Abstract
Introduction
Diagnosis of persistent erythematous, scaly patches, or plaques can be complex since psoriasis (Ps), eczematous dermatitis (ED), and mycosis fungoides (MF) can be considered. Dermoscopy, which is a noninvasive diagnostic tool, is commonly used to examine blood vessels, scales, and background color; however, research on hair shaft evaluation in inflammatory dermatoses remains scarce. The aim of the study was dermoscopic evaluation of hair shafts in skin lesions localized on the non-scalp skin areas in patients diagnosed with MF, Ps, and ED.
Methods
This was a retrospective evaluation of 55 patients diagnosed with MF, Ps, and ED. Photographic and dermoscopic documentation of these patients and detailed medical history were evaluated.
Results
A total of 21 patients with MF, 21 patients with Ps, and 13 patients with ED were evaluated. The examination revealed the presence of various abnormalities of hair shafts (e.g., numerous pili torti, single pili torti, 8-shaped hairs, pigtail hairs, broken hairs, hair shafts rapidly tapered over long sections, hair shafts irregular in thickness, angulated hairs, branched hairs, the presence of trichorrhexis nodosa, and monilethrix-like hairs), yellow dots, and black dots. The presence of pili torti was found in 80% of patients with MF, compared with 16% of patients with Ps and 8% of patients with ED (p < 0.005), with multiple pili torti found only in MF patients (67%) (p < 0.005). Statistically significant differences also applied to hair shafts rapidly tapering over long sections and 8-shaped hairs, which occurred only in MF patients (p < 0.005 and p = 0.035, respectively).
Conclusions
The presence of hair shaft abnormalities such as numerous pili torti, 8-shaped hairs, and hair shafts rapidly tapering over long sections is an important criterion that should be considered in the dermoscopic differentiation of the patchy/plaque mycosis fungoides and inflammatory dermatoses, such as psoriasis and eczematous dermatitis localized on the non-scalp skin areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Nowadays dermoscopic features of mycosis fungoides (MF) include evaluation of the type and arrangement of blood vessels and background appearance. |
There are few papers showing the presence of hair shaft abnormalities in trichoscopy of scalp involvement in the course of cutaneous T-cell lymphomas (CTCLs). |
The aim of the study was dermoscopic evaluation of hair shafts in skin lesions localized on the non-scalp skin areas in patients diagnosed with MF, psoriasis (Ps), and eczematous dermatitis (ED). |
Statistically significant differences in the occurrence of abnormal hair shafts (such as pili torti, 8-shaped hairs, and hair shafts rapidly tapering over long sections) in the skin lesions localized on the non-scalp skin areas were found between patients from the study groups. |
Assessment of hair shaft abnormalities seems to be another important dermoscopic parameter for differentiating between patients with inflammatory skin diseases. |
Introduction
The term “inflammoscopy” refers to dermoscopy analysis of inflammatory dermatoses. It enables fast initial differential diagnosis between skin lesions that clinically look alike. It focuses primarily on the type and distribution of blood vessels and background appearance (color and scaling) [1].
The differential diagnosis between patchy and plaque variants of mycosis fungoides (MF), psoriasis (Ps), and eczematous dermatitis (ED) may be challenging in everyday practice.
According to Errichetti et al. [2], typical dermoscopic features of MF include: linear/linear curved vessels and orange structureless areas for the patch stage, as well as unspecific distributed dotted/linear vessels and focal bright white areas for the plaque phase. In dermoscopy, Ps is characterized by regularly distributed dotted vessels over a light or dull red background with diffuse white scale [1]. ED in dermoscopy reveals nonspecifically arranged dotted vessels, yellow scale, serum crust, and hemorrhage in the case of the acute and subacute phases with a severe itching sensation [1].
There are few papers showing the presence of hair shaft abnormalities in trichoscopy of scalp involvement in the course of cutaneous T-cell lymphomas (CTCLs), such as short hairs with split-ends [3, 4], broken hairs, pigtail hairs [3, 5], black dots, numerous pili torti, 8-shaped hairs, and visible anagen bulbs [4, 5].
However, there are no descriptions of hair shaft abnormalities if the lesion is localized on the non-scalp skin area.
The study aimed to assess whether the presence of pili torti or other hair shaft abnormalities found with the dermoscope approach can help differentiate patchy variants of MF, Ps, and ED located outside the scalp. The assessment was focused on the evaluation of the hair shafts.
Methods
This retrospective analysis was conducted in accordance with consensus ethics principles derived from international ethics guidelines, including the Declaration of Helsinki and the International Ethical Guidelines by the Council for International Organizations of Medical Sciences. The study protocol and tools were reviewed and ethically approved by the Bioethics Committee at the Medical University of Warsaw.
The database was searched for patients diagnosed with MF, who underwent dermoscopic assessment. The evaluation included 21 patients diagnosed with MF on the basis of the clinical appearance, histopathology, and immunohistochemistry. The clonality of atypical T CD4+ was confirmed by polymerase chain reaction [PCR; T-cell receptor (TCR) rearrangement] in all patients. The comparison group included 21 patients with Ps and 13 patients with ED. As a routine procedure in our setting, dermoscopy was performed using a Fotofinder digital dermoscope, obtaining from 20 to 80 photographs (20–70-fold magnification) of skin lesions localized in different anatomical locations for each subject.
In total, 1480 photographs were assessed for the presence of specific hair shaft abnormalities by two of the contributing authors (M.J. and A.R.). A third investigator was involved in the case of disagreement (M.K.). The revision of the images was blind to the investigators.
After evaluation, the results were unblinded, and compatible dermoscopic features were assigned to respective patient groups.
The obtained data were statistically evaluated using the analysis of variance (ANOVA) test, Scheffe’s method, and the Games–Howell post-hoc test. A p-value less than 0.05 was considered statistically significant.
Results
The study included 21 patients diagnosed with MF (8 women and 13 men; age range 45–91 years; mean age 70 years), 21 with Ps (5 women and 16 men; age range 23–86 years; mean age 54 years) and 13 with ED (6 women and 7 men; age range 25–74 years; mean age 46 years).
The hair shaft abnormalities revealed with dermoscopy included: numerous pili torti, single pili torti, 8-shaped hairs, pigtail hairs, broken hairs, hair shafts rapidly tapered over long sections, hair shafts irregular in thickness, angulated hairs (defined as fractured hairs forming a sharp angle along the hair shaft) [6, 7], branched hairs (defined as hair shafts with obliquely detached pieces) [6], the presence of trichorrhexis nodosa, monilethrix-like hairs, and, additionally, yellow dots and black dots.
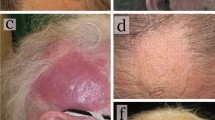
Among 21 patients with MF, hair shafts were not observed within the patches in six cases (29%). In those with hairs, the hair shaft abnormalities mainly manifested as pili torti, visible in 80% (12/15) of patients. In the majority of these cases, they were numerous (67%; 8/12; p < 0.001; Fig. 1).
In contrast, single pili torti were observed in 16% (3/19) of patients with Ps and in 8% (1/12) of patients with ED (p < 0.001). Two patients with Ps and one patient with ED were excluded from this analysis due to the absence of hair shafts within the skin lesions.
Hair shafts that were irregular in thickness were present in all assessed patient groups: 47% (7/15) of patients with MF, 50% (6/12) of patients with ED, and 58% (11/19) of patients with Ps. The main difference, however, was observed in patients with MF, where the hair shafts were rapidly tapered over long sections, giving a fairly regular appearance (Fig. 2). Such a feature was not observed in patients with Ps and ED (p < 0.001 and p = 0.001, respectively).
Other features regarding hair shafts, which were observed in patients with MF, Ps, and ED, were: angulated hairs [53% (8/15) versus 53% (10/19) versus 42% (5/12), respectively; p = 0.808], broken hairs [53% (8/15) versus 53% (10/19) versus 50% (6/12), respectively; p = 0.985] and branched hairs [13% (2/15) versus 0% (0/19) versus 17% (2/12), respectively; p = 0.215].
In MF patients, 8-shaped hairs were observed in 20% (3/15) of cases and were absent in other groups (p = 0.035; Fig. 3).
Interestingly, only patients with Ps presented trichorrhexis-nodosa-like hairs (10.5%; 2/19) and monilethrix-like hairs [5% (1/19)]. However, these findings were not statistically significant (p = 0.238 and p = 0.502, respectively). Yellow dots [81% (17/21) versus 57% (12/21) versus 54% (7/13), respectively] and black dots [5% (1/21) versus 5% (1/21) versus 8% (1/13), respectively] were observed in the MF, Ps, and ED groups. The differences among groups were not statistically significant (p = 0.168 and p = 0.925).
Detailed results are presented in Table 1.
Discussion
Adult human hairs can be divided into hairs located in hormone-dependent areas of the body (e.g., the scalp, beard, chest, armpits, and genital area) and androgen-independent hairs (e.g., eyebrows and eyelashes). In this case, we are referring to terminal hair shafts, which are long, thick, pigmented, and medullated. This type of hair usually extends more than 3 mm into the hypodermis. The rest of the body is covered with vellus hairs (short, thin, devoid of pigment, and located more shallowly in the skin compared with terminal hairs) [8].
In the case of scalp diseases, the evaluation of the hair shafts is often crucial for the correct diagnosis. For this purpose, Rudnicka et al. [9] proposed a trichoscopic classification of abnormalities in the structure of the hair shafts, distinguishing six main groups of disorders: (1) hair shafts with fractures, (2) hair narrowing, (3) hairs with node-like structures, (4) curls and twists, (5) bands, and (6) short hair [9, 10].
To our knowledge, no previous studies have been published on the dermoscopic assessment of hair shaft abnormalities in dermatological diseases where the lesions are located outside the scalp. In this context, based on the assumption that the neoplastic process in the non-scalp skin areas could affect the skin appendages, we decided to assess the usefulness of dermoscopic evaluation of hair shafts in the differential diagnosis of patients with MF.
Previous publications on dermoscopy in MF have focused on the detailed assessment of blood vessels and the background of the affected skin [2, 11,12,13,14,15,16].
Rakowska et al. observed significant hair shaft abnormalities in the trichoscopy (scalp dermoscopy) of patients with erythrodermic variants of CTCL compared with patients with psoriatic erythroderma and erythrodermic atopic dermatitis (AD). The most sensitive and specific trichoscopic symptoms for erythrodermic CTCLs were numerous pili torti and 8-shaped hairs [5].
Pili torti is a form of inherited or acquired hair shaft disorder connected with increased hair fragility. It is characterized by the presence of the hair shafts, which are flattened at irregular intervals and twisted 180° along their long axis [5, 9, 17]. The unequal development of the outer root sheath cells is the cause of hair shaft abnormality in inherited forms of pili torti [5, 17, 18]. Perifollicular inflammation, accompanied by fibrosis, is responsible for the distortion of the hair follicle in acquired forms of pili torti, mainly in cicatricial alopecias [5, 17].
Trichoscopy at high magnifications (70× or more) usually reveals typical hair shaft twisting along its long axis with a tendency to break at these points; however, lower magnifications may reveal only hair shafts bent irregularly in different directions [19].
In the presented study, pili torti were present in 80% (p < 0.001) of MF lesions (67% of patients had numerous pili torti, and 33% exhibited single pili torti). Although pili torti were also observed in patients with Ps (16%) and ED (8%), this type of abnormality concerned single hair shafts. In patients with MF, pili torti were present within skin lesions of various locations, while in the group of patients with Ps and ED, they were mainly observed in skin lesions of the body regions most prone to repeated injuries (e.g., elbows, knees) and probably as a result of the mechanical trauma.
The percentage of the complete hair loss within the affected skin was highest in the group of patients with MF at 29%, compared to 9.5% in the Ps group and 8% in the ED group (p = 0.162), which appears to be an important sign for the differentiation between the above mentioned entities. However, it did not reach statistical significance in our analysis.
Irregularity in hair shaft thickness was present in all evaluated patient groups. The main difference between them was that the narrowing of the hair shaft in MF patients was sudden and extended over a long section—referred to as hair shafts rapidly tapering over long sections. This particular abnormality was observed in 47% of patients with MF (p < 0.001). Rakowska et al. [5] did not observe this type of hair shafts in the scalp trichoscopy of patients with the erythrodermic variant of CTCL, Ps, and AD.
Angulated hairs were present in patients from all study groups (53% MF, 53% Ps, and 42% ED; p = 0.808). They were described by Khunkhet et al. in alopecia areata (AA) and defined as partially fractured hairs forming a sharp angle along the hair shaft. According to the authors, the presence of angulated hairs is connected to the weakening of the hair shafts caused by peribulbar lymphocytic infiltrate [7]. A very similar trichoscopic sign, so-called trichoclasis (transverse fracture of the hair shaft with an intact cuticle), was earlier mentioned by Rudnicka et al. in AA patients [20]. In 2020, Elmas et al. also found angulated hairs in patients with trichotillomania (TTM), concluding that this type of hair shaft abnormality is neither a specific sign for AA nor for TTM [6].
We hypothesize that the presence of the angulated hairs in our patients may be attributed to the weakening of the hair shafts by the subepidermal/subepithelial inflammatory infiltrate of the neoplastic lymphocytes in the dermis and recurrent mechanical injuries, such as scratching.
The term “branched hairs” was first used by Elmas et al. [6] to describe a hair shafts with obliquely detached pieces. In the presented study, this type of hair shaft abnormality was observed in 13% of MF patients and 17% of ED patients. They were not present in patients with Ps (p = 0.215). Elmas et al. observed this type of hairs in patients with TTM, suggesting that the branched hairs may be caused by pushing and manipulating the hair shaft linearly from the distal end to the proximal end [6]. We suspect that these kinds of hair shaft abnormalities may be caused by repetitive scratching and rubbing of itchy skin by those patients due to pruritus.
Similarly to angulated hairs, no branched hairs were observed in the scalp trichoscopy of patients with erythrodermic CTCL, as well as in patients from the comparative groups (psoriatic erythroderma and erythrodermic atopic dermatitis) in the study conducted by Rakowska et al. [5]. We might hypothesize that these differences are attributable to greater hair shaft thickness in the scalp in comparison to other body areas.
Pigtail hairs, also known as circle hairs, are short, regularly twisted hairs with tapered distal parts. Such a disturbance is most likely the result of the rapid hair regrowth that precedes the complete regeneration of the hair follicle [20]. Sławińska et al. [21] described the case of an 88-year-old woman with folliculotropic mycosis fungoides (FMF) on the scalp, where trichoscopic examination revealed pigtail-like hairs. In the presented study, pigtail hairs were observed in 13% of MF patients and in 17% with ED. However, their presence was not statistically significant (p = 0.215).
In contrast, 8-shaped hairs (thin, long, pigmented hair shafts, repeatedly rolled in two opposite directions resembling number 8) were described by Rakowska et al. [5] as pathognomonic (if numerous) for the scalp involvement in the course of erythrodermic CTCL. Formation of 8-shaped hairs may result from the folliculotropic lymphocytic infiltrate effecting the hair epithelium keratinocytes. In the presented study, 8-shaped hairs were foundonly in MF patients (20%).
Broken hairs were present in at least half of the patients in each of the study groups (53% MF, 53% Ps, and 50% ED; p = 0.985), while black dots were present in these patients only in a small percentage (5% MF, 5% Ps, and 8% ED; p = 0.925). In contrast, Rakowska et al. [5] described the presence of multiple broken hairs (due to breakage of pili torti) in trichoscopy only in patients with erythrodermic CTCL. Sławińska et al. [21] found broken hairs and black dots, but within yellow dots, in the scalp trichoscopy of folicullotropic MF patients.
The limitations of our study are its retrospective design and the restricted number of patients. Further investigation is needed.
Conclusions
The presence of hair shaft abnormalities, such as numerous pili torti, 8-shaped hairs, and hair shafts rapidly tapering over long sections, is an important criterion that should be considered in the dermoscopic differentiation of the patchy MF and inflammatory dermatoses, like Ps and ED, localized on the non-scalp skin areas.
Data Availability
The datasets analyzed in the current study are not publicly available, because they include the sensitive data of hospitalized patients that are subject to regional data protection laws. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Errichetti E. Dermoscopy of inflammatory dermatoses (inflammoscopy): an up-to-date overview. Dermatol Pract Concept. 2019;9(3):169–80.
Errichetti E, Apalla Z, Geller S, Slawinska M, et al. Dermoscopic spectrum of mycosis fungoides: a retrospective observational study by the International Dermoscopy Society. J Eur Acad Dermatol Venereol. 2022;36(7):1045–53.
Gallo G, Pileri A, Starace M, et al. Clinical and trichoscopic features in 18 cases of folliculotropic mycosis fungoides with scalp involvement. Sci Rep. 2021;11(1):10555.
Rudnicka L, Chrostowska S, Kaminski M, et al. The role of trichoscopy beyond hair and scalp diseases. A review. J Eur Acad Dermatol Venereol. 2023. https://doi.org/10.1111/jdv.19047.
Rakowska A, Jasinska M, Sikora M, et al. Cutaneous T-cell lymphoma in erythrodermic cases may be suspected on the basis of scalp examination with dermoscopy. Sci Rep. 2021;11(1):282.
Elmas OF, Metin MS. Trichoscopic findings of trichotillomania: new observations. Postepy Dermatol Alergol. 2020;37(3):340–5.
Khunkhet S, Vachiramon V, Suchonwanit P. Trichoscopic clues for diagnosis of alopecia areata and trichotillomania in Asians. Int J Dermatol. 2017;56(2):161–5.
Buffoli B, Rinaldi F, Labanca M, et al. The human hair: from anatomy to physiology. Int J Dermatol. 2014;53(3):331–41.
Rudnicka L, Olszewska M, Waskiel A, Rakowska A. Trichoscopy in hair shaft disorders. Dermatol Clin. 2018;36(4):421–30.
Kremer N, Martinez H, Leshem YA, et al. The trichoscopic features of hair shaft anomalies induced by epidermal growth factor receptor inhibitors: a case series. J Am Acad Dermatol. 2021;85(5):1178–84.
Ghahramani GK, Goetz KE, Liu V. Dermoscopic characterization of cutaneous lymphomas: a pilot survey. Int J Dermatol. 2018;57(3):339–43.
Lallas A, Apalla Z, Lefaki I, et al. Dermoscopy of early stage mycosis fungoides. J Eur Acad Dermatol Venereol. 2013;27(5):617–21.
Ozturk MK, Zindanci I, Zemheri E. Dermoscopy of stage llA mycosis fungoides. North Clin Istanb. 2020;7(2):174–9.
Slawinska M, Sokolowska-Wojdylo M, Sobjanek M, Golinska J, Nowicki RJ, Rudnicka L. The significance of dermoscopy and trichoscopy in differentiation of erythroderma due to various dermatological disorders. J Eur Acad Dermatol Venereol. 2021;35(1):230–40.
Toncic RJ, Rados J, Curkovic D, Ilic I, Caccavale S, Bradamante M. Dermoscopy of syringotropic and folliculotropic mycosis fungoides. Dermatol Pract Concept. 2020;10(4): e2020069.
Xu C, Liu J, Wang T, Luo Y, Liu Y. Dermoscopic patterns of early-stage mycosis fungoides in a Chinese population. Clin Exp Dermatol. 2019;44(2):169–75.
Hoffmann A, Waskiel-Burnat A, Zolkiewicz J, et al. Pili torti: a feature of numerous congenital and acquired conditions. J Clin Med. 2021;10(17):3901.
Maruyama T, Toyoda M, Kanei A, Morohashi M. Pathogenesis in pili torti: morphological study. J Dermatol Sci. 1994;7(Suppl):S5-12.
Rudnicka L, Rakowska A, Kurzeja M, Olszewska M. Hair shafts in trichoscopy: clues for diagnosis of hair and scalp diseases. Dermatol Clin. 2013;31(4):695–708.
Rudnicka L, Rakowska A, Olszewska M, Slowinska M, Czuwara J, Rusek M, Pinheiro AMC. Hair shafts. In: Rudnicka L, Olszewska M, Rakowska A, editors. Atlas of trichoscopy. Springer: London; 2012. p. 11–45.
Slawinska M, Sobjanek M, Olszewska B, Nowicki R, Sokolowska-Wojdylo M. Trichoscopic spectrum of folliculotropic mycosis fungoides. J Eur Acad Dermatol Venereol. 2018;32(3):e107–8.
Acknowledgements
The authors thank all study participants for their invaluable contribution to the study.
Funding
No funding or sponsorship was received for this study or publication of this article.
Author information
Authors and Affiliations
Contributions
Conceptualization: Jasińska Magdalena, Rakowska Adriana, and Lortkipanidze Nino. Methodology: Jasińska Magdalena and Rakowska Adriana. Formal analysis and investigation: Jasińska Magdalena, Rakowska Adriana, Kurzeja Marta, Czuwara Joanna, Michalczyk Agnieszka, and Borkowska Barbara, Gajda-Mroz Patrycja. Writing—original draft preparation: Jasińska Magdalena. Writing—review and editing: Rakowska Adriana, Jasińska Magdalena, Lortkipanidze Nino, Rudnicka Lidia, Olszewska Malgorzata, and Czuwara Joanna. Supervision: Rakowska Adriana. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Adriana Rakowska is an Editorial Board member of Dermatology and Therapy. Adriana Rakowska was not involved in the selection of peer reviewers for the manuscript nor any of the subsequent editorial decisions. Magdalena Jasińska, Joanna Czuwara, Nino Lortkipanidze, Agnieszka Michalczyk, Barbara Borkowska, Patrycja Gajda-Mróz, Marta Kurzeja, Małgorzata Olszewska, and Lidia Rudnicka declare no conflict of interest regarding this paper.
Ethical Approval
The study was conducted in accordance with consensus ethics principles derived from international ethics guidelines, including the Declaration of Helsinki and the International Ethical Guidelines by the Council for International Organizations of Medical Sciences. The study protocol and tools were reviewed and ethically approved by Bioethics Committee at the Medical University of Warsaw.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Jasińska, M., Czuwara, J., Lortkipanidze, N. et al. Hair Shaft Abnormalities as a Dermoscopic Feature of Mycosis Fungoides: Pilot Results. Dermatol Ther (Heidelb) 14, 2119–2126 (2024). https://doi.org/10.1007/s13555-024-01206-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01206-z