Abstract
Stem canker (SC), caused by Diaporthe helianthi, is the most serious sunflower disease in Uruguay. Yield losses have been estimated up to 75%. Chemical control is one of the strategies used to manage this disease, but fungicide application should be done before symptoms are visible. Ascospores are the primary source of inoculum, they are produced in perithecia which develop in infected stubble and are dispersed by wind to infect plants. As in other monocyclic diseases, quantifying primary inoculum is essential to predict an epidemic. In this study, ascospores were trapped on microscope slides with solid petroleum jelly which were placed on top of flat open cages filled with natural infected stubble. Cages were placed outdoors, slides where replaced twice a week and stained ascospores were counted under the microscope. Our objective was to develop weather-based models to predict ascospore release levels of D. helianthi from infected stubble. Explanatory weather variables were calculated during the seven-day periods prior to each field weekly ascospore count using daily weather station data from La Estanzuela, Uruguay. Then, logistic models were fit to estimate probabilities of having severe or moderate to light levels of ascospore counts. The best models included variables associated to the precipitation and dew-induced wetness frequency, combinated with the simultaneous occurrence of high relative humidity or low thermal amplitude records. Estimating the evolution of ascospore release through the weather-based models might help to guide preventive fungicide applications to control stem canker in Uruguay.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stem canker (SC) is a sunflower (Helianthus annuus L.) disease caused by multiple Diaporthe/Phomopsis species. Diaporthe helianthi Munt.-Cvetk was first reported in former Yugoslavia in 1980 (Muntañola-Cvetkovic et al. 1981), and it soon became the most serious disease in European sunflower producing countries, where yield losses were estimated up to 50% and losses in oil content exceeded 10% (Laville 1986; Masirevic and Gulya 1992). Until recently, D. helianthi was considered the only species causing the disease. However, three different species, D. gulyae, D. kongii and D. kochmanii, were discovered causing disease outbreaks in Australia in 2009 (Thompson et al. 2011), additionally since 2010, D. masirevicii and D. novem have been added to list in Australia and Europe (Harveson et al. 2016). Conversely, in United States SC was also reported in Ohio in 1980, but the identity of the Diaporthe species was not defined precisely (Herr et al. 1983), until D. gulyae, D. helianthi and D. stewartii were reported (Mathew et al. 2015; Olson et al. 2017). In South America, it was first reported in Argentina in 1992 (Fálico de Alcaraz et al. 1994), later in Uruguay in the growing season 2002–2003 but the species involved were not studied. In Uruguay, SC was assumed to be caused by D. helianthi as it was considered, at that time, to be the only species causing the disease globally.
The first disease epiphyte was determined in a localized sunflower sowing region in Uruguay called Mercedes (department of Soriano), nevertheless by the next growing season it was widespread across the country. Maximum yield losses in the country have been estimated in 75% for grain, 21% for oil content and 26% for thousand kernel weights (Stewart and Rodríguez 2018).
The fungus survives on stubble where perithecia are developed (Herr et al. 1983) and produce ascospores, which function as primary source of inoculum (Delos et al. 1995). SC is known to be a monocyclic disease, the absence of secondary cycles during the growing season and the fact that there is no spread from plant to plant, determine that the amount of primary inoculum is essential for epidemics to occur (Delos et al. 1995). Madden et al. (2007) stated that monocyclic diseases are controlled by reducing the amount of primary inoculum or the efficacy of inoculum. Thus, determinants for SC development would be, among others, amount of stubble lying on the soil surface, number of perithecia per gram of stubble, successive ascospore releases, and favorable weather conditions.
Ascospore production from perithecia is a continuous phenomenon, successive emissions from the same perithecia are observed every time favorable conditions are met (Delos et al. 1995). Ascospores discharge is active through specialized apical structure or elastic ring that acts as a pressure release valve in the ascus (Hawksworth et al. 1995; Kendrick 2000). As the ascus reaches maturity it begins to take up water and exert great pressure on its walls, this pressure becomes so great that it finally causes a rupture at the apical pore. Asci send out ascospores with a burst of kinetic energy obtained from internal pressure, releasing ascospores into airflow (Kendrick 2000) which are then dispersed by wind to infect plants. Infection takes place under favorable conditions and disease symptoms are seen approximately three to four weeks later (Delos et al. 1995; Fálico et al. 2003). The fungus generally penetrates the leaf edge, enters via hydathode pores, and progresses systemically via leaf veins, down main vein, until it reaches the stem through petiole (Delos et al. 1995; Harveson et al. 2016). Elongated grey to brown lesions are formed in stem centered on the axil, as the pathogen progresses the pith is destroyed, stems soften, plants tip over and generally break.
Higher incidence of SC has been associated to mean minimal temperature over 18.8 °C and longer incubations periods associated to high precipitations (Fálico et al. 2003). Climate effect on fungus dynamic and plant receptivity as a function of plant growth stage and variety, has been gathered in the model developed in France called ASPHODEL decision-support (Delos and Moinard 1996). The model simulates the effect of climate on fungus biology, maturation of ascospores, projection and infection. It was developed to improve farmers spraying decisions, optimizing time of application, and helping farmers decide whether to spray or not, since fungicide applications to control SC should be done preventive. Nevertheless, such model can not always be extrapolated to other regions. In fact, erratic predictions using ASPHODEL were carried out using local meteorological conditions in other countries, these were attributed to differences in the pathogen’s population (Battilani et al. 2003).
Quantifying primary inoculum is essential to predict development of monocyclic diseases such as SC. Ascospore release could be used to predict epidemics and might help to guide preventive fungicide applications to control SC in Uruguay. Thus, the objective of this study was to develop weather-based logistic regression models to predict binary levels of ascospore releases of Diaporthe helianthi from infected stubble.
Materials and methods
Development of weather-based logistic regression models
Ascospore counts from infected stubble
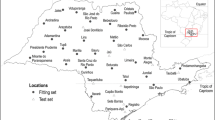
During two growing season (2005/06 and 2006/07) an ascospore trap, consisting of 12 to 18 pieces of natural infected stubble from the previous season, was accommodated into a flat wooden cage (44 cm length × 34 cm wide and 5 cm height) with mesh (2.5 × 2.5 cm) on both sides. The stubble was fitted in to cover the entire bottom of the cage. The cage (LE-trap) was placed outdoors at La Estanzuela Research Station (coordinates: 34° 20′ 23.72” S – 57° 41′ 39.48” W) (Fig. 1). Throughout October to March, microscope slides with solid petroleum jelly pasted thinly on one side where placed lying down on the cage, over the mesh with petroleum jelly downwards. Slides where replaced twice a week, taken into the laboratory, stained with a drop of cotton blue dilution, and sealed with 2 × 2 cm cover slide. The whole surface of the cover slide was examined under the microscope and ascospores were counted. Ascospores were recognized by its size and characteristic peanut-shape, being two-celled and constricted at the septum.
Binary response variable
Ascospore counts (two counts per week) were weekly accumulated (Asc7). These weekly ascospore counts (n = 34) were binary coded as 1 (Asc7 > 148; S: severe ascospore release level) or 0 (Asc7 ≤ 148; L: moderate to light level). The threshold (148 ascospores) was defined following a statistical criterion and corresponded to 70% percentile respect to the 34 weekly (Asc7) values analyzed.
Explanatory weather variables
Daily records of maximum temperature (MxT: °C), minimum temperature (MnT: °C), precipitation (Prec: mm) and relative humidity (RH: %, average of three observations: 9:00, 15:00 and 21:00 h) were collected from an automatic weather station (Campbell Sci CR 1000) located at La Estanzuela research station (INIA) beside the experimental field. Daily thermal amplitude (Ta:°C) was calculated as the range or difference between MxT and MnT. From these daily meteorological data, the following secondary weather variables were calculated at successive 7-day intervals including twice a week ascospore counts (n = 34): number of days with MaxT<30 °C and MinT>15 °C (DT); mean maximum temperature (MMxT); mean minimum temperature (MMnT); days with Prec≥27 mm (DPrec1); days with Prec≥0.2 mm and Prec<27 mm (DPrec2); total accumulated millimeters of daily Prec (>0 mm) (TPrec); days with simultaneous occurrence of Prec≥0.2 mm and Prec<27 mm and Ta < 14 °C (DPrecTa); days without Prec (Prec<0.2 mm) (DryD) and Ta < 7 °C (DryDTa); sum of wet days = DPrecTa+DryDTa (WetDTa); days with simultaneous occurrence of Prec≥0.2 mm and Prec<27 mm and RH > 76% (DPrecRH); days without Prec (Prec<0.2 mm) (DryD) and RH > 81% (DryDRH); sum of wet days = DPrecRH+DryDRH (WetDRH); days with RH >85% (DRH).
Both, temperature/moisture thresholds of the above weather variables and the length of the time interval (in which weather variables were calculated), were finally defined by iteratively maximizing their correlations with ascospore release levels. A computer program using the language available from SAS (Statistical Analysis Systems, version 8.0; SAS Institute, Inc., Cary, NC, USA) was written to calculate the weather variables and to perform the iterative process (partial results of this iterative preliminary analysis are not provided). The Freq procedure in SAS was used to calculate Kendall Tau-b nonparametric correlation coefficients (rk) between binary levels of weekly ascospore counts (Asc7 > 148: severe ascospore counts (S) and Asc7 ≤ 148: moderate to light (L), coded as 1 and 0 respectively) and explanatory weather variables. Using the SAS Logistic procedure, weather-based logistic regression models were fit (maximum likelihood method) to estimate the probabilities of occurrence of the binary weekly ascospore release levels. Each logistic model fits one equation for estimating ln[PrS/(1-PrS)] = β0 + β1X, where PrS is the probability of observing a severe (S) weekly ascosopore release level, β0 and β1 are the parameter estimates and X is a weather predictor. The logit function [ln(PrS/(1-PrS)], where ln is the natural logarithm, joins the stochastic component with the weather variables. Solving Exp{ln[PrS/(1-PrS)]}/{1 + Exp{ln[PrS/(1-PrS)]}}, the probability PrS is obtained. The probability of having a moderate-light ascospore release level (PrL) results from subtracting PrS to 1.
Stepwise logistic regression was used as one of the methods to select the most appropriate weather-based model (significance level of 0.01 was used as entry (SLE) and retention (SLS) criteria for the variables). The predictive ability of the models was also calculated based on the number of pairs of observations (t) with different outcome ascospore release level (240 pairs = 10S * 24 L). A pair of input observations with different responses is said to be concordant (or discordant) if the larger ordered value of the response has a higher (or lower) predicted event probability than the smaller response. If the pair is neither concordant nor discordant, it is a tie. Somers’D and Gamma correlation indices were calculated from the number of concordant (nc) and discordant (nd) pairs of observations. The values of both indices range from −1.0 (all pairs disagree, no association) to 1.0 (all pairs agree, perfect association). Finally, prediction accuracy of each of the analyzed models was also calculated as the percentage of cases analyzed in which there was agreement between the observed weekly ascospore release level and that predicted by the logistic models.
Validation
For validation purposes, two other ascospore traps were placed at Rodó, department of Soriano (Rodo-trap) and at Paysandú, department of Paysandú (Pay-trap) (Fig. 1), at 2005/06 cropping season. Both ascospore traps were manipulated in a similar manner as LE-trap (described previously). Distance from Pay-trap to Rodo-trap is approximately 148 km, and from Rodo-trap to LE-trap 74 km. In Rodó, daily data of maximum and minimum temperature and relative humidity was collected from the conventional weather station of Mercedes (National Weather Service from Uruguay). Daily precipitation data was collected from a conventional precipitation gauge located in the experimental field. The distance between Rodó and Mercedes (Department of Soriano) is 68 km. In Paysandú, daily meteorological data was collected from an automatic weather station (Campbel Sci CR 1000) located in the experimental field. Probabilities of weekly ascospore levels estimated by selected binary logistic regression models were validated against observed Asc7 values in Rodó (n = 9) and Paysandú (n = 10) traps. Prediction accuracy was calculated as the number of all correct model predictions divided by the total weekly ascospore counts evaluated in each location.
Results
The highest correlations (rk: coefficient of Kendall Tau-b) between binary levels of weekly ascospore counts and the weather variables analyzed were found when the latter were calculated in seven-day intervals prior to each weekly ascospore count (Asc7) (Table 1). Simple variables associated with temperature showed low correlations with Asc7 binary levels, moreover, correlations presented by variables only related to the precipitation, such as DPrec1 (rk = −0.05), DPrec2 (rk = 0.37) and TPrec (rk = 0.21), were also low (Table 1). When weather variables were constructed to estimate both precipitation-based wet days (DPrecRH) and dew-induced wet days (DryDRH), requiring simultaneous occurrence of high relative humidity records, better correlations were obtained. DPrecRH combines the simultaneous daily occurrence of precipitation (ranging from 0.2 mm to 27 mm) and high air relative humidity (RH > 76%), resulting highly correlated (rk = 0.56) to weekly ascospore counts. In contrast, dew-induced wet days associated to the days without precipitation (Prec<0.2 mm) and simultaneous high relative humidity (RH > 81%), was not as highly correlated as the latter (DryDRH; r k = 0.31). Nonetheless by adding up these last two variable, WetDRH was assembled, and yielded the highest correlation with ascospore release levels (rk = 0.64). Using daily thermal amplitude records for estimating precipitation-based wet days (DPrecTa) and dew-induced (DryDTa) wet days, the variable WetDTa was calculated, reaching a high correlation (rk = 0.58) with ascospore release levels.
The binary response logistic regression model (model I) using the explanatory variable WetDRH correctly classified 30 out of the 34 weekly ascospore counts, with a prediction accuracy of 88% (Table 2). The stepwise logistic regression run with all the analyzed weather variables selected model I as the most appropriate. Other predictive indices, such as Somers’D and Gamma, were also the highest (Table 2). Clear correspondence resulted between the probability of occurrence of a high level of ascospore counts (PrS) estimated by model I, and its correspondent observed weekly ascospore counts (Asc7) at La Estanzuela (Uruguay), during seasons 2005/06 and 2006/07 (Fig. 2). Model II presented slightly weaker predictive ability indices than model I, but the former has the advantage that can be run only with daily temperature and precipitation data. It is worth mentioning that daily relative humidity records are not always available in historical weather data.
Weekly evolution of the probability of occurrence of a severe level (PrS) of weekly ascospore counts (Asc7 > 148 ascospores) estimated by model I (Table 2) for 2005/06 and 2006/07 cropping seasons. Weekly ascospore counts (LE-trap; n = 34) observed at La Estanzuela (Uruguay) were also presented (bars)
Validation
Probabilities of weekly ascospore levels estimated by both binary response logistic regression model I and II, were validated against observed Asc7 values in Rodó (n = 9) and Paysandú (n = 10) traps. Both models miss-classified three out of nine Asc7 counts in Rodó (Fig. 3) and one out of ten in Paysandú (Fig. 4).
Weekly evolution of the probability of occurrence of a severe level (PrS) of weekly ascospore counts (Asc7 > 148 ascospores) estimated by model I (above) and II (bottom) (Table 2) for 2005/06 cropping season. Weekly ascospore counts (Rod-trap; n = 9) observed at Rodó (Uruguay) were also presented (bars)
Weekly evolution of the probability of occurrence of a severe level (PrS) of weekly ascospore counts (Asc7 > 148 ascospores) estimated by model I (above) and II (bottom) (Table 2) for 2005/06 cropping season. Weekly ascospore counts (Pay-trap; n = 10) observed at Paysandú (Uruguay) were also presented (bars)
Discussion
Other studies have used logistic regression techniques in order to develop weather-based models to predict ordinal and binary levels of crop diseases (De Wolf et al. 2003; Moschini et al. 2014), mycotoxin contamination (Sancho et al. 2018) and weekly binary release of Guignardia citricarpa ascospores (Dummel et al. 2015). Similarly, the current study developed weather-based logistic models to predict weekly binary ascospore release levels of D. helianthi from infected stubble.
Ascospores are actively released into airflow with a burst of kinetic energy obtained from internal pressure of the ascus (Hawksworth et al. 1995; Kendrick 2000). Our results show that precipitations, dew, relative humidity, thermal amplitude or more likely a combination of all, are key variables in determining ascospore release, thus probably affecting the internal pressure of the ascus. Simple weather variables associated with temperature and precipitation presented low correlation with weekly ascospore count (Asc7) levels. Interestingly, number of days with precipitation equal or higher than 27 mm was correlated negatively to Asc7, and so excessive precipitations could act washing down ascospores from perithecium apical structure, or even prevent the ascus from building up the internal pressure necessary to send out the ascospores into the airflow above the debris. Excessive precipitations causing washing down of ascospores and preventing them from reaching the sunflower leaves has already been mentioned by Fálico et al. (2003).
Some weather variables identified in this study explained most of the variability in weekly ascospore counts during 2005–2006 and 2006–2007 cropping season at La Estanzuela. Variables like WetDTa and WetDRH which sum up wet days induced by rain plus those originated by dew, can be understood as long duration of wet period. According to our results, the fungus requires long precipitation-induced wet duration for the ascospore discharge, but not all days with precipitation can lead to that. The correlation showed by the variable DPrec2 (rk = 0.37) increased significantly when the occurrence of days with precipitation ranging from 0.2 mm to 27 mm was combined with high relative humidity records (RH > 76%), as expressed by DPrecRH (rk = 0.56). It is so those days with precipitation originated by fast passing of front systems or short-lived convective systems, frequently accompanied by low daily records of relative humidity, were discarded (short wetness periods). In concordance, Dummel et al. (2015) in Montecarlo (Argentina) measured the daily wetness duration, finding that the variable which only counted the days with long wetness periods (greater than 10 h) reached the highest correlation with weekly ascospore releases of Guignardia citricarpa (citrus field). Moreover, in the current study, dew expressed in variables such as DryDTa and DryDRH had a minor effect on ascospore discharge, but both contributed to increase correlations in variables WetDTa and WetDRH. Coincidently, the key environmental component of the best logistic model for predicting weekly ascospore releases of Guignardia citricarpa (Dummel et al. 2015) was the variable WetDTa, which counts the days with simultaneous occurrence of precipitation (> = 0.2 mm) and daily thermal amplitude <14.2 °C and the days without precipitation and thermal amplitude <7 °C, in 7-day periods previous to each observation.
It has been mentioned that successive ascospore discharges from perithecium can occur, and can last from 10 to 17 days, and that these are stimulated by strong fluctuation of water content in debris tissue produced either by rain or by lack of rain (Le page 1994). In the current study, the best fitting variables in the models were those that consider both the number of days with precipitations and the number of dry days combined with the simultaneous occurrence of high values of relative humidity (WetDRH) or low values of thermal amplitude (WetDTa).
Probabilities of weekly ascospore levels estimated by both binary response logistic regression models I and II, misclassified 33% of the observed weekly ascospore counts (Asc7) in Rodó (n = 9) (Fig. 3). Using Pay-trap observations (n = 10) (Fig. 4), both models miss-classified only 10% of the observed weekly ascospore counts (Asc7). Rodó was the only location where the weather station was not in situ (except the precipitation gauge), so weather variables were probably not as precise as in the other two locations (Paysandú and La Estanzuela), and this might be the reason why predictions were not as accurate. Both logistic models run in both locations (Rod-trap and Pay-trap) had similar performance in order to predict weekly ascospore release levels. In Rodó, it is worth pointing out that model II, which used thermal amplitude values instead of relative humidity records, could estimate correctly the high ascospore release (757 ascospores) accumulated at 344 julian day (week number: 49). Model I underestimated this observed ascospore count (Fig. 3).
The weather-based predictive models developed in this study simulate ascospore release of Diaporthe helianthi. Agreement between observed and estimated Asc7 confirms the value of the fitted models. Estimating the evolution of weekly ascospore release using the selected weather-based models might help farmers, together with other tools, to generate risk alarms and create an efficient monitoring system and sustainable SC chemical control in Uruguay.
References
Battilani P, Rossi V, Girometta B, Delos M, Rouzet J, André N, Esposito S (2003) Estimating the potential development of Diaporthe helianthi epidemics in Italy. Bulletin OEPP/EPPO Bulletin 33:427–431
De Wolf ED, Madden LV, Lipps PE (2003) Risk assessment models for wheat fusarium head blight epidemics based in within-season weather data. Phytopathology 93:428–435
Delos M, Moinard J (1996) ASPHODEL: modele de simulation des épidémies de Phomopsis du tournesol (Diaporthe helianthi). Med Fac Landbouww Univ Gent 61(2b):555–565
Delos M, Moinard J, Jacquin D (1995) Etude et surveillance du Phomopsis, des pieges au modele. Phytoma 473:25–27
Dummel DM, Agostini JP, Moschini R (2015) Predictive model for ascospore release of Guignardia citricarpa using climatological data. Acta Hortic 1065:953–963. https://doi.org/10.17660/ActaHortic.2015.1065.119
Fálico de Alcaraz L, Visintín G, Alcaraz Fálico ME (1994) El cancro del tallo del girasol Diaporthe helianthi Munt. Cvet In: Ciencia, Docencia y Tecnología 8:19–26
Fálico L, Alcaraz Fálico ME, Visintín G, García B (2003) Relación entre las condiciones climáticas, aparición de síntomas e incidencia del cancro del tallo del girasol (Diaporthe helianthi). Ciencia, Docencia y Tecnología 27:167–177
Harveson RM, Markell SG, Block CC, Gulpa TJ (2016) Compendium of sunflower diseases and pests. APS press, St Paul, Minnesota, U.S.A.
Hawksworth DL, Kirk PM, Sutton BC, Pegler DN (1995) Ainsworth & Bisby’s dictionary of fungi. 8th edition. International Mycological Institute, CAB International. University Press, Cambridge, UK
Herr LJ, Lipps PE, Watters BL (1983) Diaporthe stem canker of sunflower. Plant Dis 67:911–913
Kendrick B (2000) The fifth kingdom. 3rd edition. Focus publishing, R. Pullins Co., Newburyport, MA, USA
Laville J (1986) Cahier technique tournesol: maladie. Cetiom, Paris
Le page R (1994) Le Phomopsis du tournesol. Numero special Oleoscope, Cetiom N 14, vol 67
Madden LV, Hughes G, van den Bosch F. (2007) The study of plant disease epidemics. The American Phytopathological society. St. Paul, Mn, USA. 421 pp.
Masirevic S, Gulya TJ (1992) Sclerotinia and Phomopsis—two devastating sunflower pathogens. Field Crops Res 30:271–300
Mathew FM, Alananbeh MK, Jordahl JG, Meyer SM, Castlebury LA, Gulya TJ, Markell SG (2015) Phomopsis stem canker: a reemerging threat to sunflower (Helianthus annuus) in the United States. Phytopathology 105:990–997
Moschini RC, Canteros BI, Martínez MI, De Ruyver R (2014) Quantification of the environmental effect on citrus canker intensity at increasing distances from a natural windbreak in northeastern Argentina. Australas Plant Pathol 43:653–662
Muntañola-Cvetkovic M, Mihaljcevic M, Petrov M (1981) On the identity of the causative agent of a serious Phomopsis-Diaporthe disease in sunflower plants. Nova Hedwigia 34:417–435
Olson TR, Kontz B, Markell SG, Gulya TJ, Mathew FM (2017) First report of Diaporthe stewartii causing phomopsis stem canker of sunflower (Helianthus annuus) in Minnesota. Plant Dis 101:382–382
Sancho AM, Moschini RC, Filippini S, Rojas D, Ricca A (2018) Weather-based logistic models to estimate total fumonisin levels in maize kernels at export terminals in Argentina. Tropical Plant Pathology 43:99–108
Stewart S, Rodríguez M (2018) Cancro del tallo: epidemiología y control en Uruguay. 1° Taller de Enfermedades de Cultivos Extensivos, Escuela de Ciencias Agrarias Naturales y Ambientales de la UNNOBA. Pergamino, Argentina
Thompson SM, Tan YP, Young AJ, Neate SM, Aitken EAB, Shivas RG (2011) Stem cankers on sunflower (Helianthus annuus) in Australia reveal a complex of pathogenic Diaporthe (Phomopsis) species. Persoonia 27:80–89
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors of the manuscript submitted to Australasian Plant Pathology, point out that we have no potential conflict of interest.
Rights and permissions
About this article
Cite this article
Moschini, R.C., Rodríguez, M.J., Martínez, M.I. et al. Weather-based predictive models for Diaporthe helianthi ascospore release in Uruguay. Australasian Plant Pathol. 48, 519–527 (2019). https://doi.org/10.1007/s13313-019-00655-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-019-00655-x