Abstract
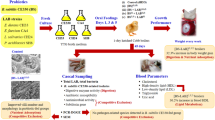
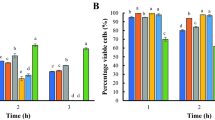
This study sought to isolate lactic acid bacteria (LAB) from chicken intestines and caeca and apply them as probiotics in broilers. Out of the 247 isolates, 14 LAB were selected based on their tolerance to pH 3 and 0.5% bile salt conditions and were tested against Salmonella serovars using two assay methods: (1) bacterial cells and double layers and (2) cell-free supernatants and agar well diffusion. The chicken isolates CA4, CH24 and CH33 strongly inhibited Salmonella Typhimurium ATCC13311 and S. Enteritidis. The selected strains were identified via 16S rDNA sequencing as Enterococcus faecium CA4, Enterococcus durans CH33 and Lactobacillus salivarius CH24. Only CH33 survived in simulated gastric juice and intestinal juice with survival rates of 90 and 18%, respectively. All three chicken LAB strains as well as food-originating Pediococcus acidilactici SH8 and bacteriocin-producing Bacillus subtilis KKU213 were tested in broilers. Single strains and mixed cultures of KKU213 and the four LAB strains were orally fed to 1-day-old male Cobb broilers, which were then raised for 45 days. Broilers fed LAB strains demonstrated higher numbers of LAB than the groups fed only B. subtilis KKU213 or mixed cultures. Among all treatments, the broilers fed B. subtilis KKU213 on days 1 and 3 and LAB on day 5 (T8) had the highest body weights and high-density lipid levels and the lowest uric acid levels. Therefore, a combination of bacterial species originating from various sources exhibits potential as a probiotic mixture to promote health in broilers.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Boonthai, T.; Vuthiphandchai, V.; Nimrat, S.: Probiotic bacteria effects on growth and bacterial composition of black tiger shrimp (Penaeus monodon). Aquac. Nutr. 17, 634–644 (2011)
Iaconelli, C.; Lemetais, G.; Kechaou, N.; Chain, F.; Bermudez-Humaran, L.G.; Langella, P.; Gervais, P.; Beney, L.: Drying process strongly affects probiotics viability and functionalities. J. Biotechnol. 214, 17–26 (2015)
Pringsulaka, O.; Rueangyotchanthana, K.; Suwannasai, N.; Watanapokasin, R.; Amnueysit, P.; Sunthornthummas, S.; Sukkhum, S.; Sarawaneeyaruk, S.; Rangsiruji, A.: In vitro screening of lactic acid bacteria for multi-strain probiotics. Livest. Sci. 174, 66–73 (2015)
FAO/WHO: FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food. World Health Organization, Geneva (2002)
Matamoros, S.; Pilet, M.F.; Gigout, F.; Prévost, H.; Leroi, F.: Selection and evaluation of seafood-borne psychrotrophic lactic acid bacteria as inhibitors of pathogenic and spoilage bacteria. Food Microbiol. 26, 638–644 (2009). https://doi.org/10.1016/j.fm.2009.04.011
Lay, C.L.; Mounier, J.; Vasseur, V.; Weill, A.; Blay, G.L.: In vitro and in situ screening of lactic acid bacteria and propionibacteria antifungal activities against bakery product spoilage molds. Food Control 60, 247–255 (2016). https://doi.org/10.1016/j.foodcont.2015.07.034
Victoria Moreno-Arribas, M.; Carmen Polo, M.; Jorganes, F.; Muñoz, R.: Screening of biogenic amine production by lactic acid bacteria isolated from grape must and wine. Int. J. Food Microbiol. 84, 117–123 (2003). https://doi.org/10.1016/S0168-1605(02)00391-4
Raghavendra, P.; Halami, P.M.: Screening, selection and characterization of phytic acid degrading lactic acid bacteria from chicken intestine. Int. J. Food Microbiol. 133(1), 129–134 (2009). https://doi.org/10.1016/j.ijfoodmicro.2009.05.006
Oh, Y.J.; Jung, D.S.: Evaluation of probiotic properties of Lactobacillus and Pediococcus strains isolated from Omegisool, a traditionally fermented millet alcoholic beverage in Korea. LWT Food Sci. Technol. 63, 437–444 (2015)
García-Ruiz, A.; Gonzalez de Llano, D.; Esteban-Fernandez, A.; Requena, T.; Bartolome, B.; Moreno-Arribas, M.V.: Assessment of probiotic properties in lactic acid bacteria isolated from wine. Food Microbiol. 44, 220–225 (2014). https://doi.org/10.1016/j.fm.2014.06.015
Öz, E.; Kaban, G.; Baris, Ö.; Kaya, M.: Isolation and identification of lactic acid bacteria from pastırma. Food Control 77, 158–162 (2017). https://doi.org/10.1016/j.foodcont.2017.02.017
Dertli, E.; Mercan, E.; Arıcı, M.; Yılmaz, M.T.; Sağdıç, O.: Characterisation of lactic acid bacteria from Turkish sourdough and determination of their exopolysaccharide (EPS) production characteristics. LWT Food Sci. Technol. 71, 116–124 (2016). https://doi.org/10.1016/j.lwt.2016.03.030
Sakaridis, I.; Soultos, N.; Dovas, C.I.; Papavergou, E.; Ambrosiadis, I.; Koidis, P.: Lactic acid bacteria from chicken carcasses with inhibitory activity against Salmonella spp. and Listeria monocytogenes. Anaerobe 18, 62–66 (2012). https://doi.org/10.1016/j.anaerobe.2011.09.009
Abushelaibi, A.; Al-Mahadin, S.; El-Tarabily, K.; Shah, N.P.; Ayyash, M.: Characterization of potential probiotic lactic acid bacteria isolated from camel milk. LWT Food Sci. Technol. 79, 316–325 (2017). https://doi.org/10.1016/j.lwt.2017.01.041
Benavides, A.B.; Ulcuango, M.; Yépez, L.; Tenea, G.N.: Assessment of the in vitro bioactive properties of lactic acid bacteria isolated from native ecological niches of Ecuador. Rev. Argent. Microbiol. 48(3), 236–244 (2016). https://doi.org/10.1016/j.ram.2016.05.003
Löfström, C.; Hintzmann, A.; Sørensen, G.; Baggesen, D.L.: Outbreak of Salmonella enterica serovar Typhimurium phage type DT41 in Danish poultry production. Vet. Microbiol. 178, 167–172 (2015). https://doi.org/10.1016/j.vetmic.2015.04.017
Wang, Y.; Yang, B.; Wu, Y.; Zhang, Z.; Meng, X.; Xi, M.; Wang, X.; Xia, X.; Shi, X.; Wang, D.; Meng, J.: Molecular characterization of Salmonella enterica serovar Enteritidis on retail raw poultry in six provinces and two National cities in China. Food Microbiol. 46, 74–80 (2015). https://doi.org/10.1016/j.fm.2014.07.012
Mani-López, E.; García, H.S.; López-Malo, A.: Organic acids as antimicrobials to control Salmonella in meat and poultry products. Food Res. Int. 45, 713–721 (2012). https://doi.org/10.1016/j.foodres.2011.04.043
Awad, W.A.; Ghareeb, K.; Abdel-Raheem, S.; Bohm, J.: Effects of dietary inclusion of probiotic and synbiotic on growth performance, organ weights, and intestinal histomorphology of broiler chickens. Poult. Sci. 88, 49–55 (2009)
Cao, G.T.; Zeng, X.F.; Chen, A.G.; Zhou, L.; Zhang, L.; Xiao, Y.P.; et al.: Effects of a probiotic, Enterococcus faecium, on growth performance, intestinal morphology, immune response, and cecal microflora in broiler chickens challenged with Escherichia coli K88. Poult. Sci. 92, 2949–2955 (2013)
Ashayerizadehl, A.; Dabiri, N.; Mirzadeh, K.; Ghorbani, M.R.: Effects of dietary inclusion of several biological feed additives on growth response of broiler chickens. Cell Anim. Biol. 5, 61–65 (2011)
Olnood, C.G.; Beski, S.S.; Iji, P.A.; Choct, M.: Delivery routes for probiotics: effects on broiler performance, intestinal morphology and gut microflora. Anim. Nutr. 1, 192–202 (2015b). https://doi.org/10.1016/j.aninu.2015.07.002
Olnood, C.G.; Beski, S.S.M.; Mingan Choct, M.; Iji, P.A.: Novel probiotics: their effects on growth performance, gut development, microbial community and activity of broiler chickens. Anim. Nutr. 1, 184–191 (2015a). https://doi.org/10.1016/j.aninu.2015.07.003
Mirmomeni, M.H.; Sajjadi Majd, S.; Sisakhtnezhad, S.; Doranegard, F.: Comparison of the three methods for DNA extraction from paraffim-embedded tissues. J. Biol. Sci. 10, 261–266 (2010)
Babot, J.D.; Argañaraz-Martínez, E.; Saavedra, L.; Apella, M.C.; Chaia, A.P.: Selection of indigenous lactic acid bacteria to reinforce the intestinal microbiota of newly hatched chicken–relevance of in vitro and ex vivo methods for strains characterization. Res. Vet. Sci. 97, 8–17 (2014)
Khochamit, N.; Siripornadulsil, S.; Sukon, P.; Siripornadulsil, W.: Antibacterial activity and genotypic-phenotypic characteristics of bacteriocin-producing Bacillus subtilis KKU213: potential as a probiotic strain. Microbiol. Res. 170, 36–50 (2015). https://doi.org/10.1016/j.micres.2014.09.004
Favaro, L.; Todorov, S.D.: Bacteriocinogenic LAB strains for fermented meat preservation: perspectives, challenges, and limitations. Probiotics Antimicrob. Proteins 9(4), 444–458 (2017). https://doi.org/10.1007/s12602-017-9330-6
Apajalahti, J.; Vienola, K.: Interaction between chicken intestinal microbiota and protein digestion. Anim. Feed Sci. Technol. 221, 323–330 (2016)
Özcelik, S.; Kuley, E.; Özogul, F.: Formation of lactic, acetic, succinic, propionic, formic and butyric acid by lactic acid bacteria. LWT Food Sci. Technol. 73, 536–542 (2016). https://doi.org/10.1016/j.lwt.2016.06.066
Maragkoudakis, P.A.; Mountzouris, K.C.; Psyrras, D.; Cremonese, S.; Fischer, J.; Cantor, M.D.; Tsakalidou, E.: Functional properties of novel protective lactic acid bacteria and application in raw chicken meat against Listeria monocytogenes and Salmonella enteritidis. Int. J. Food Microbiol. 130, 219–226 (2009). https://doi.org/10.1016/j.ijfoodmicro.2009.01.027
Swetwiwathana, A.; Visessanguan, W.: Potential of bacteriocin-producing lactic acid bacteria for safety improvements of traditional Thai fermented meat and human health. Meat Sci. 109, 101–105 (2015). https://doi.org/10.1016/j.meatsci.2015.05.030
Bartkiene, E.; Bartkevics, V.; Mozuriene, E.; Krungleviciute, V.; Novoslavskij, A.; Santini, A.; Rozentale, I.; Juodeikiene, G.; Cizeikiene, D.: The impact of lactic acid bacteria with antimicrobial properties on biodegradation of polycyclic aromatic hydrocarbons and biogenic amines in cold smoked pork sausages. Food Control 71, 285–292 (2017). https://doi.org/10.1016/j.foodcont.2016.07.010
Abriouel, H.; Franz, C.M.A.P.; Omar, N.B.; Glvezm, A.: Diversity and applications of Bacillus bacteriocins. FEMS Microbiol. Rev. 45, 201–32 (2011). https://doi.org/10.1111/j.1574-6976.2010.00244.x
Argyri, A.; Zoumpopoulou, G.; Karatzas, K.G.; Tsakalidou, E.; Nychas, G.E.; Panagou, E.Z.; Tassou, C.C.: Selection of potential probiotic lactic acid bacteria from fermented olives by in vitro tests. Food Microbiol. 33, 282–291 (2013). https://doi.org/10.1016/j.fm.2012.10.005
Sonsa-Ard, N.; Rodtong, S.; Chikindas, M.L.; Yongsawatdigul, J.: Characterization of bacteriocin produced by Enterococcus faecium CN-25 isolated from traditionally Thai fermented fish roe. Food Control 54, 308–316 (2015). https://doi.org/10.1016/j.foodcont.2015.02.010
Filho, R.A.C.P.; Díaz, S.J.A.; Fernando, F.S.; Chang, Y.-F.; Filho, R.L.A.; Junior, A.B.: Immunomodulatory activity and control of Salmonella Enteritidis colonization in the intestinal tract of chickens by Lactobacillus based probiotic. Vet. Immunol. Immunopathol. 167(1–2), 64–69 (2015)
Carter, A.; Adams, M.; La Ragione, R.M.; Woodward, M.J.: Colonisation of poultry by Salmonella Enteritidis S1400 is reduced by combined administration of Lactobacillus salivarius 59 and Enterococcus faecium PXN-33. Vet. Microbiol. 199, 100–107 (2017). https://doi.org/10.1016/j.vetmic.2016.12.029
Brisbin, J.T.; Gong, J.; Parvizi, P.; Sharif, S.: Effects of lactobacilli on cytokine expression by chicken spleen and cecal tonsil cells. Clin. Vaccine Immunol. 17, 1337–1343 (2010)
Marranzino, G.; Villena, J.; Salva, S.; Alvarez, S.: Stimulation of macrophages by immunobiotic Lactobacillus strains: influence beyond the intestinal tract. Microbiol. Immunol. 56, 771–781 (2012)
Acknowledgements
We thank Assistant Professor Dr. Peerapol Sukon and his colleagues from the Research Group for Preventive Technology in Livestock, Faculty of Veterinary Medicine, Khon Kaen University, Khon Kaen, Thailand, for their assistance with the care and use of laboratory animals according to the standard guidelines. This work was supported by Research and Researcher for Industry (RRi), the Thailand Research Fund (TRF), and the Master Program (Grant Nos. MSD58I0016, 590053) and Teacher Aood Local Farm, Sakon Nakhon, Thailand.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buahom, J., Siripornadulsil, S. & Siripornadulsil, W. Feeding with Single Strains Versus Mixed Cultures of Lactic Acid Bacteria and Bacillus subtilis KKU213 Affects the Bacterial Community and Growth Performance of Broiler Chickens. Arab J Sci Eng 43, 3417–3427 (2018). https://doi.org/10.1007/s13369-017-3045-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-017-3045-6