Abstract
Many viruses first isolated in honey bees can also infect and circulate in wild bee species. While most common bee viruses are generally less prevalent in wild bees than honey bees, they are occasionally locally common. However, most studies have not assessed prevalence of viruses in honey bees and wild bees at the same sites, making it difficult to determine how viruses may spread across species. We addressed this by surveying the prevalence of six viruses in honey bees and bumble bees at sites across the Pacific Northwest, USA. These data were used to assess covariance in virus prevalence between bee groups across multiple ecotypes and to assess how virus prevalence in bumble bees correlated with prevalence in honey bees. We show deformed wing virus (DWV) and black queen cell virus (BQCV) were more common in honey bees than bumble bees, while Israeli acute paralysis virus (IAPV) was found at similar levels. Prevalence of DWV and IAPV were correlated with landscape context, with both most common at urban sites. However, BQCV prevalence in bumble bees was best predicted by BQCV prevalence in honey bees and by apiary density. Our study provides evidence that several viruses are found in both honey bees and wild bumble bees at the same sites, although prevalence was not necessarily correlated among the two groups. Understanding the prevalence and impacts of bee viruses on both managed and wild bees is key for disease mitigation and pollinator conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The pervasive spread of viruses infecting bees is a major factor implicated in global bee losses (Berényi et al. 2006; Singh et al. 2010; Cornman et al. 2012). While many common bee viruses were first identified in managed honey bees, subsequent studies found these pathogens in wild bees, wasps, and ants (Singh et al. 2010; Levitt et al. 2013; Grozinger and Flenniken 2019; Schläppi et al. 2019; Nanetti et al. 2021). Virus spread from honey bees to wild bees is thought to occur especially for wild species like bumble bees that broadly overlap in resource use with honey bees (Woolhouse 2001; Li et al. 2011; Fürst et al. 2014; Alger et al. 2019a, b; Purkiss and Lach 2019; Gusachenko et al. 2020). Pathogen transmission from wild bees to honey bees may also take place, especially if viruses are more common in wild species (McMahon et al. 2015).
Assessing pathogen prevalence across communities in which virus transmission occurs will allow for more effective management of bee viruses in wild and managed species. Specifically, understanding the factors that mediate virus prevalence in multiple bee species within the same communities can address complexities of multispecies disease transmission (Alger et al. 2019a, b; Dalmon et al. 2021). Moreover, a landscape and community perspective in epidemiology can aid in assessing factors that shape the distribution and dynamics of multi-host pathogens (Figure 1). For example, by investigating the prevalence of viruses across both managed honey bees and wild bumble bees at the same sites, studies can assess host co-occurrence patterns, identify drivers of heterogeneities among genera and regions, and quantify how mechanisms may co-vary or deviate across scales of biological organization (Johnson et al. 2015). Studies assessing pathogens in both wild and honey bees can also aid in inferring which viruses are transmitted between these groups and for creating management strategies that consider the potential for spread among and within different taxonomic groups (Peng et al. 2011; Dalmon et al. 2021).
Cultural and environmental factors also affect the accumulation of bee viruses (Dalmon et al. 2021). For example, a high density of managed pollinators can promote pathogen spread in wild bees (Otterstatter and Thomson 2008; Alger et al. 2019a, b). In cultivated landscapes managed for agricultural pollination, honey bees are often kept at high density and moved frequently to pollinate different crops, increasing exposure to multiple plant communities throughout a year. Similarly, honey bees are often kept at high density in urban landscapes by hobbyist beekeepers. As several common viruses are far more prevalent in managed compared to wild bees, pathogen spread from honey bees to wild bees is often inferred, yet studies have also confirmed virus replication in wild bumble bees (Tehel et al. 2016; Gusachenko et al. 2020). Common viruses have also been isolated from blooming flowers, suggesting the density and identity of pollinator resources may affect rates of virus transmission (Alger et al. 2019a, b; Gusachenko et al. 2020).
Here, we investigated local, community, and landscape factors affecting the prevalence and distribution of six viruses in managed honey bees and wild bumble bees in the Pacific Northwest USA (Figure 1). Our survey included agricultural sites supporting commercial apiaries, urban sites with a high density of hobbyist beekeepers, and other sites in natural and residential landscapes. We compared the prevalence of viruses in managed honey bees to wild bumble bees. We then used statistical models to assess effects of community and landscape factors on wild bumble bee virus prevalence. Finally, we used the survey data, contextualized with local information, to identify the strongest local and landscape predictors of virus co-occurrence in honey bees and bumble bees. By assessing the patterns and processes that mediate the distribution of six bee viruses in the Pacific Northwest, USA, we hoped to identify areas where multi-species transmission is likely and where disease assessment and control measures should be prioritized.
2 Materials and methods
2.1 Study area
The western parts of the study extent experience high levels of precipitation between October and April, which sustains dense evergreen forest and a heterogeneous mix of smaller farms in agricultural regions. A dramatic rain shadow restricts precipitation on the eastern side of the Cascade mountains where irrigated agriculture dominates around the Columbia River Basin. Steadily increasing elevation moving further east across Washington state towards Idaho permits increasing precipitation that allows very productive dryland agricultural systems comprised of larger farms that cultivate a mix of wheat, peas, lentils, canola, and other small grains.
2.2 Bee sampling
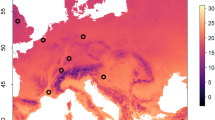
We searched for honey bees and bumble bees at 101 sites across the Pacific Northwest USA (Washington, Idaho, and Oregon States) to evaluate the prevalence of common bee viruses in both genera. We were able to sample honey bees at 86 sites and bumble bees at 86 sites, and both honey bees and bumble bees were collected at 71 sites across our study extent. At each sites, we hand netted honey bees near apiaries and bumble bees foraging on flowers. The bumble bees sampled included primarily Bombus vosnesenskii, B. centralis, B. huntii, B. griseocollis, B. nevadensis, and B. bifarious. All tested samples are shown in Figure 2, while the 71 sites where both bumble bees and honey bees were surveyed were included as replicates in our statistical analyses including both genera. Sampling occurred between 10 July and 29 August, 2020. Netted bees were deposited in 5-mL centrifuge tubes and euthanized in dry ice in the field and then stored at − 80 °C until RNA extraction. Nets were sanitized between sites.
Map of virus prevalence across study sites for Apis mellifera and Bombus spp. Pearson correlations were calculated based on the 71 sites where both honey bees and bumble bees were sampled. For the Pearson correlations between honey bee and bumble bee prevalence, “*” denotes a significance level 0.01 ≤ P < 0.05, while “ + ” represents a significance level 0.05 ≤ P < 0.10.
2.3 Bee virus assessment
Bee thoraxes were isolated from each bee for RNA extractions (Boncristiani et al. 2011). For each of the 71 sites with both honey bees and bumble bees, we pooled the 30 honey bees into three samples of 10 bees each. As bumble bees at each site may represent unique wild colonies, we did not pool bumble bees but completed RNA extraction using individual thoraxes. RNA was extracted from bee thoraxes by placing them in a centrifuge tube (2 mL), after which glass beads and TRIzol Reagent (Sigma-Aldrich, USA; 1 mL per tube) were added before homogenization in a Bead Ruptor Elite (OMNI International) for two 30-s intervals at 4 m/s and 6 m/s. After homogenization, 200 µL of chloroform was added, and tubes were vortexed for 15 s and put on ice for 15 min. After settling, samples were centrifuged at 14,000 gravity (g) for 20 min. The aqueous phase was then transferred into a fresh tube, and isopropanol (0.5 mL per mL of TRIzol reagent) was added and mixed by inverting the tube. Samples were left on ice for 40 min and then centrifuged at 14,000 × g for 10 min to precipitate and separate the RNA in a small pellet. RNA pellets were washed with 1 mL 75% ethanol twice and centrifuged at 7500 × g for 5 min. The ethanol was poured off, and pellets were allowed to air-dry before resuspending in 1 mL nuclease-free water and stored at − 80 °C. The concentration of the extracted RNA was measured on a NanoDrop 2000 c (Thermo Fisher, Waltham, MA).
Complementary DNA (cDNA) was synthesized through reverse transcriptase PCR. One microgram of RNA diluted in 16 μL of water and 4 μL cDNA iScript master mix (Bio-Rad, Hercules, CA, USA) were combined. The cDNA was synthesized in a thermocycler program: one cycle at 94 °C for 5 min followed by 56 °C for 30 s, and 72 °C for 45 s. cDNA products were stored at − 20 °C. We used multiplex RT-PCR, an efficient technique for simultaneous detection of different viruses, to detect the six viruses: (i) acute bee paralysis virus (ABPV), (ii) black queen cell virus (BQCV), (iii) chronic bee paralysis virus (CBPV), (iv) deformed wing virus (DWV), (v) Israeli acute paralysis virus (IAPV), and (vi) sacbrood virus (SBV) in a 25 μL reaction with 0.5 uL of each of the 10 mM oligonucleotide primers, 12.5 Taq mastermix (supplied with the enzyme), and 1.5 μL of cDNA. PCR was done with one cycle at 94 °C for 5 min followed by 35 cycles at 94 °C for 30 s, 56 °C for 30 s, and 72 °C for 45 s and a final extension cycle at 72 °C for 10 min. PCR products were analyzed by electrophoresis on a 1.5% agarose gel (100 V for 60 min).
2.4 Statistical analysis
We calculated virus prevalence for honey bees and bumble bees at each study site and measured correlations between the prevalence of each virus across these groups (n = 71) using Pearson correlation tests with the R package “Hmisc” (function rcorr). We also applied Fisher’s exact test to assess differences in prevalence of BQCV, DWV, and IAPV between honey bees and bumble bees. To assess spatial autocorrelation in virus prevalence, we fitted spatial correlograms to the honey bee and bumble bee data using the R package, ncf. Spatial correlograms depict the correlation (Moran’s I) between pairs of observations spaced at progressively longer lag distance.
We next assessed whether honey bee virus prevalence and landscape context affected virus prevalence in bumble bees for the three most common viruses identified (IAPV, DWV, BQCV) using binomial generalized linear regression models. These regression models were fit using base R function glm and included the causal factors of urban context (yes/no), agricultural with managed pollination (yes/no), and the prevalence of the virus in honey bees from the same site.
Finally, to test which local and landscape factors predicted BQCV co-occurrence in honey bees and bumble bees, we isolated a subset of 51 sites for which we obtained apiary management data from participating beekeepers. In these models, we included two additional factors, the number of active hives and the regional density (variable ranked 1 to 4) based on observations by local beekeepers, beekeeping associations, and the researchers (Pfeiffer and Crowder 2022).
3 Results
All six viruses surveyed were found in honey bees, and four of the six were also found in bumble bees (BQCV, CBPV, DWV, IAPV); ABPV and SBV were found only in honey bees. BQCV was the only virus that had a significant correlation in prevalence between honey bees and bumble bees at the same sites (Figure 2). Significant local spatial autocorrelation of proportional virus prevalence in wild bumble bees was also observed at the shortest 20 km (mean of class) distance class for each of the viruses, indicating that sites between 0 and 40 km were more similar than expected given the variation in the rest of dataset (Figure 3).
3.1 Virus prevalence, local pervasiveness, and co-occurrence
The most common honey bee viruses (BQCV, DWV) were observed at lower prevalence in bumble bees than honey bees (Figure 2) (BQCV: χ2 = 37.3, P < 0.01; DWV: χ2 = 35.7, P < 0.01), but IAPV had similar prevalence (χ2 = 1.62, P = 0.20). BQCV, DWV, and IAPV were found in honey bees at 60.5%, 54.7%, and 7.0% of sites, and in bumble bees at 13.8%, 10.3%, and 13.8% of sites. IAPV and BQCV were observed in 34 and 27 bumble bees from 12 sites, which made them the most prevalent viruses. CBPV was found in honey bees at 12 sites and bumble bees at two locations (one co-occurrence). BQCV (r = 0.28, P = 0.017) and CBPV (r = 0.22, P = 0.062) prevalences were positive correlated across honey bees and bumble bees at the same sites (n = 71).
3.2 Factors predicting bumble bee virus occurrence
The most influential factor affecting BQCV prevalence in bumble bees was the prevalence of BQCV in honey bees at the same site, while urban landscape context was the strongest predictor of DWV and IAPV in bumble bee populations (Figure 4; Table S1). When looking at the most common virus (BQCV), BQCV co-occurrence in honey bees and bumble bees was only affected by regional bee density; a unit increase in regional apiary density led to a predicted 2.96-fold increase in BQCV (95% CI 1.76 to 42.5) (Figure 5, Table S2).
Log odds ratios for bumble bee virus logistic regression models for BQCV (A), DWV (B), and IAPV (C). Logistic regression models include three explanatory variables: the proportion of the virus in honey bees, urban landscape context, and agricultural landscape with honey bee apiaries for pollination.
Fitted model effects and observed data elucidating the relationship between regional honey bee apiary density and BQCV co-occurrence. The projected model curve is based on the top AICc selected model, and model fit statistics are provided in Table S2.
4 Discussion
Our study identified covariance (and lack thereof) in prevalence of three viruses between bee groups in a study area inclusive of urban, agricultural, residential, and natural landscapes of the Pacific Northwest USA. While all viruses surveyed were observed in managed honey bees, only four were seen in wild bumble bees. The dominant honey bee viruses (DWV, BQCV, SBV) were far less common in wild bumble bees, but IAPV was the most common virus in bumble bees and prevalence was similar in honey bees and bumble bees. While community-level differences between bee taxa were observed, landscape factors also impacted the distribution of viruses in wild bees. IAPV and DWV were more common in urban sites, while the prevalence of BQCV in bumble bees was best predicted by BQCV prevalence in honey bees and by regional honey bee apiary density.
Interest in the widespread occurrence of DWV and Varroa mite (Varroa destructor), an ectoparasitic viral vector in managed honey bees, has led to research illustrating the directional spread of DWV into wild bee communities from managed honey bees (Manley et al. 2019). The overall tendency of bee viruses to accumulate in managed honey bee populations yields support to the hypothesis that other viruses are spread to wild bees in a similar manner. The prevalence of common honey bee viruses seen in our study were much lower in bumble bee populations than honey bees, yet other studies have observed highly variable wild bee virus levels in different North American regions. For example, DWV observations range from very common (> 50%) to rather low (9%) prevalence, and BQCV ranges from infrequent (< 10%) to high (73%) prevalence in other studies (Dolezal et al. 2016; Alger et al. 2019a, b). It is additionally important to consider that our field sampling methodology may reduce our record of virus incidence, because individuals impaired by severe illness would not be sampled.
IAPV was the only virus observed more commonly in bumble bees than honey bees across sites, although the difference was not significant (13.8% of bumble bee sites vs. 7% of honey bee sites). Similar results have been observed in other studies, where bumble bees exhibited higher prevalence of IAPV (Dolezal et al. 2016; Dalmon et al. 2021). In a greenhouse study, infected honey bees were observed to transmit IAPV to bumble bees, and bumble bees were observed to transmit IAPV to honey bees within a week, demonstrating environmental transmittance in both directions. This provides support for further investigation of community-level virus transmission research for IAPV and other viruses (Singh et al. 2010). While it seems likely that virus spread from managed honey bees to wild bees is the dominant transmission direction for DWV, BQCV, and SBV in most parts of the world, given the accumulation of much higher prevalence in honey bee populations, virus transmission from wild bee reservoirs into managed honey bees may also be occurring, especially for IAPV (Ravoet et al. 2014; Dolezal et al. 2016; Dalmon et al. 2021).
While BQCV in bumble bees (13.8% of sites) was predicted by BQCV in honey bees (60% of sites), the other two common viruses (IAPV, DWV) were most affected by landscape context. Both viruses were more common in urban sites around Seattle and Portland compared to the arid eastern sites. These more mesic ecosystems sustain high bumble bee species richness, and likely higher abundance than more arid landscapes. This abundance could allow more coincidence and temporal continuity of hosts facilitating virus transmission; however, other rationales coexist. Less floral resources could also result in higher coincidence of bees on shared floral blooms. Rapid growth of urban beekeeping may also result in less experienced bee keepers with less disease management skills (Bruckner et al. 2023). We also expect that urban beekeepers maintain more constant occupancy of apiaries compared to transitory commercial apiaries in agricultural landscapes. However, despite recent advances in understanding factors affecting virus prevalence in bees, there remains much to be learned about impacts of bee viruses on wild bees. While studies show similar wing deformation symptoms in bumble bees compared to honey bees with DWV, more studies could inform the discussion of pathogen spread from managed apiaries, the degree to which wild bees act as reservoirs, and potential negative impacts of RNA viruses across bee species (Genersch et al. 2006; Fürst et al. 2014; Tehel et al. 2020).
Bee viruses are a major threat, and common bee viruses isolated first in honey bees are often observed at higher prevalence in managed apiaries compared to wild bees, increasing the chances of virus spread from managed apiaries to wild bees (Goulson and Hughes 2015). However, this is not always the case, as in this study and others, IAPV virus was observed with higher prevalence in wild bumble bee populations (see also Dolezal et al. 2016; Dalmon et al. 2021). Correlation in the presence of BQCV in honey bees and bumble bees suggests that a high level of environmental transmittance across taxa is likely within our study extent. These results also suggest that improved disease mitigation efforts could improve pollinator health broadly, rather than solely benefit of managed bee populations. The proliferation and concentration of bee virus infections and co-infections are a widespread concern in pollinator conservation, and attention to local incidence rates in managed and wild bee communities may promote more cautious hive movement and attention to pest control.
Data availability
Data and code are available at GitHub, https://github.com/verawp.
References
Alger SA, Burnham PA, Boncristiani HF, Brody AK (2019a) RNA virus spillover from managed honey bees (Apis mellifera) to wild bumble bees (Bombus spp.). PLoS ONE 14:e0217822
Alger SA, Burnham PA, Brody AK (2019b) Flowers as viral hot spots: honey bees (Apis mellifera) unevenly deposit viruses across plant species. PLoS ONE 14:e0221800
Bruckner S, Wilson M, Aurell D, Rennich K, vanEngelsdorp D, Steinhauer N, Williams GR (2023) A national survey of managed honey bee colony losses in the USA: results from the Bee Informed Partnership for 2017–18, 2018–19, and 2019–20. J Apic Res 1–15. https://doi.org/10.1080/00218839.2022.2158586
Berényi O, Bakonyi T, Derakhshifar I, Köglberger H, Nowotny N (2006) Occurrence of six honey bee viruses in diseased Austrian apiaries. Appl Environ Microbiol 72:2414–2420
Boncristiani H, Li J, Evans JD, Pettis J, Chen Y (2011) Scientific note on PCR inhibitors in the compound eyes of honey bees, Apis mellifera. Apidologie 42:457–460
Cornman RS, Tarpy DR, Chen Y, Jeffreys L, Lopez D, Pettis JS, vanEngelsdorp D, Evans JD (2012) Pathogen webs in collapsing honey bee colonies. PLoS ONE 7:e43562
Dalmon A, Diévart V, Thomasson M, Fouque R, Vaissière BE, Guilbaud L, Le Conte Y, Henry M (2021) Possible spillover of pathogens between bee communities foraging on the same floral resource. InSects 12:122
Dolezal AG, Hendrix SD, Scavo NA, Carrillo-Tripp J, Harris MA, Wheelock MJ, O’Neal ME, Toth AL (2016) Honey bee viruses in wild bees: viral prevalence, loads, and experimental inoculation. PLoS ONE 11:e0166190
Fürst MA, McMahon DP, Osborne JL, Paxton RJ, Brown MJF (2014) Disease associations between honey bees and bumble bees as a threat to wild pollinators. Nature 506:364–366
Genersch E, Yue C, Fries I, de Miranda JR (2006) Detection of deformed wing virus, a honey bee viral pathogen, in bumble bees (Bombus terrestris and Bombus pascuorum) with wing deformities. J Invert Pathol 91:61–63
Goulson D, Hughes WOH (2015) Mitigating the anthropogenic spread of bee parasites to protect wild pollinators. Biol Conserv 191:10–19
Grozinger CM, Flenniken ML (2019) Bee viruses: ecology, pathogenicity, and impacts. Annu Rev Entomol 64:205–226
Gusachenko ON, Woodford L, Balbirnie-Cumming K, Ryabov EV, Evans DJ (2020) Evidence for and against deformed wing virus spillover from honey bees to bumble bees: a reverse genetic analysis. Sci Reports 10:16847
Johnson PTJ, de Roode JC, Fenton A (2015) Why infectious disease research needs community ecology. Science 349:1259504–1259504
Levitt AL, Singh R, Cox-Foster DL, Rajotte E, Hoover K, Ostiguy N, Holmes EC (2013) Cross-species transmission of honey bee viruses in associated arthropods. Virus Res 176:232–240
Li J, Peng W, Wu J, Strange JP, Boncristiani H, Chen Y (2011) Cross-species infection of deformed wing virus poses a new threat to pollinator conservation. J Econ Entomol 104:732–739
Manley R, Temperton B, Doyle T, Gates D, Hedges S, Boots M, Wilfert L (2019) Knock-on community impacts of a novel vector: spillover of emerging DWV-B from Varroa-infested honey bees to wild bumble bees. Ecol Lett 22:1306–1315
McMahon DP, Fürst MA, Caspar J, Theodorou P, Brown MJF, Paxton RJ (2015) A sting in the spit: widespread cross-infection of multiple RNA viruses across wild and managed bees. J Anim Ecol 84:615–624
Nanetti A, Bortolotti L, Cilia G (2021) Pathogens spillover from honey bees to other arthropods. Pathogens 1:1044
Otterstatter MC, Thomson JD (2008) Does pathogen spillover from commercially reared bumble bees threaten wild pollinators? PLoS ONE 3:e2771
Peng W, Li J, Boncristiani H, Strange JP, Hamilton M, Chen Y (2011) Host range expansion of honey bee black queen cell virus in the bumble bee, Bombus huntii. Apidologie 42:650–658
Pfeiffer VW, Crowder DW (2022) Factors affecting virus prevalence in honey bees in the Pacific-Northwest, USA. J Invertebr Pathol 187:107703
Purkiss T, Lach L (2019) Pathogen spillover from Apis mellifera to a stingless bee. Proc Roy Soc London Series B 286:20191071
Ravoet J, De Smet L, Meeus I, Smagghe G, Wenseleers T, de Graaf DC (2014) Widespread occurrence of honey bee pathogens in solitary bees. J Invertebr Pathol 122:55–58
Schläppi D, Lattrell P, Yañez O, Chejanovsky N, Neumann P (2019) Foodborne transmission of deformed wing virus to ants (Myrmica rubra). InSects 10:394
Singh R, Levitt AL, Rajotte EG, Holmes EC, Ostiguy N, van Engelsdorp D, Lipkin WI, dePamphilis CW, Toth AL, Cox-Foster DL (2010) RNA viruses in hymenopteran pollinators: evidence of inter-taxa virus transmission via pollen and potential impact on non-Apis hymenopteran species. PLoS ONE 5:e14357
Tehel A, Brown MJ, Paxton RJ (2016) Impact of managed honey bee viruses on wild bees. Curr Opin Virol 19:16–22
Tehel A, Streicher T, Tragust S, Paxton RJ (2020) Experimental infection of bumble bees with honey bee-associated viruses: no direct fitness costs but potential future threats to novel wild bee hosts. Roy Soc Open Sci 7:200480
Woolhouse MEJ (2001) Population biology of multihost pathogens. Science 292:1109–1112
Acknowledgements
We thank O. Forrester for helping with the data analysis and Western SARE (grant SW18-031) for the funding.
Funding
The research was funded by Western SARE (grant SW18-031).
Author information
Authors and Affiliations
Contributions
VP and DC contributed to the study conception and design. Lab analysis of samples was carried out by VP and SB. VP analyzed the data and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
No ethics approval was needed for this study.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Manuscript editor: Michelle L Flenniken
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pfeiffer, V.W., Basu, S. & Crowder, D.W. Patterns of virus coincidence between honey bees and bumble bees in the Pacific Northwest, USA. Apidologie 55, 30 (2024). https://doi.org/10.1007/s13592-024-01072-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13592-024-01072-w