Abstract
Plants produce a number of biological active substances with healthy benefits. Resveratrol (3,5,4′-trihydroxystilbene), a polyphenol produced by plants has been associated with many health beneficial properties, including its ability to induce mitochondrial biogenesis and fight against health problems such as obesity, inflammation, heart diseases, cancers among others. Mitochondrial dysfunction is recognized as central to the pathogenesis and development of many diseases. Thus the present review describes how resveratrol (RSV) may counteract physiological and age-related diseases/disorders through its effect on mitochondrial biogenesis and function. In addition, we discuss the chemistry, main sources, and the doses of RSV shown in previous studies to be efficient for the prevention and treatment of different diseases. Through its ability to improve mitochondrial dysfunction, RSV can be used in the prevention and/or treatment of human physiological diseases. However, more research for optimal dose in a human scale is still relevant. This review brings new hope to the therapy of physiological diseases as it will provide useful future perspectives for the planning of clinical studies on RSV and mitochondrial dysfunction-related diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mitochondria are organelles responsible for several vital functions in our cells. Except for red blood cells, each of our cells has between 200 and 2000 mitochondria, and they are responsible for producing about 90 percent of the energy we use to live and grow (Siekevitz 1957). When these important structures fail, our cells produce less energy, which can injure and even cause cell death. Chronic fatigue and low-energy symptoms of many diseases can be attributed to mitochondrial dysfunction. Mitochondrial biogenesis is an increase in the cell’s mitochondrial mass resulting from numerous interactions between information and constituents of nuclear origin, e1aborated by the cytoplasmic protein synthesis system, and components e1aborated within the mitochondria, by the intramitochondrial protein synthesis system (Fabian Sanchis-Gomar et al. 2014).
RSV is a plant polyphenolic micro-constituent with numerous protective potentials. It decreases mitochondrial superoxide anion generation by stimulating mitochondria biogenesis (Zhang et al. 2017). It has been previously demonstrated that oxidative stress and inflammation also contribute to premature aging and chronic diseases (Liguori et al. 2018). As an antioxidant, RSV provides the body with essential support to prevent, control, and repair oxidative stress induced by free radicals (Ning Xia et al. 2017). It also maintains levels of intracellular antioxidants, such as superoxide peroxidase (SOD), catalase, and glutathione reductase. This helps protect the mitochondria and tissues from the damaging effects of free radicals. (Leonard et al. 2003). RSV, with pleiotropic effects, has also the ability to activate sirtuin-1 (SIRT1); a protein that is associated with longevity (Lagouge et al. 2006). By activating SIRT1, resveratrol maintains the health and concentration of mitochondria. Besides oxidative damage, mitochondrial dysfunction also plays an important role in aging and disease (Amarendranath Choudhury et al. 2018). SIRT1 cooperates with the co-activator of peroxisome proliferator-activated receptor-gamma coactivator 1-alpha (PGC-1α) in promoting adaptation to caloric restriction (Krek 2006).
It increases the number and size of mitochondria in non-oxidative muscle fibers and increases their oxidative metabolism by transforming them into type 1 fibers, which probably explains the improvement in endurance (Muhammad and Allam 2018). The interest in using RSV as a bioactive compound was raised for the first time by a study conducted by Siemann and Creasy which has reported the presence of trans-resveratrol in red wine (Siemann 1992) but it has been long used in popular medicine as a treatment for hyperlipemia, arteriosclerosis, allergic diseases and inflammatory diseases (Yoshiyuki Kimura 1985). Until 2006, there was no study interested in the effect of RSV on mitochondrial biogenesis but in that year Lagouge et al. reported that RSV may improve mitochondrial function and protects against metabolic disease (Lagouge et al. 2006).
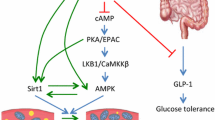
RSV also activates adenosine monophosphate-activated protein kinase (AMPK) (Timmers et al. 2011). Like SIRT1, AMPK regulates metabolism during energy stress to restore cellular homeostasis (Hardie et al. 2012). Thus, AMPK inhibits the anabolic pathways (synthesis of lipids, proteins, etc.) and activates catabolism (β-oxidation of fatty acids, autophagy, etc.). RSV activation of AMPK has been shown to result from direct inhibition of cyclic adenosine monophosphate (cAMP)-specific phosphodiesterases (PDE) (Park et al. 2012). Interestingly, several studies have revealed that activation of PGC-1α by SIRT1 (Canto et al. 2010; El-Khamisy et al. 2005), or inhibition of HIF-1α (hypoxia-inducible factor 1-alpha) by AMPK (Saiko et al. 2008) blocks aerobic glycolysis. Thus, it is tempting to think that the activation of the AMPK-SIRT1 axis by RSV induces metabolic reprogramming (Timmers et al. 2011; Wang et al. 2014).
RSV increases mitochondrial function by inducing the expression of genes controlling energy homeostasis, including PGC-1α, which in turn induces genes facilitating beta-oxidation of fatty acids and uncoupling protein 1 (UCP-1) mRNA, an uncoupling protein involved in the production of heat by brown adipose tissue (Lagouge et al. 2006). This proves the potential of RSV in the treatment of obesity and insulin resistance or at least reinforce resistance to weight gain and its related physiological imbalances. Thus, there is an evident compulsion for more clinical studies addressing the optimum doses of RSV for improving mitochondrial dysfunction and increasing mitochondrial biogenesis.
Applications of RSV in the treatment of physiological diseases have previously been presented by other authors (Catalgol et al. 2012; Rauf et al. 2017). Furthermore, some authors also focused on the effects of RSV on mitochondrial function (de Oliveira et al. 2016; Ning Xia et al. 2017) but did not detail their associations with different diseases and effective doses of RSV for their treatment.
In the present review, we discuss the effects of resveratrol on mitochondrial biogenesis and multiple diseases such as cancer, diabetes, obesity, CVDs, erectile dysfunction, neurodegenerative diseases, liver steatosis and Psoriasis that have been previously observed in animal and human studies. The solubility of RSV is very limited which may affect its bioavailability. Against this background, this review presented a different route of RSV administration and its efficacy to regulate and control various diseases. Additionally, the dose and time dependence effects were enumerated. The information provided in this review will help future researchers to plan and design preclinical and clinical studies on RSV and lifestyle or age-related diseases. At the conclusion, we supply hints for prospective directions of future resveratrol research.
Chemical structure of resveratrol
Resveratrol (3,5,4′-trihydroxystilbene) is a polyphenolic compound synthesized by a varied assortment of plants (Saiko et al. 2008). In nature, it is found in two forms of isomers, these are cis-isomer and trans-isomer (Fig. 1), the trans-isomer has the greatest biological activity and is abundant (Vian et al. 2005). RSV is a very light-sensitive compound disposed to ultraviolet-induced isomerization, over 80% of the trans-resveratrol in solution is transformed to cis-isomer when exposed to UV light for an hour (Langkake 1976; Vian et al. 2005).
Sources of resveratrol
RSV was identified and isolated at the first time from the roots of white hellebore (Veratrum grandiflorum O. Loes) in 1940 (Baur and Sinclair 2006) and afterward, RSV has been found in a variety of plants species and foods these include grapes, peanuts, berries, cocoa, dates, groundnuts, tomatoes, pines among others (Counet et al. 2006). In grapes, particularly when affected by Botrytis cinerea, it is elaborated in the crust and its proportion is high just before the ripeness of the grapes (Jubilee and Stewart 2003). Accordingly, the highest amount of RSV is found in their crust and seeds (50–100 µg per gram) (Counet et al. 2006).
The concentrations of RSV vary significantly in comestible plants (Table 1). As an example, the level of RSV rises in the grapes affected by biotic and abiotic stresses, for example, bacteria, yeast, cold, or ultraviolet (UV) radiation (Weiskirchen 2016). The entire quantity of RSV consumed by humans might be augmented by more RSV-rich foods intake (e.g., red grapes, berries, itadori, peanuts) or their derivatives such as red wine, dried fruits, tea, jams, and juices.
Biological activities of resveratrol
Many researchers have reported that RSV has several biological activities (Fig. 2), comprising anti-oxidative, anti-diabetes, anti-aging, anti-cancer, anti-inflammatory cardioprotective activities, as well as other health benefits such as induction of apoptosis, mitochondrial biogenesis among others.
Anti-inflammatory effects of resveratrol
A number of human diseases are mainly due to chronic inflammation. The inflammatory progressions may cause mutations of DNA in cells through nitrosative or oxidative stress, which may affect normal functions of cells and hence result in inflammatory diseases (Sanchez-Fidalgo et al. 2010).
Recent studies have demonstrated the dynamic role of mitochondria in the regulation of inflammatory processes. Mitochondrial stress and the buildup of damaged and dysfunctional mitochondria caused by defects in the PTEN-induced kinase 1 (PINK1) or PARKIN genes mediated mitophagy may contribute to the pathology of mitochondria-induced inflammatory diseases such as Parkinson’s disease (Newman and Shadel 2018; Sliter et al. 2018). In response to infection and stress, mitochondria release danger signals (mtDNA) in the cytoplasm that promote the formation and activation of nod-like receptor protein 3 (NLRP3) inflammasomes (inflammatory signaling platform), resulting in inflammation (Shimada et al. 2012; Zhang et al. 2010). Activation of inflammasomes by damaged mitochondria results in the caspase-1-dependent secretion of the inflammatory cytokines IL-1β and IL-18 (Nakahira et al. 2011).
In their experiment, Cui et al. have proven that RSV considerably repressed inflammation indicators, for example, inducible nitric oxide synthase (iNOS), tumor necrosis factor-α (TNF-α), and cyclooxygenase-2 (COX-2) (Cui et al. 2010). Sanchez-Fidalgo and co-workers’ study was confirmed that its supplementation diminished chronic colonic inflammation by decreased proinflammatory cytokines, comprising prostaglandin E synthases-1 (PGES-1), interleukin-1 beta (IL-1b), IL-10, TNF-α, COX-2, and iNOS, through reduction of the p38 MAP kinase (mitogen-activated protein kinases) signaling pathway (Sanchez-Fidalgo et al. 2010).
The potential of RSV in prevention and treatment of inflammatory and autoimmune diseases is elucidated by its ability to initiate apoptosis in activated T cells and downregulate IL-2, IL-9, IL-12, IL-17, TNF-α, interferon-g, monocyte chemoattractant protein-1 (MCP-1) and macrophage inflammatory protein-1 alpha (Kjaer et al. 2015).
In recent years, the study conducted by Lee et al. (2015) recommended that RSV would rise apigenin (a strong anti-inflammation) bioavailability. Cotreatment of RSV and apigenin augmented the concentrations of plasma apigenin up to 2.39 times comparing to the group treated by apigenin only (Lee et al. 2015). Zhang et al. (2017) suggested that RSV can improve mitochondrial dysfunction through stimulation of mitochondrial biogenesis, improvement of energy homeostasis, and a decrease in oxidative damage (Zhang et al. 2017). Oxidative damage to cells (their membranes, lipids, enzymes, mitochondria, and DNA) affects the functions of tissues and organs, thus causing unwanted immune responses and inflammation (Lugrin et al. 2014).
Anti-cancer effects
A chemopreventive approach to cancer is a promising strategy in the control of the disease. The idea of chemoprevention is to stop, delay, or reverse the progression of malignant transformation of cells, through physiological mechanisms that do not affect the healthy cells (Sporn 2011). In this process, a particular interest is brought to phytochemical compounds. Their anti-carcinogenic effect is attributed to their anti-mutagenic, antioxidant, and anti-inflammatory properties (Catalgol et al. 2012).
During the process of tumorigenesis, mitochondria undergo a series of alterations that notably contribute to the metabolic reprogramming of cancer cells (Gogvadze et al. 2008). Reddivari et al. reported that two weeks of resveratrol-grapeseed extract (RSV-GSE) treatment resulted in suppression of Wnt/β-catenin signaling and elevation of mitochondrial-mediated apoptosis in colon cancer stem cells (Reddivari et al. 2016). RSV enhances mitochondrial functions via SIRT1 (Lagouge et al. 2006). When a calorie restriction SIRT1 is activated, it regulates the metabolism including mitophagy (Pietrocola et al. 2012), the process by which damaged mitochondria are destroyed.
Resveratrol activates SIRT1 via AMPK and in the same way as SIRT1, AMPK regulates metabolism during energy stress, to restore cellular homeostasis (Hardie et al. 2012; Park et al. 2012). AMPK inhibits anatomical pathways (synthesis of lipids, proteins, etc.) and activates catabolism (autophagy, beta-oxidation ...). It has been shown that the activation of AMPK by resveratrol is the result of direct inhibition of PDE specific for cAMP (Park et al. 2012). The rise in the cAMP level then induces a signaling cascade involving the exchange of protein directly activated by cAMP 1 (Epac1) and Ca2+-calmodulin-dependent-protein kinase kinase beta (CamKKbeta). In increasing the NAD+/NADH ratio, AMPK activates SIRT1 which deacetylates and activates the PGC-1α, leading to an increase in oxidative phosphorylation and the synthesis of new mitochondria. Several studies have shown that the activation of PGC-1α by the SIRT1 (Canto et al. 2010; El-Khamisy et al. 2005) or inhibition of hypoxia-inducible factor 1-alpha (HIF-1 alpha) by AMPK (Faubert et al. 2013) blocks aerobic glycolysis. It can thus be supposed that the activation of the AMPK-SIRT1 axis by RSV induces a metabolic reprogramming preventing tumor development. Recently a study conducted by Blanquer-Rossello et al. has described the ability of RSV to target cancer cell metabolism and enhance chemotherapy effects by promoting mitochondrial electron transport chain overload and, ultimately, increasing ROS production (Blanquer-Rossello et al. 2017).
Impact of resveratrol on obesity, diabetes and cardiovascular diseases
Obesity is a global societal health problem and it has been associated with high risks of contracting a number of diseases including type 2 diabetes and cardiovascular diseases. Its development has been strongly associated with mitochondrial imbalances (de Mello et al. 2018) which lead the development of many cardiac diseases such as Hypertension, ischemia-reperfusion injury, diabetes, cardiac hypertrophy, and heart failure and atherosclerosis, due to the uncontrolled production of ROS (Siasos et al. 2018).
Recent studies on obese animals showed that RSV treatment increased mitochondrial biogenesis and function, improvement of energy homeostasis, and a decrease in oxidative damage (de Oliveira et al. 2016; Zhang et al. 2017). These effects can be accredited to the activation of the AMPK/SIRT1/PGC-1α axis. It was confirmed that most cases of diabetes are due to oxidative stress caused especially by sedentary behavior and an undesirable eating routine. RSV counteract mitochondrial dysfunction by activating Nrf2 (nuclear respiratory factor 2) (Bonnefont-Rousselot 2016). RSV has been shown to activate Nrf2 in both mice heart (RSV at 10 mg/kg/day for 3 days before exposure to lipopolysaccharides (LPS) ) and human neonatal cardiomyocyte (HCM) cells (that were treated with RSV at 3 µM for 4 h before exposure to LPS) (Enkui Hao et al. 2013).
Several human studies have demonstrated that RSV is effective for people with diabetes, overweight, and/or hypertension (Campbell et al. 2019; Sahebkar 2013). RSV restores blood sugar levels due to its ability to protect against high production of glucose in diabetic patients and eliminate free radicals, which can damage mitochondria (Naudi et al. 2012; Zhang et al. 2010). RSV promotes the loss of body fat and lowers cholesterol and triglyceride levels (Zhang et al. 2010). Treatment with RSV at concentrations of 30 mg/kg for 10 weeks significantly reduced body weight, liver weight, subcutaneous, and epididymal adipose tissue weight in obese mice (Chih-Chun Chang 2016). Mitochondrial dysfunction also plays an important role in the pathophysiology of cardiovascular diseases (Bayeva et al. 2013). In addition, RSV reduces the levels of different markers of risk of cardiovascular disease, which is particularly important with diabetes (Satya Dash 2013). A very recent study showed that RSV treatment (500 mg/day for 4 weeks) (Table 2) upregulated PPAR-γ and SIRT1 and had beneficial effects on total antioxidant capacity (TCA), high density lipoprotein-cholesterol (HDL-c) and malondialdehyde (MDA) levels in patients with T2DM, coronary artery disease, hyperlipidemia with other cardiovascular risks (Hoseini et al. 2019). A meta-analysis indicated that treatment with RSV (≥ 150 mg/day) lowered systolic blood pressure with no effect on diastolic blood pressure (Liu et al. 2015). Timmers et al. showed in 2011 that RSV mimics the effect of a low-calorie diet (Timmers et al. 2011) and thus facilitates weight loss. Thus RSV is an excellent solution even for people who cannot do physical exercises like those with a physical disability, heart disease, neurological problem, etc. and all too often suffer from overweight or diabetes.
Recently research findings demonstrated that RSV can control the development of T2DM via SIRT1 by regulating the expression of mitochondrial genes involving in biogenesis, β-cells differentiation, and lipid metabolism (Cao et al. 2018). Diao et al. revealed that RSV ameliorates mitochondrial function and consequently improves cardiac function in diabetic rats, and uncoupling protein 2 (UCP2) was involved in the protective effects of RSV on diabetic cardiomyopathy (DCM) (Diao et al. 2019).
The above results have proven that mitochondrial dysfunction was closely linked to the pathophysiology of T2DM and clarified the ability of RSV to prevent and control obesity, diabetes, and cardiovascular diseases.
Antioxidant effects of resveratrol
As described above, mitochondrial dysfunction arises as a consequence of the excessive production of ROS which oxidizes cellular lipids, proteins, and DNA (Esra Birben et al. 2012). One of the antioxidative effects of RSV is its ability to directly capture free radicals (scavenger effect of RSV) (Leonard et al. 2003).
In their study, Li et al. described that RSV induces the expression of mitochondrial genes and manganese superoxide dismutase (Mn-SOD) via SIRT1 and mitochondrial biogenesis signaling pathways, thus inhibits ROS production in cardiomyocytes (Li et al. 2013; Ning Xia et al. 2017).
RSV is capable to induce antioxidant enzymes such as Mn-SOD involved in the dismutation of the superoxide ion (O2, very reactive radical species) in oxygen and hydrogen peroxide (Campbell et al. 2019; Nakata et al. 2012). RSV can also prevent oxidation of low-density lipoprotein cholesterol (LDL-C) by chelating copper and capturing ROS (Andrea Markus 2008). The activity of deacetylase is required for the activation of the PGC-1α co-activator. Once activated, PGC-1α may interact with transcription factors such as Nrf1/Nrf2 to induce transcription of antioxidant genes such as quinone oxidoreductase 1, γ-glutamylcysteine synthetase, or heme oxygenase-1 (Alissa and Ferns 2012). It has been shown that the exposure of blood platelets to oxidative stress, for example, the exposure of the cells to peroxynitrite (ONOO-), induces the inhibition of their essential activities (Pawel Nowak 2001). According to Olas and Wachowicz, the antioxidant effect of RSV is due to 4’-hydroxyl group but also of the meta configuration of the two hydroxyl groups of the first benzene group (Olas et al. 2006).
Briefly, RSV is an effective sensor of free radicals (ROS), a lipid peroxidation reducing agent, and an inhibitor of protein oxidation through regulating mitochondrial biogenesis.
Neuroprotective effects of resveratrol
RSV also appears as a neuroprotective agent because of its anti-inflammatory and antioxidant effects. Previous studies have shown that RSV can be used in treatment of neurodegenerative and neurological diseases and/or disorders (Parkinson’s disease, Alzheimer disease, epilepsy, etc.) via promoting activation of AMPK and SIRT1 ensuing PGC-1α activation which lead to an increase of mitochondrial biogenesis and a decrease of ROS thus relieve mitochondrial dysfunction a characteristic of neurodegenerative diseases (Anna Ferretta et al. 2014; Chuang et al. 2019). Additionally, through AMPKα-SIRT1 activation, RSV decreases nuclear factor-κB (NF-κB) signaling (Tian et al. 2016). The activation of NF-κB in glial and immune cells initiates inflammatory processes thus RSV supplementation could decrease the inflammatory level (Tian et al. 2016; Vingtdeux et al. 2010).
RSV has been revealed to decrease plaque formation related to neurodegeneration (Karuppagounder et al. 2009). RSV has also been shown to significantly prevent intracerebroventricular streptozotocin (ICV)-induced cognitive impairment in rats (Monisha Sharma 2002). In rodent models with similar symptoms as those of Huntington’s disease, RSV has expressively improved cognitive and motor impairment thanks to its inhibitory activity of COX-1 (Puneet Kumar 2006). Another study demonstrated that RSV prevents memory impairment induced by diabetes and the increase in Acetylcholinesterase (AChE) activity (Schmatz et al. 2009).
Resveratrol and erectile dysfunction
Erectile dysfunction (ED) is an inability to have and maintain an erection of sufficient quality to obtain satisfactory sexual activity. ED may be a reflection of a pathological condition, such as diabetes, a hormonal disease, a neurological disorder, a heart or vascular condition (Boydens et al. 2016). In addition, ED is common among heavy smokers, regular drug users and alcoholics (Zhong et al. 2019). RSV, due to its strong antioxidative effects, was able to preserve the important metabolic pathways involved in erectile function (Faid et al. 2015). Supplementation of RSV (10 mg/kg/day for 28 days) to nicotine-treated Wistar rats reversed contractile activity of the bladder and corpus cavernosum strips impairments (Toklu 2010). Other reports have shown that RSV downregulated the expression of PDE5, p53 and FOXO3a, which regulate apoptosis and oxidative stress and contrarily increased blood testosterone level, expression of nNOS, eNOS, cavernous cyclic guanosine monophosphate (cGMP) and SIRT1, an activator of mitochondrial biogenesis and thus, restored erectile function in experimental animals with diabetes (Bai and An 2015; Sener et al. 2018; Shin et al. 2008; Wen Yu et al. 2013). As we have described above, physiological diseases are closely linked to mitochondrial dysfunction. Therefore, supplementation with RSV may help not only to improve these pathologies but also erectile dysfunction as well as infertility.
Resveratrol, sirtuins and aging
Throughout aging, the decline of mitochondrial activity may impair the production of adequate adenosine triphosphate (ATP) for homeostasis, eventually initiating apoptosis (Biala et al. 2015), involving to the beginning and progression of aging and aging-related disorders. Mitochondrial dysfunction results in the decline of mitochondrial biogenesis and is expected to be the main cause of aging (Lopez-Lluch et al. 2008). Researchers demonstrated that RSV has a lifespan extension potential in various animal models (Kasiotis et al. 2013). An experiment carried by Muhammad and Allam (2018) demonstrated that RSV supplementation increased the endurance of aged mice in comparison with the aged control group (Muhammad and Allam 2018).
The anti-aging activity of resveratrol is clarified by its action on SIRT1 a molecule of longevity that can reverse the decline of mitochondrial biogenesis that occurs in senescent cells (Lagouge et al. 2006). The sirtuins are NAD-dependent deacetylase proteins able to modulate the transcriptional activity of many genes. Some of the best-characterized effects of SIRT1 include increased oxidative stress resistance and altered metabolism mediated by changes in the activity of the transcriptional factor FOXO (Anne Brunet 2004), suppression of NF-κB-dependent inflammatory responses (Fan Yeung et al. 2004) and the promotion of gluconeogenesis, fatty acid oxidation and mitochondrial biogenesis, through PGC-1α (Lagouge et al. 2006). According to Zhang et al. (2017), the treatment of intrauterine growth retarded suckling piglets with RSV increased hepatic SIRT1 activity, aided by increased NAD + concentration. Moreover, PGC-1α mRNA and its protein levels were significantly induced upon resveratrol administration. This was resembled by the increased mRNA abundance of downstream target genes associated with mitochondrial biogenesis (Zhang et al. 2017). The activation of SIRT1 by RSV is substrate-dependent and many of its effects are consistent with the modulation of its target on genes (Purushotham et al. 2009). As described above resveratrol may grant various health benefits that may increase longevity to mammals and humans.
Conclusion
Resveratrol has been shown to improve mitochondrial function and prevention and/or treatment human physiological diseases through its ability to activate AMPK, PGC-1α, and SIRT1, the main drivers of mitochondrial biogenesis. One of the ways used to deal with the consequences of high-fat diet intake such as diabetes, obesity, cardiovascular diseases among others is to exercise regularly and confine the intake of calories which would result in a reduction in endoplasmic reticulum stress. In this review, we have described how RSV seems to mimic numerous biochemical effects of restriction of calories. Interestingly, several studies have shown that activation of PGC-1α by SIRT1 blocks aerobic glycolysis. Thus, the activation of the AMPK-PGC-1α-SIRT1 by RSV may induce metabolic reprogramming hence preventing or improving metabolic diseases. However, the research for the “optimal dose” is still relevant. Resveratrol has multiple action targets and beneficial effects that are however controversial in some studies. Most of the recent studies have been done on cell cultures or animal models but very little on a human scale. The very low bioavailability of resveratrol taken orally is one of the major limiting factors. Therefore, future clinical trials and meta-analyses should focus on obtaining more accurate and consensual data on resveratrol.
References
Alissa EM, Ferns GA (2012) Functional foods and nutraceuticals in the primary prevention of cardiovascular diseases. J Nutr Metab 2012:569486. https://doi.org/10.1155/2012/569486
Amarendranath Choudhury RB, Adapa D, Chakraborty I, Banerjee TS, Vana DR (2018) Understanding the role of resveratrol in major neurological and lifestyle diseases: an insight into molecular mechanisms and druggability. Pharm Bioprocess 6(2):064–083
Andrea Markus M (2008) Resveratrol in prevention and treatment of common clinical conditions of aging. Clin Interv Aging 3(2):331–339
Anna Ferretta AG, Tanzarella P, Piccoli C, Capitanio N, Nico B, Annese T, Paola MD, Dell’Aquila C, De Mari M, Ferranini E, Bonifati V, Pacelli C, Cocco T (2014) Effect of resveratrol on mitochondrial function: Implications in parkin-associated familiar Parkinson’s disease. Biochim Biophys Acta 1842:902–915. https://doi.org/10.1016/j.bbadis.2014.02.010
Anne Brunet LBS, Sturgill JF, Chua KF, Greer PL, Lin Y, Tran H, Ross SE, Mostoslavsky R, Haim Y, Cohen LS, Hu H-L, Cheng MP, Jedrychowski SP, Gygi DA, Sinclair FW, Alt ME, Greenberg (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303:2011–2015
Arslan G, Yilmaz N (2013) Determination of trans-resveratrol levels in different fruits, vegetables and their skin by HPLC. Asian J Chem 25(3):1225–1228. https://doi.org/10.14233/ajchem.2013.12599
Bai Y, An R (2015) Resveratrol and sildenafil synergistically improve diabetes-associated erectile dysfunction in streptozotocin-induced diabetic rats. Life Sci 135:43–48. https://doi.org/10.1016/j.lfs.2015.04.020
Baur JA, Sinclair DA (2006) Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5(6):493–506. doi:https://doi.org/10.1038/nrd2060
Bayeva M, Gheorghiade M, Ardehali H (2013) Mitochondria as a therapeutic target in heart failure. J Am Coll Cardiol 61(6):599–610. https://doi.org/10.1016/j.jacc.2012.08.1021
Biala AK, Dhingra R, Kirshenbaum LA (2015) Mitochondrial dynamics: orchestrating the journey to advanced age. J Mol Cell Cardiol 83:37–43. https://doi.org/10.1016/j.yjmcc.2015.04.015
Blanquer-Rossello MD, Hernandez-Lopez R, Roca P, Oliver J, Valle A (2017) Resveratrol induces mitochondrial respiration and apoptosis in SW620 colon cancer cells. Biochim Biophys Acta Gen Subj 1861(2):431–440. https://doi.org/10.1016/j.bbagen.2016.10.009
Bonnefont-Rousselot D (2016) Resveratrol and cardiovascular diseases. Nutrients. https://doi.org/10.3390/nu8050250
Boydens C, Pauwels B, Vanden Daele L, Van de Voorde J (2016) Protective effect of resveratrol and quercetin on in vitro-induced diabetic mouse corpus cavernosum. Cardiovasc Diabetol 15:46. https://doi.org/10.1186/s12933-016-0366-9
Burns J, Yokota T, Ashihara H, Lean MEJ, Crozier A (2002) Plant foods and herbal sources of resveratrol. J Agric Food Chem 50:3337–3340
Callemien D, Jerkovic V, Rozenberg R, Collin S (2005) Hop as an interesting source of resveratrol for brewers: optimization of the extraction and quantitative study by liquid chromatography/atmospheric pressure chemical ionization tandem mass spectrometry. J Agric Food Chem 53:424–429
Campbell CL, Yu R, Li F, Zhou Q, Chen D, Qi C et al (2019) Modulation of fat metabolism and gut microbiota by resveratrol on high-fat diet-induced obese mice. Diabetes Metab Syndr Obes 12:97–107. https://doi.org/10.2147/DMSO.S192228
Canto C, Jiang LQ, Deshmukh AS, Mataki C, Coste A, Lagouge M et al (2010) Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab 11(3):213–219. https://doi.org/10.1016/j.cmet.2010.02.006
Cao MM, Lu X, Liu GD, Su Y, Li YB, Zhou J (2018) Resveratrol attenuates type 2 diabetes mellitus by mediating mitochondrial biogenesis and lipid metabolism via Sirtuin type 1. Exp Ther Med 15(1):576–584. https://doi.org/10.3892/etm.2017.5400
Catalgol B, Batirel S, Taga Y, Ozer NK (2012) Resveratrol: French paradox revisited. Front Pharmacol 3:141. https://doi.org/10.3389/fphar.2012.00141
Chalimoniuk M, Głowacka J, Zabielna A, Eckert A, Strosznajder JB (2006) Nitric oxide alters arachidonic acid turnover in brain cortex synaptoneurosomes. Neurochem Int 48:1–8. https://doi.org/10.1016/j.neuint.2005.08.011
Chen M, Yi L, Jin X, Liang X, Zhou Y, Zhang T, Xie Q, Zhou X, Chang H, Fu Y, Zhu J, Zhang Q, Mi M (2013) Resveratrol attenuates vascular endothelial inflammation by inducing autophagy through the cAMP signaling pathway. Autophagy 9(12):2033–2045. https://doi.org/10.4161/auto.26336
Chih-Chun Chang K-YL, Peng K-Y, Day Y-J, Hung L-M (2016) Resveratrol exerts anti-obesity effects in high-fat diet obese mice and displays differential dosage effects on cytotoxicity, differentiation, and lipolysis in 3T3-L1 cells. Endocr J 63(2):169–178
Chiva-Blanch G, Urpi-Sarda M, Rotches-Ribalta M, Zamora-Ros R, Llorach R, Lamuela-Raventos RM, Andres-Lacueva C (2011) Determination of resveratrol and piceid in beer matrices by solid-phase extraction and liquid chromatography-tandem mass spectrometry. J Chromatogr A 1218(5):698–705. https://doi.org/10.1016/j.chroma.2010.12.012
Chuang YC, Chen SD, Hsu CY, Chen SF, Chen NC, Jou SB (2019) Resveratrol promotes mitochondrial biogenesis and protects against seizure-induced neuronal cell damage in the hippocampus following status epilepticus by activation of the PGC-1alpha signaling pathway. Int J Mol Sci. https://doi.org/10.3390/ijms20040998
Counet C, Callemien D, Collin S (2006) Chocolate and cocoa: new sources of trans-resveratrol and trans-piceid. Food Chem 98(4):649–657. https://doi.org/10.1016/j.foodchem.2005.06.030
Cui X, Jin Y, Hofseth AB, Pena E, Habiger J, Chumanevich A et al (2010) Resveratrol suppresses colitis and colon cancer associated with colitis. Cancer Prev Res (Phila) 3(4):549–559. https://doi.org/10.1158/1940-6207.CAPR-09-0117
Cvejic JM, Djekic SV, Petrovic AV, Atanackovic MT, Jovic SM, Brceski ID, Gojkovic-Bukarica LC (2010) Determination of trans- and cis-resveratrol in Serbian commercial wines. J Chromatogr Sci 48:229–234
Diao J, Wei J, Yan R, Fan G, Lin L, Chen M (2019) Effects of resveratrol on regulation on UCP2 and cardiac function in diabetic rats. J Physiol Biochem 75(1):39–51. https://doi.org/10.1007/s13105-018-0648-7
de Oliveira MR, Nabavi SF, Manayi A, Daglia M, Hajheydari Z, Nabavi SM (2016) Resveratrol and the mitochondria: from triggering the intrinsic apoptotic pathway to inducing mitochondrial biogenesis, a mechanistic view. Biochim Biophys Acta 1860(4):727–745. https://doi.org/10.1016/j.bbagen.2016.01.017
de Mello AH, Costa AB, Engel JDG, Rezin GT (2018) Mitochondrial dysfunction in obesity. Life Sci 192:26–32. https://doi.org/10.1016/j.lfs.2017.11.019
El-Khamisy SF, Saifi GM, Weinfeld M, Johansson F, Helleday T, Lupski JR, Caldecott KW (2005) Defective DNA single-strand break repair in spinocerebellar ataxia with axonal neuropathy-1. Nature 434(7029):108–113. https://doi.org/10.1038/nature03314
Enkui Hao FL, Chen Y, Zhang H, Cong X, Shen X, Su G (2013) Resveratrol alleviates endotoxin-induced myocardial toxicity via the Nrf2 transcription factor. PLoS ONE 8(7):e69452. https://doi.org/10.1371/journal.pone.0069452.g001
Esra Birben UMS, Sackesen C, Erzurum S, Kalayci O (2012) Oxidative stress and antioxidant defense. WAO J 5:9–19
Fabian Sanchis-Gomar JLG-G, Gómez-Cabrera MC, Pallardó FV (2014) Mitochondrial biogenesis in health and disease. Molecular and therapeutic approaches. Curr Pharm Des 20:5619–5633
Faid I, Al-Hussaini H, Kilarkaje N (2015) Resveratrol alleviates diabetes-induced testicular dysfunction by inhibiting oxidative stress and c-Jun N-terminal kinase signaling in rats. Toxicol Appl Pharmacol 289(3):482–494. https://doi.org/10.1016/j.taap.2015.10.012
Fan Yeung JE, Hoberg CS, Ramsey MD, Keller DR, Jones, Mayo MW (2004) Modulation of NF-jB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23:2369–2380
Faubert B, Boily G, Izreig S, Griss T, Samborska B, Dong Z et al (2013) AMPK is a negative regulator of the Warburg effect and suppresses tumor growth in vivo. Cell Metab 17(1):113–124. https://doi.org/10.1016/j.cmet.2012.12.001
Gogvadze V, Orrenius S, Zhivotovsky B (2008) Mitochondria in cancer cells: what is so special about them? Trends Cell Biol 18(4):165–173. https://doi.org/10.1016/j.tcb.2008.01.006
Haohao Z, Guijun Q, Juan Z, Wen K, Lulu C (2015) Resveratrol improves high-fat diet induced insulin resistance by rebalancing subsarcolemmal mitochondrial oxidation and antioxidantion. J Physiol Biochem 71(1):121–131. https://doi.org/10.1007/s13105-015-0392-1
Hardie DG, Ross FA, Hawley SA (2012) AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 13(4):251–262. https://doi.org/10.1038/nrm3311
Hoseini A, Namazi G, Farrokhian A, Reiner Ž, Aghadavod E, Bahmani F, Asemi Z (2019) The effects of resveratrol on metabolic status in patients with type 2 diabetes mellitus and coronary heart disease. Food Funct. https://doi.org/10.1039/c9fo01075k
Hurst WJ, Glinski JA, Miller KB, Apgar J, Davey MH, Stuart DA (2008) Survey of the trans-resveratrol and trans-piceid content of cocoa-containing and chocolate products. J Agric Food Chem 56:8374–8378
Jubilee R, Stewart MCA (2003) Resveratrol: a candidate nutritional substance for prostate cancer prevention. J Nutr 133:2440–2443
Karuppagounder SS, Pinto JT, Xu H, Chen HL, Beal MF, Gibson GE (2009) Dietary supplementation with resveratrol reduces plaque pathology in a transgenic model of Alzheimer’s disease. Neurochem Int 54(2):111–118. https://doi.org/10.1016/j.neuint.2008.10.008
Kasiotis KM, Pratsinis H, Kletsas D, Haroutounian SA (2013) Resveratrol and related stilbenes: their anti-aging and anti-angiogenic properties. Food Chem Toxicol 61:112–120. https://doi.org/10.1016/j.fct.2013.03.038
Khurana S, Venkataraman K, Hollingsworth A, Piche M, Tai TC (2013) Polyphenols: benefits to the cardiovascular system in health and in aging. Nutrients 5(10):3779–3827. https://doi.org/10.3390/nu5103779
Kisbenedek A, Szabo S, Polyak E, Breitenbach Z, Bona A, Mark L, Figler M (2014) Analysis oftrans-resveratrol in oilseeds by high-performance liquid chromatography. Acta Alimentaria 43(3):459–464. https://doi.org/10.1556/AAlim.43.2014.3.13
Kjaer TN, Thorsen K, Jessen N, Stenderup K, Pedersen SB (2015) Resveratrol ameliorates imiquimod-induced psoriasis-like skin inflammation in mice. PLoS ONE 10(5):e0126599. https://doi.org/10.1371/journal.pone.0126599
Krek DA, a. W (2006) SIRT1: linking adaptive cellular responses to aging-associated changes in organismal physiology. Physiology 21:404–410. https://doi.org/10.1152/physiol.00031.2006
Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Auwerx J (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell 127(6):1109–1122. https://doi.org/10.1016/j.cell.2006.11.013
Langkake P (1976) The production of resveratrol by Wis vinifera and other members of the Vitaceae as a response to infection or injury. Physiol Plant Pathol 9:77–86
Lee JA, Ha SK, Cho E, Choi I (2015) Resveratrol as a bioenhancer to improve anti-inflammatory activities of apigenin. Nutrients 7(11):9650–9661. https://doi.org/10.3390/nu7115485
Leonard SS, Xia C, Jiang B-H, Stinefelt B, Klandorf H, Harris GK, Shi X (2003) Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochem Biophys Res Commun 309(4):1017–1026. https://doi.org/10.1016/j.bbrc.2003.08.105
Li YG, Zhu W, Tao JP, Xin P, Liu MY, Li JB, Wei M (2013) Resveratrol protects cardiomyocytes from oxidative stress through SIRT1 and mitochondrial biogenesis signaling pathways. Biochem Biophys Res Commun 438(2):270–276. https://doi.org/10.1016/j.bbrc.2013.07.042
Liguori I, Russo G, Curcio F, Bulli G, Aran L, Della-Morte D, Abete P (2018) Oxidative stress, aging, and diseases. Clin Interv Aging 13:757–772. https://doi.org/10.2147/CIA.S158513
Liu Y, Ma W, Zhang P, He S, Huang D (2015) Effect of resveratrol on blood pressure: a meta-analysis of randomized controlled trials. Clin Nutr 34(1):27–34. https://doi.org/10.1016/j.clnu.2014.03.009
Lopez-Lluch G, Irusta PM, Navas P, de Cabo R (2008) Mitochondrial biogenesis and healthy aging. Exp Gerontol 43(9):813–819. https://doi.org/10.1016/j.exger.2008.06.014
Lugrin J, Rosenblatt-Velin N, Parapanov R, Liaudet L (2014) The role of oxidative stress during inflammatory processes. Biol Chem 395(2):203–230. https://doi.org/10.1515/hsz-2013-0241
Magyar K, Halmosi R, Palfi A, Feher G, Czopf L, Fulop A, Szabados E (2012) Cardioprotection by resveratrol: A human clinical trial in patients with stable coronary artery disease. Clin Hemorheol Microcirc 50(3):179–187. https://doi.org/10.3233/CH-2011-1424
Monisha Sharma YKG (2002) Chronic treatment with trans resveratrol prevents intracerebroventricular streptozotocin induced cognitive impairment and oxidative stress in rats. Life Sci 71:2489–2498
Muhammad MH, Allam MM (2018) Resveratrol and/or exercise training counteract aging-associated decline of physical endurance in aged mice; targeting mitochondrial biogenesis and function. J Physiol Sci 68(5):681–688. https://doi.org/10.1007/s12576-017-0582-4
Nakahira K, Haspel JA, Rathinam VA, Lee SJ, Dolinay T, Lam HC,.. . Choi AM (2011) Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat Immunol 12(3):222–230. doi:https://doi.org/10.1038/ni.1980
Nakata R, Takahashi S, Inoue H (2012) Recent advances in the study on resveratrol. Biol Pharm Bull 35(3):273–279. https://doi.org/10.1248/bpb.35.273
Naudi A, Jove M, Ayala V, Cassanye A, Serrano J, Gonzalo H et al (2012) Cellular dysfunction in diabetes as maladaptive response to mitochondrial oxidative stress. Exp Diabetes Res 2012:696215. https://doi.org/10.1155/2012/696215
Newman LE, Shadel GS (2018) Pink1/Parkin link inflammation, mitochondrial stress, and neurodegeneration. J Cell Biol 217(10):3327–3329. https://doi.org/10.1083/jcb.201808118
Ning Xia AD, Förstermann U, Li H (2017) Antioxidant effects of resveratrol in the cardiovascular system. Br J Pharmacol 174:1633–1646. https://doi.org/10.1111/bph.13492
Olas B, Nowak P, Ponczek MP, Wachowicz B (2006) Resveratrol, a natural phenolic compound may reduce carbonylation proteins induced by peroxynitrite in blood platelets. Gen Physiol Biophys 25:215–222
Park SJ, Ahmad F, Philp A, Baar K, Williams T, Luo H, Chung JH (2012) Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 148(3):421–433. https://doi.org/10.1016/j.cell.2012.01.017
Pawel Nowak BW (2001) Studies on pig blood platelet responses to peroxynitrite action. Platelets 12:376–381. https://doi.org/10.1080/09537100120068161
Pietrocola F, Marino G, Lissa D, Vacchelli E, Malik SA, Niso-Santano M, Kroemer G (2012) Pro-autophagic polyphenols reduce the acetylation of cytoplasmic proteins. Cell Cycle 11(20):3851–3860. https://doi.org/10.4161/cc.22027
Puneet Kumar SSVP, Naidu PS, Kumar A (2006) Effect of resveratrol on 3-nitropropionic acid-induced biochemical and behavioural changes: possible neuroprotective mechanisms. Behav Pharmacol 17:485–492
Purushotham A, Schug TT, Xu Q, Surapureddi S, Guo X, Li X (2009) Hepatocyte-specific deletion of SIRT1 alters fatty acid metabolism and results in hepatic steatosis and inflammation. Cell Metab 9(4):327–338. https://doi.org/10.1016/j.cmet.2009.02.006
Ragab AS, Van Fleet J, Jankowski B, Park J-H, Bobzin SC (2006) Detection and quantitation of resveratrol in tomato fruit (Lycopersicon esculentum Mill.). J Agric Food Chem 54:7175–7179
Rauf A, Imran M, Suleria HAR, Ahmad B, Peters DG, Mubarak MS (2017) A comprehensive review of the health perspectives of resveratrol. Food Funct 8(12):4284–4305. https://doi.org/10.1039/c7fo01300k
Reddivari L, Charepalli V, Radhakrishnan S, Vadde R, Elias RJ, Lambert JD, Vanamala JK (2016) Grape compounds suppress colon cancer stem cells in vitro and in a rodent model of colon carcinogenesis. BMC Complement Altern Med 16:278. https://doi.org/10.1186/s12906-016-1254-2
Romero-Perez AI, Ibern-Gomez M, Lamuela-Raventós RM, de la Torre-Boronat MC (1999) Piceid, the major resveratrol derivative in grape juices. J Agric Food Chem 47:1533–1536
Romero-Pérez AI, Lamuela-Raventós RM, Andrés-Lacueva C, de la Torre-Boronat MC (2001) Method for the quantitative extraction of resveratrol and piceid isomers in grape berry skins. Effect of powdery mildew on the stilbene content. J Agric Food Chem 49:210–215
Sahebkar A (2013) Effects of resveratrol supplementation on plasma lipids: a systematic review and meta-analysis of randomized controlled trials. Nutr Rev 71(12):822–835. https://doi.org/10.1111/nure.12081
Saiko P, Szakmary A, Jaeger W, Szekeres T (2008) Resveratrol and its analogs: defense against cancer, coronary disease and neurodegenerative maladies or just a fad? Mutat Res 658(1–2):68–94. https://doi.org/10.1016/j.mrrev.2007.08.004
Sanchez-Fidalgo S, Cardeno A, Villegas I, Talero E, de la Lastra CA (2010) Dietary supplementation of resveratrol attenuates chronic colonic inflammation in mice. Eur J Pharmacol 633(1–3):78–84. https://doi.org/10.1016/j.ejphar.2010.01.025
Sanders TH, McMichael RW Jr, Hendrix KW (2000) Occurrence of resveratrol in edible peanuts. J Agric Food Chem 48:1243–1246
Satya Dash CX, Morgantini C, Szeto L, Gary F, Lewis (2013) High-dose resveratrol treatment for 2 weeks inhibits intestinal and hepatic lipoprotein production in overweight/obese men. Arterioscler Thromb Vasc Biol 33:2895–2901. https://doi.org/10.1161/atvbaha.113.302342/-/dc1
Schmatz R, Mazzanti CM, Spanevello R, Stefanello N, Gutierres J, Correa M, Morsch VM (2009) Resveratrol prevents memory deficits and the increase in acetylcholinesterase activity in streptozotocin-induced diabetic rats. Eur J Pharmacol 610(1–3):42–48. https://doi.org/10.1016/j.ejphar.2009.03.032
Sebastia N, Montoro A, Léon Z, Soriano JM (2017) Searching trans-resveratrol in fruits and vegetables: a preliminary screening. J Food Sci Technol 54(3):842–845. https://doi.org/10.1007/s13197-016-2474-7
Sener TE, Tavukcu HH, Atasoy BM, Cevik O, Kaya OT, Cetinel S, Sener G (2018) Resveratrol treatment may preserve the erectile function after radiotherapy by restoring antioxidant defence mechanisms, SIRT1 and NOS protein expressions. Int J Impot Res 30(4):179–188. https://doi.org/10.1038/s41443-018-0042-6
Shimada K, Crother TR, Karlin J, Dagvadorj J, Chiba N, Chen S, Arditi M (2012) Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 36(3):401–414. https://doi.org/10.1016/j.immuni.2012.01.009
Shin S, Jeon JH, Park D, Jang M-J, Choi JH, Choi B-H, Kim Y-B (2008) trans-resveratrol relaxes the corpus cavernosum ex vivo and enhances testosterone levels and sperm quality in vivo. Arch Pharmacal Res 31(1):83–87. https://doi.org/10.1007/s12272-008-1124-7
Siasos G, Tsigkou V, Kosmopoulos M, Theodosiadis D, Simantiris S, Tagkou NM, Papavassiliou AG (2018) Mitochondria and cardiovascular diseases-from pathophysiology to treatment. Ann Transl Med 6(12):256. https://doi.org/10.21037/atm.2018.06.21
Siekevitz P (1957) Powerhouse of the cell. Sci Am 197(1):131–140
Siemann EH, a. C. L. L (1992) Concentration of the phytoalexin resveratrol in wine. Am J Enol Vitic 43:49–52
Simao F, Matte A, Pagnussat AS, Netto CA, Salbego CG (2012) Resveratrol preconditioning modulates inflammatory response in the rat hippocampus following global cerebral ischemia. Neurochem Int 61(5):659–665. https://doi.org/10.1016/j.neuint.2012.06.009
Sliter DA, Martinez J, Hao L, Chen X, Sun N, Fischer TD, Youle RJ (2018) Parkin and PINK1 mitigate STING-induced inflammation. Nature 561(7722):258–262. https://doi.org/10.1038/s41586-018-0448-9
Sporn MB (2011) The big C - for chemoprevention. Nature 471(11):S10–S11
Tian Y, Ma J, Wang W, Zhang L, Xu J, Wang K, Li D (2016) Resveratrol supplement inhibited the NF-kappaB inflammation pathway through activating AMPKalpha-SIRT1 pathway in mice with fatty liver. Mol Cell Biochem 422(1–2):75–84. https://doi.org/10.1007/s11010-016-2807-x
Timmers S, Konings E, Bilet L, Houtkooper RH, van de Weijer T, Goossens GH, Schrauwen P (2011) Calorie restriction-like effects of 30 days of resveratrol supplementation on energy metabolism and metabolic profile in obese humans. Cell Metab 14(5):612–622. https://doi.org/10.1016/j.cmet.2011.10.002
Toklu H (2010) Resveratrol supplementation protects against chronic nicotine-induced oxidative damage and organ dysfunction in the rat urogenital system. Marmara Pharm J 1(14):29–40. https://doi.org/10.12991/201014462
Um JH, Park SJ, Kang H, Yang S, Foretz M, McBurney MW, Chung JH (2010) AMP-activated protein kinase-deficient mice are resistant to the metabolic effects of resveratrol. DIABETES 59(3):554–563. https://doi.org/10.2337/db09-0482
Vian MA, Tomao V, Gallet S, Coulomb PO, Lacombe JM (2005) Simple and rapid method for cis- and trans-resveratrol and piceid isomers determination in wine by high-performance liquid chromatography using Chromolith columns. J Chromatogr A 1085(2):224–229. https://doi.org/10.1016/j.chroma.2005.05.083
Vingtdeux V, Giliberto L, Zhao H, Chandakkar P, Wu Q, Simon JE, Marambaud P (2010) AMP-activated protein kinase signaling activation by resveratrol modulates amyloid-beta peptide metabolism. J Biol Chem 285(12):9100–9113. https://doi.org/10.1074/jbc.M109.060061
Wang Y, Catana F, Yang Y, Roderick R, Van Breemen RB (2002) An LC-MS method for analyzing total resveratrol in grape juice, cranberry juice, and in wine. J Agric Food Chem 50:431–435
Wang B, Sun J, Ma Y, Wu G, Tian Y, Shi Y, Le G (2014) Resveratrol preserves mitochondrial function, stimulates mitochondrial biogenesis, and attenuates oxidative stress in regulatory T cells of mice fed a high-fat diet. J Food Sci 79(9):H1823–H1831. https://doi.org/10.1111/1750-3841.12555
Weiskirchen SWR (2016) Resveratrol: how much wine do you have to drink to stay healthy? Adv Nutr 7(4):706–718. https://doi.org/10.3945/an.115.011627
Wen Yu ZW, Qiu X-F, Chen Y, Dai Y-T (2013) Resveratrol, an activator of SIRT1, restores erectile function in streptozotocin-induced diabetic rats. Asian J Androl 15(5):646–651. https://doi.org/10.1038/aja.2013.60
Yoshiyuki Kimura HOASA (1985) Effects of stilbenes on arachidonate metabolism in leukocytes. Biochim Biophys Acta 834:275–278
Zhang H, Li Y, Su W, Ying Z, Zhou L, Zhang L, Wang T (2017) Resveratrol attenuates mitochondrial dysfunction in the liver of intrauterine growth retarded suckling piglets by improving mitochondrial biogenesis and redox status. Mol Nutr Food Res. https://doi.org/10.1002/mnfr.201600653
Zhang Q, Raoof M, Chen Y, Sumi Y, Sursal T, Junger W, Hauser CJ (2010) Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464(7285):104–107. https://doi.org/10.1038/nature08780
Zhong BL, Xu YM, Xie WX, Lu J (2019) Cigarette smoking is significantly linked to sexual dissatisfaction in chinese heroin-dependent patients receiving methadone maintenance treatment. Front Psych 10:306. https://doi.org/10.3389/fpsyt.2019.00306
Acknowledgements
This review was supported State Key Laboratory of Food Science and Technology, Jiangsu, P.R. China, and National First-class discipline program of Food Science and Technology (JUFSTR201180201) and Collaborative Innovation Center of Food Safety and Quality Control in Jiangsu Province, Jiangnan University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
Angelo Uriho has no conflict of interest. Xue Tang has no conflict of interest. Guowei Le has no conflict of interest. Shaojun Yang has no conflict of interest. Yves Harimana has no conflict of interest. Steven Papy Ishimwe has no conflict of interest. Lu Yiping has no conflict of interest. Kai Zhang has no conflict of interest. Shuhua Ma has no conflict of interest. Bertrand Muhoza has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uriho, A., Tang, X., Le, G. et al. Effects of resveratrol on mitochondrial biogenesis and physiological diseases. ADV TRADIT MED (ADTM) 21, 1–14 (2021). https://doi.org/10.1007/s13596-020-00492-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-020-00492-0