, Walsh PJ, Ram A, Farrell RL (2002) The invasion of Pinus radiata seedlings by sapstain fungi, following attack by the Black Pine Bark Beetle, Hylastes ater (Coleoptera: Scolytidae). For Ecol Manage 165:47–56. https://doi.org/10.1016/S0378-1127(01)00628-4
Ruiz C, Lanfranco D (2008) Los escarabajos de corteza en Chile: una revisión de la situación actual e implicancias en el comercio internacional. Bosque 29:109–114
SAG (2010) Informativo Fitosanitario Forestal No 4. Santiago, Chile
SAG (2022) Sistema de Información de Sanidad Vegetal. Santiago, Chile
Schedl KE (1957) Bark- and timber-beetles from South Africa. Ann Mag Nat Hist 10:149–159. https://doi.org/10.1080/00222935708655940
Seebens H, Blackburn TM, Dyer EE et al (2017) No saturation in the accumulation of alien species worldwide. Nat Commun 8:14435
Six DL, Wingfield MJ (2010) The role of phytopathogenicity in bark beetle–fungus symbioses: a challenge to the classic paradigm. Annu Rev Entomol 56:255–272. https://doi.org/10.1146/annurev-ento-120709-144839
Tiranti SI (2010) Observations on pine scolytids from Andean Patagonia, with the first record of the genus Orthotomicus for Argentina. Bol Sanid Veg Plagas 36:87–90
Tribe GD (1990) Phenology of Pinus radiata log colonization and reproduction by the European bark beetle Orthotomicus erosus (Wollaston) (Coleoptera: Scolytidae) in the south-western Cape Province. J Entomol Soc South Afr 53:117–126
Turner RM, Brockerhoff EG, Bertelsmeier C et al (2021) Worldwide border interceptions provide a window into human-mediated global insect movement. Ecol Appl 31:e02412. https://doi.org/10.1002/eap.2412
USDA FS (2019) Mediterranean Pine Engraver Found In Urban Pines. USDA Forest Service, Arizona Forest Health Alert
Vilardo G, Faccoli M, Corley JC, Lantschner MV (2022) Factors driving historic intercontinental invasions of European pine bark beetles. Biol Invasions 24:2973–2991. https://doi.org/10.1007/s10530-022-02818-2
Villacide, Gómez, Perez CA, Corley JC et al (2023) Forest health in the Southern Cone of America: state of the art and perspectives on regional efforts. Forests 14:756. https://doi.org/10.3390/f14040756
Wang K, Xu ML, Liu PH et al (2018) Interception situation and suggestion of Scolytidae at China ports. Plant Quarentine 32:65–68
Wingfield MJ, Brockerhoff EG, Wingfield BD, Slippers B (2015) Planted forest health: the need for a global strategy. Science (1979) 349:832–836
Wingfield MJ, Barnes I, de Beer ZW et al (2017) Novel associations between ophiostomatoid fungi, insects and tree hosts: current status—future prospects. Biol Invasions 19:3215–3228. https://doi.org/10.1007/s10530-017-1468-3
Wood SL (2007) Bark and ambrosia beetles of South America (Coleoptera: Scolytidae). Provo, Bean Life Science Museum
Wood SL, Bright DE (1992) A catalog of Scolytidae and Platypodidae (Coleoptera), Part 2. Great Basin Nat Mem 13:1–835
Funding
This work was supported by a grant from Agencia Nacional de Promoción Científica y Tecnológica of Argentina (PICT 2019–235) and CONICET (PIP 11220200100764CO). SAE thanks to ANID PIA/BASAL FB0002. Insect sampling in Uruguay was supported by project INIA Bosque30.
Author information
Authors and Affiliations
Contributions
Victoria Lantschner and Juan Corley conceptualized and designed the study. All authors actively contributed to data collection. Victoria Lantschner led the analysis and interpretation of the results and the drafting of the article. All authors collaborated in reviewing and approving the final version for publication.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Edited by Pedro HB Togni
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lantschner, V., Gomez, D.F., Vilardo, G. et al. Distribution, Invasion History, and Ecology of Non-native Pine Bark Beetles (Coleoptera: Curculionidae: Scolytinae) in Southern South America. Neotrop Entomol 53, 351–363 (2024). https://doi.org/10.1007/s13744-023-01125-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-023-01125-2