Abstract
Objective
Evaluation of tolerability, safety and immunogenicity of a two-dose series of a quadrivalent meningococcal polysaccharide diptheria toxoid conjugate (ACYW-D) vaccine in Indian and Russian infants/toddlers.
Design
Open-label, single-arm, phase III multi-national trial.
Study participants
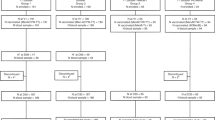
300 children aged 9–17 months, previously unvaccinated against meningococcal disease from four sites each in India (n=200) and the Russian Federation (n=100).
Intervention
Two 0.5 mL doses of ACYW-D by intramuscular injection, 3–6 months apart.
Main outcome measures
Meningococcal antibody titers to serogroups A, C, W-135 and Y, determined using a serum bactericidal assay in the presence of human complement before vaccination and 28 days after the second vaccination. Titers ≥1:8 against either/all of the A, C, W-135 or Y were considered seroprotective.
Results
After dose 2, 95.7–99.5% and 92.9–99.0% of infants/ toddlers achieved seroprotection across the four serogroups in India and the Russian Federation, respectively. No immediate adverse events were reported after any dose of ACYW-D. Solicited reactions were reported in 49.2% of participants, and were mainly of Grade 1 severity, and resolved within three days. Unsolicited adverse events were reported in 19.1% of infants: one event (Grade 3 diarrhea, resolving within one day) was considered related to study vaccine. No non-serious adverse events led to premature withdrawal from the study. Four serious adverse events were reported; none were considered related to study vaccine. No deaths occurred during the study.
Conclusions
A two-dose series of ACYW-D vaccine in Indian and Russian children (9-17 month) was well-tolerated with no safety concerns, and induced robust bactericidal antibody responses against the meningococcal serogroups contained in the vaccine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Manchanda V, Gupta S, Bhalla P. Meningococcal disease: History, epidemiology, pathogenesis, clinical manifestations, diagnosis, antimicrobial susceptibility and prevention. Indian J Med Microbiol. 2006;24:7–19.
Pace D, Pollard AJ. Meningococcal disease: Clinical presentation and sequelae. Vaccine. 2012;30:062.
Titova L, Samodova O, Buzinov R, Gordienko T. Epidemiology of meningococcal infection in arkhangelskoblast. Available from: http://wwwepinorthorg/eway/defaultaspx?pid=230&trg=Area_5268&MainArea_5 260=5263:0:15,2946:1:0:0:::0:0&Area_5263=5268:44984::1: 5264: 1:::0:0&Area_5268=5273:47185::1:5266:3:::0:0. Accessed June 19, 2017.
Koroleva IS, Maxina TA, Zakroeva M, Beloshitsky GV, Lytkina IN, Pyaeva AP. Epidemiology of invasive meningococcal disease in Moscow, 2005–2010. Available from: www.meningitis.org/assets/x/53942. Accessed June 19, 2017.
Sinclair D, Preziosi MP, Jacob John T, Greenwood B. The epidemiology of meningococcal disease in India. Trop Med Int Health. 2010;15:1421–35.
John TJ, Gupta S, Chitkara AJ, Dutta AK, Borrow R. An overview of meningococcal disease in India: knowledge gaps and potential solutions. Vaccine. 2013;31:2731–7.
Borrow R, Lee JS, Vazquez JA, Enwere G, Taha MK, Kamiya H, et al. Meningococcal disease in the Asia–Pacific region: Findings and recommendations from the Global Meningococcal Initiative. Vaccine. 2016;34: 5855–62.
Jafri RZ, Ali A, Messonnier NE, Tevi–Benissan C, Durrheim D, Eskola J, et al. Global epidemiology of invasive meningococcal disease. Popul Health Metr. 2013;11:17.
Borrow R, Alarcon P, Carlos J, Caugant DA, Christensen H, Debbag R, et al. The Global Meningococcal Initiative: global epidemiology, the impact of vaccines on meningococcal disease and the importance of herd protection. Expert Rev Vaccines. 2017;16:313–28.
Koroleva I, Beloshitskij G, Zakroeva I, Melnikova A, Koroleva M, Shipulin G, et al. Invasive meningococcal disease in Russian Federation. In: European Meningococcal Disease Society. 20 years EMGM. Bad Loipersdorf, Austria. 17–19 September 2013. p71. Available from: http://emgm.eu/meetings/emgm2013/Abstract_Book_EMGM_2013.pdf. Accessed April 13, 2017.
Robertson CA, Greenberg DP, Hedrick J, Pichichero M, Decker MD, Saunders M. Safety and immunogenicity of a booster dose of meningococcal (groups A, C, W, and Y) polysaccharide diphtheria toxoid conjugate vaccine. Vaccine. 2016;34:5273–8.
Pina LM, Bassily E, Machmer A, Hou V, Reinhardt A. Safety and immunogenicity of a quadrivalent meningococcal polysaccharide diphtheria toxoid conjugate vaccine in infants and toddlers: three multicenter phase III studies. Pediatr Infect Dis J. 2012; 31:1173–83.
Pelton SI, Gilmet GP. Expanding prevention of invasive meningococcal disease. Expert Rev Vaccines. 2009;8: 717–27.
Borrow R, Balmer P, Miller E. Meningococcal surrogates of protection–serum bactericidal antibody activity. Vaccine. 2005;23:2222–7.
Choudhuri D, Huda T, Theodoratou E, Nair H, Zgaga L, Falconer R, et al. An evaluation of emerging vaccines for childhood meningococcal disease. BMC Public Health. 2011;11:S29.
Halperin SA, Diaz–Mitoma F, Dull P, Anemona A, Ceddia F. Safety and immunogenicity of an investigational quadrivalent meningococcal conjugate vaccine after one or two doses given to infants and toddlers. European J Clin Microb Infect Dis. 2010;29:259–67.
Santos GF, Deck RR, Donnelly J, Blackwelder W, Granoff DM. Importance of complement source in measuring meningococcal bactericidal titers. Clin Diagn Lab Immunol. 2001;8:616–23.
Hansen J, Zhang L, Eaton A, Baxter R, Robertson CA, Decker MD, et al. Post–licensure safety surveillance study of routine use of quadrivalent meningococcal diphtheria toxoid conjugate vaccine (MenACWY–D) in infants and children. Vaccine. 2018;36:2133–8.
Noya F, McCormack D, Reynolds DL, Neame D, Oster P. Safety and immunogenicity of two doses of quadrivalent meningococcal conjugate vaccine or one dose of meningococcal group C conjugate vaccine, both administered concomitantly with routine immunization to 12–to 18–month–old children. Can J Infect Dis Med Microbiol. 2014;25:211–6.
Yadav S, Manglani MV, Narayan DA, Sharma S, Ravish HS, Arora R, et al. Safety and immunogenicity of a quadrivalent meningococcal conjugate vaccine (MenACYW–DT): a multicenter, open–label, nonrandomized, phase III clinical trial. Indian Pediatr. 2014; 51:451–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Javadekar, B., Ghosh, A., Kompithra, R.Z. et al. Safety and Immunogenicity of Two Doses of a Quadrivalent Meningococcal Polysaccharide Diphtheria Toxoid Conjugate Vaccine in Indian and Russian Children Aged 9 to 17 Months. Indian Pediatr 55, 1050–1055 (2018). https://doi.org/10.1007/s13312-018-1440-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-018-1440-z