Abstract
The detrimental effects of brother–sister and nephew–aunt matings on the colony foundation success of Bombus terrestris (Linnaeus, 1758) queens were investigated under controlled laboratory conditions. Among 86 hibernated queens, 43 were mated with randomly selected males from the same colony (inbred group), and 43 were mated with unrelated males from other colonies (outbred group). Nests were observed twice a week, and some developmental determinants of colony quality were recorded. The results suggest that inbreeding adversely affects the time taken to initiate a colony, the time taken for the first male to emerge, the number of workers in the first brood, and the total number of workers and young queens. The percentage of the queens that produced at least 50 workers significantly differed between the inbred and outbred groups (13.95 and 62.79 %, respectively). In the inbred group, 48.83 % of the queens produced both workers and males in their first brood, versus 4.65 % in the outbred group. Although the total number of individuals in the first brood was not significantly different for the two groups, the inbred group produced a higher proportion of males in the first brood (43.99 versus 12.74 % in the outbred group). Thus, inbreeding has a substantial deleterious effect on captive B. terrestris populations, so it is necessary to maintain genetic diversity and prevent inbreeding when aiming for the sustainable laboratory rearing of B. terrestris.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The large-scale laboratory rearing of Bombus terrestris (Linnaeus, 1758) has been promoted around the world since 1985, and is crucial to greenhouse tomato pollination. Annually, more than one million B. terrestris colonies are commercially produced by over 30 producers worldwide, with most of the market share covered by a few global companies (Reade et al. 2014; Velthuis and van Doorn 2006). Colonies have been shipped throughout the world to supply the constant demand (Lecocq et al. 2016a).
The year round rearing procedure of B. terrestris involves three stages: the rearing of colonies, the mating of queens and males, and diapause (Velthuis and van Doorn 2006). Some losses occur during each of these stages, depending on the mating ratio, survival ratio, egg-laying ratio, and finally the ratio of colony-founding queens (Amin et al. 2010; Gurel and Gosterit 2008). Significant variations are seen in the time taken to lay eggs, the proportion of queens that lay eggs, the total number of workers, queens, and males produced, and (especially) whether the B. terrestris colony reaches a sufficient size to carry out pollination. Many factors relating to environmental conditions, genetic structure, and the physiological properties of the queens affect colony foundation success (Velthuis and van Doorn 2006). The proportion of the colonies that reach a sufficient size to perform pollination, which varies from 40 to 50 % even under optimal conditions, is an important criterion in the mass-rearing of B. terrestris (Gosterit and Gurel 2009). One of the most important requirements to improve the colony foundation success rate is to prevent inbreeding. Because bumblebees present single-locus complementary sex determination (sl-CSD) (Cook and Crozier 1995; Zayed and Packer 2005), they are particularly vulnerable to inbreeding costs such as the production of diploid males and reductions in the size of the worker force, the growth, and the reproductive capacity of the colony. Therefore, successful colony production depends on the mating of unrelated queens and males.
Like honeybees (Apis mellifera Linnaeus) and several other hymenopteran species, the sex of bumblebees is determined by haplodiploidy (Duchateau et al. 1994). Fertilized eggs laid by mated queens develop into diploid females (workers or young queens) if they are heterozygous at a single sex locus or diploid males if they are homozygous at this sex locus (sl-CSD) (Heimpel and de Boer 2008; Whitehorn et al. 2009a). The production of diploid males increases dramatically with inbreeding. Matings such as brother–sister, niece–nephew, and nephew–aunt increase the frequencies of homozygous sex alleles by varying degrees (Duchateau and Marien 1995; Gerloff and Schmid-Hempel 2005). Towards the end of the colony life cycle, founder queens and reproductive workers lay unfertilized hemizygote eggs which develop into haploid males (Alaux et al. 2007).
Diploid male production (DMP) rarely occurs in large haplodiploid populations such as honeybees. Honeybee queens mate in flight with numerous males from various genetic sources, and the sex of their offspring depends on multiple alleles at the sex locus (Cobey 2007). A honeybee colony comprises a superfamily of three worker groups: supersisters, half-sisters, and full sisters (Harbo and Rinderer 1980). This complicated family structure and the mating behavior of queens reduces inbreeding costs in A. mellifera populations (Oldroyd et al. 1992; Stockley et al. 1993).
In contrast to A. mellifera, B. terrestris queens mate once, and their colonies only include supersisters (Schmid-Hempel and Schmid-Hempel 2000). In the annual reproductive cycle of B. terrestris, the low number of sex alleles increases the possibility of mating with close relatives. Inbreeding in haplodiploid species can lead to the production of homozygotes with the male phenotype (Duchateau et al. 1994; Duchateau and Marien 1995). When a queen mates with a related male, the fertilized eggs yield diploid males at the beginning of the colony’s life cycle, reducing the size of the worker force, the growth, and the reproductive capacity of the colony (Ayabe et al. 2004; Maebe et al. 2014). Therefore, early male production can hinder successful colony foundation (Hingston 2005; Whitehorn et al. 2009b). Mating with a brother also significantly reduces the likelihood that the queen will survive hibernation (Gerloff and Schmid-Hempel 2005). Viability and immune response are generally lower in diploid males than in haploid males (Gerloff et al. 2003). Queens can mate with diploid males to produce triploid workers and progeny queens, but the triploid queens are infertile (Ayabe et al. 2004; Darvill et al. 2012). These negative effects of inbreeding reduce the size of the worker population and make it less likely that a colony will become sufficiently large to carry out pollination as expected, and therefore determine the ability of colonies to act as pollinators as well as the success of mass rearing if the bumblebees are being reared by a breeder. In the study reported here, I investigated the adverse effects of brother–sister and nephew–aunt matings of B. terrestris under controlled laboratory conditions.
Materials and methods
Young queens (gynes) and males were obtained from 43 maternal B. terrestris colonies that produced both sexes; 22 colonies from one bumblebee breeder (Bio Group, Antalya, Turkey) and another 21 colonies from a second bumblebee breeder (Agrobay, İzmir, Turkey). These maternal colonies were considered to be genetically diverse because each breeder had reared colonies on a large scale and needed to maintain sufficient genetic diversity to allow sustainable rearing. The maternal colonies were kept in captivity under standard laboratory rearing conditions (28–29 °C; 50 % RH). To establish the inbred group (brother–sister or nephew–aunt matings), young queens from these colonies were mated with randomly selected males produced by founder queens and/or reproductive workers from the same colony. The queens and males produced in colonies from the two different bumblebee breeders were hybridized to generate an outbred group. In both groups, queens were mated with males in mating cages in an illuminated mating room at 23 ± 1 °C and 50 ± 5 % RH (Amin et al. 2010) and then placed in a cold room at 2.5 °C for 2 months to artificially induce diapause (Gosterit and Gurel 2009). The mating pair was removed during copulation to avoid harassment from other males (Lhomme et al. 2013). After the hibernation period, the 86 surviving queens (43 inbred and 43 outbred matings; only one queen was randomly selected from each maternal colony for both groups) were placed into individual starting boxes (8 × 8 × 6 cm) and transferred to the same rearing room (28–29 °C; 50 % RH). To stimulate egg laying, all queens were subjected to CO2 narcosis according to protocols reported by Lhomme et al. (2013). One honeybee worker was placed next to each queen instead of a B. terrestris worker (Gurel and Gosterit 2008). This process prevented the possibility of male production by these B. terrestris workers in the first brood. When all of the workers in the first brood had emerged, the colonies were transferred to larger plastic boxes (20 × 30 × 25 cm) obtained from a commercial bumblebee breeder. The queens and their colonies were fed ad libitum with freshly thawed pollen and sugar solutions (50 Brix) (Riberio et al. 1996).
Nests were observed twice a week, and developmental traits which are affected by inbreeding—e.g., the timing of egg laying by queens (colony initiation), the number of egg cells in the first brood, the time taken for the first worker to emerge (the beginning of the social phase; Duchateau and Velthuis 1988), the time taken for the first male to emerge, the numbers of workers and males in the first brood, the time taken to produce a young queen (progeny queen or gyne), and the total number of workers, males, and young queens produced by each colony—were recorded. These traits are also related, directly and indirectly, to the colony foundation success of a queen and the ability of a colony to perform pollination. The total number of workers, males, and queens produced included the number of dead bees in the colony, which were removed and noted. Furthermore, the numbers of workers and males in all of the colonies were counted weekly to determine population growth. Queens that produced more than 10 workers were considered to have successfully founded colonies, and colonies that included at least 50 workers were considered to be sufficiently large to perform pollination (Gurel and Gosterit 2008; Velthuis and van Doorn 2006). The time taken to produce a gyne was counted from the beginning of the social phase; other traits were measured from when the queens were placed in their starting boxes. To calculate the gyne production time, the developmental time of an average gyne (30 days) was subtracted from the emergence date of the first offspring queen (Duchateau and Velthuis 1988).
One-way analysis was performed to determine the effects of inbreeding on colony foundation success (Minitab statistical software, version 16.2.4). All data were tested for normality and homogeneity of variances using the Shapiro–Wilk and Levene tests before analysis. Data that did not meet the assumptions of ANOVA were square-root transformed. The percentages of the queens that laid eggs and produced 10 and 50 workers were compared by two-proportion z-tests.
Results
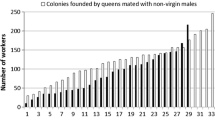
Egg laying was accomplished by 42 queens in the inbred group and 40 queens in the outbred group. The percentage of the queens that produced at least 10 workers was not significantly different in the inbred and outbred groups (62.79 and 79.06 %, respectively; z = −1.69, p = 0.153). However, the percentage of the queens that produced at least 50 workers was significantly lower in the inbred group (13.95 % in the inbred group versus 62.79 % in the outbred group; z = −4.66, p < 0.001).
The mating of queens with randomly selected males from the same colony significantly affected characteristics that were directly related to yearly rearing success (Table 1), such as colony initiation (F = 7.49, p < 0.01), time taken for the first male to emerge (F = 42.93, p < 0.001), number of workers in the first brood (F = 6.09, p < 0.05), total number of workers (F = 50.57, p < 0.001), and the total number of young queens (F = 16.53, p < 0.001). The first males emerged earlier in the inbred than in the outbred colonies. Although the inbred and outbred colonies yielded almost the same total numbers of individuals in their first broods, the ratio of males in the first brood was 43.99 % in the inbred group and 12.74 % in the outbred groups (z = 5.39, p < 0.001). In the inbred group, 48.83 % of the queens produced both workers and males in their first broods; the corresponding percentage was 4.65 % in the outbred group (z = 8.07, p < 0.001).
In both the inbred and outbred colonies, the first workers emerged in the sixth week. However, unlike the first-worker emergence time, the first-male emergence time significantly differed between the groups. Specifically, the first males emerged at the beginning of week 7 and in week 10 in the inbred and outbred colonies, respectively (Table 2).
Discussion
To ensure that high-quality colonies are obtained in successive generations, young B. terrestris queens must be reared in sufficient numbers each year, and they must be mated with unrelated males. However, in a closed breeding system, the low number of sex alleles present increases the probability of queens mating with related males (Duchateau et al. 1994). As queens and related males share the same alleles, the resulting worker population is half the size of the worker population that would be produced by an unrelated mating (Cook and Crozier 1995; Hingston 2005). Therefore, in B. terrestris, inbreeding adversely affects the success rate of colony production, the survival of the colony, and the immune response of individuals (Gerloff et al. 2003; Whitehorn et al. 2009b). The same effects have also been reported for A. mellifera (Bienefeld et al. 1989).
The most potentially disastrous effects occur when queens mate with males from the same colony. Early in its life cycle, a B. terrestris colony is headed by a single monandrous queen which is solely responsible for egg laying (Schmid-Hempel and Schmid-Hempel 2000). After the competition point (the observed time at which worker oviposition, oophagy, and egg-cup destruction occur; Bloch and Hefetz 1999; Duchateau and Velthuis 1988), workers can lay eggs that produce male offspring (Alaux et al. 2004). Thus, when randomly selected males mate with queens from the same colony, we can expect three mating combinations: (1) brother–sister only, (2) nephew–aunt only, and (3) both brother–sister and nephew–aunt matings. For each of these mating combinations, Table 3 lists the probability of diploid male production (DMP) in future generations.
In this experiment, the negative effect of inbreeding on B. terrestris colonies was investigated by randomly selecting young queens and males from the same colony and mating them. The results confirmed that inbreeding has a strong negative effect on several colony traits that determine the pollination quality of the colony. The adverse effects of inbreeding appeared early in the colony’s life. The first males emerged earlier in the inbred group than in the outbred group, reducing the worker force in colonies of the former group. Although a method of distinguishing haploid from diploid males was reported by Gerard et al. (2015), any test was not employed to distinguish diploid and haploid males in the present experiment. For this reason, it is not known for certain whether the males that emerged the earliest in inbred colonies were diploid. In both groups, helper B. terrestris workers were not used to stimulate the oviposition of queens, so the males produced in the first brood developed only from eggs laid by the queens. Inbreeding resulting from mating between siblings reduces sex alleles in hymenopteran populations with sl-CSD (Heimpel and de Boer 2008). If B. terrestris has only a single sex locus, half of the brood produced in the growth or ergonomic phase of the colony (during which only workers are produced) develop into diploid males instead of workers (Duchateau et al. 1994). Whitehorn et al. (2009b) investigated the costs of brother–sister mating in B. terrestris under field conditions and suggested that diploid male colonies exhibit significantly reduced fitness, slower growth rates, and significantly fewer offspring than haploid male colonies. Unlike Whitehorn et al. (2009b), the present study was conducted to investigate brother–sister and nephew–aunt matings of B. terrestris under controlled laboratory conditions. However, I did not categorize inbred colonies on the basis of whether they produced diploid males in this study. Our results showed that the total number of workers produced during the lifetime of the colony was significantly reduced by the decreased size of the worker force during early developmental stages. Moreover, the total number of workers determines whether the colony reaches a sufficient size to carry out pollination. The total number of young queens that developed from diploid heterozygote eggs also significantly differed for the inbred and outbred colonies.
In conclusion, inbreeding has a substantial deleterious effect on captive B. terrestris populations. Diploid males exhibit low viability (they are sterile or unable to mate), and are more likely to emerge in small or inbreeding populations (Duchateau and Marien 1995; Stouthamer et al. 1992). No data were saved that would have allowed us to determine the diapause performance in this experiment. However, Gerloff and Schmid-Hempel (2005) reported that inbreeding significantly reduces the likelihood of the queen surviving hibernation. Inbreeding or mating with relatives also yields infertile triploid queens and negatively affects the population’s reproductive success in future generations (Ayabe et al. 2004; Darvill et al. 2012). Although pre-mating mechanisms that prevent mating with relatives have been reported for some Hymenoptera (van Wilgenburg et al. 2006), a B. terrestris queen will mate with her brother or nephew (Lecocq et al., 2016b), as seen in the present study. The results of study clearly show that mating stage is a crucial influence on the ability to sustain rearing activities all year round. The reduction in the size of the worker force and the very low pollinator colony production ratio observed for the inbred group are serious problems in mass rearing. Therefore, a mating program that avoids inbreeding and maintains genetic diversity should be implemented for laboratory rearing. Inbreeding can be prevented when pedigrees are known and recorded. Additionally, colonies that produce males early should be eliminated, and the mating of queens with males from such colonies should be avoided. Males and queens from different commercial stocks could also be used to diversify the genetic pool and prevent adverse effects of inbreeding, but this option requires disease and parasite control. Bumblebee breeders could also divide their stocks into different groups, and then queens and males from different groups could be mated.
References
Alaux C, Savarit F, Jaisson P, Hefetz A (2004) Does the queen win it all? Queen-worker conflict over male production in the bumblebee, Bombus terrestris. Naturwissenchaften 91:400–403. doi:10.1007/s00114-004-0547-3
Alaux C, Boutot M, Jaisson P, Hefetz A (2007) Reproductive plasticity in bumblebee workers (Bombus terrestris)—reversion from fertility to sterility under queen influence. Behav Ecol Sociobiol 62:213–222. doi:10.1007/s00265-007-0455-6
Amin MR, Than KK, Kwon YJ (2010) Mating status of bumblebees, Bombus terrestris (Hymenoptera: Apidae) with notes on ambient temperature, age and virginity. Appl Entomol Zool 45:363–367. doi:10.1303/aez.2010.363
Ayabe T, Hoshiba H, Ono M (2004) Cytological evidence for triploid males and females in the bumblebee, Bombus terrestris. Chromosome Res 12:215–223
Bienefeld K, Reinhardt F, Pirchner F (1989) Inbreeding effects of queen and workers on colony traits in the honey bee. Apidologie 20:439–450
Bloch G, Hefetz A (1999) Regulation of reproduction by dominant workers in bumblebee (Bombus terrestris) queenright colonies. Behav Ecol Sociobiol 45:125–135
Cobey SW (2007) Comparison studies of instrumentally inseminated and naturally mated honey bee queens and factors affecting their performance. Apidologie 38:390–410. doi:10.1051/apido:2007029
Cook JM, Crozier RH (1995) Sex determination and population biology in the Hymenoptera. Trends Ecol Evol 10:281–286
Darvill B, Lepais O, Woodall LC, Goulson D (2012) Triploid bumblebees indicate a direct cost of inbreeding in fragmented populations. Mol Ecol 21:3988–3995. doi:10.1111/j.1365-294X.2012.05679.x
Duchateau MJ, Marien J (1995) Sexual biology of haploid and diploid males in the bumble bee Bombus terrestris. Insectes Soc 42:255–266
Duchateau MJ, Velthuis HHW (1988) Development and reproductive strategies in Bombus terrestris colonies. Behaviour 107:186–207
Duchateau MJ, Hoshiba H, Velthuis HHW (1994) Diploid males in the bumble bee Bombus terrestris, sex determination, sex alleles and viability. Entomol Exp Appl 71:263–269
Gerard M, Michez D, Fournier D, Maebe K, Smagghe G, Biesmeijer JC, de Meulemeester T (2015) Discrimination of haploid and diploid males of Bombus terrestris (Hymenoptera; Apidae) based on wing shape. Apidologie 46:644–653. doi:10.1007/s13592-015-0352-3
Gerloff CU, Schmid-Hempel P (2005) Inbreeding depression and family variation in a social insect, Bombus terrestris (Hymenoptera: Apidae). Oikos 111:67–80
Gerloff CU, Ottmer BK, Schmid-Hempel P (2003) Effects of inbreeding on immune response and body size in a social insect, Bombus terrestris. Funct Ecol 17:582–589
Gosterit A, Gurel F (2009) Effect of different diapause regimes on survival and colony development in the bumble bee, Bombus terrestris. J Apic Res 48:279–283. doi:10.3896/IBRA.1.48.4.08
Gurel F, Gosterit A (2008) Effects of different stimulation methods on colony initiation and development of Bombus terrestris L. (Hymenoptera: Apidae) queens. Appl Entomol Zool 43:113–117. doi:10.1303/aez.2008.113
Harbo J, Rinderer TE (1980) Breeding and genetics of honey bees. USDA/ARS Agriculture Handbook 335:49–57
Heimpel GE, de Boer JG (2008) Sex determination in the Hymenoptera. Ann Rev Entomol 53:209–230. doi:10.1146/annurev.ento.53.103106.093441
Hingston AB (2005) Inbreeding in the introduced bumblebee Bombus terrestris causes uncertainty in predictions of impacts on native ecosystems. Ecol Manag Restor 6:151–153
Lecocq T, Rasmont P, Harpke A, Schweiger O (2016a) Improving international trade regulation by considering intraspecific variation for invasion risk assessment of commercially traded species: the Bombus terrestris case. Conserv Lett. doi:10.1111/conl.12215
Lecocq T, Gerard M, Maebe K, Brasero N, Dehon L, Smagghe G, Valterova I, de Meulemeester T, Rasmont P, Michez D (2016b) Chemical reproductive traits of diploid Bombus terrestris males: consequences on bumblebee conservation. Insect Sci. doi:10.1111/1744-7917.12332
Lhomme P, Sramkova A, Kreuter K, Lecocq T, Rasmont P, Ayasse M (2013) A method for year-round rearing of cuckoo bumblebees (Hymenoptera: Apoidea: Bombus subgenus Psithyrus). Ann Soc Entomol Fr 49:117–125. doi:10.1080/00379271.2013.774949
Maebe K, Meeus I, Ganne M, Wackers F, Smagghe G (2014) Scientific note on microsatellite DNA analyses revealing diploid and haploid drones in bumblebee mass breeding. Apidologie 45:189–191. doi:10.1007/s13592-013-0237-2
Oldroyd BJ, Rinderer TE, Harbo JR, Buco SM (1992) Effects of intracolonial genetic diversity on honey bee (Hymenoptera: Apidae) colony performance. Ann Entomol Soc Am 85:335–343
Reade C, Goka K, Thorp R, Mitsuhata M, Wasbauer M (2014) CSR, biodiversity and Japan’s stakeholder approach to the global bumble bee trade. J Corp Citizsh 56:53–66. doi:10.9774/GLEAF.4700.2014.de.00006
Riberio MF, Duchateau MJ, Velthuis HHW (1996) Comparison of the effects of two kinds of commercially pollen on colony development and queen production in bumble bee Bombus terrestris L (Hymenoptera, Apidae). Apidologie 27:133–144
Schmid-Hempel R, Schmid-Hempel P (2000) Female mating frequencies in Bombus spp. from Central Europe. Insectes Soc 47:36–41
Stockley P, Searle JB, Macdonald DW, Jones CS (1993) Female multiple mating behaviour in the common shrew as a strategy to reduce inbreeding. Biol Sci 254:173–179
Stouthamer R, Luck RF, Werren JH (1992) Genetics of sex determination and the improvement of biological control using parasitoids. Environ Entomol 21:427–435
van Wilgenburg Driessen G, Beukeboom LW (2006) Single locus complementary sex determination in Hymenoptera: an “unintelligent” design? Front Zool 3:1. doi:10.1186/1742-9994-3-1
Velthuis HHW, van Doorn A (2006) A century of advances in bumblebee domestication and the economic and environmental aspects of its commercialization for pollination. Apidologie 37:421–451. doi:10.1051/apido:2006019
Whitehorn PR, Tinsley MC, Goulson D (2009a) Kin recognition and inbreeding reluctance in bumblebees. Apidologie 40:627–633. doi:10.1051/apido/2009050
Whitehorn PR, Tinsley MC, Brown MJF, Darvill B, Goulson D (2009b) Impacts of inbreeding on bumblebee colony fitness under field conditions. BMC Evol Biol 9:152. doi:10.1186/1471-2148-9-152
Zayed A, Packer L (2005) Complementary sex determination substantially increases extinction proneness of haplodiploid populations. Proc Natl Acad Sci USA 102:10742–10746. doi:10.1073/pnas.0502271102
Acknowledgments
This study was supported by the Scientific Research Projects Unit of Süleyman Demirel University (project number: SDU-05427) and the Scientific and Technical Research Council of Turkey (project number: 114O645).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gosterit, A. Adverse effects of inbreeding on colony foundation success in bumblebees, Bombus terrestris (Hymenoptera: Apidae). Appl Entomol Zool 51, 521–526 (2016). https://doi.org/10.1007/s13355-016-0427-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-016-0427-2