Abstract
Research on the etiology and treatment of diabetes has made substantial progress. As a result, several new classes of anti-diabetic drugs have been introduced in clinical practice. Nonetheless, the number of patients achieving glycemic control targets has not increased for the past 20 years. Two areas of unmet medical need are the restoration of insulin sensitivity and the reversal of pancreatic beta cell failure. In this review, we integrate research advances in transcriptional regulation of insulin action and pathophysiology of beta cell dedifferentiation with their potential impact on prospects of a durable “cure” for patients suffering from type 2 diabetes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The prevalence of type 2 diabetes continues to rise, albeit at a lower rate compared to the 1980–2000 period [1]. In the last decade, several additions have been made to the diabetes pharmacopeia [2]. However, the percentage of patients achieving target glycemic control has not increased [3]. Thiazolidinedione-type insulin sensitizers [4], Sglt2 inhibitors [5], and Glp1 receptor agonists (RA) have all shown partial protection from macrovascular complications [6], achieving a milestone that had thus far eluded practitioners. Furthermore, Glp1-RA and dual-acting Glp1-RA and Gip-RA can potentially achieve a weight loss effect comparable to bariatric surgery [7] in the treatment of the main antecedent of type 2 diabetes in non-East-Asian populations, i.e., obesity [8]. However, all these drugs suffer from secondary failures [9, 10], and thus type 2 diabetes is still approached using a “treat to fail” paradigm, in which drugs are added in stepwise fashion to achieve a therapeutic glucose-lowering effect. Except for sulfonylureas, it’s unclear whether secondary drug failures are the results of mechanism-based desensitization to the drug, or reduced patient adherence [11, 12].
When sulfonylurea-type insulin secretagogues were first-line treatment for diabetes, this pattern was more pronounced [13]. But secondary failure will occur regardless of treatment used [9, 10, 14]. In this regard, it bears emphasizing that the GRADE study, designed at the behest of the U.S. Center for Medicare as a comparative effectiveness study to help lower cost and increase durability of combination therapy [15], did not find paradigm-shifting differences between different classes of drugs as add-ons to metformin treatment that justify the steep cost of newer agents. Thus, a $200/year sulfonylurea was nearly as effective at delaying escalation of care as a 20-fold more expensive DPP4 inhibitor or Glp1-RA (Fig. 1) [16]. While this may not be a burning issue in Japan, where a national agency establishes country-wide drug prices, it is a key question to contain the escalating costs of treating diabetes in North-America and Europe. Therefore, there is a need for research aimed at identifying mechanism-based interventions to modify the course of the disease, or better yet to cure it. This review summarizes our work in this area.
Comparative effectiveness and cost of metformin add-on treatments. Top-line results from the GRADE study summarizing the percentage of patients requiring addition of medications at 5 years and experiencing major cardiovascular adverse events. Medication costs are from [16]
Genes of type 2 diabetes
Type 2 diabetes shows familiar clustering and heritability; yet attempts to define its genetic underpinning have consistently demonstrated that there is no “diabetes” gene, but rather many genes with small effects in large populations and relatively few genes with large effects in individual patients [17]. With the exception of monogenic forms of diabetes [18,19,20], identification of which preceded the glorious revolution of genome-wide studies, examples of gene variations affecting diabetes predisposition are primarily associated with coding sequence variants in which the functional impact on a gene product can be assessed by standard methodologies [21], as compared to the hundreds of genomic loci that have been associated with different disease traits. Sequence variants with strong genetic evidence of linkage to variously defined diabetes traits are indeed common, but their pathogenic significance is unclear [22]. This can be explained by the fact that many variants land in introns, and elude functional characterization, while coding-sequence variants require interrogation in specific cellular contexts that are not easy to replicate. Another recurring problem is that gene function is generally imputed based on experimental animal work but uncovering patterns of gene expression and function in humans can be challenging. There are obvious and not-so-obvious reasons for this fallacy. First, changes in mRNA levels may or may not be associated with altered gene function, yet they are virtually universally assumed to do so. Conversely, protein function can be regulated independent of mRNA levels, as is the case with the FoxO genes, of which more will be said later. In addition, the cell type in which these changes occur may be elusive, either because it is a transient population (e.g., progenitor cells or cells that undergo fate transition), or because it exerts its effects in a cell-independent fashion.
Our work on C2CD4A, a protein of unknown function that we have successfully decoded as a diabetes-susceptibility gene associated with a widely replicated locus [23], provides an example of studies needed to delve into gene regions showing strong cross-population evidence of linkage to diabetes traits. Transcription factors direct pancreatic β-cell maintenance and function and are thought to underlie the genetic predisposition to type 2 diabetes, often acting through transcriptional hubs, also known as super-enhancers or stretch-enhancers that are thought to exert hierarchical control over complex gene networks [24, 25]. Despite its well-documented role in β-cell maintenance and dysfunction in humans and experimental animals [26,27,28], FoxO1 had not been associated with β-cell super-enhancers or GWAS loci [29, 30]. This gap in knowledge can be attributed to the fact that FoxO1 is primarily regulated by cellular localization (nuclear vs. cytoplasmic) and not at the mRNA level, at least in the critical early stages of β-cell failure [31, 32]. Thus, looking for changes in FoxO1 mRNA levels is generally inadequate to interrogate its function. During a genome-wide survey of FoxO1 targets, we discovered a strong FoxO1 super-enhancer conserved in β-cells of humans and experimental animals near C2CD4A on human chromosome 15 [33]. This locus, first identified by the Kadowaki group in Japanese type 2 diabetics, harbors several single nucleotide polymorphisms conferring susceptibility to human type 2 diabetes in multiple ethnic groups, and is thus one of the most widely replicated diabetes loci [34,35,36]. We performed cell biological and molecular analyses of C2CD4A, because its expression, rather than that of the neighboring gene C2CD4B, was altered in mouse models of diabetes generated by targeted inactivation of FoxO1 in β-cells [37]. Key evidence of a role of C2CD4A in β-cell function arose from the generation of a β-cell-specific loss-of-function model in mice, showing that C2cd4a ablation was associated with glucose intolerance and decreased insulin secretion, impaired glucose-induced insulin release, and altered β-cell gene expression patterns. To evaluate these changes at a functional level, we performed gain-of-function studies, and found a striking transcriptional regulation of the glycolytic cascade, including β-cell disallowed genes [23] that enforce coupling of nonoxidative glycolysis to oxidative phosphorylation by preventing monocarboxylate uptake and conversion of lactate to pyruvate (Fig. 2) [38]. An impairment of this process can result in metabolic inflexibility via increased lipid utilization [39]. The Rutter group independently identified the neighboring C2CD4B gene as the potential diabetes-susceptibility locus [40]. Given that the genes are physically close and structurally related, it’s possible that they both contribute and that functional studies that involve both will be required to fully assess their contribution to diabetes pathogenesis and β-cell function. These findings illustrate how cellular and molecular physiology, together with physiologic evidence in experimental animals can help disentangle the genetics of human type 2 diabetes [23].
C2CD4A couples nonoxidative glycolysis with oxidative phosphorylation. Genes involved in the closed coupling of glycolysis to oxidative phosphorylation in β-cells are regulated by C2CD4A, providing a potential mechanism by which this gene product is related to diabetes susceptibility. These genes are normally suppressed (“disallowed”) to prevent β-cell monocarboxylate uptake (MCT) and conversion to pyruvate (LDH). This process is altered in diabetes, and a reduction of C2CD4A levels can lead to impaired stimulus/secretion coupling
Another aspect of human genetics that can be deconvoluted using experimental approaches is the interaction between genes and environment. The best characterized examples of genetic predisposition to type 2 diabetes are the MODY genes, acting in autosomal dominant fashion to impair insulin secretion [20]. The predisposition to impaired insulin secretion or β-cell function is generally less marked in common forms of diabetes and requires environmental factors such as diet, physical activity, and comorbidities like obesity to result in β-cell failure. FoxO1 is an example of acquired transcriptional abnormalities leading to altered β-cell function. FoxO1 is inactive in a healthy β-cell and is activated by nuclear translocation in response to nutritional or inflammatory cues. FoxO1 nuclear translocation reduces lipid utilization as a source of energy for mitochondrial oxidative phosphorylation and increases the expression of genes related to β-cell differentiation [41]. However, this protective mechanism is only effective in the short-term. Prolonged activation of FoxO1 increases its deacetylation-dependent degradation [42], triggering an acquired loss of protein that is commonly found in different models of experimental diabetes as well as human islets from type 2 diabetics [27]. Loss of FoxO1 leads to metabolic inflexibility, i.e., the inability of the β-cell to select glucose vs. lipids as an energy source, and increases lipid oxidation [43] and β-cell dedifferentiation [27, 32, 44]. These mechanisms are discussed in greater depth in the section on β-cell failure. Interestingly, acquired FoxO1 loss-of-function, as observed in these circumstances, is associated with abnormalities of the HNF4α transcriptional network [43, 45]. To understand whether these two factors cooperate in regulating β-cell function, we performed an extensive analysis of their interactions and tested them in vivo using animal models deficient for one or both protein products. At the genomic and epigenetic level, we found that FoxO1 inactivation–a common feature of human and murine diabetic β-cells– resulted in characteristic changes to β-cell enhancers, with increased occupancy by HNF4α. We reckoned that this was compensatory in nature, and that in the absence of FoxO1, HNF4α compensates for some of its functions. But when we tested the hypothesis in vivo we found the opposite result [46]. Combined ablation of FoxO1 and HNF4α reversed the β-cell defects associated with single mutations of either gene, indicating that their interactions are antagonistic rather than compensatory. Thus, in glucose tolerance, hyperglycemic clamps, and ex vivo insulin secretion experiments, defects associated with FoxO1 or HNF4α ablation were normalized when both genes were inactivated. This unexpected interaction illustrates another problem related to interpreting genomic data, namely the potential for antagonistic interactions between different loci. In our experiments with FoxO1 and HNF4α, gene expression analyses revealed that different gene families were regulated by these two transcription factors with different modalities. For example, the glycolysis pathway was regulated through antagonistic epistasis (i.e., the two factors had opposite effects), and the protocadherin gene family by synergistic epistasis (i.e., the two factors had additive effects) [46].
Modeling these interactions, as progressively larger association studies and genome sequencing experiments result in an ever-expanding set of diabetes-susceptibility loci, presents a nearly unsurmountable challenge, suggesting that genetic heterogeneity is a critical factor in individual patients. Thus, what can be the goal of human diabetes genetics? There are two areas that could benefit from this knowledge: diabetes staging and pharmacogenomics [47]. For the time being, they are both in their infancy, as evidence that genetic staging of diabetes outperforms accurate history-taking and physical exam is weak [48]. The future of this work hinges on whether it can be converted into pharmacogenomics. Given the expanding number of options available to practitioners to treat diabetes, and the manifold combinations, it would indeed be helpful to know whether gene variants predispose to certain combinations being more effective [47].
Insulin resistance
Insulin resistance is a complex condition, entailing biochemical and molecular abnormalities in different cell types and target tissues [49]. It differs from β-cell dysfunction in a fundamental way. The latter is dynamic: in pre-diabetes the insulin response to nutrients is increased [50] but it deteriorates over time, leading to hyperglycemia [51]. Insulin resistance worsens over time in individuals progressing to type 2 diabetes [52], although comparatively less than β-cell dysfunction [51].
Throughout the last two decades of the last Century, insulin resistance held sway as the key pathogenetic mechanism of type 2 diabetes [53]. The introduction of the first true insulin sensitizer, troglitazone, into the diabetes pharmacopeia in 1994, and its remarkable glucose-lowering, insulin-like metabolic effects, vindicated the notion that insulin resistance is key to developing diabetes and insulin sensitization necessary to its treatment [54]. However, with the emergence of serious side effects to treatment with various thiazolidinediones [55], these drugs have fallen out of favor despite their proven benefits not only on glucose control [56], but also on secondary prevention of macrovascular disease [4], and β-cell function [57].
A potential solution to this conundrum is to separate the clinical benefits of thiazolidinediones from their adverse effects on weight gain, fluid balance, and bone loss [56]. Leveraging knowledge on post-translational modifications of the target of thiazolidinediones, PPARγ, we identified sites of deacetylation induced by treatment with rosiglitazone and introduced mutations in PPARγ that mimicked the effect of this compound in experimental animals. Interestingly, the glucose-lowering and insulin-sensitizing effects of PPARγ were maintained in this model, but the adverse effects on bone formation and fluid balance were prevented, demonstrating that it is theoretically possible to synthesize designer thiazolidinediones that improve insulin sensitivity without affecting bone and cardiovascular or renal function [58, 59]. Whether the liabilities of this approach outweigh the potential benefits is a commercial and legal, rather than a scientific and medical decision.
Because insulin has pleiotropic actions on virtually every tissue, there are many different definitions of insulin resistance. Is there is a core biochemical defect or target tissue that causes disease progression? Let’s examine this question at the cellular and organismal level. At the cellular level, to the extent that we can define a “core” defect in insulin action in the pathogenesis of diabetes, it is hyperinsulinemia-induced insulin receptor downregulation, exacerbating the impairment of insulin signaling [60]. The onset of hyperinsulinemia is indeed the earliest marker of insulin resistance and recedes with treatment [61]. It can also arise from impaired insulin clearance [62], but the prediction is that it would quickly give rise to receptor downregulation, bringing about cellular insulin resistance.
Indeed, in the golden era of receptor radio-immunoassays, there were many publications demonstrating impaired insulin binding to cells from patients with diabetes [63]. Enthusiasm for an intrinsic impairment of insulin receptor function as the cause of diabetes was dampened by the realization that this defect could be reversed by fasting, exercise, and drug treatment [64]. Thereafter, a dogma arose that receptor defects are immaterial to the pathogenesis of insulin resistance, and that the fundamental defect occurs at a distal step in cellular insulin signaling. Hence, the last two decades have witnessed an explosion of alternative explanations for the onset of insulin resistance involving cytokines, tissue-derived “-kines” and sundry hormones, inflammation, neurotransmitters, microbiota, and other factors [53]. The implied rationale of these studies is that these factors affect insulin signaling downstream of insulin receptor, potentially acting on a specific metabolic pathway (amino acids, lipids, glucose) that leads to disease. Although all these factors are likely to be present in vivo, in our opinion they play a supporting but not a central role in the pathogenesis of diabetes. The reasons for this assertion are principally twofold: one is that none of these factors appears to be as widespread, early, or as widely replicated in patient studies as hyperinsulinemia. But more importantly, although the notion that a selective impairment of insulin action plays a causal role in the progression of diabetes has been considered from the early days of radio receptor assays, evidence has remained circumstantial. Instead, evidence from several laboratories indicates that insulin signaling, at least within the IRS-Akt-FoxO axis, is a homeostatic module [65,66,67]. A clear illustration of this finding is that, when the key transcriptional mediator of insulin signaling, FoxO1 (and 3a and 4), is genetically inactivated, insulin signaling is predicted to increase, but in fact decreases due to the lack of transcription of the genes encoding IRS1 and IRS2. As a result, Akt phosphorylation is decreased. Thus, even in the event of a single defective signaling pathway, the entire module would be affected by either increasing or decreasing flow of information at other chokepoints along the path [65]. Against this explanation, it can be argued that the entire path can be circumvented by accessory or ancillary pathways causing insulin resistance independent of insulin receptor-IRS-Akt-FoxO. But then there should not be hyperinsulinemia in these patients, and this does not happen in practice. This makes an isolated defect in insulin signaling less likely.
A similar conclusion can be reached about whether there is a core tissue defect at the organismal level [68]. In the 1980s, the idea that the primary defect in insulin action was secondary to impaired glucose utilization in muscle gained popularity through the use of hyperinsulinemic glucose clamps, demonstrating a reduced effect of insulin on glucose utilization [69]. However, this defect can be overcome by glucose mass action in hyperglycemic individuals [70], suggesting that additional metabolic disturbances contribute to disease progression. A secondary line of evidence supporting an organism-wide as opposed to tissue-specific mechanism of insulin resistance in the pathogenesis of diabetes arose from experimental animal studies assessing the effects of tissue-specific conditional knockouts of genes involved in insulin action, primarily but not exclusively the insulin receptor. In those instances, localized insulin resistance is generally associated with specific sub-phenotypes, but falls short of overt diabetes [71]. Even allowing for the self-evident argument of species-specific differences between rodents and humans, the broad conclusion appears to favor a generalized, rather than organ-specific origin of insulin resistance.
It is in this context that the potential role of hyperinsulinemia itself emerged. As articulated in a classic essay by McGarry [72], hyperinsulinemia can promote rather than impair a subset of insulin actions, driving excess lipid/lipoprotein synthesis in liver and storage in skeletal muscle, which in turn leads to impaired glucose utilization, increased glucose production, and β-cell failure [72]. Although many aspects of this hypothesis have been experimentally validated [73, 74], and the adverse role of free fatty acids and triglycerides on insulin action and atherogenic lipoprotein production have been well documented [75], it falls short of explaining the onset of hyperinsulinemia [71, 76]. That compensatory hyperinsulinemia may make things worse by promoting a subset of insulin actions is not only possible but borne out by the side effects of insulin treatment in diabetes, primarily but not exclusively weight gain.
What the debate about the role of hyperinsulinemia highlights is not the specificity of insulin resistance, but the need to separate insulin sensitization from its untoward effects. In fact, as reviewed above, thiazolidinediones are potent insulin sensitizers, but cause assorted liabilities that can be imputed to excessive, rather than impaired, insulin sensitivity. This question loomed large in our research trajectory, and we sought to investigate it in different ways. Ideally, one would want to restore the missing actions of insulin without promoting weight gain, generation of atherogenic lipoproteins, and assorted vascular effects. There have been suggestions that endocrine members of the FGF family can act as selective insulin sensitizers [77,78,79]. Consistent with the McGarry hypothesis that hyperinsulinemia has heterogeneous effects, we were struck to discover that the same transcription factor, FoxO1, regulated at once the genes encoding the rate-limiting steps of glucose utilization (glucokinase) and glucose production (glucose-6-phosphatase), but in opposite directions, i.e., it activated the former and inhibited the latter. This discovery highlighted the fundamental conundrum of insulin action, namely that carbon atoms must be metabolized somehow, so if we suppress glucose production to reduce fasting hyperglycemia, we will promote lipid synthesis with attendant weight gain and secretion of atherogenic lipoproteins. On the other hand, this discovery provided us with an opportunity to dissect the two processes. We leveraged the single-gateway regulation of glucokinase and glucose-6-phosphatase by FoxO1 [80] as a screening assay to probe the ability of chemical small molecule FoxO1 inhibitors to selectively regulate two key functions of insulin in the liver, lipid synthesis and glucose production. We were indeed able to identify two classes of FoxO1 inhibitors that differed by their abilities to regulate one vs. the other response, consistent with the idea that it is possible to achieve selectivity in the clinic to modulate insulin action in a more targeted fashion, e.g., by promoting glucose disposal without increasing lipid synthesis [81]. Not coincidentally, these chemical inhibitors appear to have synergistic effects with FGF-21 [82]. Thus, a profitable area of future research is the pursuit of clinically validated compounds that can achieve selective sensitization to insulin by acting on FoxO1 or germane mediators of insulin signaling.
β-cell failure
β-cell dysfunction was originally thought of primarily as a defect of glucose-induced insulin secretion [83]. However, we now recognize that the β-cell response to other secretagogues may not be entirely normal [84]. We also recognize that reduced insulin secretion may arise from either a functional defect, or from a reduced number of insulin-producing cells [85]. Since biology does not read our papers, it’s unlikely that the two aspects of this problem can be as neatly segregated in vivo as they are in a publication. More likely, at any given time during the progression of diabetes, the endocrine pancreas contains a combination of fewer and lesser-functioning β-cells. In any event, it is apparent that insulin secretory function deteriorates with time [86], much more rapidly so after the onset [51] of even mild hyperglycemia [87].
Insulin secretion has a KATP channel-dependent first-phase and a KATP channel-independent second-phase [88]. The molecular architecture of first-phase has been genetically validated by loss- and gain-of-function mutations of glucokinase and the KATP channel in humans, leading respectively to hyper- and hypoglycemia [89]. Persistent activation of this pathway, as caused for example by sulfonylureas, leads to its desensitization through membrane depolarization [90]. This mechanism underlies secondary failure to sulfonylureas. The pathway underpinning second-phase is more elusive, and different second messengers have been proposed to mediate it, including glutamate, NADPH, NAD shuttle, and Acyl-CoA [91]. This pathway is leveraged by GLP1-RA and requires a permissive glucose level–hence the lack of hypoglycemia as a side effect of treatment with these agents. Although not as rapid as sulfonylurea-induced failure, therapeutic failure to GLP1-RA also occurs [9].
Attaching a functional meaning to changes in β-cell number has proved more difficult. Of course, having fewer β-cells is not helpful. But does the progression of β-cell failure, as outlined above, entail changes in cell number? Extensive morphometric analyses of β-cell mass (not area, which can be misleading if pancreas weight is altered), show that the normal range in non-diabetic and diabetic subjects can vary by as much as six-fold [85]. While there is a decrease with diabetes, many diabetics still have more β-cells than most non-diabetics, so reduced mass alone is unlikely to promote the transition from euglycemia to hyperglycemia. Since we do not know if changes to β-cell mass are dynamic in nature, the relative contribution of mass vs. function remains an open question. It is likely to differ not only in different individuals but also depending on disease stage.
Regardless of the relative contributions of one vs. the other, it must be recognized that the endocrine pancreas is a staid organ. It does not renew itself with the same vigor as the intestine, blood, liver; it’s indeed more like the brain. β-cells turn over slowly [92]. We do not study comparative physiology, but published data allow to extrapolate that β-cell proliferation rates in humans are like rodents, in which most functional studies have been performed [93, 94]. In addition, work from the Melton laboratory has conclusively demonstrated that neogenesis of β-cells from non- β-cell progenitors does not occur to any significant, or indeed detectable effect [92]. Thus, regardless of the animal species, β-cells do not divide unless they must, such as in extreme insulin resistance. When they do, they appear to be more susceptible to cell death. Based on the refractoriness of β-cells to replication, the sensitivity of replicating cells to toxicity, and the intrinsic difficult of assessing the effects β-cell growth factors in vivo, we are not optimistic that promoting β-cell growth be a viable option as a future treatment of diabetes.
Death of β-cells, by various mechanisms, has been invoked as a cause of diabetes [95]. Although a stressed cell, as the insulin-overproducing cell is likely to be, can be more susceptible to death cues, a key argument against death-based mechanisms as a cause of β-cell failure is the latter’s rapid reversibility under appropriate treatment conditions. Classically, few weeks of low-calorie diet can partly restore β-cell function even after many years from diagnosis, and this benefit can be long-lasting [96, 97]. The rapid restoration of β-cell function in response to pharmacological treatment is also more easily explained if there is not a permanent loss of cells. It can be argued that treatment simply increases the function of residual β-cells. This is possible, but less likely since cells were already under stress, for example in patients treated with a sulfonylurea, and it bears to reason that further stimulation of insulin release is unlikely to relieve this cellular stress.
This fundamental clinical observation drove our initial foray into the concept of β-cell dedifferentiation as a cause of β-cell failure. We were surprised when we investigated the fate of islet β-cells in animal models of diabetes and discovered that β-cells had not died but had become devoid of insulin. In fact, they had reverted to a progenitor-like stage, and some of them had converted into α-like or mesenchymal cells (Fig. 3) [32]. In due course, many investigators confirmed the findings [32, 98,99,100,101,102,103]. Dor and colleagues specifically investigated whether re-activation of the progenitor cell marker Neurogenin3 was required for the process of dedifferentiation and found that it occurred even in Neurogenin3 knockouts [104]. These data indicate that Neurogenin3 protein is not required for dedifferentiation or redifferentiation, but do not change the overall conclusions.
Our view of how β-cell failure develops. This diagram illustrates a schematic progression of β-cell failure and its relationship to the function of different β-cell transcription factors. Under conditions of nutrient excess or related metabolic abnormalities, FoxO1 is activated by nuclear translocation (bottom) and promotes lipid rather than glucose utilization (“metabolic inflexibility”). This activation cannot be maintained chronically, as FoxO1 degradation outruns its synthesis. When FoxO1 levels falls, the state of differentiation of the β-cell becomes impaired, and the activity of other transcription factors is also reduced. β-cells can convert to α-like cells (elevation of Arx) or progenitor-like cells (re-activation of Neurog3) [32, 43, 110]
An interesting feature of the dedifferentiation process was the activation of the PPARα and PPARα pathways, leading to a combined state of increased lipid turnover, i.e., more synthesis and more oxidation [43]. This seemingly paradoxical observation resonated with the beneficial effect of PPARα agonists on β-cell function in diabetic patients [105]. A possible explanation for the combined activation of both pathways is that the increase in lipid synthesis somehow offsets the potential toxic effects of excessive lipid oxidation in mitochondria. But the reason why increased lipid utilization would be conducive to β-cell failure remains speculative. As we mapped the progression of dedifferentiation in mouse models, we detected distinctive changes in mitochondrial complex function. Thus, in the early stages of β-cell compensation, we saw selective dysfunction of complexes I, IV, and V [37]. With the onset of hyperglycemia, we saw the additional impairment of complex III, a critical site of ROS formation in response to lipids [106], which can account for the cellular toxicity of increased lipid oxidation, as seen in our model [43]. When we interrogated the gene expression patterns corresponding to each stage, we found that the transition to the diabetic stage was accompanied by impaired expression of Cyb5r3, a gene with multiple potential functions, including as a complex III oxidoreductase. Indeed, upon genetic or functional ablation of CYB5R3, we were able to bring about a blunted respiratory response to glucose and extensive mitochondrial and secretory granule morphological abnormalities, consistent with altered expression of key differentiation markers [107]. In vivo, CYB5R3 deficiency resulted in impaired insulin secretion, glucose intolerance and diet-induced hyperglycemia, suggesting that CYB5R3 is required for proper β-cell function [107]. These data support both the idea of metabolic inflexibility as a key steppingstone in the progression of β-cell failure, and the role CYB5R3 in the progression of mitochondrial dysfunction, leading to impaired ATP synthesis and insulin production. We have not yet fully mapped the functions of CYB5R3 in β-cells, and more work will be forthcoming.
In the wake of these observations, we set out to address three questions: (i) does dedifferentiation occur in human diabetes, (ii) is dedifferentiation reversible, and if so, (iii) are there pharmacological pathways that can be leveraged to reverse it? Dedifferentiation has been found in islets from diabetic donors in European, North-American [27], Japanese [108], and Chinese populations [44], including lean type 2 diabetics [109]. The rates of increase vary between 2.5- and fourfold. Amo-Shiinoki and colleagues were also able to determine progression of dedifferentiation according to disease stage, indicating that there is a correlation between the anatomical findings and clinical features [108].
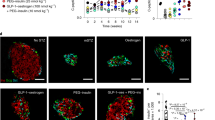
We then sought to address the question of whether the mechanism of dedifferentiation observed in animals held true in humans and, if so, whether it could be reversed (Fig. 4). To perform mechanism-based studies, we employed a systems biology approach to transform mRNA data derived from single-cell sequencing of islets from either normal or diabetic donors and identify master regulator proteins associated with diabetic vs. non-diabetic islet cell types [110]. As we discussed in the section on diabetes genetics, changes to mRNA levels, or lack thereof, are not very informative to interrogate gene function. Our colleagues Ding and Califano developed an algorithm that uses thousands of reporter gene activities to identify drivers of specific cell states. The predictions from this systems analysis (metaVIPER) were entirely consistent with the findings in animal models. In addition to distinct cell types, representing physiologic β– and α–cells, we found cell types highly enriched in diabetic patients, characterized by: (a) metabolic inflexibility, (b) mixed α/β-cell identity, and (c) endocrine progenitor/stem cell features (Fig. 4). Thus, the unbiased human islet survey fully vindicated the experimental animal findings. Moreover, it provided a generally applicable method that allowed us to detect similar cell types in all other published studies on this topic, through a re-analysis of gene expression databases [110].
A systems approach to understand β-cell failure. The diagram illustrates the path from determining mRNA expression levels in islet cells to identifying actionable target for pharmacological treatment of β-cell failure [110]
We ranked putative regulators of these states and used CRSPR-based single-cell expression in human islets to test the analytical predictions by gain-of-function studies in normal β-cells, as well as loss-of-function studies in diabetic β-cells. Remarkably, we found that inhibition of transcription factor BACH2, which we had first encountered as an ectopically activated gene in animal experiments with FoxO1 knockouts in β-cells [37], resulted in restoration of β-cell function in diabetic mice and islets of diabetic humans. In contrast, its gain-of-function reproduced the different stages of dedifferentiation, cell conversion, activation of PPARs, and progenitor-like features. Moreover, using BACH inhibitors (not specific to BACH2 but also inhibiting the related isoform BACH1), we were able to lower glycemia and increase insulin secretion in diabetic mice as well as human diabetic islets [110]. Since BACH inhibitors are FDA-approved for use in multiple sclerosis [111], there is an immediate opportunity to test this pathway in human trials. These studies advanced research in multiple ways: first, they demonstrated that single-cell mRNA data can be used to identify diabetes-specific cell types; second, they showed that key pathogenic features of the disease are conserved in humans and animal models; third, they provided a mechanism for reversibility of β-cell failure; and fourth, they identified an actionable therapeutic target for disease modification [110].
Prospects for patients
Where does this leave patients? In an uncomfortably tight place, constrained by an increasingly prescriptive and unreasonably expensive regimen of multiple drugs, none of which is either long-lasting or especially effective, because none of them addresses the key pathogenic factors of the disease. While bio-engineered compounds (Glp1-RA and their brethren) outperform plant-derived compounds (metformin, sulfonylureas, and gliflozins) especially regarding weight loss, both groups of medications still hew to a “treat to fail” approach. Insulin remains necessary for many patients, with the attendant complications. Lifestyle modifications, the only regimen found to have a preventive effect, are beyond the cultural, economic, and implementation capabilities of most patients, and are only given lip service by busy medical professionals. The pharmaceutical industry is recalcitrant to embrace innovation because the burdensome regulatory process has made failure expensive indeed. Stockholders insist not only on generous returns, but also on immediate ones. This attitude promotes busywork as opposed to transformative thinking. Although there has been much progress in treating the disease and inroads have been made in the prevention of complications, we do not agree with the premise that we have an effective treatment for type 2 diabetes. A permanent, or at least more durable solution can be found and when it does, we hope that our contribution will help.
References
IDF. IDF Diabetes Atlas, 10th edn. In: Federation ID (eds). IDF Diabetes Atlas. Brussels: International Diabetes Federation, 2021
Taylor SI, Yazdi ZS, Beitelshees AL. Pharmacological treatment of hyperglycemia in type 2 diabetes. J Clin Invest. 2021;131:e142243
Fang M, Wang D, Coresh J, Selvin E. Trends in diabetes treatment and control in U.S. adults, 1999–2018. N Engl J Med. 2021;384:2219–28.
Kernan WN, Viscoli CM, Furie KL, et al. Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med. 2016;374:1321–31.
Ferrannini E, DeFronzo RA. Impact of glucose-lowering drugs on cardiovascular disease in type 2 diabetes. Eur Heart J. 2015;36:2288–96.
Holst JJ. From the incretin concept and the discovery of GLP-1 to today’s diabetes therapy. Front Endocrinol (Lausanne). 2019;10:260.
Frias JP, Fernandez Lando L, Brown K. Tirzepatide versus semaglutide once weekly in type 2 diabetes. Reply N Engl J Med. 2022;386: e17.
Drucker DJ. GLP-1 physiology informs the pharmacotherapy of obesity. Mol Metab. 2022;57: 101351.
Zinman B, Nauck MA, Bosch-Traberg H, et al. Liraglutide and glycaemic outcomes in the LEADER trial. Diabetes Ther. 2018;9:2383–92.
Home PD, Ahren B, Reusch JEB, et al. Three-year data from 5 HARMONY phase 3 clinical trials of albiglutide in type 2 diabetes mellitus: long-term efficacy with or without rescue therapy. Diabetes Res Clin Pract. 2017;131:49–60.
Cai J, Wang Y, Baser O, Xie L, Chow W. Comparative persistence and adherence with newer anti-hyperglycemic agents to treat patients with type 2 diabetes in the United States. J Med Econ. 2016;19:1175–86.
Mody R, Huang Q, Yu M, et al. Adherence, persistence, glycaemic control and costs among patients with type 2 diabetes initiating dulaglutide compared with liraglutide or exenatide once weekly at 12-month follow-up in a real-world setting in the United States. Diabetes Obes Metab. 2019;21:920–9.
Karam JH, Sanz N, Salamon E, Nolte MS. Selective unresponsiveness of pancreatic beta-cells to acute sulfonylurea stimulation during sulfonylurea therapy in NIDDM. Diabetes. 1986;35:1314–20.
Jones AG, McDonald TJ, Shields BM, et al. Markers of beta-cell failure predict poor glycemic response to GLP-1 receptor agonist therapy in type 2 diabetes. Diabetes Care. 2016;39:250–7.
Rasouli N, Younes N, Utzschneider KM, et al. Association of baseline characteristics with insulin sensitivity and beta-cell function in the glycemia reduction approaches in diabetes: a comparative effectiveness (GRADE) study cohort. Diabetes Care. 2021;44:340–9.
Taylor SI. The high cost of diabetes drugs: disparate impact on the most vulnerable patients. Diabetes Care. 2020;43:2330–2.
Barbetti F, Rapini N, Schiaffini R, Bizzarri C, Cianfarani S. The application of precision medicine in monogenic diabetes. Expert Rev Endocrinol Metab. 2022;17:111–29.
Yamagata K, Furuta H, Oda N, et al. Mutations in the hepatocyte nuclear factor-4alpha gene in maturity-onset diabetes of the young (MODY1). Nature. 1996;384:458–60.
Taylor SI, Arioglu E. Syndromes associated with insulin resistance and acanthosis nigricans. J Basic Clin Physiol Pharmacol. 1998;9:419–39.
Fajans SS, Bell GI. MODY: history, genetics, pathophysiology, and clinical decision making. Diabetes Care. 2011;34:1878–84.
Sarhangi N, Sharifi F, Hashemian L, et al. PPARG (Pro12Ala) genetic variant and risk of T2DM: a systematic review and meta-analysis. Sci Rep. 2020;10:12764.
Florez JC, Udler MS, Hanson RL. Genetics of Type 2 Diabetes. In: rd, Cowie CC, Casagrande SS, et al., eds. Diabetes in America. Bethesda (MD), 2018
Kuo T, Kraakman MJ, Damle M, Gill R, Lazar MA, Accili D. Identification of C2CD4A as a human diabetes susceptibility gene with a role in beta cell insulin secretion. Proc Natl Acad Sci USA. 2019;116:20033–42.
Gaulton KJ, Ferreira T, Lee Y, et al. Genetic fine mapping and genomic annotation defines causal mechanisms at type 2 diabetes susceptibility loci. Nat Genet. 2015;47:1415–25.
Guo S, Dai C, Guo M, et al. Inactivation of specific beta cell transcription factors in type 2 diabetes. J Clin Invest. 2013;123:3305–16.
Talchai SC, Accili D. Legacy effect of Foxo1 in pancreatic endocrine progenitors on adult beta-cell mass and function. Diabetes. 2015;64:2868–79.
Cinti F, Bouchi R, Kim-Muller JY, et al. Evidence of beta-cell dedifferentiation in human type 2 diabetes. J Clin Endocrinol Metab. 2016;101:1044–54.
Sun J, Ni Q, Xie J, et al. Beta cell dedifferentiation in T2D patients with adequate glucose control and non-diabetic chronic pancreatitis. J Clin Endocrinol Metab. 2018;104:83.
Pasquali L, Gaulton KJ, Rodriguez-Segui SA, et al. Pancreatic islet enhancer clusters enriched in type 2 diabetes risk-associated variants. Nat Genet. 2014;46:136–43.
Tennant BR, Robertson AG, Kramer M, et al. Identification and analysis of murine pancreatic islet enhancers. Diabetologia. 2013;56:542–52.
Kitamura YI, Kitamura T, Kruse JP, et al. FoxO1 protects against pancreatic beta cell failure through NeuroD and MafA induction. Cell Metab. 2005;2:153–63.
Talchai C, Xuan S, Lin HV, Sussel L, Accili D. Pancreatic beta cell dedifferentiation as a mechanism of diabetic beta cell failure. Cell. 2012;150:1223–34.
Warton K, Foster NC, Gold WA, Stanley KK. A novel gene family induced by acute inflammation in endothelial cells. Gene. 2004;342:85–95.
Yamauchi T, Hara K, Maeda S, et al. A genome-wide association study in the Japanese population identifies susceptibility loci for type 2 diabetes at UBE2E2 and C2CD4A-C2CD4B. Nat Genet. 2010;42:864–8.
Grarup N, Overvad M, Sparso T, et al. The diabetogenic VPS13C/C2CD4A/C2CD4B rs7172432 variant impairs glucose-stimulated insulin response in 5,722 non-diabetic Danish individuals. Diabetologia. 2011;54:789–94.
Mehta ZB, Fine N, Pullen TJ, et al. Changes in the expression of the type 2 diabetes-associated gene VPS13C in the beta-cell are associated with glucose intolerance in humans and mice. Am J Physiol Endocrinol Metab. 2016;311:E488-507.
Kim-Muller JY, Fan J, Kim YJ, et al. Aldehyde dehydrogenase 1a3 defines a subset of failing pancreatic beta cells in diabetic mice. Nat Commun. 2016;7:12631.
Ishihara H, Wang H, Drewes LR, Wollheim CB. Overexpression of monocarboxylate transporter and lactate dehydrogenase alters insulin secretory responses to pyruvate and lactate in beta cells. J Clin Invest. 1999;104:1621–9.
Buteau J, Shlien A, Foisy S, Accili D. Metabolic diapause in pancreatic beta-cells expressing a gain-of-function mutant of the forkhead protein Foxo1. J Biol Chem. 2007;282:287–93.
Mousavy Gharavy SN, Owen BM, Millership SJ, et al. Sexually dimorphic roles for the type 2 diabetes-associated C2cd4b gene in murine glucose homeostasis. Diabetologia. 2021;64:850–64.
Kim-Muller JY, Kim YJ, Fan J, et al. FoxO1 deacetylation decreases fatty acid oxidation in beta-cells and sustains insulin secretion in diabetes. J Biol Chem. 2016;291:10162–72.
Ido-Kitamura Y, Sasaki T, Kobayashi M, et al. Hepatic FoxO1 integrates glucose utilization and lipid synthesis through regulation of Chrebp O-glycosylation. PLoS ONE. 2012;7: e47231.
Kim-Muller JY, Zhao S, Srivastava S, et al. Metabolic inflexibility impairs insulin secretion and results in MODY-like diabetes in triple FoxO-deficient mice. Cell Metab. 2014;20:593–602.
Sun J, Ni Q, Xie J, et al. beta-cell dedifferentiation in patients with T2D with adequate glucose control and nondiabetic chronic pancreatitis. J Clin Endocrinol Metab. 2019;104:83–94.
Kuo T, Damle M, Gonzalez BJ, Egli D, Lazar MA, Accili D. Induction of alpha cell-restricted Gc in dedifferentiating beta cells contributes to stress-induced beta-cell dysfunction. JCI Insight. 2019;5:e128351
Kuo T, Du W, Miyachi Y, et al. Antagonistic epistasis of Hnf4alpha and FoxO1 metabolic networks through enhancer interactions in beta-cell function. Mol Metab. 2021;53:101256.
Chung WK, Erion K, Florez JC, et al. Precision medicine in diabetes: a consensus report from the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2020;43:1617–35.
Skyler JS, Bakris GL, Bonifacio E, et al. Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes. 2017;66:241–55.
Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiol Rev. 2018;98:2133–223.
Weyer C, Hanson RL, Tataranni PA, Bogardus C, Pratley RE. A high fasting plasma insulin concentration predicts type 2 diabetes independent of insulin resistance: evidence for a pathogenic role of relative hyperinsulinemia. Diabetes. 2000;49:2094–101.
Weyer C, Bogardus C, Mott DM, Pratley RE. The natural history of insulin secretory dysfunction and insulin resistance in the pathogenesis of type 2 diabetes mellitus. J Clin Invest. 1999;104:787–94.
Ferrannini E, Natali A, Muscelli E, et al. Natural history and physiological determinants of changes in glucose tolerance in a non-diabetic population: the RISC Study. Diabetologia. 2011;54:1507–16.
Haeusler RA, McGraw TE, Accili D. Biochemical and cellular properties of insulin receptor signalling. Nat Rev Mol Cell Biol. 2018;19:31–44.
Nolan JJ, Ludvik B, Beerdsen P, Joyce M, Olefsky J. Improvement in glucose tolerance and insulin resistance in obese subjects treated with troglitazone [see comments]. N Engl J Med. 1994;331:1188–93.
Rosen CJ. Revisiting the rosiglitazone story–lessons learned. N Engl J Med. 2010;363:803–6.
Davidson MA, Mattison DR, Azoulay L, Krewski D. Thiazolidinedione drugs in the treatment of type 2 diabetes mellitus: past, present and future. Crit Rev Toxicol. 2018;48:52–108.
Leahy JL. Thiazolidinediones in prediabetes and early type 2 diabetes: what can be learned about that disease’s pathogenesis. Curr DiabRep. 2009;9:215–20.
Kraakman MJ, Liu Q, Postigo-Fernandez J, et al. PPARgamma deacetylation dissociates thiazolidinedione’s metabolic benefits from its adverse effects. J Clin Invest. 2018;128:2600–12.
Liu L, Fan L, Chan M, et al. PPARgamma deacetylation confers the antiatherogenic effect and improves endothelial function in diabetes treatment. Diabetes. 2020;69:1793–803.
Gavin JR 3rd, Roth J, Neville DM Jr, de Meyts P, Buell DN. Insulin-dependent regulation of insulin receptor concentrations: a direct demonstration in cell culture. Proc Natl Acad Sci USA. 1974;71:84–8.
Ferrannini E, Muscelli E, Natali A, et al. Association of fasting glucagon and proinsulin concentrations with insulin resistance. Diabetologia. 2007;50:2342–7.
Poy MN, Yang Y, Rezaei K, et al. CEACAM1 regulates insulin clearance in liver. Nat Genet. 2002;30:270–6.
Olefsky JM. Decreased insulin binding to adipocytes and circulating monocytes from obese subjects. J Clin Invest. 1976;57:1165–72.
Freidenberg GR, Reichart D, Olefsky JM, Henry RR. Reversibility of defective adipocyte insulin receptor kinase activity in non-insulin-dependent diabetes mellitus. Effect of weight loss. J Clin Invest. 1988;82:1398–406.
Tsuchiya K, Tanaka J, Shuiqing Y, et al. FoxOs integrate pleiotropic actions of insulin in vascular endothelium to protect mice from atherosclerosis. Cell Metab. 2012;15:372–81.
Dong XC, Copps KD, Guo S, et al. Inactivation of hepatic Foxo1 by insulin signaling is required for adaptive nutrient homeostasis and endocrine growth regulation. Cell Metab. 2008;8:65–76.
Kubota N, Kubota T, Itoh S, et al. Dynamic functional relay between insulin receptor substrate 1 and 2 in hepatic insulin signaling during fasting and feeding. Cell Metab. 2008;8:49–64.
Accili D. Insulin action research and the future of diabetes treatment: The 2017 Banting Medal for Scientific Achievement lecture. Diabetes. 2018;67:1701–9.
Golay A, DeFronzo RA, Ferrannini E, et al. Oxidative and non-oxidative glucose metabolism in non-obese type 2 (non-insulin-dependent) diabetic patients. Diabetologia. 1988;31:585–91.
Kelley DE, Mandarino LJ. Fuel selection in human skeletal muscle in insulin resistance: a reexamination. Diabetes. 2000;49:677–83.
Nandi A, Kitamura Y, Kahn CR, Accili D. Mouse models of insulin resistance. Physiol Rev. 2004;84:623–47.
McGarry JD. What if Minkowski had been ageusic? An alternative angle on diabetes. Science. 1992;258:766–70.
Haeusler RA, Accili D. The double life of Irs. Cell Metab. 2008;8:7–9.
Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metab. 2008;7:95–6.
Bergman RN, Iyer MS. Indirect regulation of endogenous glucose production by insulin: the single gateway hypothesis revisited. Diabetes. 2017;66:1742–7.
Biddinger SB, Hernandez-Ono A, Rask-Madsen C, et al. Hepatic insulin resistance is sufficient to produce dyslipidemia and susceptibility to atherosclerosis. Cell Metab. 2008;7:125–34.
Inagaki T, Dutchak P, Zhao G, et al. Endocrine regulation of the fasting response by PPARalpha-mediated induction of fibroblast growth factor 21. Cell Metab. 2007;5:415–25.
Qiang L, Accili D. FGF21 and the second coming of PPARgamma. Cell. 2012;148:397–8.
Kharitonenkov A, Shiyanova TL, Koester A, et al. FGF-21 as a novel metabolic regulator. J Clin Invest. 2005;115:1627–35.
Haeusler RA, Pratt-Hyatt M, Welch CL, Klaassen CD, Accili D. Impaired generation of 12-hydroxylated bile acids links hepatic insulin signaling with dyslipidemia. Cell Metab. 2012;15:65–74.
Langlet F, Haeusler RA, Linden D, et al. Selective inhibition of FOXO1 activator/repressor balance modulates hepatic glucose handling. Cell. 2017;171(824–835): e818.
Lee YK, Diaz B, Deroose M, et al. FOXO1 inhibition synergizes with FGF21 to normalize glucose control in diabetic mice. Mol Metab. 2021;49: 101187.
Cerasi E, Luft R. Insulin response to glucose infusion in diabetic and non-diabetic monozygotic twin pairs. Genetic control of insulin response? Acta Endocrinol (Copenh). 1967;55:330–45.
Consortium R. Lack of durable improvements in beta-cell function following withdrawal of pharmacological interventions in adults with impaired glucose tolerance or recently diagnosed type 2 diabetes. Diabetes Care. 2019;42:1742–51.
Rahier J, Guiot Y, Goebbels RM, Sempoux C, Henquin JC. Pancreatic beta-cell mass in European subjects with type 2 diabetes. Diabetes Obes Metab. 2008;10(Suppl 4):32–42.
Trico D, Natali A, Arslanian S, Mari A, Ferrannini E. Identification, pathophysiology, and clinical implications of primary insulin hypersecretion in nondiabetic adults and adolescents. JCI Insight. 2018;3:e124912
Brunzell JD, Robertson RP, Lerner RL, et al. Relationships between fasting plasma glucose levels and insulin secretion during intravenous glucose tolerance tests. J Clin Endocrinol Metab. 1976;42:222–9.
Matschinsky FM. Glucokinase as glucose sensor and metabolic signal generator in pancreatic beta-cells and hepatocytes. Diabetes. 1990;39:647–52.
Greeley SA, Tucker SE, Worrell HI, Skowron KB, Bell GI, Philipson LH. Update in neonatal diabetes. Curr Opin Endocrinol Diabetes Obes. 2010;17:13–9.
Remedi MS, Nichols CG. Chronic antidiabetic sulfonylureas in vivo: reversible effects on mouse pancreatic beta-cells. PLoS Med. 2008;5: e206.
Seino S, Sugawara K, Yokoi N, Takahashi H. beta-Cell signalling and insulin secretagogues: a path for improved diabetes therapy. Diabetes Obes Metab. 2017;19(Suppl 1):22–9.
Dor Y, Brown J, Martinez OI, Melton DA. Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature. 2004;429:41–6.
Xuan S, Kitamura T, Nakae J, et al. Defective insulin secretion in pancreatic beta cells lacking type 1 IGF receptor. J Clin Invest. 2002;110:1011–9.
Gregg BE, Moore PC, Demozay D, et al. Formation of a human beta-cell population within pancreatic islets is set early in life. J Clin Endocrinol Metab. 2012;97:3197–206.
Halban PA, Polonsky KS, Bowden DW, et al. beta-cell failure in type 2 diabetes: postulated mechanisms and prospects for prevention and treatment. Diabetes Care. 2014;37:1751–8.
Savage PJ, Bennion LJ, Flock EV, et al. Diet-induced improvement of abnormalities in insulin and glucagon secretion and in insulin receptor binding in diabetes mellitus. J Clin Endocrinol Metab. 1979;48:999–1007.
Taylor R, Adamson AJ, Sattar N, Lean MEJ, Mathers JC. Di Rt. VLCD for weight loss and remission of type 2 diabetes?—Authors’ reply. Lancet. 2018;392:1307.
Fiori JL, Shin YK, Kim W, et al. Resveratrol prevents beta-cell dedifferentiation in nonhuman primates given a high-fat/high-sugar diet. Diabetes. 2013;62:3500–13.
Blum B, Roose AN, Barrandon O, et al. Reversal of beta cell de-differentiation by a small molecule inhibitor of the TGFbeta pathway. eLife. 2014;3:e02809.
Chera S, Baronnier D, Ghila L, et al. Diabetes recovery by age-dependent conversion of pancreatic delta-cells into insulin producers. Nature. 2014;514:503–7.
Lenz A, Toren-Haritan G, Efrat S. Redifferentiation of adult human beta cells expanded in vitro by inhibition of the WNT pathway. PLoS ONE. 2014;9: e112914.
Wang Z, York NW, Nichols CG, Remedi MS. Pancreatic beta cell dedifferentiation in diabetes and redifferentiation following insulin therapy. Cell Metab. 2014;19:872–82.
Sheng C, Li F, Lin Z, et al. Reversibility of beta-cell-specific transcript factors expression by long-term caloric restriction in db/db mouse. J Diabetes Res. 2016;2016:6035046.
Van de Casteele M, Leuckx G, Baeyens L, et al. Neurogenin 3+ cells contribute to beta-cell neogenesis and proliferation in injured adult mouse pancreas. Cell Death Dis. 2013;4: e523.
Berkowitz K, Peters R, Kjos SL, et al. Effect of troglitazone on insulin sensitivity and pancreatic beta-cell function in women at high risk for NIDDM. Diabetes. 1996;45:1572–9.
Brand MD. Mitochondrial generation of superoxide and hydrogen peroxide as the source of mitochondrial redox signaling. Free Radic Biol Med. 2016;100:14.
Fan J, Du W, Kim-Muller JY, et al. Cyb5r3 links FoxO1-dependent mitochondrial dysfunction with beta-cell failure. Mol Metab. 2020;34:97–111.
Amo-Shiinoki K, Tanabe K, Hoshii Y, et al. Islet cell dedifferentiation is a pathologic mechanism of long-standing progression of type 2 diabetes. JCI Insight. 2021;6:e143791
Md Moin AS, Dhawan S, Cory M, Butler PC, Rizza RA, Butler AE. Increased frequency of hormone negative and polyhormonal endocrine cells in lean individuals with type 2 diabetes. J Clin Endocrinol Metab. 2016;101:3628–36.
Son J, Ding H, Farb TB, et al. BACH2 inhibition reverses beta cell failure in type 2 diabetes models. J Clin Invest. 2021;131:e153876
Ahuja M, Ammal Kaidery N, Yang L, et al. Distinct Nrf2 signaling mechanisms of fumaric acid esters and their role in neuroprotection against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced experimental Parkinson’s-like disease. J Neurosci. 2016;36:6332–51.
Acknowledgements
This review is intended to summarize work in the Authors’ laboratory. The reference list reflects this limitation, with apologies to the many colleagues whose contributions could not be cited. This work has been supported by grants from NIH (DK64819, DK57539, DK58282, DK63608, HL87123), Astra-Zeneca, Merck, Takeda, Servier, and the JPB Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest relevant to the material covered in this review.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Accili, D., Du, W., Kitamoto, T. et al. Reflections on the state of diabetes research and prospects for treatment. Diabetol Int 14, 21–31 (2023). https://doi.org/10.1007/s13340-022-00600-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-022-00600-2