Abstract
Earlier studies showed environmentally friendly farming (EFF) increased the populations of various organisms in agricultural landscapes, but the mechanisms of these increases were not well studied, due to a lack of knowledge on their temporal dynamics. Here, we examined spatio-temporal dynamics of Tetragnatha spiders, natural enemies of rice pests, in EFF and conventional farming (CF). Field surveys were conducted in 15 paddy fields under CF and 18 under EFF during three seasons in Tochigi Prefecture, central Japan. The results showed that Tetragnatha spiders were more abundant in paddy fields than in ditches during the growing season, but this tendency was reversed during the non-crop season. Thus, complementary utilization of ditches and paddy fields during different seasons appeared to maintain Tetragnatha populations. Both Tetragnatha spider and flying insect abundance increased in paddy fields under only EFF when the surrounding forest cover was high. There was a significant positive correlation between the density of flying insects and the population growth rate of Tetragnatha spiders, suggesting a bottom-up effect of flying insects emerging from paddy fields. We concluded that the high potential productivity in paddy fields, coupled with the presence of ditches as important habitats, maintained the high abundance of Tetragnatha spiders in paddy-dominated landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Farmlands are not only sources of food for people, but also provide habitat for many organisms (Katoh et al. 2009; Pimentel et al. 1992). Paddy fields are created in at least 114 countries around the world (Maclean et al. 2002), and provide breeding and feeding habitats for various organisms as alternative to wetlands (Elphick 2000; Katano et al. 2003). However, the Green Revolution during the 1960s and 1970s enhanced rice productivity through the utilization of agrochemicals and consolidation of ditches (Hazell 2010), which decreased the species diversity of paddy-inhabiting organisms (Katayama et al. 2015; Kobori and Primack 2003). To ameliorate these negative effects, environmentally friendly farming (EFF) has become an increasingly popular option among farmers. Previous studies have shown that EFF enhances species diversity and abundance of organisms in paddy fields (Nakanishi et al. 2009; Tanaka and Ihara 2012; Ushio et al. 2014), but it may decrease crop productivity due to the prevalence of diseases, pests, and weeds of rice paddies. Thus, it is crucial to identify environmental conditions that could mitigate this tradeoff, such as the identification of landscape structures maintaining high densities of natural enemies in paddy fields under EFF (Kiritani 2000; Miyashita et al. 2014; Takada et al. 2012).
Spiders are dominant predators of insect pests in paddy ecosystems. Spiders of the genus Tetragnatha are common web-building spiders that appear to have potential in pest control (Amano et al. 2011; Kobayashi et al. 2011; Takada et al. 2012). In addition, these spiders were selected as indicators that can be used to evaluate the effect of EFF on the conservation and enhancement of biodiversity in paddy fields in Japan (Tanaka and Ihara 2013).
Earlier studies showed that the abundance of Tetragnatha spiders inhabiting grasslands, paddy fields and creeks was dependent on the surrounding forest cover (Amano et al. 2011; Miyashita et al. 2012; Yoshida 1981). Since the abundance of spiders is generally affected by food availability (Diehl et al. 2013; Miyashita et al. 2003; Polis et al. 1998), the high abundance of Tetragnatha spiders was supposed to be due to the bottom-up effect of prey insects, which emerge in high densities from paddy fields or grasslands with high surrounding forest cover (Amano et al. 2011; Miyashita et al. 2012; Yoshida 1981). Especially, dipterans may be the key prey for Tetragnatha spiders in paddy fields (Kiritani et al. 1972; Yoshida 1987). In order to test the bottom-up effect, it is necessary to demonstrate correlations between the abundance of dipterans and the population growth rate of Tetragnatha spiders, using time-series data, rather than snapshot correlations between the prey and predator abundance. It is also important to consider not only the abundance of spiders in paddy fields, but also in ditches, as Tetragnatha spiders prefer damp habitats (Yoshida 1981). In the non-crop season, paddy fields have little water and vegetation to support webs, and consequently are probably unsuitable habitats for Tetragnatha spiders, while ditches adjacent to paddy fields often hold water and vegetation almost year-round and may function as an important habitat for spiders. It is therefore necessary to examine spider population dynamics in landscapes including paddy fields and ditches to identify any limiting factors for their population growth rate.
The objective of this study was to identify the mechanisms that increase the abundance of Tetragnatha spiders in paddy fields under EFF by surveying population dynamics in a paddy-dominated landscape. Specifically we focused on testing:
-
1.
The population of Tetragnatha spiders, which was maintained by complementary utilization of paddy fields and ditches through the seasons.
-
2.
The positive effect of EFF on Tetragnatha spiders and flying insects, which was enhanced when the surrounding forest cover was higher.
-
3.
The population growth rate of Tetragnatha spiders, which was increased with an increasing density of flying insects.
-
4.
The density of Tetragnatha spiders in ditches in the non-crop season, which was also high when the surrounding forest cover was high and nearby paddy fields were under EFF.
Methods
Study area
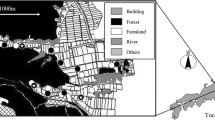
Field surveys were conducted in Shioya Town, Tochigi Prefecture, central Japan (36°44′N, 139°51′E). This study area is characterized by paddy-dominated landscapes mixed with cedar plantations (Fig. 1), and there were no natural wetlands or abandoned wet paddy fields that could serve as source habitats for Tetragnatha spiders and their major prey. Some of these paddy fields were under EFF, where the use of agrochemicals and chemical fertilizers was limited compared to conventional farming (CF), i.e., no use of insecticides since 2010, and an almost 50 % reduction, for at least 10 years, in the use of other chemicals, including fungicides, herbicides, and chemical fertilizers (see details in Table 1). Most of the paddy fields had both irrigation and drainage concrete ditches on opposite sides, with a similar width (about 40 cm). Moreover, all paddy fields located opposite, across ditches adjacent to the focal paddy fields, were managed by CF. These conditions enabled us to test whether farming methods and surrounding forest cover affected the abundance of Tetragnatha spiders and associated prey.
Data sampling
We selected 15 paddy fields under CF and 18 paddy fields under EFF with a varying degree of forest cover. Tetragnatha spiders were surveyed three times, i.e., mid-June, late July, and late October (28 October to 8 November) 2013. Tetragnatha spiders were expected to migrate from ditches to paddy fields in June, when rice plants started to grow, providing physical support for spider webs, and dipterans were abundant. From early September to early October when rice was harvested, Tetragnatha spiders were expected to migrate from paddy fields back to ditches.
In each paddy field, Tetragnatha spiders were captured from the head of rice plants by 30–70 sweeps, using an insect net with a 50-cm rod and a 36-cm diameter. Sweeping was conducted both at the center (5 m away from the levee) and the edge (from the levee up to 1 m) of each paddy field in July and October. Note that, in October, sweeping was conducted on tillers grown from rice stock. In June, sweeping was conducted only at the paddy edges to avoid damage to young rice plants. This did not seem to affect the main results, as Tetragnatha spiders were scarce in this season. Samples captured by sweeping were preserved in 80 % ethanol, and the number of Tetragnatha spiders was counted in the laboratory using a stereomicroscope (sweep count). In both irrigation and drainage ditches, the number of Tetragnatha spiders was determined visually (visual count) in a 5 to 10-m-long transect, depending on the size of the adjacent paddy field. Although sweeping was also conducted on grasses in levees, spiders were rarely captured here, so we focused only on paddy fields and ditches in our study.
Flying insects were collected with sticky transparent card traps (5 cm × 10 cm) coated with adhesive chemicals on both sides. Three to four sticky traps were placed in each paddy field (just above the rice plants) and in each ditch (at ground level), and left for 5 days in June and for 8 days in July. The number of flying insects was counted on both sides of a 5-cm × 5-cm area at the center of each plate using a stereomicroscope.
Conversion of sweep counts into visual counts
To allow direct comparison of spider densities between paddy fields and ditches it is necessary to convert sweep counts into visual counts by estimating the sampling efficiency of sweeping. A total of 26 and 29 quadrats (1 m × 2 m) were established in paddy fields when the average height of rice plants was low (June; 42 cm) and high (July; 87 cm), respectively. For each quadrat, visual counting of spiders followed by sweeping was conducted. The ratio of sweep count to visual count (p i ) was calculated, and its log odds ratio (β 1) was estimated by the following generalized liner model with a binomial error term:
where i is the trial and β 1 is then intercept to be estimated.
Sampling efficiency of sweep to visual counting (S) was estimated by the following formula:
As a result, β 1 was estimated to be −1.222 and S to be 0.231 in June, while β 1 was estimated to be 0.016 and S to be 0.504 in July. There was no strong relation between p i and visual count.
Finally, sweep counts were converted to visual counts by dividing the sweep counts by sampling efficiency of sweeping (S).
Next, the number of spiders in paddy fields and ditches on a visual-count basis was also estimated. The average area of paddy fields and the average length of ditches in all study sites were calculated, and these values were multiplied by the spider density (average per squared meter) in each paddy field or ditch adjacent to the paddy field, which is termed hereafter “spider abundance.” Note that spider abundance in ditches was the sum of irrigation and drainage ditches, by this way, it was possible to evaluate the relative importance of paddy fields and ditches as habitats for spiders.
Landscape variable
Surrounding forest cover was used as a landscape variable, as it influenced the abundance of spider in paddy-dominated landscapes (Amano et al. 2011; Miyashita et al. 2012). Forest cover within a 200-m-radius buffer around a study paddy field was calculated by using a geographic information system (GIS; Arc GIS10.1, ESRI). This spatial scale was so determined because overlaps of buffers of different paddy fields were minimal at this scale, and changing the buffer size to 200 or 400 m did not have large effects on the results of subsequent statistical analysis (results not shown). Land-use data were provided by the Japan Integrated Biodiversity Information System (J-IBIS; http://www.biodic.go.jp/J-IBIS.html; Ministry of the Environment, Japan).
Statistical analysis
A two-way ANOVA was used to analyze seasonal and habitat-type differences in the spider abundance. Here, the square-root-transformed spider abundance was the response variable, while season (June, July, or October) and habitat type (paddy fields or ditches) were the independent variables. The interaction of two independent variables was tested to identify complementary utilization of paddy fields and ditches by spiders. Abundance of dipterans was also analyzed in the same way as for spiders, except that the season included only June and July. If the interaction term of the two-way ANOVA was significant (p < 0.05), the abundance between paddy fields and adjacent ditches was compared for each season by a paired data t-test.
The effect of farming type and surrounding forest cover on the density of Tetragnatha spiders and the abundance of flying insects were analyzed using a generalized linear model. A negative binomial distribution was basically used for error terms, but a zero-inflated Poisson model was used when zero values were included in more than half of all the data. Note that sweep count was used for paddy fields while visual count was used for ditches because conversion should increase the uncertainty of the estimates and a direct comparison of densities between paddy fields and ditches was not the focus of this analysis. The density of Tetragnatha spiders or the abundance of flying insects was the response variable, while farming type (CF or EFF), surrounding forest cover, and their interaction were the independent variables. If the interaction term was not significant (p < 0.05), it was excluded from the model.
The effects of the abundance of flying insects on the population increase of Tetragnatha in paddy fields was examined using a general linear model. The response variable was log[(N in July)/(N in June)], where N is the density of spiders in a paddy fields. The independent variable was the density of flying insect in June or July.
All statistical analyses were performed with R-3.0.1 (R Development Core Team 2013).
Results
Seasonal population dynamics of Tetragnatha spiders and flying insects
A total of five Tetragnatha species were collected from paddy fields and ditches through three seasons. Among these, three species, Tetragnatha extensa Linnaeus, Tetragnatha maxillosa Thorell and Tetragnatha caudicula Karsch, comprised more than 90 % of the total.
For all Tetragnatha species, there is a clear difference in seasonal fluctuations of abundance between paddy fields and ditches (Fig. 2). Two-way ANOVA revealed that the seasons-habitat type interaction had significant effects on the abundance of all Tetragnatha species (Table 2). In July, the abundance of all Tetragnatha species was significantly higher in paddy fields than in ditches [T. extensa, t(31) = 2.056, p = 0.049; T. maxillosa, t(31) = 2.501, p = 0.018; T. caudicula, t(31) = 8.122, p < 0.001; Fig. 2]. In October, however, the abundance of T. extensa and T. maxillosa was higher in ditches than in paddy fields [p < 0.001, t(17) = −5.909; p < 0.001, t(17) = −5.740, respectively], while the abundance of T. caudicula was significantly higher in paddy fields than that in ditches throughout the study period [p < 0.001, t(17) = 5.740; Fig. 2].
Seasonal changes in the abundance of Tetragnatha spiders (Tetragnatha extensa, Tetragnatha maxillosa, and Tetragnatha caudicula) in paddy fields (grey bars) and ditches (open bars). Error bars indicate SE. For abbreviations, see Fig. 1
For flying insects, the interaction between season and habitat type was not significant, while the effect of habitat type was significant (Table 3), with flying insects more abundant in paddy fields.
Effects of surrounding forest cover and farming type on Tetragnatha spiders and flying insects
Farming type and surrounding forest cover generally affected the density of three Tetragnatha species in paddy fields, but not as much those in ditches. In July, there was a significant interaction effect of farming type and forest cover on the density of three spider species (Figs. 3, 4, 5). Spider density in paddy fields under EEF increased with increasing surrounding forest cover, whereas the density in paddy fields under CF remained at a relatively low level. In other seasons, EFF had a positive effect only on T. maxillosa in June (Fig. 4). In ditches, forest cover and farming type had no effect on the density of Tetragnatha spiders, except for the effect of surrounding forest cover on the density of T. maxillosa in October and T. caudicula in June (Figs. 4, 5).
Relationship between the density of Tetragnatha extensa and surrounding forest cover. Regression lines for paddies under EFF (solid line) and CF (broken line), respectively. Gray line is statistically not significant. Density of T. extensa in paddy fields under EFF (filled circles) and CF (open circles), respectively. Differences are considered significant at p < 0.05. For abbreviations, see Fig. 1
Relationship between the density of Tetragnatha maxillosa and surrounding forest cover. Regression lines for paddies under EFF (solid lines) and CF (broken lines), respectively. Density of T. maxillosa in paddy fields under EFF (filled circles) and CF (open circles), respectively. Differences are considered significant at p < 0.05. For abbreviations, see Fig. 1
Relationship between the density of Tetragnatha caudicula and surrounding forest cover. Regression lines for paddies under EFF (solid lines) and CF (broken lines), respectively. Density of T. caudicula in paddy fields under EFF (filled circles) and CF (open circles), respectively. Differences are considered significant at p < 0.05. For abbreviations, see Fig. 1
Flying insects captured from paddy fields and ditches combined in June were mainly Nematocera (90.5 %), followed by Hemiptera (3.4 %), Brachycera (2.8 %); percentages of these insects were quite similar in July (89.6 % Nematocera, 4.1 % Brachycera, and 3.5 % Hemiptera). The compositions of flying insects were similar between paddy fields and ditches (results not shown).
In paddy fields, the interaction effect of the surrounding forest cover and farming type on the abundance of flying insects was significant in June and July, (Fig. 6). In ditches, forest cover and farming type had no effect on the density of flying insects.
Relationship between the abundance of flying insect and surrounding forest cover. Regression lines in paddies under EFF (solid lines) and CF (broken lines), respectively. Abundance of flying insects in paddy fields under EFF (filled circles) and CF (open circles), respectively. Differences are considered significant at p < 0.05. For abbreviations, see Fig. 1
Correlation between the density of flying insects and the population growth rate of Tetragnatha spiders
As shown in Fig. 7, a significant positive correlation was identified between the population growth rate of two Tetragnatha species (T. extensa and T. maxillosa) and the density of flying insects in paddy fields in June. No significant correlation was identified in paddy fields in July, nor in ditches in any season.
Discussion
Our study revealed that the abundance of two Tetragnatha species (T. extensa and T. maxillosa) was higher in paddy fields than in ditches in the growing season, but was higher in ditches than in paddy fields in the non-crop season. Thus, the complementary utilization of ditches and paddy fields through the seasons was indicated as an important mechanism for the maintenance of Tetragnatha populations in paddy fields.
Since the abundance of dipteran in paddy fields in the growing season was relatively high, especially when the surrounding forest cover was high, the population increase of Tetragnatha spiders was probably due to the bottom-up effect of such prey insects. However, in the non-crop season, paddy fields were dry with little vegetation to support spider webs, which might have forced spiders to move to adjacent ditches for the non-crop season (fall–spring), either by walking or ballooning. The seasonal population trends of T. caudicula in paddy fields were different from those of the other two species. The abundance of T. caudicula in paddy fields was high even in the non-crop season, while in ditches it was very low in all seasons. T. caudicula appeared to prefer grasslands to water habitats.
Our study also revealed that the density of Tetragnatha spiders and the abundance of flying insects were increased by EFF, when the surrounding forest cover was high. Moreover, a significant positive correlation was identified between the abundance of dipterans and the population growth rate of two Tetragnatha spider species (T. extensa and T. maxillosa). Although a positive correlation between dipterans and Tetragnatha spiders has been already shown by snapshot data (Takada et al. 2014), we provided more direct evidence that dipterans exerted a bottom-up effect on Tetragnatha spiders in paddy fields.
The other reason for the increase in Tetragnatha spiders was that the forest might function as a refuge from disturbance or provide prey subsidy to paddy-dwelling spiders. However, Tetragnatha spiders were hardly observed at forest edges (M. H. Tsutsui, personal observation), so it is unlikely that the forest functioned as a refuge or source habitat for these spider species. It is also unlikely that dipterans which emerged from paddy fields originated from forests because forest-dwelling species may not be able to live in wet paddy fields. Although the reason for the high abundance of flying insects in paddy fields with a high surrounding forest cover was unclear, two mechanisms were considered. First, the presence of surrounding forests may ameliorate harsh microclimatic conditions in paddy fields in mid-summer, resulting in an increased abundance or temporal stability of emerging dipterans. Second, forest edges per se may function as overwintering or swarming sites for dipterans.
In conventional paddy fields, however, both the density of Tetragnatha spiders and the abundance of dipterans remained at low levels, irrespective of the surrounding forest cover. This was probably due to the strong effect of insecticides on dipterans living in paddy water during their larval stage, which indirectly decreased the density of spiders via the reduced bottom-up effect. As insecticides were applied to nursery boxes immediately before rice planting, their residue in paddy soil might have decreased the number of aquatic dipteran larvae, not Tetragnatha spiders living on paddy vegetation. It is unlikely that insecticides had a direct effect in June and July because insecticide application after rice planting was in August (Table 1). Earlier studies indicated that the effects of insecticides are different among invertebrate taxa and types of insecticides used (Hayasaka et al. 2012a, b; Takamura and Yasuno 1986). However, as these results were based on short-term experiments, they may not have captured cumulative effects of insecticide application at the natural community level. Further research is required to uncover the general effects of insecticide application on dipterans in the future.
Our study finally revealed that the density of Tetragnatha spiders in ditches in the non-crop season was little affected by the surrounding forest cover, despite their high density in the nearby paddy fields in the growing season. We consider that density-dependent population regulation might play a role in this because the area of ditches was much smaller than that of paddy fields, resulting in severe space limitation for spiders. Nevertheless, even when local density regulation occurred, EFF might have enhanced the density of Tetragnatha spiders at a larger spatial scale through their dispersal into local low-density areas. To test this possibility, it is necessary to use bigger study areas, comprising multiple landscape units, which will enable us to adopt the true “landscape approach” (McGarigal and Cushman 2002).
Our results indicated that Tetragnatha spider populations were maintained by complementary utilization of paddy fields and ditches through the seasons, as well as the bottom-up effect of dipterans emerging from paddy fields. This is in contrast with earlier studies in European agricultural croplands, where the spillover effect from surrounding source habitats was the main process that enhanced the abundance and/or species diversity in dry croplands (Blitzer et al. 2012; Schmidt et al. 2008; Tscharntke et al. 2012), although the importance of spillover from croplands to surrounding habitats is hypothetical (Rand et al. 2006). This difference may be attributed to the higher productivity in still, shallow-water environments (paddy fields) compared to dry terrestrial environments (cereal croplands) (Cebrian 1999; Kikuchi et al. 2012). Thus, in paddy-dominated ecosystems, croplands per se are high-quality habitats, whereas surrounding components, such as ditches, levees, and forest edges, provide alternative habitats in the non-crop season, or they ameliorate environmental conditions inside the paddy fields. It is possible that agronomic management practices in paddy fields, such as the utilization of agrochemicals, may have a stronger impact on organisms than that in dry arable lands. Furthermore, we suggest that the combined existence of paddy fields and ditches may be important for the maintenance of predator abundance in paddy fields. These results provide novel insights into the management of paddy-dominated ecosystems. Recent agricultural intensification has led to changes in water-management systems. Although such water management has contributed to an increased rice yield, the construction of pipelines and underground drains has resulted in the loss of open ditches that has disrupted the complementary utilization of alternative water habitats by marsh predators, including Tetragnatha spiders, and consequently might have reduced pest-control services. It is therefore important to pay more attention to these indirect effects in the near future.
References
Amano T, Kusumoto Y, Okamura H, Baba YG, Hamasaki K, Tanaka K, Yamamoto S (2011) A macro-scale perspective on within-farm management: how climate and topography alter the effect of farming practices. Ecol Lett 14:1263–1272
Blitzer EJ, Dormann CF, Holzschuh A, Klein AM, Rand TA, Tscharntke T (2012) Spillover of functionally important organisms between managed and natural habitats. Agric Ecosyst Environ 146:34–43
Cebrian J (1999) Patterns in the fate of production in plant communities. Am Nat 154:449–468
Diehl E, Mader VL, Wolters V, Birkhofer K (2013) Management intensity and vegetation complexity affect web-building spiders and their prey. Oecologia 173:579–589
Elphick CS (2000) Functional equivalency between rice fields and seminatural wetland habitats. Conserv Biol 14:181–191
Hayasaka D, Korenaga T, Sánchez-Bayo F, Goka K (2012a) Differences in ecological impacts of systemic insecticides with different physicochemical properties on biocenosis of experimental paddy fields. Ecotoxicology 21:191–201
Hayasaka D, Korenaga T, Suzuki K, Saito F, Sanchez-Bayo F, Goka K (2012b) Cumulative ecological impacts of two successive annual treatments of imidacloprid and fipronil on aquatic communities of paddy mesocosms. Ecotox Environ Safe 80:355–362
Hazell PBR (2010) The Asian green revolution. In: Spielman DJ, Pandya-Lorch R (eds) Proven successes in agricultural development: a technical compendium to Millions Fed. International Food Policy Research Institute, Washington, DC, pp 67–98
Katano O, Hosoya K, Iguchi K, Yamaguchi M, Aonuma Y, Kitano S (2003) Species diversity and abundance of freshwater fishes in irrigation ditches around rice fields. Environ Biol Fish 66:107–121
Katayama N, Baba YG, Kusumoto Y, Tanaka K (2015) A review of post-war changes in rice farming and biodiversity in Japan. Agric Syst 132:73–84
Katoh K, Sakai S, Takahashi T (2009) Factors maintaining species diversity in satoyama, a traditional agricultural landscape of Japan. Biol Conserv 1429:1930–1936
Kikuchi E, Takagi Y, Shikano S (2012) Surveys of plankton (aquatic microorganisms) communities in rice fields. Res Bull Environ Educ Cent Miyagi Univ Educ 14:7–15 (in Japanese with English summary)
Kiritani K (2000) Integrated biodiversity management in paddy fields: shift of paradigm from IPM toward IBM. Integr Pest Manage Rev 5:175–183
Kiritani K, Kawahara S, Sasaba T, Nakasuji F (1972) Quantitative evaluation of predation by spiders on the green rice leafhopper, Nephotettix cincticeps Uhler, by a sight-count method. Res Popul Ecol 13:187–200
Kobayashi T, Takada M, Takagi S, Yoshioka A, Washitani I (2011) Spider predation on a mirid pest in Japanese rice fields. Basic Appl Ecol 12:532–539
Kobori H, Primack RB (2003) Participatory conservation approaches for Satoyama, the traditional forest and agricultural landscape of Japan. AMBIO J Hum Env 32:307–311
Maclean JL, Dawe DC, Hardy B, Hettel GP (2002) Rice almanac: source book for the most important economic activity of earth. CABI, Wallingford, pp 59–236
McGarigal K, Cushman SA (2002) Comparative evaluation of experimental approaches to the study of habitat fragmentation effects. Ecol Appl 12:335–345
Miyashita T, Takada M, Shimazaki A (2003) Experimental evidence that aboveground predators are sustained by underground detritivores. Oikos 103:31–36
Miyashita T, Chishiki Y, Takagi RS (2012) Landscape heterogeneity at multiple spatial scales enhances spider species richness in an agricultural landscape. Popul Ecol 54:573–581
Miyashita T, Yamanaka M, Tsutsui HM (2014) Distribution and abundance of organisms in paddy-dominated landscapes with implication for wildlife-friendly farming. In: Miyashita T, Nishikawa U (eds) Social-ecological restoration in paddy-dominated landscapes. Springer, Japan, pp 45–65
Nakanishi K, Tawa K, Kanbara B, Noma N, Sawada H (2009) Comparisons of macro-aquatic animal communities among paddy fields under different cultivation management systems. Jpn J Environ Entomol Zool 20:103–114
Pimentel D, Stachow U, Takacs DA, Brubaker HW, Dumas AR, Meaney JJ, Oneil JAS, Onsi DE, Corzilius DB (1992) Conserving biological diversity in agricultural/forestry systems. Bioscience 42:354–362
Polis GA, Hurd SD, Jackson CT, Sanchez-Piñero F (1998) Multifactor population limitation: variable spatial and temporal control of spiders on Gulf of California islands. Ecology 79:490–502
R Development Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, https://cran.r-project.org/bin/windows/base/old/3.0.1/. Accessed 5 Nov 2013
Rand TA, Tylianakis JM, Tscharntke T (2006) Spillover edge effects: the dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats. Ecol Lett 9:603–614
Schmidt MH, Thies C, Nentwig W, Tscharntke T (2008) Contrasting responses of arable spiders to the landscape matrix at different spatial scales. J Biogeogr 35:157–166
Takada MB, Yoshioka A, Takagi S, Iwabuchi S, Washitani I (2012) Multiple spatial scale factors affecting mirid bug abundance and damage level in organic rice paddies. Biol Control 60:169–174
Takada MB, Takagi S, Iwabuchi S, Mineta T, Washitani I (2014) Comparison of generalist predators in winter-flooded and conventionally managed rice paddies and identification of their limiting factors. SpringerPlus 3:418
Takamura K, Yasuno M (1986) Effects of pesticide application on chironomid larvae and ostracods in rice fields. Appl Entomol Zool 21:370–376
Tanaka K, Ihara H (2012) Biodiversity research for the development of indicator organisms in environment-preserving agriculture. In: Nakano S, Yahara T, Nakashizuka T (eds) The biodiversity observation networks in the Asia-Pasific Region. Springer, Japan, pp 95–113
Tanaka K, Ihara H (2013) Outline of the research project on biodiversity and the manual of indicator animals. JATAFF J 1:2–8 (in Japanese with English summary)
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF et al (2012) Landscape moderation of biodiversity patterns and processes—eight hypotheses. Biol Rev 87:661–685
Ushio N, Saito R, Akanuma H, Watanabe R (2014) Effectiveness of wildlife-friendly farming on aquatic macroinvertebrate diversity on Sado Island in Japan. In: Miyashita T, Nishikawa U (eds) Social-ecological restoration in paddy-dominated landscapes. Springer, Japan, pp 95–113
Yoshida M (1981) Preliminary study on the ecology of three horizontal orb weavers, Tetragnatha praedonia, T. japonica, and T. pinicola (Araneae: Tetragnathidae). Acta Arachnol 30:49–64
Yoshida M (1987) Predatory behavior of Tetragnatha praedonia (Araneae: Tetragnathidae). Acta Arachnol 35:57–75
Acknowledgments
We thank S. Sugiyama and all the farmers who permitted us to conducted surveys in their paddy fields, and we also thank G. Fujita, S. Naoe, S, Nishijima and other members of the Laboratory of Biodiversity Science for their help with the fieldwork and helpful suggestions. We would like to thank T. Kagaya for long-term support of our fieldwork, A. Tanikawa for advice on spider identification and Y. Osada for valuable advice on statistical analysis. This study was supported by the Global COE programme (the Center of Excellence for Asian Conservation Ecology) and JSPS KAKENHI (Grant No. 25292210, 15K14600).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsutsui, M.H., Tanaka, K., Baba, Y.G. et al. Spatio-temporal dynamics of generalist predators (Tetragnatha spider) in environmentally friendly paddy fields. Appl Entomol Zool 51, 631–640 (2016). https://doi.org/10.1007/s13355-016-0440-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-016-0440-5