Abstract
Aerosol inhalation of amphotericin B (AmB) can be a clinically compliant way to administer the drug directly to the pulmonary route for treatment as well as prophylaxis of invasive pulmonary aspergillosis (IPA). We report aerosol formulation of AmB using sodium deoxycholate sulfate (SDCS), a lipid carrier synthesized in-house using natural precursor deoxycholic acid. In vitro toxicity was determined by MTT assay. Biodistribution and histopathology in rats were evaluated in targeted organs including the lungs, kidneys, spleen, and liver. No toxicity was observed when lung and kidney cells treated with AmB-SDCS formulations up to 8 μg/mL and minimal toxicity at higher concentration 16 μg/mL, while the Fungizone®-like formulation induced toxicity to lung and kidney cells with viability decreasing from 86 to 41% and 100 to 49%, respectively, when compared with an equivalent concentration of AmB-SDCS. Renal and hepatic markers were raised for Fungizone®-like formulation–treated rats but not for AmB-SDCS formulations following 7 days of regular dosing by intratracheal instillation. AmB concentrations were highest in the lungs (5.4–8.3 μg/g) which were well above minimum inhibitory concentration (MIC) of all Aspergillus species. Plasma concentration was also above MIC (> 2 μg/mL) for all AmB-SDCS formulations in comparison with Fungizone®-like formulation. No evidence of abnormal histopathology was observed in the lungs, liver, spleen, and kidneys for all AmB-SDCS formulations but was observed for the group treated with Fungizone®-like formulation. It is concluded that AmB-SDCS formulations can be efficiently administered via intratracheal instillation with no evidence of toxicity and may find great value in the treatment as well as prophylaxis of IPA through inhalation route.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive pulmonary aspergillosis (IPA) is considered a major cause of morbidity and mortality in immune-compromised individuals, such as transplant recipients, HIV-infected individuals, cancer patients with prolonged neutropenia, or those with inherited immune-deficiency [1]. The mortality rate in these patients is more than 90% in severe cases owing to the difficulty in early diagnosis and unavailability of effective treatment options [2].
Amphotericin B (AmB), a polyene macrolide antifungal, is the drug of choice for systemic fungal infections and is usually administered intravenously. The most serious adverse effect of AmB is nephrotoxicity and AmB induces hematological side effects and infusion-related reactions [3]. In order to increase the therapeutic potential and overcome the toxicity, AmB has been incorporated in various lipid-based preparations. Three FDA-approved lipid-based preparations are AmBisome™ (a liposomal formulation), Abelecet™ (a lipid complex), and Amphotec™ (a colloidal dispersion) [4] whereas Fungizone® (a deoxycholic acid complex) is the conventional formulation. These lipid-based formulations have several advantages in the context of solubility and reduced adverse effects while maintaining therapeutic activity. However, some drawbacks associated with these lipid-based formulations are the high cost of therapy and varying pharmacokinetic profiles which limit their use [5, 6]. Therefore, AmB has been formulated to various other dosage forms, such as nanoparticles and emulsions to obtain higher efficacy with minimal adverse effects [7].
Antifungal drug administration via aerosol formulations has been of great interest during the last two decades. Pulmonary delivery of therapeutic agents through inhalation is a non-invasive method to obtain local as well as systemic effects with optimum bioavailability due to reduced first-pass metabolism in the liver and has the additional advantages of decreased adverse effects and better patient compliance [8]. Therefore, enormous efforts are in progress to develop a targeted system to alveolar macrophages using various carriers attached to the drug molecules to get an improved efficacy with the added benefit of minimal adverse effects [9]. Aerosol formulations of liposomal as well as non-liposomal AmB have promising results and are well documented for the prophylaxis as well as treatment of fungal infections [10]. Various aerosol formulations of AmB have shown to be effective in the prophylaxis of AmB in animal models [11, 12]. Following aerosol administration of liposomal AmB, a very negligible amount of the drug was deposited at organs other than the lungs which suggested minimal systemic toxicity of aerosol formulations [13,14,15].
Pulmonary delivery of AmB is pivotal for lung fungal infections and fungal prophylaxis for transplant recipients. This demands further research into renally safe and effective pulmonary delivery and carrier systems for AmB. In this study, sodium deoxycholate sulfate (SDCS) (Fig. 1) was used as the lipid carrier to develop aerosol formulations of AmB. SDCS was found to be less toxic to erythrocytes and formed a stable micelle system following reconstitution with a high zeta potential, and the particle size was appropriate as a nanomicellar system [16]. AmB-SDCS formulations were found to be less toxic to lung tissue as well as to embryonic kidney cells and were effectively phagocytosed by alveolar macrophages [17]. These formulations show higher membrane permeability and retard the unwanted release of free AmB; therefore, it is quite evident that SDCS can be used as a safe lipid carrier for aerosol formulations of AmB and can be highly effective in treating IPA by targeting alveolar macrophages [16, 17]. Aerodynamic parameters of AmB-SDCS including mass median aerodynamic diameter (MMAD), fine particle fraction (FPF), and geometric standard deviation (GSD) were studied to be suitable for pulmonary delivery. Thus, from these three studies, it was evident that SDCS can be employed as a safe and effective carrier for pulmonary delivery of AmB and can be highly effective in treating invasive pulmonary aspergillosis by targeting alveolar macrophages [16,17,18].
The present study was carried out to determine the biodistribution and histopathology of AmB-SDCS formulations in rats, after intra-tracheal instillation. Targeted organs in the study were the lungs, kidneys, spleen, and liver. The kidney and liver markers were also investigated to assess the toxic effects of AmB delivered as SDCS formulations. The Fungizone®-like formulation was also prepared and used as the control. In addition, in vitro toxicity was determined using respiratory cell lines including immortalized human proximal tubular epithelium HK-2 (ATCC® CRL-2190™), immortalized human bronchial epithelial cells HBE1 (ATCC® CRL-2741), and the monocyte cell line RAW 267.4 (ATCC® TIB-71™).
Materials and methods
Materials
AmB was generously donated by BioLab Co. Ltd. (Samustraprakan, Thailand). Deoxycholic acid, sodium borohydride, and sodium deoxycholate were supplied by Sigma-Aldrich (St. Louis, MO, USA). Ethyl acetate, hexane, acetonitrile, dichloromethane, hydrochloric acid 37%, tetrahydrofuran (THF), chloroform, methanol, and acetic acid were obtained from RCI Labscan Ltd. (Bangkok, Thailand). Anhydrous sodium sulfate was supplied by Fischer Scientific (Leicestershire, UK). Sodium acetate hydrated was obtained from Ajax Finechem Pty Ltd. (Auckland, New Zealand). Dimethyl sulfoxide (DMSO) was purchased from Riedel-de Haean (Germany). Polyamides membranes of pore size 0.22 μm were obtained from Sartorius (Gottingen, Germany). Sodium deoxycholate sulfate (SDCS) was synthesized in-house using a previously reported method by [18]. All the chemicals were of analytical grade and used without further purification. THF was freshly distilled before use.
Preparation of AmB-SDCS formulations
AmB and SDCS formulations were prepared using various concentrations of SDCS. Briefly, AmB (50 mg) and SDCS (26 mg) in a 1:1 M ratio were stirred in double distilled water until completely dissolved. An appropriate quantity of 0.2 M sodium hydroxide solution was slowly added with continuous stirring at room temperature to obtain a clear yellowish solution. The pH of the solution was adjusted back to 7.4 using phosphoric acid (0.2 M) for an in situ phosphate buffer. The final volume of the solution was made to 50 mL by adding double-distilled water. The solution was lyophilized afterwards and was reconstituted using distilled water for further studies. No cryoprotectant was used during freeze-drying process to prevent subsequent influence on intratracheal instillation. Using similar methods, various formulations of AmB with different molar ratios of SDCS were prepared: 1:2 (50 and 52 mg), 1:3 (50 and 78 mg), 1:4 (50 and 104 mg), and 1:5 (50 and 130 mg). A similar method was employed for the preparation of the control formulation of AmB using sodium deoxycholate in a 1:2 M ratio mimicking the commercially available Fungizone®, i.e., referred as “the Fungizone®-like formulation”. Freeze-dried formulations were stored at 4 °C and protected from light.
Cytotoxicity assay using respiratory and kidney cell lines
The 3-(4,5 dimethylthiazole-2yl)-2 diphenyltetrazolium bromide (MTT) colorimetric assay as described by Mosmann (1983) and modified by Edmondson et al. (1988) was used to assess the cytotoxicity of various formulations of AmB-SDCS as well as SDCS on kidney and respiratory cell lines including HK-2 (ATCC® CRL-2190™) and HBE1 (ATCC® CRL-2741), respectively, and the monocyte cell line RAW 267.4 (ATCC® CRL-2190™). RAW 267.4 cell line is effective in studying the response against various fungal strains and microbial products. Macrophages play a key role in scavenging fungi and other foreign microbial contaminants, acting as the first defense and restricting the infection at a local site. Therefore, biocompatibility against RAW 267.4 cells was also studied. RAW 267.4 cells are monocyte-like macrophages. The HK-2, HBE1, and RAW 267.4 cells were distributed in 96-well plates at a density of 1 × 105 cells/well in 100 μL of complete medium and allowed to attach overnight at 37 °C and 5% CO2 with 95% relative humidity. After 24 h, the medium (100 μL) was replaced with medium containing various concentrations of different formulations of AmB-SDCS (volume, 100 μL; concentrations, 1 to 16 μg/mL for each AmB to SDCS formulation) or the equivalent AmB concentrations of Fungizone®-like formulation. After incubation for 24 h, 50 μL of a solution of MTT at 1.25 mg/mL was added and incubated further for 4 h at 37 °C in 5% CO2 and 95% humidity, covered with an aluminum foil. The solutions were removed from the 96-well plate after incubation and 100 μL of DMSO was added to dissolve the formazan crystals. The optical densities (OD) were measured at 570 nm using a microplate reader. Control cells were incubated in medium without AmB. The number of viable cells in the treated wells was compared with those in the untreated wells and estimated as percent viability. The percentage of surviving cells was calculated from the following formula.
Biodistribution and histopathology studies using experimental animals
The animal studies were performed strictly following the ethical guidelines approved by the animal ethics committee, Prince of Songkla University (MOE 0521.11/522, Ref. 08/2015).
Animal’s husbandry
Male Wistar rats (250–300 g, 6–8 weeks old) used in this study were obtained from the National Laboratory Animal Center, Mahidol University, Nakorn Pathom, Thailand, and supplied with the standard feed protocol by the Southern Laboratory Animal Facility, Faculty of Sciences, Prince of Songkla University, Hat Yai, Songkhla, Thailand. The rats were outbred and specified as pathogen-free. The animals were put on rest for 7 days before the experiment. The rats were kept in stainless steel cages of dimensions 25 × 96 × 15 cm with 4 rats/cage at 25 °C and subjected to light for 8 h/day. The animals were kept at 50% relative humidity with no noise disturbance, good ventilation, and continuous electrical power supply. All of the animals were allowed ad libitum access to a standard rodent diet and water. Strict hygienic conditions were maintained throughout the study duration for all of the animals with proper disinfection of the cage floors.
Collection of blood and tissue samples from animals
The animals used for this study were randomly divided into 7 groups (n = 5). The control group was instilled with normal saline (NS) solution through the intratracheal route. Group 1 was instilled with the reference drug formulation (Fungizone®-like formulation). Groups 2 through 6 were instilled with different formulations of AmB-SDCS of various concentrations of SDCS as described earlier. All instillations were performed into the trachea and the rats were anesthetized with pentobarbitone (50 mg/kg, intraperitoneally) and atropine (0.2 mg/kg, subcutaneously) as pre-medication. The rats were then positioned vertically and the drug was administered by inserting a polyethylene tube into the trachea via the mouth and injected using a 100-mm tube (Corning, USA) for 1–2 min. AmB-SDCS formulations equivalent to 1.5 mg/kg of AmB was administered in a total volume of 1 mL and instilled through the trachea. In order to investigate the effects of AmB-SDCS formulations on the kidney and liver physiology and toxicity, blood samples (1 mL) were taken from the tail of each rat pre- and post-experiment.
After 7 days of regular dosing (1.5 mg/kg/day), a blood specimen (1 mL) was collected from the tail of each animal. Based on cytotoxicity data against various cell lines, it was evident that AmB-SDCS formulations were not toxic even at higher doses. Therefore, a slightly higher dose was used for in vivo studies as described in the “Introduction” section that higher doses of lipid-based formulations of AmB can be used in order to lower their toxicity. The animals were then euthanized using pentobarbitone (100–150 mg/kg intraperitoneally). The lung, liver, spleen, and kidney tissue specimens were collected. The tissue samples were washed in ice-cold saline, blotted with a paper towel to remove excess fluid, and weighed. Tissue samples were stored at − 20 °C until assessed for AmB concentration using liquid chromatography technique coupled with tandem mass spectrometry (LC-MS/MS). To investigate histological characterization, the lung, liver, spleen, and kidney organs were dissected and trimmed to remove excess fat. All organ tissues were fixed using 10% buffered formalin for 3 days. The tissues were then processed in gradually increasing concentrations of ethanol till 100%, using a tissue processor (LEICA TP 1020, Leica Microsystems GmbH, Wetzlar, Germany) for 24 h to remove any water. The processed tissues were then embedded in Paraplast blocks using a tissue embedder (LEICA EG 1160, Leica Microsystems GmbH, Wetzlar, Germany). Sections of 5-μm thickness were cut from the tissues using a digital microtome. The tissue slides were stained with Harris hematoxylin and eosin (H&E) and examined using a microscope (Olympus DP73 equipped with cellSens software, version 6.1.4.2).
Determination of serum creatinine, blood urea nitrogen, SGOT, and SGPT
In order to investigate the effects of AmB-SDCS formulations on kidney and liver physiology, blood (1 mL) was taken from the tail of each rat prior to the experiment to obtain baseline values for blood urea nitrogen (BUN), creatinine (Cr), serum glutamate oxaloacetate transaminase (SGOT), and serum glutamate pyruvate transaminase (SGPT). After 7 days of regular dosing (1.5 mg/kg/day), blood collection was done again to measure the BUN, Cr, SGOT, and SGPT levels to investigate the toxicity of the control Fungizone®-like formulation and the AmB-SDCS formulations on the organs. The data from each treated rat were compared with that from the control using the one-way analysis of variance (ANOVA). The level of significance was defined as p < 0.05.
LC-MS/MS analysis
AmB was quantified in the plasma and tissue samples with LC-MS/MS (API 3200™ AB SCIEX, Singapore) analysis. The analytical method was adopted from Al-Quadeib et al. (2014) [19] using rat plasma in place of human plasma. Briefly, chromatographic conditions were carried out using gradient elution consisting of two solvents with these compositions: solvent A, methanol-acetonitrile (50:50 v/v) containing 0.1% formic acid, and solvent B, 10 mM ammonium formate (pH 3 ± 0.02) containing 0.2% formic acid and 1% acetonitrile. The flow rate was set to 0.3 mL/min with a total run time of 3.2 min. The data were processed using Analyst version 1.6 software. The high-performance liquid chromatograph (Agilent 1260, CA, USA) was connected to a triple quadrupole tandem mass detector (AB Sciex) with an electrospray ionization (ESI) source for mass spectroscopy detection. The ESI source was set in a positive mode and quantification was performed using the multiple reaction mode for suitable mass transfer. The optimal conditions for mass spectrometry were set: capillary voltage 3.5 kV, cone voltage 15 V, extractor 2.0 V, source temperature 115 °C, desolvation temperature 400 °C, and collision energy 15 eV. The protonated parent ion of AmB was quantified using multiple reaction monitoring of the transition of m/z 925. No significant difference (P < 0.05) was observed for the standard curve best-fit equations on intercept, slope, or correlation coefficient between the rat and human plasma. The precision and accuracy of the LC-MS/MS method were measured in the concentration range of 100 to 4000 ng/mL and no significant difference was observed between inter-day and intra-day analysis (P < 0.05) for rat plasma. The correlation coefficient r2 was more than 0.995 showing excellent linearity for the concentration ranges used. The method was able to detect AmB concentrations for all AmB-SDCS formulations used in the various organs. Limit of quantification (LOQ) and limit of detection (LOD) were 50 and 5 ng/mL, respectively, for the instrument used.
Statistical analysis
The data was statistically processed to determine the level of significance. Data is presented as mean ± standard deviation (SD) from at least three samples unless indicated. The data was evaluated using the ANOVA followed by any other suitable statistical test if required. All statistical comparisons were determined using SPSS software version 16.0 (SPSS Inc., Chicago, IL, USA). Statistical significance was designated as P < 0.05.
Results and discussion
Invasive pulmonary aspergillosis (IPA) is one of the major fungal diseases of great concern due to its major role as a direct or indirect cause of death at bone marrow and solid organ transplantation centers as well as leukemia treatment centers. In our present study, we evaluated the efficacy of aerosol formulations of AmB in the form of AmB-SDCS using an in-house synthesized lipid carrier SDCS from a natural precursor (deoxycholic acid) in experimental animals. Intratracheal instillation is an effective route for pulmonary drug administration with only loss of the drug retained in the device. It provides efficient pulmonary targeting, even drug distribution to four lobes, and non-compromised lung function during anesthesia [20]. According to our previous report, AmB-SDCS formulation exists in the form of nanomicelles [16]. The development of drug targeting systems to target specific tissues is receiving great popularity in pharmacotherapy and pharmacology. This strategy has benefits of reduced dose administration to achieve the desired therapeutic effects with reduced adverse events and toxicity associated with the drug molecule.
Cell viability by MTT assay
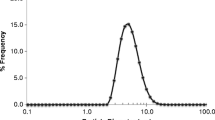
AmB-SDCS lipid formulations as well as Fungizone®-like formulation were evaluated for toxicity on kidney and lung cell lines including HK-2 and HBE1 (ATCC® CRL-2741), respectively, and RAW 267.4 following a 24-h exposure using MTT reduction assay. Figure 2 shows the percent viability against the three cell lines at concentrations that ranged from 1 to 16 μg/mL. The effective concentration of AmB is 1 μg/mL which is also quite similar to the AmB concentration for the lungs. The typical in vitro AmB concentration for drug delivery should be 8–16 times the normal in vivo therapeutic concentration in the bloodstream; therefore, similar concentrations were used in the experiments [21]. Lipid carrier SDCS in its pure form was also employed for MTT reduction assay at all concentrations and found to be non-toxic with cell viability at nearly 100% for all concentrations used.
Viability was nearly 100% in the case of the HK-2 cells for AmB-SDCS formulations 1:1 and 1:2 at 1–2 μg/mL AmB, whereas it was more than 90% for all other formulations. All of the formulations showed more than 80% viability even up to 8 μg/mL AmB, except formulations 1:4 and 1:5 with cell viability of 70–80%. In the case of formulation 1:2, viability was even higher (nearly 90%) at 8 μg/mL AmB. In the case of Fungizone®-like formulation, a significant decrease in viability was observed with increasing concentrations, and viability reduced to 62% at 8 μg/mL AmB. Cell death was even more evident at 16 μg/mL AmB where cell survival was below 50% which was substantially less than all AmB-SDCS formulations. The data indicated that AmB-SDCS formulations 1:4 and 1:5 were slightly toxic at higher concentrations but all the formulations were better than the Fungizone®-like formulation.
No toxicity was observed for AmB-SDCS formulations (1:1, 1:2, and 1:3) in the case of HBE1 cells at a concentration range of 1–4 μg/mL AmB with cell viability of nearly 100%, whereas in the case of other formulations (1:4 and 1:5), viability was slightly below 90%. Even at the higher concentrations of 8 and 16 μg/mL AmB, all AmB-SDCS formulations, except formulation 1:5, cell damage was also not too high with viabilities of 80% and 70%, respectively, whereas for formulation 1:5, the viabilities were 76% and 68%, respectively, at the concentrations of 8 and 16 μg/mL AmB. Fungizone®-like formulation depicted higher cell toxicity with percent viability reduced to 57% and 41% at 8 and 16 μg/mL AmB, respectively, which were significantly higher toxicities than all AmB-SDCS formulations.
AmB-SDCS formulations evaluated against RAW 267.4 cells showed no evidence of cell death with nearly 100% viability at concentrations 1–4 μg/mL AmB except AmB-SDCS 1:5, where viability was slightly below 90%. All formulations except 1:5 showed no significant cell damage even at the higher concentrations of 8 and 16 μg/mL AmB with cell viabilities in the range of 80–90% and 70–85%, respectively. For AmB-SDCS (1:5), slight toxicity was observed with decreased viability to nearly 70% at both concentrations used. Significant toxicity was evident for Fungizone®-like formulation where the viabilities of cells decreased to below 80% and 70% at 4 and 8 μg/mL AmB, respectively. Cell death was even more at the highest concentration with reduced cell viability to nearly 50% at 16 μg/mL AmB. Toxicity to cells by Fungizone®-like formulation was higher than that by all AmB-SDCS formulations.
A relatively weaker barrier is usually provided to the lipid bilayers by the micellar system in comparison with lipid nanoparticles (LPNs) as well as liposomes. The release of AmB in this case may be faster from the micelles in comparison with LPNs or even AmBisome™. However, the release of AmB can be slow from AmBisome™ or LPNs owing to the presence of strong interaction between AmB and lipids like cholesterol and phospholipids with a subsequent decrease in the cytotoxicity of the entrapped drugs in the LPNs and AmBisome™ [22]. In the AmB-SDCS formulations, it is quite possible that they formed a more stable micellar system compared with Fungizone®-like formulation resulting in a slower release of the AmB, as evident from spectrofluorometric data from previously published literature [16], and ultimately lower toxicity to the kidney cells. Similarly, lower toxicity was observed for lung cells as well as monocyte cell lines for AmB-SDCS formulations compared with that for Fungizone®-like formulation. The higher toxicity by the Fungizone®-like formulation can be attributed to the micelles as these present a relatively weaker barrier in comparison with lipid bilayers [16]. Due to the aforesaid reasons, lower toxicity was evident with AmB-SDCS formulations even at the higher concentration of 8 μg/mL.
Kidney and liver toxicity
Renal and hepatic toxicity of Fungizone®-like formulation as well as various AmB-SDCS formulations was investigated after intratracheal instillation. After 7 days of regular dosing (1.5 mg/kg/day), blood serum of the animals under study was analyzed for BUN, Cr, SGOT, and SGPT (Fig. 3). Fungizone®-like formulation–treated rats had an increased Cr as well as BUN compared with the control group. The Cr value increased from 0.81 to 1.16 mg% whereas BUN increased from 19.6 to 21.8 mg% for Fungizone®-like formulation–treated rats. However, in the case of AmB-SDCS formulations, except AmB-SDCS 1:5, stable Cr and BUN levels were observed. For AmB-SDCS formulation 1:5, a very slight increase in Cr was observed from 0.85 to 0.94 mg% and the BUN level increased to 21.3 from 18.8 mg%. A quite similar increase in SGOT and SGPT was also observed for the Fungizone®-like formulation–treated groups compared with the groups treated with the AmB-SDCS formulations. In the Fungizone®-like formulation–treated group, SGPT and SGOT increased to 54 and 119 U/L from the baseline values of 37 and 69.5 U/L, respectively. The very slight increases in Cr and BUN observed for AmB-SDCS formulation 1:5 were not significant (P < 0.05).
The results indicated that AmB in the form of an AmB-SDCS formulation presented with normal BUN and Cr values, whereas in the case of Fungizone®-like formulation, nephrotoxicity was observed even at a lower administered dose with an increase in the value of Cr. The lower toxicity observed with AmB-SDCS formulation in comparison with Fungizone®-like formulation is probably due to the slower release of AmB from the AmB-SDCS micelle system compared with the deoxycholate micelle release from Fungizone®-like formulation with the resultant controlled levels of the drug in the blood plasma with AmB-SDCS nanomicelles. Chitosan has also been shown to demonstrate some degree of renal toxicity with increased BUN levels but this effect was more prominent at higher doses. An AmB nanoparticulate system using chitosan and dextran sulfate also exhibited lower nephrotoxicity in comparison with Fungizone®-like formulation probably due to the modification of the aggregation state of AmB [23]. It has been demonstrated that low-molecular-weight chitosan was safe for intravenous administration but the important aspect is the release of AmB from the micelle system, which results in reduced toxicity in the kidneys due to the interaction of AmB with the lipids in the cell membranes. Renal failure is also related to the aggregation state of AmB with the monomeric form showing reduced toxicity compared with the dimeric or oligomeric form. AmB-SDCS micelles are expected to control the release of AmB mostly in monomeric form, thus showing little or no toxicity to the kidney cells on the basis of molecular aggregation changes as observed previously [18, 24] by spectrophotometry and dynamic light scattering. Various lipid formulations have also demonstrated decreased or a non-significant increase in the Cr values using various doses in comparison with the Fungizone ®-like formulation. The increase in Cr was greater than 80% of the baseline value following a dose of 1 mg/kg [25].
Histopathology of tissue samples for nephrotoxicity and hepatotoxicity
The most serious adverse effect of AmB following chronic usage is nephrotoxicity with a rise in the Cr level > 80% in treated patients. Histopathological analysis of kidney tissues following intra-tracheal instillation of AmB-SDCS formulations did not present any unusual signs of tissue necrosis for all formulations as well as the NS-treated group, except for the Fungizone®-like formulation treated–groups. The kidneys of the control group treated with NS (Fig. 4, 1A) showed normal histological structures of glomeruli, tubules, and vessels. Similar normal kidney architecture was also observed in animals treated with AmB-SDCS formulation (1:2) (Fig. 4, 3A). However, dramatic injury was evident in Fungizone®-like formulation–treated animals. The kidneys showed wider capillaries with the space filled by red blood cells showing evidence of increased hemolysis compared with those in the control group. Glomeruli showed hyperemia and widened Bowman’s capsule as evidenced by the deformation of the glomerulus outline (Fig. 4, 2A).
Histopathology of kidneys: 1A) NS-treated rats showing normal histological structure of glomeruli, tubules, vessels; 2A) Fungizone®-like formulation–treated rats with wider capillaries with space filled by erythrocytes and glomeruli showing hyperemia with widened Bowman’s capsule; 3A) AmB-SDCS (1:2)-treated rats with normal histology of glomerulus and tubules. Liver of 1B) NS-treated rats with normal hepatic parenchyma and hepatic lobules with normal portal triad and normal chord-like arrangement of hepatocytes; 2B) Fungizone®-like formulation–treated rats with shrinkage of hepatocytes and some cell death and deranged hepatocytes with disturbed normal chord-like arrangement of hepatocytes; 3B) AmB-SDCS (1:2)-treated rats with normal morphology and chord-like arrangement of hepatocytes with normal nuclei between sinusoids and hepatocytes. Lungs of 1C) NS-treated rats with normal bronchioles and differentiable type I and type II cells; 2C) Fungizone®-like formulation–treated rats with hypertrophy of bronchioles due to thickening of bronchial epithelium and also thickened alveolar walls; 3C) AmB-SDCS (1:2)-treated rats with normal alveoli and aggregation of inflammatory cells. Spleen of 1D) NS-treated rats with normal tissue parenchyma and no evidence of fibrosis, inflammation, and fatty infiltration; 2D) Fungizone®-like formulation–treated rats with loss of normal architecture and evidence of foreign bodies and giant cells; 3D) AmB-SDCS (1:2)-treated rats with normal parenchyma and no evidence of inflammation with normal cell morphology in both white and red pulp
Considering the histology of the liver, the hepatic parenchyma was seen to consist of numerous hepatic lobules separated from each other by soft connective tissue septa housing the portal triad. Each hepatic lobule incorporated a thin-walled central vein surrounded by hepatic cords radiating towards the periphery in the NS-treated group (Fig. 4, 1B). The animal groups treated by AmB-SDCS formulation (1:2) (Fig. 4, 3B) depicted quite normal morphology including the thickness of cells and normal blood vessels as well as normal nuclei between the sinusoids and hepatic cells. These results were comparable with the animals in the control (NS) group. However, the Fungizone®-like formulation–treated animals exhibited cell apoptosis and death of some cells. Shrinkage of hepatocytes was observed and the normal chord-like arrangement of hepatocytes was disturbed. The hepatocytes did not show any typical oval shape and some necrosis of the liver macrophages (Kupffer cells) in the liver was observed. Derangement of the hepatocytes was also observed with some level of deformity of cells (Fig. 4, 2B).
In addition, normal morphology of the lungs was observed in the NS-treated animals showing normal bronchioles and epithelial cells (Fig. 4, 1C). Type I and II cells were quite normal and differentiable. The animals treated with AmB-SDCS formulation (1:2) showed aggregations of inflammatory cells that were most probably monocytes. The inflammatory cells engulfed the drug particles which indicated an excellent immune response (Fig. 4, 3C). The Fungizone®-like formulation–treated group lacked clear orientation around the terminal bronchioles and hypertrophy was observed due to thickening of the bronchial epithelium at both the right and left sides. Thickening of alveolar walls was also observed (Fig. 4, 2C). No such abnormality was observed for the AmB-SDCS formulations.
The sections of spleen tissue revealed discrete lymphoid masses (white pulp) embedded in a highly vascular matrix (red pulp). The spleen has vascular as well as lymphoid elements and is a place for hematopoiesis besides being involved in the clearance of aged and degenerated red blood cells and circulating bacteria and particulate matter from the blood supply. The spleen can be a site for direct as well as indirect toxicity and can be a target for various carcinogens. The histology of the spleen from the NS-treated rats as well as AmB-SDCS formulation (1:2)–treated groups depicted normal parenchyma with no evidence of any fibrosis, inflammation, or fatty infiltration. Morphology of the cells in both the white pulp and red pulp was normal and showed no toxicity to the formulations of the treated rats (Fig. 41D, 3D). The Fungizone®-like formulation–treated rats showed loss of the normal architecture structure besides marked depletion of lymphoid tissue. Evidence of foreign bodies and giant cells was also observed in the Fungizone®-like formulation–treated group showing some evidence of toxicity (Fig. 4, 2D).
Biodistribution of drug to tissues
The kidneys, lungs, spleen, and liver were investigated for the amount of AmB following 7 days of regular dosing of AmB-SDCS formulations and Fungizone®-like formulation by intratracheal instillation. The total treated dose for AmB-SDCS formulations and control Fungizone®-like formulation was 10.5 mg/kg (1.5 mg/kg/day) equivalent to AmB. The drug was calculated as microgram per gram for each tissue under this study. The concentrations of AmB obtained in various tissues as well as plasma following 7 days of intratracheal instillation are shown in Table 1. AmB-SDCS formulations show a low drug concentration in the kidney tissues, whereas an increased concentration was observed in the case of Fungizone®-like formulation. The lungs showed high AmB concentrations for all AmB-SDCS formulations and similarly, high concentrations were also observed in the spleen. The concentration of AmB in the liver was less compared with that in the spleen but was still high enough. The liver is also as important as the spleen because it is a reservoir organ for AmB due to high accumulation in the reticuloendothelial system (RES).
In the kidneys, fungal burden can be reduced by either poly-aggregated AmB lipid formulations or dimeric Fungizone®-like formulation with more or lower levels of nephrotoxicity [26]. The developed AmB-SDCS formulations depicted lower concentrations in the kidneys compared with the reference Fungizone®-like formulation (Table 1). In the study of various lipid complexes of AmB, similar renal concentrations were reported by liposomal AmB formulations [27, 28]. The deposition of AmB in the kidney at the 1.5 mg/kg-administered AmB-SDCS formulations was a very low amount (50–100 ng/g). This is expected to reduce the renal toxicity of AmB more effectively in comparison with the Fungizone®-like formulation. In real life, there was an incidence of nephrotoxicity in more than 50% of patients at the normally administered doses [29].
The spleen and liver are considered to be AmB reservoirs due to the relatively higher drug accumulation in the RES [30, 31]. AmB-SDCS formulations show higher concentrations of the drug in the spleen following intratracheal instillation into the lungs. The concentration of AmB in the spleen for liposomal formulations was above the normal MIC values of 0.5–2 μg/mL. The high drug accumulation for AmB-SDCS formulations in the organs with RES was probably related to the appropriate size of micellar formulations of AmB which is enough to be less opsonized by the contents of the plasma [32]. The decrease in concentration that was observed with an increase of the lipid carrier SDCS was probably due to the lower release of the drug owing to the strong interaction between the lipid and drug molecule and the formation of a stable micellar system. Liposomal AmB formulations also obtained higher levels in the spleen and liver in comparison with Fungizone®-like formulation, and the activity of liposomal formulations was related to both the increased tissue penetration and the sustained bioactivity of AmB levels in the target organs. Biodistribution data presented an increase in drug accumulation with formulations 1:1 to 1:3 and a decrease with formulations 1:4 and 1:5. This can be explained on the basis of size distribution of the AmB-SDCS formulations. The hydrodynamic radius of AmB-SDCS formulations 1:1 to 1:5 is 203, 269, 171, 65, and 63 nm, respectively. The smaller sized micelles might have faced rapid clearance compared with larger micelles. The rapid decrease of size with formulations 1:4 and 1:5 can be explained as the availability of more SDCS promotes the formation of smaller sized micelles instead of depositing on the surface of the large micelle [16, 33].
The concentrations of AmB following intratracheal instillation of the AmB-SDCS formulations are shown in Table 1. It was evident that high concentrations of AmB were observed following inhalation and the pulmonary concentrations were more than 5 μg/g for all AmB-SDCS formulations. These higher concentrations were significantly higher than Fungizone®-like formulation and were adequate to treat pulmonary aspergillosis for which the MIC is about 1–2 μg/mL for all three strains of Aspergillus and is also helpful in treating mucormycosis with a MIC of 0.5–1 μg/mL [34, 35]. The highest AmB concentration in the lungs was observed with AmB-SDCS (1:2 and 1:3) which was almost twice the concentration achieved with Fungizone®-like formulation. The different concentrations distributed in the various organs are given in Table 1. The micelle size can be a significant factor as well as a comparison of the bound and unbound fractions of the drug. AmB-SDCS formulations have shown to form a stable micellar system with lower drug release resulting in lower toxicity compared with Fungizone®-like formulation [16]. The benefit associated with AmB-SDCS formulations is higher accumulation in the lungs in comparison with liposomal formulations with lower drug levels in the lungs. The concentration in the plasma was above 2 μg/mL for all AmB-SDCS formulations which was also higher than that for Fungizone®-like formulation. This was also higher than the reported AmB lipid formulations as well as Fungizone®-like formulation [4, 36]. The concentration was in the range to kill the fungus as it was relevant to the MIC needed for fungicidal activity.
It is evident that aerosol formulations for oral inhalation have lower systemic concentrations and the tissue distribution is also negligible, except for the lungs, in comparison with the extensive distribution following intravenous administration [13]. The mortality rates associated with IPA are still alarmingly high despite the availability of various AmB formulations and other new antifungal drugs like caspofungin and voriconazole [37]. The failure associated with intravenous AmB administration is the result of various factors like fungus proliferation to other body tissues despite its predominant residence in the lung airways. The penetration of AmB following intravenous administration is inadequate and it is also evident that only a small amount of the drug administered by the IV route is actually delivered to the lungs. Therefore, inhalation of the drug is the best way to obtain a higher concentration at the lungs which are the direct target of interest [11]. The available data for inhaled administration of AmB are limited but it is pertinent to mention that inhaled liposomal AmB formulations were well tolerated in humans [38]. However, adverse effects like mild bronchospasm, nausea, and cough were observed with inhaled Fungizone® which in some cases resulted in the discontinuation of the study mainly due to the dose of drug administered [39, 40]. The major problem was cough which was probably due to the deoxycholate rather than the AmB [41]. In our study, no toxicity was observed in the rats taking the AmB-SDCS formulations as evidenced by the histopathological results of the tissues. Furthermore, all AmB-SDCS formulations were well tolerated by the animals showing no detrimental effects of the carrier lipid SDCS on the natural surfactants of the lungs.
Conclusions
AmB-SDCS formulations in various molar ratios were found suitable as aerosol formulations for delivery into the lungs through inhalation. In vitro cytotoxicity study results demonstrated that SDCS was a safe lipid carrier for pulmonary delivery of AmB with no evidence of toxicity for the lung as well as kidney cells. The BUN and Cr levels were observed to be normal before and after 7 days of regular dosing which indicated no nephrotoxicity. Furthermore, normal SGPT and SGOT values showed no hepatotoxicity. Histopathology of the lung, liver, spleen, and kidney tissues showed no evidence of toxicity. Higher concentrations of the drug were observed in the lungs, spleen, and liver with minimal AmB concentration in the kidneys. Plasma and lung concentrations were well above the MIC required for treatment of aspergillosis. It is concluded that an AmB-SDCS-based nanomicellar system is better than the conventional Fungizone®-like formulation in the context of safety and biodistribution in animals. This study demonstrates the potential of the SDCS micellar system which can find future implications in designing therapeutically safe and cost-effective formulations for pulmonary delivery of AmB by aerosol administration for treatment of fungal infections of the lung, especially for the treatment of IPA.
References
Walsh TJ, Anaissie EJ, Denning DW, Herbrecht R, Kontoyiannis DP, Marr KA, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327–60.
Oren I, Goldstein N. Invasive pulmonary aspergillosis. Curr Opin Pulm Med. 2002;8:195–200.
Hamill RJ. Amphotericin B formulations: a comparative review of efficacy and toxicity. Drugs. 2013;73:919–34.
Torrado JJ, Espada R, Ballesteros MP, Torrado-Santiago S. Amphotericin B formulations and drug targeting. J Pharm Sci. 2008;97:2405–25.
Slain D. Lipid-based amphotericin B for the treatment of fungal infections. Pharmacotherapy. 1999;19:306–23.
Walsh TJ, Finberg RW, Arndt C, Hiemenz J, Schwartz C, Bodensteiner D, et al. Liposomal amphotericin B for empirical therapy in patients with persistent fever and neutropenia. National Institute of Allergy and Infectious Diseases Study Group. N Engl J Med. 1999;340:764–71.
Tang X, Zhu H, Sun L, Hou W, Cai S, Zhang R, et al. Enhanced antifungal effects of amphotericin B-TPGS-b-(PCL-ran-PGA) nanoparticles in vitro and in vivo. Int J Nanomedicine. 2014;9:5403–13.
Gelperina S, Kisich K, Iseman MD, Heifets L. The potential advantages of nanoparticle drug delivery systems in chemotherapy of tuberculosis. Am J Respir Crit Care Med. 2005;172:1487–90.
Vyas S, Quraishi S, Gupta S, Jaganathan K. Aerosolized liposome-based delivery of amphotericin B to alveolar macrophages. Int J Pharm. 2005;296:12–25.
Vyas S, Kannan M, Jain S, Mishra V, Singh P. Design of liposomal aerosols for improved delivery of rifampicin to alveolar macrophages. Int J Pharm. 2004;269:37–49.
Allen SD, Sorensen KN, Neial MJ, Durrant C, Proffit RT. Prophylactic efficacy of aerosolized liposomal (AmBisome) and non-liposomal (Fungizone®) amphotericin B in murine pulmonary aspergillosis. J Antimicrob Chemother. 1994;34:1001–13.
Cicogna CE, White MH, Bernard EM, Ishimura T, Sun M, Tong WP, et al. Efficacy of prophylactic aerosol amphotericin B lipid complex in a rat model of pulmonary aspergillosis. Antimicrob Agents Chemother. 1997;41:259–61.
Koizumi T, Kubo K, Kaneki T, Hanaoka M, Hayano T, Miyahara T, et al. Pharmacokinetic evaluation of amphotericin B in lung tissue: lung lymph distribution after intravenous injection and airspace distribution after aerosolization and inhalation of amphotericin B. Antimicrob Agents Chemother. 1998;42:1597–600.
Lambros MP, Bourne DWA, Abbas SA, Johnson DL. Disposition of aerosolized liposomal amphotericin B. J Pharm Sci. 1997;86:1066–9.
Ruijgrok EJ. Efficacy of aerosolized amphotericin B desoxycholate and liposomal amphotericin B in the treatment of invasive pulmonary aspergillosis in severely immunocompromised rats. J Antimicrob Chemother. 2001;48:89–95.
Usman F, Ul-Haq Z, Khalil R, Tinpun K, Srichana T. Pharmacologically safe nanomicelles of amphotericin B with lipids: nuclear magnetic resonance and molecular docking approach. J Pharm Sci. 2017;106:3574–82.
Adhikari K, Buatong W, Thawithong E, Suwandecha T, Srichana T. Factors affecting enhanced permeation of amphotericin B across cell membranes and safety of formulation. AAPS PharmSciTech. 2016;17:820–8.
Gangadhar KN, Adhikari K, Srichana T. Synthesis and evaluation of sodium deoxycholate sulfate as a lipid drug carrier to enhance the solubility, stability and safety of an amphotericin B inhalation formulation. Int J Pharm. 2014;471:430–8.
Al-Quadeib BT, Radwan MA, Siller L, Mutch E, Horrocks B, Wright M, et al. Therapeutic monitoring of amphotericin B in Saudi ICU patients using UPLC MS/MS assay: monitoring amphotericin B in patients using UPLC. Biomed Chromatogr. 2014;28:1652–9.
Yu B, Okano T, Kataoka K, Kwon G. Polymeric micelles for drug delivery: solubilization and haemolytic activity of amphotericin B. J Control Release. 1998;53:131–6.
Bhamra R, Sa’ad A, Bolcsak LE, Janoff AS, Swenson CE. Behavior of amphotericin B lipid complex in plasma in vitro and in the circulation of rats. Antimicrob Agents Chemother. 1997;41:886–92.
Tiyaboonchai W, Limpeanchob N. Formulation and characterization of amphotericin B–chitosan–dextran sulfate nanoparticles. Int J Pharm. 2007;329:142–9.
Risovic V, Boyd M, Choo E, Wasan KM. Effects of lipid-based oral formulations on plasma and tissue amphotericin B concentrations and renal toxicity in male rats. Antimicrob Agents Chemother. 2003;47:3339–42.
Jung SH, Lim DH, Jung SH, Lee JE, Jeong K-S, Seong H, et al. Amphotericin B-entrapping lipid nanoparticles and their in vitro and in vivo characteristics. Eur J Pharm Sci. 2009;37:313–20.
Clark JM, Whitney RR, Olsen SJ, George RJ, Swerdel MR, Kunselman L, et al. Amphotericin B lipid complex therapy of experimental fungal infections in mice. Antimicrob Agents Chemother. 1991;35:615–21.
Wasan KM, Grossie VB, Lopez-Berestein G. Concentrations in serum and distribution in tissue of free and liposomal amphotericin B in rats during continuous intralipid infusion. Antimicrob Agents Chemother. 1994;38:2224–6.
Puri A, Loomis K, Smith B, Lee J-H, Yavlovich A, Heldman E, et al. Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic. Crit Rev Ther Drug Carrier Syst. 2009;26:523–80.
Caldeira LR, Fernandes FR, Costa DF, Frézard F, Afonso LCC, Ferreira LAM. Nanoemulsions loaded with amphotericin B: a new approach for the treatment of leishmaniasis. Eur J Pharm Sci. 2015;70:125–31.
Zhao M, Hu J, Zhang L, Zhang L, Sun Y, Ma N, et al. Study of amphotericin B magnetic liposomes for brain targeting. Int J Pharm. 2014;475:9–16.
Serrano DR, Hernández L, Fleire L, González-Alvarez I, Montoya A, Ballesteros MP, et al. Hemolytic and pharmacokinetic studies of liposomal and particulate amphotericin B formulations. Int J Pharm. 2013;447:38–46.
Diekema DJ, Messer SA, Hollis RJ, Jones RN, Pfaller MA. Activities of caspofungin, itraconazole, posaconazole, ravuconazole, voriconazole, and amphotericin B against 448 recent clinical isolates of filamentous fungi. J Clin Microbiol. 2003;41:3623–6.
Salas V, Pastor FJ, Calvo E, Alvarez E, Sutton DA, Mayayo E, et al. In vitro and in vivo activities of posaconazole and amphotericin B in a murine invasive infection by Mucor circinelloides: poor efficacy of posaconazole. Antimicrob Agents Chemother. 2012;56:2246–50.
Dupont B. Overview of the lipid formulations of amphotericin B. J Antimicrob Chemother. 2002;49:31–6.
Denning DW. Invasive Aspergillosis. Clin Infect Dis. 1998;26:781–803.
Purcell IF, Corris PA. Use of nebulised liposomal amphotericin B in the treatment of Aspergillus fumigatus empyema. Thorax. 1995;50:1321–3.
Diot P, Rivoire B, Le Pape A, Lemarie E, Dire D, Furet Y, et al. Deposition of amphotericin B aerosols in pulmonary aspergilloma. Eur Respir J. 1995;8:1263–8.
Dubois J, Bartter T, Gryn J, Pratter MR. The physiologic effects of inhaled amphotericin B. Chest. 1995;108:750–3.
Gryn J, Goldberg J, Johnson E, Siegel J, Inzerillo J. The toxicity of daily inhaled amphotericin B. Am J Clin Oncol. 1993;16:43–6.
Funding
This research was supported by a grant from the 2014 scholarship awards for Masters and Ph.D. studies under Thailand’s Education Hub for Southern Region of ASEAN countries (TEH-AC) and National Research Council of Thailand (PHA610372S), Department of Pharmaceutical Technology, Faculty of Pharmaceutical Sciences, Prince of Songkla University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Biodistribution and histopathology studies were performed that strictly followed the ethical guidelines approved by the animal ethics committee, Prince of Songkla University (MOE 0521.11/522, Ref. 08/2015).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Usman, F., Nopparat, J., Javed, I. et al. Biodistribution and histopathology studies of amphotericin B sodium deoxycholate sulfate formulation following intratracheal instillation in rat models. Drug Deliv. and Transl. Res. 10, 59–69 (2020). https://doi.org/10.1007/s13346-019-00662-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-019-00662-x