Abstract
Achyranthes aspera L. (Prickly Chaff flower) is an imperative Asian herb that has been known for numerous healing powers. The aim of this research is to investigate the pharmacological i.e., antidepressant and anxiolytic properties of the methanol extract of aerial part of A. aspera (MEAA) by in vivo and in silico approaches. Additionally, thrombolytic potential of the crude extract has also been explored. The antidepressant and anxiolytic investigations were performed by force swimming test (FST), tail suspension test (TST), and hole board test (HBT) in mice, whereas clot lysis activity test was conducted using thrombolytic experimental method. Furthermore, in silico studies were appraised with Discovery studio, PyRx, UCSF Chimera whereas, PASS prediction, and ADME/T was implemented using online tools. Treatment with MEAA revealed significant (p < 0.05) reduction of immobility time in FST and TST, increased head dipping and decreased the latency of time in HBT. Besides, MEAA produced significant (p < 0.001) clot lysis activity at a minimum dose. In the computer-aided approach, among all the proteins, the docking score has been revealed ranging from − 2.31 kcal/mol to − 7.4 kcal/mol. Spathulenol, diazepam and hydroquinone showed the best binding affinity and all compounds were found safe in ADME/T study. The outcomes of this research demonstrate that MEAA may provide potential for mitigating depression, anxiety, and coagulation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human neurological disorders in the public health sector is growing fear and significantly affecting the normal function and quality of life. Depression affects the individual significantly and results in physical disability including increased morbidity and mortality. Anxiety, however, is another common psychological disorder globally (Möller et al. 2016). Furthermore, depressive disorders listed under World Health Organization (WHO) contribute significantly to non-fatal diseases worldwide and are considered as the sixth-largest contributing disease worldwide (Depression 2017). Anxiety is a normal emotion, but if it happens too often, it becomes a terrible psychiatric disorder. The presence of depression-related anxiety leads to complex symptoms, decreases in actual prognosis, regression in response to medication or treatment, and increases the risk of suicide (Kara et al. 2000). Stress classified as pathogenesis of anxiety is very versatile and includes depression, mood swings, and other psychological disorders as most predominant cases. Distortion in brain monoamine neurotransmitters such as norepinephrine, serotonin, and dopamine is a causative factor behind depression which is a chronic illness and can affect a person’s mood, thoughts, physical health, and behavior (Gold et al. 1988). Clinically significant depression in 15% or more of patients is observed during depletion of neuronal storage granules of norepinephrine, serotonin, and dopamine by Reserpine which is well-known as an antihypertensive drug (Goodwin and Bunney 1971). The stressful condition also results in oxidative stress, which has been reported as a potential agent leading to the pathogenesis of a number of chronic illnesses such as liver damage, diabetes, obesity, inflammation, neurological diseases, and cancer (Hassan et al., 2014). However, numerous researches show that, depression and anxiety are connected but are not fully distinct forms of the disease. In fact, about half of people with depression are also diagnosed with anxiety.
Correspondingly, blood clots (thrombosis) formed by homeostasis contribute to coronary blood disorders such as acute myocardial infarction, hemorrhage in the brain, which may lead to death (Emon et al. 2020a, b). Intravenous heparin is the first line of therapy for thrombus treatment because of its safety, potency, and viability (Prasad et al. 2006). While streptokinase and urokinase are widely used because of their lower costs than other thrombolytic drugs, but they are also dangerous since they can cause severe bleeding, re-occlusion, and re-infarction (Haines and Bussey 1995).
The pharmacological operation of phytoconstituents can be well characterized by virtual screening (Parasuraman 2011). The application of molecular docking is also an essential in silico methods for the design and preparation of new drug molecules using Computer Aided Drug Discovery (CADD) techniques. An effective molecular docking may allow the native ligand to detect the binding site of the three-dimensional protein structure and to correlate with physiochemical interactions (Guedes et al.2014).
The utilization of plants and plant-based natural products is a vital health concern in respect to their wide range of nutritional values as they are good source of vitamins, minerals, phenolic, fibers, antioxidants and bioactive metabolites (Haque et al. 2017b). Since ancient times different parts of plants, plant extracts, and plant-derived natural products have been used by all cultures and civilizations for the treatment of various ailments (Haque et al. 2017a). The traditional use of medicinal plants is largely focused on experimental evidence that is scientifically validated. Besides, several secondary bioactive metabolites and phytochemicals with potential therapeutic use offered by Amaranthaceae family include alkaloids, tannins and flavonoids (Scott et al. 2005). Prickly Chaff Flower or A. aspera L., belonging to family Amaranthaceae are familiar as a weed plant on roadside and garbage places throughout India, Bangladesh, Myanmar, Pakistan as well as other Asian regions. Various parts including seeds, stem, leaves and root of A. aspera incorporate potential effects in asthmatic cough, snakebite, hydrophobia, urinary calculi, rabies, influenza, piles, bronchitis, diarrhea, renal dropsy, gonorrhea, and abdominal pain (Bhoomika et al. 2007; Jain and Puri 1984). In spite of evaluating different biological activities of A. aspera, not much attempt was taken to assess the antidepressant, anti-anxiolytic and thrombolytic properties and therefore, the neuropharmacological and thrombolytic behavior of the methanol soluble extract of the A. aspera’s aerial part (MEAA) was investigated using biological and computational approaches. These include predictions of activity of substances spectra (PASS), molecular docking, absorption, distribution, metabolism, excretion, and toxicity (ADME/T).
Materials and methods
Drug and chemicals
Diazepam (Square Pharmaceutical Ltd, Bangladesh), Commercially available lyophilized Streptokinase (SK) vials (Sanofi Bangladesh Ltd), methanol (Sigma-Aldrin, MO, St. Louis, USA) tween–80 (Goyal Chem Associates, New Delhi, India), saline water (Orion Pharmaceuticals Ltd, Bangladesh) have been purchased from mentioned suppliers.
Collection and identification of the plant
Prickly aerial part (Achyranthes aspera L.) were collected from Hobigonj, Sylhet, Bangladesh. After that, the taxonomy of the plant was identified by Dr. Sheikh Bokhtear Uddin, Professor, Department of Botany, University of Chittagong, Chittagong-4331, Bangladesh and stored for the further analysis (Accession No: CTGUH-SR-2787). After washing, the aerial part of the plant was separated, and plant materials were dried throughout shade at low temperatures and thrashed using a suitable blender machine. The substance was then put in an impermeable container and kept until a further examination in a cool, safe, and warm place.
Extraction of plant material
The granulated powder substance (approximately 800 g) was soaked in methanol at a ratio of 1:6 (sample: solvent) (w/v) for 3 days at room temperature, with frequent shaking and mixing for the most intense absorption (Haque et al. 2019). Finally, it was filtered by the Whatman filter paper (Bibby RE200, Sterilin Ltd, UK) and concentrated by using a water bath. Finally, 5.8 g crude extracts have been extracted from the powdered materials and the concentrate was then kept at 4 °C for the reservation as well as for the investigation.
Experimental animals
For the experiment, Swiss-albino mice of either sex and with a weight value of 25–30 g were obtained from the Animal Resources Facility of International Centre for Diarrheal Diseases (ICDDR, B). Before the study, all animals were accustomed to the new environment for one week. The animals were kept at 25 °C temperature, comparative humidity (55–65%), and 12 h day/light cycle in a well-ventilated animal house during the experimental phase. Standard laboratory food and drinking water (Ad libitum) were given to the experimental animals.
In vivo study
Acute toxicity test
The study was conducted under standard laboratory conditions guided by Organization for Environmental Control Development (Viran et al. 2003). Each Group (n = 10) received oral doses of either 1000, 2000 or 3000 mg/kg of MEAA while the control group was treated with only vehicle (water). The groups were monitored for 48 h. The animals were weighed and physical, psychological and lack of any signs of harmfulness were noted every day.
Forced swimming test
The force swimming test (FST) was conducted to assess the antidepressant activity of MEAA in mice following the previously described method (Cryan et al. 2005). The day before the final experiment, a preliminary study was carried out to adapt the animals to the environment. An experimental swimming device consisting clear glass tank (25 ± 15 ± 25 cm) was filled up to 15 cm of water (25 ± 1 °C) thus to facilitate the swimming. Animals were divided into five groups where each group consists of five mice and Group I received the vehicle, Group II received standard drug (diazepam 5 mg/kg, b.w; i.p), Group III–V received test sample (MEAA 100, 200 and 400 mg/kg, b.w; p.o.). After 30 min, each mouse was placed in the tank for 6 min where the first 2 min was considered as initial adjustment time and the next 4 min was recorded as the immobility duration.
Tail suspension test
The tail suspension test (TST) is a convenient and effective way to evaluate the action of antidepressants (Wang et al. 2019). Mice were divided into five groups where each group consists of five mice and Group I received the vehicle, Group II received standard drug (diazepam 5 mg/kg, b.w; i.p), Group III–V received crude drug sample (MEAA 100, 200 and 400 mg/kg, b.w; p.o.). After test samples were administered, mice were led to a depressive condition (immobility), which was held with adhesive tape at the end of the tail (almost 1 cm from the tip of the tail). For each mouse of all groups, only the last 4-min of immobility time were recorded in a total of 6 min of observation.
Hole-board test
The hole-board test (HBT) was done by the previously mentioned procedure following Moniruzzaman et al. (Moniruzzaman et al. 2015). A hole-board apparatus was designed for the research. The device is composed of an evenly distributed wooden box (40 × 40 cm) with 16 holes (each 3 cm in diameter). The device was lifted to 25 cm in height. Mice were divided into five groups where each group consists of five mice and Group I received the vehicle, Group II received standard drug (diazepam 5 mg/kg, b.w; i.p), Group III–V received test sample (MEAA 100, 200 and 400 mg/kg, b.w; p.o.). Mice were put in the apparatus after 30 min administration. The head dips were reported over 5 min. Diazepam (5 mg/kg, i.p.) has been used as a standard anxiolytic agent.
In vitro study
Thrombolytic study
Streptokinase solution preparation
Commercially available Streptokinase (SK)-15, 00,000 I.U. (Polamin Werk GmbH) was homogenously mixed up with 5 mL of clean distilled water and preserved as a stock solution to conduct the in vitro thrombolytic property screening. After that 30,000 IU (100 µL) was used from this stock solution to attain thrombolysis activity (Prasad et al. 2007; Sajib Rudra et al. 2020).
Specimen for thrombolytic test
The experiment was performed using the previously established protocol (Emon et al. 2020a, b). For this research, blood (5 mL) was taken from the physically strong human volunteers (n = 10) having no history of taking NSAID and anticoagulants for 10 days. About 500 µL of blood has been transferred to the early measured Eppendorf and sustained in the incubator at 37 °C for 45 min. After the formation of coagulation, the serum was completely separated from the Eppendorf. Each tube containing only coagulation has been weighed further in order to determine the exact weight of the coagulation. Then 100 µL of plant extract was added to the tubes. The test tube containing plant extract was suspended in the incubator and fixed the temperature at 37 °C for 90 min. The blood serum has been withdrawn after the clot lysis and the tube has been measured once again to track the weight difference after the formation of clot lysis. The percentage of the clot lysis was obtained by using the following formula.
In silico studies
Molecular docking: protein preparation
3D structures of the protein used for the experiments which includes human serotonin transporter (PDB ID: 6AWO) (Coleman et al. 2016) and potassium channel (PDB ID: 4UUJ) (Lenaeus et al. 2014) for the respective antidepressant and anxiolytic study and tissue plasminogen activator (PDB ID: 1A5H) (Renatus et al. 1997) were collected from RCBS Protein Data Bank in PDB format. All the water and the heteroatom were removed from the proteins throughout using Discovery studio 2020. Proteins were arranged by combining non-polar hydrogens and assigning the Gasteiger charge. Besides, all the proteins were brought down to the least energy state with keeping standard residues in AMBER ff14sB and other residues in Gasteiger mode in UCSF Chimera and processed for further analyses (Shapovalov and Dunbrack Jr 2011).
Molecular docking: ligand preparation
The structure of eight previously identified compounds (Fig. 1) from Achyranthes aspera L. namely: 4-triacontanone, betaine, ecdysterone, eugenol, hydroquinone, oleanolic acid, p- benzoquinone, spathulenol (Bhoomika et al. 2007) and standard drugs namely: diazepam and streptokinase were retrieved from the PubChem database. The ligands were downloaded in the 2DSDF format and minimized and converted into pdbqt format via PyRx tools to quest of best optimal hit against these selected targets. PyRx (Herowati and Widodo 2014) from MGLTools (https://ccsb.scripps.edu/mgltools/) was used for the virtual screening using default settings.
Molecular docking: docking analysis
PyRx Autodock Vina has been used for the protein-ligand linking process of the chosen protein-ligand complexes (Herowati and Widodo 2014). Initially the protein was formatted to macromolecule and the docking study was employed with a semi-flexible docking system. Using PyRx AutoDock software, PDB files of phytochemicals and proteins have been minimized and subsequently transformed to PDBQT format. This study has maintained the rigidity of proteins and flexibility of ligands. Ligand molecules had 10 degrees of liberty. AutoDock defines the steps to transform molecules into pdbqt format, sort of box, grid box creation, etc. The grid box was generated in the center of the box with an active site. Finally, BIOVIA Discovery Studio Visualizer 2020 (Biovia 2017) has been accelerated to assesses docking positions for the best linking strategies.
Pharmacokinetics and toxicity measurement
Here for determining the pharmacokinetic properties (ADME) of three major compounds, the online tool SwissADME (http://www.swissadme.ch/) was used. Lipinski’s rule of five (M.W not more than 500; H-bond donors ≤ 5; H-bond acceptors ≤ 10; Lipophilicity < 5 and molar refractivity ranging from 40 to 130) were considered to evaluate favorable drug-like properties of all compounds (Lipinski et al. 1997). Moreover, the toxicological properties of all the compounds were determined by the web tool admetSAR (http://lmmd.ecust.edu.cn/admetsar2).
PASS prediction
The identified major compounds were subjected to analyzed possible biological effects by using the PASS online. Based on compounds chemical structure analysis, this tool predicts the biological properties of a compound (up to 4000 types) as probable activity and probable inactivity; where the standard values of Pa and Pi ranges from 0.000 to 1.000 (Mojumdar et al. 2016). A compound considered biologically active when its Pa ˃ Pi. Meanwhile, Pa ˃ 0.7 denotes high pharmacological activity, 0.5 < Pa < 0.7 exhibits moderate pharmacological potentiality and Pa < 0.5 demonstrates low pharmacological potentiality (Goel et al. 2011; Khurana et al. 2011).
Statistical analysis
Data were expressed as mean ± standard error mean (SEM). Statistical comparisons were performed by using one-way ANOVA followed by Dunnett’s test. The values obtained were compared with the control group and considered statistically significant when p < 0.001, p < 0.01, and p < 0.05. All statistical analyses were performed using GraphPad Prism Version 6.0 (GraphPad software Inc, San Diego, CA) software.
Results
Effect of MEAA on oral acute toxicity
None of the typical morphological characteristics (fur, face, eyes, and nose) has been changed in acute oral toxicity study. Tremors, hallucinations, salivations, diarrhea lethargy or irregular behavior such as self-mutation, regressive actions etc. have not been reported. Gait and attitude, controlling reactivity or sensory stimuli were all natural. The weight between the control and the treatment groups was not changed substantially. Daily fluctuations in the intake of food and water of animals were controlled. This shows that the MEAA was stable at a single bodyweight dose till 3000 mg/kg. That’s why MEAA (200 and 400 mg/kg, b.w; p.o) have been chosen for the biological study.
Effects of MEAA on FST
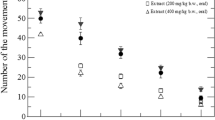
The effectiveness of the decreasing immobility time (sec) in the FST has been shown in Fig. 2. With the increase of dose, the immobility has been decreased which was found statistically significant (p < 0.001) at the dose dependent pathway. The highest number of immobility was seen 57.49 ± 1.58 (p < 0.001) (sec) for MEAA 400 (mg/kg; b.w). Moreover, standard drug (Diazepam 5 mg/kg, b.w; i.p) yielded 40.36 ± 2.37 (p < 0.001) (sec) of immobility during FST.
Effect of MEAA on the force swimming test (FST) and tail suspension test (TST). Data were presented as Mean ± SEM (n = 5), and c p < 0.05, b p < 0.01, a p < 0.001 considered as significant where # indicated the control group. Ctrl = Control; MEAA = methanol extract of the aerial part of Achyranthes aspera; DPM = Diazepam
Effects of MEAA on TST
The antidepressant effects of MEAA were studied to determine the immobilization time in the TST. The MEAA has been revealed a significant (p < 0.05, p < 0.001) reduction of immobility time in a dose-dependent manner. At the doses of 100, 200 and 400 (mg/kg, b.w; p.o), the time of immobility in TST has been found 104.59 ± 2.30, 77.80 ± 2.31 (p < 0.001), and 49.32 ± 2.28 (p < 0.001) (sec) respectively (Fig. 2).
Effects of MEAA on HBT
With the administration of MEAA 100 (mg/kg; p.o,) the number of head dips did not increase significantly. The number in the head dips were increased significantly (p < 0.01, p < 0.001) with the increasing of the doses. The initial head dip latency (sec) of head poking was significantly (p < 0.5, p < 0.01) declined by the increased dose. Correspondingly, reference drug (diazepam, 5 mg/kg, i.p.) demonstrated substantial (p < 0.01) exploratory activity (Fig. 3).
Effect of MEAA on thrombolytic test
The expansion of 100 µL SK, a positive control (30,000 I.U.) to the coagulations alongside an hour and a half of brooding at 37 °C, indicated 59.73 ± 3.52% (p < 0.001) clot lysis. In addition, saline water showed 3.23 ± 1.32% of clot lysis which is therapeutically insignificant. The summary of the clot lysis of MEAA has been shown in Fig. 4.
Molecular docking analysis for antidepressant and anxiolytic study
The docking analysis results for antidepressant and anxiolytic activity are presented in Table 1; Fig. 5. In this investigation, two receptors namely human serotonin transporter (PDB ID: 6AWO) and potassium channel (PDB ID: 4UUJ) were selected for antidepressant and anxiolytic study respectively. In the case of human serotonin transporter (PDB ID: 6AWO), the ranking of the docking score is as follows: Spathulenol > P- Benzoquinone > Diazepam > Eugenol > Hydroquinone > Ecdysterone > Betaine > Oleanolic acid > 4-triacontanone. On the contrary, the docking of the selected compound against the potassium channel (PDB ID: 4UUJ) is as follows: Diazepam > Hydroquinone > Ecdysterone > Eugenol > 4-triacontanone > Oleanolic acid > P- Benzoquinone > Spathulenol > Betaine. Spathulenol binds to the human serotonin transporter via several amino acid residues namely: trp214, arg194, tyr99. Again, diazepam obtained the highest docking score (− 7.3 kcal/mol) with a series of bonds through amino acid residues: conventional hydrogen bond (asn41), carbon hydrogen bond (gly42), alkyl and pi-alkyl bond (pro154, val93and pro41) to the potassium channel.
3D and 2D presentation of the best binding-interactions of the docked compounds and receptors: a and b spathulenol and diazepam with human serotonin transporter (PDB ID: 6AWO), c and d hydroquinone and diazepam with potassium channel (PDB ID: 6UUJ), e and f spathulenol and streptokinase with tissue plasminogen activator (PDB ID: 1A5H)
Molecular docking analysis for thrombolytic study
The docking analysis results for the thrombolytic activity have been presented in Table 1; Fig. 5. In the case of tissue plasminogen activator (PDB ID: 1A5H), the highest score has been obtained − 7.21 kcal/mol for Spathulenol. Spathulenol binds to the plasminogen activator through a series of bonds where, they interact to the receptor with a number of amino acids: pi-sigma (tyr99), alkyl and pi-alkyl (trp214, arg174). The ranking of the docking score is as follow: Spathulenol > Streptokinase > P- Benzoquinone > 4-triacontanone > Betaine > Ecdysterone > Hydroquinone > Eugenol > Oleanolic acid.
Pharmacokinetics and toxicity measurement
The pharmacokinetic characteristics of the selected substances were calculated by the online tool SwissADME and followed Lipinski’s rules. Lipinski has here declared that if a drug/compound follows the following criteria such as molecular weight < 500 amu, Hydrogen bond donor < 5, Hydrogen bond acceptor sites ≤ 10, and Lipophilicity value LogP ≤ 5, then the compound would be orally bioavailable. The study showed that all the compounds complied with the rules of Lipinski, suggesting the strong oral bioavailability of these compounds. Again, the online admetSAR server was also used to predict the toxicological properties of the seven compounds. The analyses showed that the selected compounds are non-Ames toxic and non-carcinogenic (Table 2).
PASS prediction study
Eight previously selected compounds of Achyranthes aspera were studied by the PASS online tool for antidepressant, anxiolytic and thrombolytic activities and the potent compounds displayed higher Pa value than Pi (Table 3).
Discussion
While various forms of medication have been available to treat anxiety, depression, coronary artery occlusion but the complete relief of disease symptoms remained a mystery. Typical side effects and low pharmacokinetics have limited the clinical implementation of these medications. In the meantime, some of the drugs are already in the market which have various side effects. The current research shows that MEAA has strong and effective implications on force swimming, tail suspension, head-dipping in hole board, and anticoagulant activity. While progress is made in the treatment of depression and anxiety, the clinical needs of many patients are still to be addressed. The safety, effects duration, and side effects of current drugs have become a significant issue and there is need for newer drugs. Phytomedicine is therefore promising for the treatment of these diseases due to the variety of neural targets (Fajemiroye et al. 2016).
Forced swimming test and tail suspension test are widely accepted methods for the measurement of the action of antidepressants. The characteristic behavior assessed for these tests known as immobility is thought to have been like human depression. The ability to minimize immobility times in mice is well known as the antidepressant capability of drugs (Porsolt et al. 1977). The implementation of swimming as a target allowed us to identify numerous possible antidepressant compounds. The antagonists of BDNF (brain-derived neurotrophic factor) and NT-3 (Neurotrophin-3), estradiol, kappa opioid receptor, glutamate metabotropic receptors, the synthesis of nitric oxide, melanocortin-4, GABA (gamma-aminobutyric acid) receptor and the agonists of the delta-opioid receptor, improve swimming activity. Since the 5-HT (5-hydroxytryptamine receptor) complies with the prevention of its anti-depressant effect on the swimming test, the effect of SSRI (Selective serotonin receptor inhibitors) depends on the intact synthesis of 5-HT (Cryan et al. 2005). Because of significant correlation between the reduction of immobility and effectiveness with clinical potency, rat model is given focused priority to assess antidepressant though similar to human depression, FST does not induce in rats symptomatology (Willner 1983) and the event is quite specific due to the propensity of distinguishing antidepressants from neuroleptics and anxiolytics. Though dopaminergic, anticholinergic, and GABAergic moieties are performing as antidepressants, it is quite arduous to determine their absolute effects which will cause specific action (Borsini and Meli 1988). Antidepressant-like responses of methanol extract of A. aspera probably through the restoration of brain monoamines including norepinephrine, 5-hydroxytryptamine, and dopamine levels reverses the reserpine-induced depression indicated by the decrease in the extension of the immobility period in the TST. As an elevated level of cortisol is also a causative etiology of depression in humans, there is also a possibility of reduction of serum cortisol levels by MEAA to act as an antidepressant agent.
Respectively, the number as well as the time of exploratory head-dips, were observed in HBT with the treatment of MEAA in dose-dependent manner. Because of the methodological simplicity of HBT, this experiment is beneficial and different behavioral responses can be detected and measured freely during unfamiliar circumstances. The head-dipping behavior of the animals is directly related to their psychological state (Ali et al. 2015). This finding is consistent with the claims that the frequency of head-dipping and earliness of the latency to the first head-dip has been seen in the hole-board after the administration of non-sedative doses of diazepam. Anxiolytic status has been identified with increased head dipping and earliness off head dipping. This indicates that a decrease in head dipping behavior in the hole-board test may indicate the animal’s anxiety and that both animal anxiogenic and anxiolytic behavior can be measured using the hole-board test (Takeda et al. 1998). Many first-line anxiolytic and antidepressant medications enhance the activity of monoamines by inhibiting an enzyme that breaks down monoamine (norepinephrine, serotonin, and dopamine) and blocking reuptake in the synaptic cleft. First-line medicines are often taken by the patient itself with or without prescription as an anxiolytic and/or antidepressant drug. The most widely used anxiolytic drugs are benzodiazepines that amplify GABAergic inhibitory transmission (Tatarczyńska et al. 2001). Interestingly, some of the anxiolytic medications have behaviors that may interfere with pathophysiology with antidepressant behavior. Serotonin agonists such as Buspirone and Gepirone are anticipated to acts on presynaptic and postsynaptic 5-hydroxytryptamine-1A (5-HT1A) receptors and employed both as antidepressant and anxiolytic agents (Schechter et al. 2005).
Experimental results in blood coagulation suggest that the coagulation process is divided into three stages during the thrombolytic activity test: prothrombin activator formation, thrombin training, and fibrin development. The formation of thrombus may be disrupted by antithrombotic or thrombolytic drugs. The basic activity of thrombolytic agents is to remove the fibrin from plasmin that can be activated by inactive plasminogen activators. Streptokinase (SK) and urokinase (UK) act via following the indirect pathway of clot lysis. In the indirect mechanism, the thrombolytic enzymes directly dissolve the fibrin, which was proved by our thrombolysis experiments in vitro (Yuan et al. 2012). This study has already shown that MEAA can inhibit coagulation that may be an essential element in the management of antithrombotic or thrombolytic treatment and does not affect the normal process of clotting in the body at a low dose. Therefore, in the prevention of thrombosis, low-dose MEAA is recommended.
Studies on molecular docking have been widely used to forecast ligand-target interactions and to gain a deeper understanding of natural products’ biological activity. It also provides additional insights into potential mechanisms of action and binding strategies within the binding pockets of different proteins (Khan et al. 2019). To consider the fact, a molecular docking study has been conducted to explain and validate the experimental results found in biological study. The experimental findings are confirmed in this process. Eight representative molecules of Achyranthes aspera were chosen for docking study in order to gain a deeper insight regarding the witness of biological activity (antidepressant, anxiolytic, and thrombolytic) and they were compared to the standard drugs. These compounds were then docked against three targets, namely the human serotonin transporter (PDB ID: 6AWO), and potassium channel (PDB ID: 4UUJ), tissue plasminogen activator (PDB ID: 1A5H) for antidepressant, anxiolytic and thrombolytic study respectively. Molecular docking with human serotonin transporter (PDB ID: 6AWO) showed that several amino acid residues reacted by the hydrogen bonds with selected compounds resulting in docking values between − 4.3 to -7.4 kcal/mol. From those findings, it can be inferred that, by interactions with these target proteins, the phytoconstituents are significantly responsible for the antidepressant activity. Besides, in the anxiolytic docking study, molecular docking with the potassium channel (PDB ID: 4UUJ) has been accomplished and after interactions between the bioactive compounds and enzymes, the docking score has been assumed ranging from − 2.3 to − 7.3 kcal/mol.
Among all the compounds spathulenol, 4-triacontanone, and betaine have shown the significant docking score against plasminogen tissue activator (PDB ID: 1A5H) in the thrombolytic docking study and the highest score against tissue plasminogen activator enzymes has been seen in spathulenol. This research suggests that the bioactive compounds (spathulenol, 4-triacontanone, and betaine) may be responsible for the thrombolytic activity of the MEAA.
The investigation further established the pharmacokinetic/drug likeliness and toxicological properties based on the results of the molecular docking study against human serotonin transport, potassium channel, tissue plasminogen activator. These interpretations were significantly intended as essential considerations for the development of a new drug, clinical trial, and biological review of the isolated compounds (Lipinski 2004). Throughout this case, the pharmacokinetic properties of the major compounds of Achyranthes aspera were determined by using an online method (SwissADME), which was based on the Lipinski Five Rules. Lipinski’s rule of 5 reports that the molecular weight of orally administered drug/compounds should be < 500 amu, the value of lipophilicity is ≤ 5, hydrogen bond acceptors < 5, and the hydrogen bonds donor is ≤ 10. When any drugs or compounds break this rule, the bioavailability of the compound can be questionable (Zhang and Wilkinson 2007). The findings suggest all compounds met the requirements of Lipinski’s five rules, suggesting that these compounds are effective drug candidates and potentially preferable for oral use. Besides, the admetSAR online tool was used to evaluate the toxicological properties of the selected phytochemicals, as the safety of the compounds is an important parameter for becoming a good drug (Cheng et al. 2012). In this research, no ames toxicity and carcinogens were identified as a threat.
PASS is a computer-driven online tool used to simulate various forms of bioactivity simultaneously depending on the structure of different molecules or substances (Poroikov and Filimonov 2005). For this analysis, molecules/compounds showing more Pa (probable activity) value than Pi (probable inactivity) are considered as appropriate for a biological activity. Finally, employing complementary protocols, this detailed analysis proved scientifically significant. The described results may be attributable to the combined orientation of many phytoconstituents, among those currently reported and probably other constituents that are not currently reported.
Conclusions
The outcomes of this present research reflect that the aerial part of Achyranthes aspera L. can be a conspicuous source of antidepressant and anxiolytic potentials as well as promising candidate for thrombolytic potential. Furthermore, in molecular docking analysis, various bioactive potential constituents displayed auspicious binding affinity to specific proteins, and the ADME/T study showed their druglike characters. These experimental findings have been found in consistent with the PASS predictions for bioactive constituents. So that, Achyranthes aspera L. may be a suitable candidate in the management of depression, anxiety and thrombosis related disorders. However, additional studies are required to classify and isolate the pure compounds that are responsible for the biological consequences observed. Besides, extensive mechanistic investigations are suggested to support these present findings.
Abbreviations
- ADME/T:
-
Absorption, distribution, metabolism, excretion, and toxicity
- BDNF:
-
Brain-derived neurotrophic factor
- CADD:
-
Computer aided drug discovery
- FST:
-
Force swimming test
- GABA:
-
Gamma-aminobutyric acid
- HBT:
-
Hole board test
- MEAA:
-
Methanol extract of aerial part of A. asperaMethanol extract of aerial part of A. aspera
- NT-3:
-
Neurotrophin-3
- PASSPASS:
-
Predictions of activity of substances spectra
- SKSK:
-
Streptokinase
- SSRI:
-
Selective serotonin receptor inhibitors
- TST:
-
Tail suspension test
- UKUK:
-
Urokinase
- 5-HT:
-
5-hydroxytryptamine receptor
References
Ali MS, Dash PR, Nasrin M (2015) Study of sedative activity of different extracts of Kaempferia galanga in Swiss albino mice. BMC Complement Altern Med 15:158
Bhoomika R, Ramesh K, Anita A (2007) Phyto-pharmacology of Achyranthes aspera: a review. Pharmacogn Rev 1:143
Biovia DS (2017) Discovery studio visualizer. San Diego, CA, USA 936
Borsini F, Meli A (1988) Is the forced swimming test a suitable model for revealing antidepressant activity? Psychopharmacology 94:147–160
Cheng F, Li W, Zhou Y, Shen J, Wu Z, Liu G, Lee PW, Tang Y (2012) admetSAR: a comprehensive source and free tool for assessment of chemical ADMET properties. ACS Publications, Washington
Coleman JA, Green EM, Gouaux E (2016) X-ray structures and mechanism of the human serotonin transporter. Nature 532:334–339
Cryan JF, Valentino RJ, Lucki I (2005) Assessing substrates underlying the behavioral effects of antidepressants using the modified rat forced swimming test. Neurosci Biobehav Rev 29:547–569
Depression W (2017) Other common mental disorders: global health estimates. World Health Organization, Geneva, pp 1–24
Emon NU, Jahan I, Sayeed MA (2020a) Investigation of antinociceptive, anti-inflammatory and thrombolytic activity of Caesalpinia digyna (Rottl.) leaves by experimental and computational approaches. Adv Tradit Med 20:1–9
Emon NU, Kaiser M, Islam M, Kabir MFI, Jamir M, Uddin MAJ, Tanjil SM, Rasel ANM, Alam S, Islam MN (2020b) Anxiolytic and thrombolytic investigation of methanol extract of Piper nigrum L. fruits and Sesamum indicum L. seeds. J Adv Biotechnol Exp Ther 3:158–164
Fajemiroye JO, da Silva DM, de Oliveira DR, Costa EA (2016) Treatment of anxiety and depression: medicinal plants in retrospect. Fundam Clin Pharmacol 30:198–215
Goel RK, Singh D, Lagunin A, Poroikov V (2011) PASS-assisted exploration of new therapeutic potential of natural products. Med Chem Res 20:1509–1514
Gold PW, Goodwin FK, Chrousos GP (1988) Clinical and biochemical manifestations of depression. N Engl J Med 319:413–420
Goodwin FK, Bunney WE (1971) Depressions following reserpine: a reevaluation. Seminars in psychiatry. Grune & Stratton, Inc., Philadelphia
Guedes IA, de Magalhães CS, Dardenne LE (2014) Receptor–ligand molecular docking. Biophys Rev 6:75–87
Haines ST, Bussey HI (1995) Thrombosis and the pharmacology of antithrombotic agents. Ann Pharmacother 29:892–905
Haque MA, Jantan I, Arshad L, Bukhari SNA (2017a) Exploring the immunomodulatory and anticancer properties of zerumbone. Food Funct 8:3410–3431
Haque MA, Jantan I, Bukhari SNA (2017b) Tinospora species: An overview of their modulating effects on the immune system. J Ethnopharmacol 207:67–85
Haque MA, Jantan I, Harikrishnan H, Ghazalee S (2019) Standardized extract of Zingiber zerumbet suppresses LPS-induced pro-inflammatory responses through NF-κB, MAPK and PI3K-Akt signaling pathways in U937 macrophages. Phytomedicine 54:195–205
Hassan W, Eduardo Barroso Silva C, Mohammadzai IU, Batista Teixeira da Rocha, J, Landeira-Fernandez J (2014) Association of oxidative stress to the genesis of anxiety: implications for possible therapeutic interventions. Curr Neuropharmacol 12, 120–139
Herowati R, Widodo GP (2014) Molecular Docking studies of chemical constituents of Tinospora cordifolia on glycogen phosphorylase. Procedia Chem 13:63–68
Jain S, Puri H (1984) Ethnomedicinal plants of Jaunsar-Bawar Hills, Uttar Pradesh, India. J Ethnopharmacol 12:213–222
Kara S, Yazici KM, Güleç C, Ünsal I (2000) Mixed anxiety–depressive disorder and major depressive disorder: comparison of the severity of illness and biological variables. Psychiatry Res 94:59–66
Khan S, Nazir M, Raiz N, Saleem M, Zengin G, Fazal G, Saleem H, Mukhtar M, Tousif MI, Tareen RB (2019) Phytochemical profiling, in vitro biological properties and in silico studies on Caragana ambigua stocks (Fabaceae): a comprehensive approach. Ind Crops Prod 131:117–124
Khurana N, Ishar MPS, Gajbhiye A, Goel RK (2011) PASS assisted prediction and pharmacological evaluation of novel nicotinic analogs for nootropic activity in mice. Eur J Pharmacol 662:22–30
Lenaeus MJ, Burdette D, Wagner T, Focia PJ, Gross A (2014) Structures of KcsA in complex with symmetrical quaternary ammonium compounds reveal a hydrophobic binding site. Biochemistry 53:5365–5373
Lipinski CA (2004) Lead-and drug-like compounds: the rule-of-five revolution. Drug Discov Today Technol 1:337–341
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ 1997) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 23:3–25
Mojumdar M, Kabir M, Hasan M, Ahmed T, Rahman MR, Akter Y, Rahman, MM (2016) Molecular docking and pass prediction for analgesic activity of some isolated compounds from Acalypha indica L. and ADME/T property analysis of the compounds. World J Pharmaceut Res 5:1761–1770
Möller H-J, Bandelow B, Volz H-P, Barnikol UB, Seifritz E, Kasper S (2016) The relevance of ‘mixed anxiety and depression’as a diagnostic category in clinical practice. Eur Arch Psychiatry Clin NeuroSci 266:725–736
Moniruzzaman M, Rahman A, Ferdous A (2015) Evaluation of sedative and hypnotic activity of ethanolic extract of Scoparia dulcis Linn. J Evid Based Integr Med 2015:873954
Parasuraman S (2011) Prediction of activity spectra for substances. J Pharmacol Pharmacother 2:52
Poroikov V, Filimonov D (2005) PASS: prediction of biological activity spectra for substances. Predict Toxicol 520:520
Porsolt R, Bertin A, Jalfre M (1977) Behavioral despair in mice: a primary screening test for antidepressants. Archives internationales de pharmacodynamie et de therapie 229:327–336
Prasad S, Kashyap RS, Deopujari JY, Purohit HJ, Taori GM, Daginawala HF (2006) Development of an in vitro model to study clot lysis activity of thrombolytic drugs. Thromb J 4:14
Prasad S, Kashyap RS, Deopujari JY, Purohit HJ, Taori GM, Daginawala HF (2007) Effect of Fagonia arabica (Dhamasa) on in vitro thrombolysis. BMC Complement Altern Med 7:36
Renatus M, Bode W, Huber R, Sturzebecher J, Prasa D, Fischer S, Kohnert U, Stubbs MT (1997) Structural mapping of the active site specificity determinants of human tissue-type plasminogen activator. Implications for the design of low molecular weight substrates and inhibitors. J Biol Chem 272:21713–21719
Sajib Rudra M, Sawon SU, Emon NU, Alam S, Tareq SM, Islam MN, Uddin MMR, Chowdhury AMS, Hasbe AN, Shakil M (2020) Biological investigations of the methanol extract of Tetrastigma leucostaphylum (Dennst.) Alston ex Mabb.(Vitaceae): in vivo and in vitro approach
Schechter LE, Ring RH, Beyer CE, Hughes ZA, Khawaja X, Malberg JE, Rosenzweig-Lipson S (2005) Innovative approaches for the development of antidepressant drugs: current and future strategies. NeuroRx 2:590–611
Scott IM, Puniani E, Jensen H, Livesey JF, Poveda L, Sánchez-Vindas P, Durst T, Arnason JT (2005) Analysis of Piperaceae germplasm by HPLC and LCMS: a method for isolating and identifying unsaturated amides from Piper spp extracts. J Agric Food Chem 53:1907–1913
Shapovalov MV, Dunbrack RL Jr (2011) A smoothed backbone-dependent rotamer library for proteins derived from adaptive kernel density estimates and regressions. Structure 19:844–858
Takeda H, Tsuji M, Matsumiya T (1998) Changes in head-dipping behavior in the hole-board test reflect the anxiogenic and/or anxiolytic state in mice. Eur J Pharmacol 350:21–29
Tatarczyńska E, Kłodzińska A, Chojnacka-Wójcik E, Pałucha A, Gasparini F, Kuhn R, Pilc A (2001) Potential anxiolytic‐and antidepressant‐like effects of MPEP, a potent, selective and systemically active mGlu5 receptor antagonist. Br J Pharmacol 132:1423–1430
Viran R, Erkoç F, Polat H, Koçak O (2003) Investigation of acute toxicity of deltamethrin on guppies (Poecilia reticulata). Ecotoxicol Environ Saf 55:82–85
Wang S, Liu H, Wang X, Lei K, Li G, Quan Z (2019) Synthesis and evaluation of antidepressant activities of 5-aryl-4, 5-dihydrotetrazolo [1, 5-a] thieno [2, 3-e] pyridine derivatives. Molecules 24:1857
Willner P (1983) Dopamine and depression: a review of recent evidence. I. Empirical studies. Brain Res Rev 6:211–224
Yuan J, Yang J, Zhuang Z, Yang Y, Lin L, Wang S (2012) Thrombolytic effects of Douchi fibrinolytic enzyme from Bacillus subtilis LD-8547 in vitro and in vivo. BMC Biotechnol 12:36
Zhang M-Q, Wilkinson B (2007) Drug discovery beyond the ‘rule-of-five’. Curr Opin Biotechnol 18:478–488
Acknowledgements
The authors would like to express their gratitude to the Department of Pharmacy, International Islamic University Chittagong, Chittagong-4318, Bangladesh for providing instrumental and chemical facilities. The authors also wish to acknowledge Professor Dr. Shaikh Bokhtear Uddin, (University of Chittagong, Bangladesh).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All the authors contributed equally.
Corresponding authors
Ethics declarations
Ethical statement
All biological property assessments were performed according to the ethical standards stated in the Declaration of Helsinki 2013. Animal models were held and treated in accordance with the principles of the Swiss Academy of Medical Sciences and Swiss Academy of Sciences and were euthanized following the Guidelines for the Euthanasia of Animals: 2013 edition.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Emon, N.U., Alam, S., Rudra, S. et al. Evaluation of pharmacological potentials of the aerial part of Achyranthes aspera L.: in vivo, in vitro and in silico approaches. ADV TRADIT MED (ADTM) 22, 141–154 (2022). https://doi.org/10.1007/s13596-020-00528-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-020-00528-5