Abstract
Introduction
Pivotal trials documented glycemic benefits of fixed-ratio combination of insulin glargine 100 U/mL and lixisenatide (iGlarLixi), with no weight gain and low hypoglycemia risk in type 2 diabetes (T2D). This study aimed at assessing effectiveness and patterns of use of iGlarLixi in a real-world setting.
Methods
This was a retrospective, multicenter, study, based on electronic medical records. All patients initiating iGlarLixi from May 2018 to July 2020 were considered.
Results
Overall, 25 centers provided data on 675 patients initiating iGlarLixi with the following characteristics: age 66.4 ± 10.1 years, 54.2% men, T2D duration 15.5 ± 11.5 years, HbA1c 8.6 ± 1.4%, body mass index (BMI) 30.8 ± 5.3 kg/m2, 45.1% already treated with basal insulin, and 21.9% with basal bolus (± oral hypoglycemic agents). Metformin and sodium–glucose cotransporter-2 inhibitors were used in 76.0% and 0.9% of patients, respectively. Combinations of iGlarLixi with other glucose-lowering drugs such as sulfonylureas or short-acting insulin were found in 32.4% of patients. Effectiveness of iGlarLixi (N = 184) showed that HbA1c declined by 0.77% [95% confidence interval (CI) −1.00, −0.54] after 6 months. In combination with metformin and/or SGLT-2i (N = 117), HbA1c declined by −0.92% (95% CI −1.22, −0.62) and weight significantly decreased by 1.21 kg. iGlarLixi dose was suboptimally titrated. Safety data (N = 171) showed incidence rates of blood glucose ≤ 70 and < 54 mg/mL of 0.26 and 0.05 events per person-month during 6 months, respectively, with a risk reduction of about 75% with respect the 6 months before iGlarLixi initiation. No severe hypoglycemia was reported.
Conclusion
In adults with T2D, effectiveness and safety of iGlarLixi were documented in a real-world setting; appropriateness of use and adequate titration should be urgently improved so that clinical practice outcomes become more comparable to clinical trials results. Further real-world studies on the effect of iGlarLixi therapy are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
In pivotal trials, fixed-ratio combination of insulin glargine 100 U/mL and lixisenatide (iGlarLixi) improves metabolic control, with no weight gain and low risk of hypoglycemia in type 2 diabetes (T2D). |
Data on real-world effectiveness and patterns of use of iGlarLixi are scant. |
What was learned from the study? |
Effectiveness and safety of iGlarLixi documented in pivotal trials are confirmed in a real-world setting. |
However, appropriateness of use and adequate titration should be urgently improved. |
Introduction
In recent years, a paradigm shift in the management of type 2 diabetes (T2D) has occurred: while the basal-bolus insulin regimen was generally considered the most effective treatment of advanced stage of T2D, currently, in many cases the combination of metformin, GLP-1 receptor agonists (GLP-1RA), and basal insulin is increasingly preferred to the basal-bolus regimen [1].
The main effect of basal insulin consists in reducing gluconeogenesis to achieve fasting blood glucose (FBG) control, while GLP-1RA mitigates the effect of blood sugar elevation at meals (especially prandial GLP-1RA), while also contributing to control fasting blood glucose levels [2].
One of the most widely used basal insulin is insulin glargine 100 U/mL (iGlar), whose efficacy and safety, including cardiovascular safety, are well established [3]. The short-acting GLP-1 RA lixisenatide (Lixi) has a robust postprandial glucose (PPG)-lowering effect, mainly due to the delay in gastric emptying and the reduction in glucagon release [4]. Since iGlar and Lixi act selectively on FBG and PPG, their once-daily fixed-ratio combination (FRC) iGlarLixi may offer more effective and complete glucose control than its single components owing to the complementary effect on FBG and PPG, which is evidenced by reduced average glycemia, glycemic exposure, and glucose variability [5, 6].
iGlarLixi was investigated in patients undergoing uncontrolled basal-oral therapy (BOT) (LixiLan-L phase 3 trial) and proved to be superior to iGlar in terms of glycemic control, with additional benefits on body weight and without increase in the risk of hypoglycemia [7]. In another phase 3 trial (LixiLan-O study) in patients with T2D inadequately controlled on metformin with or without a second oral glucose-lowering drug, iGlarLixi achieved greater reductions in glycated hemoglobin (HbA1c) from baseline than iGlar or Lixi, without increasing hypoglycemia or weight gain risk [8]. Similarly, the LixiLan-G trial showed that switching to iGlarLixi as compared with continuing GLP1-RA alone may improve metabolic control [9]. Furthermore, iGlarLixi was associated with a lower risk of gastrointestinal adverse effects over time compared with Lixi alone [10]. A recent postmarketing randomized clinical trial (RCT) (Solimix study) showed that, in suboptimally controlled T2D requiring treatment beyond BOT, iGlarLixi, compared with twice-daily premix BIAsp30, provided better glycemic control (−1.3% versus −1.1%, p < 0.001) with weight benefit and less hypoglycemia [11, 12].
In addition to the accumulating data from clinical trials, real-word evidence (RWE) from a larger, more heterogeneous patient population may help to inform the prescribing decisions of clinicians. Being relatively new on the market, real-world evidence on iGlarLixi is currently limited.
iGlarLixi was launched in Italy in May 2018 in two prefilled pens for once-daily injection (Suliqua Sanofi, Paris, France): the 10–40 U pen that delivers 0.50 µg of Lixi for each unit of iGlar, up to a maximum of 40 U (peach pen) and the 30–60 U pen that delivers 0.33 µg of Lixi for each unit of iGlar, up to a maximum of 60 U (olive pen).
The ENSURE (EffectiveNess of SUliqua in clinical practice: a REtrospective multinational real-world) study was designed to evaluate the effectiveness of the FRC in an adult real-world T2D population in Italy. Moreover, this study aimed at providing insights on the use of iGlarLixi in clinical setting and describe the clinical profile of patients selected for this therapeutic option.
Methods
ENSURE was a multicenter, retrospective, cohort study based on electronic medical records (EMRs) (Smart Digital Clinic, software property of METEDA s.r.l., San Benedetto del Tronto, Italy). Methods were similar to those applied to previous multicenter, retrospective, cohort study based on EMRs [14,15,16].
All patients with diagnosis of T2D, aged ≥ 18 years, with at least one prescription of iGlarLixi from May 2018 to July 2020 were eligible for the study.
A total of 25 diabetes clinics were involved from different areas of Italy, providing a representative picture of the national approach of use of iGlarLixi. Data were recorded on EMRs as part of routine clinical practice. Data relative to the 6-month period before and after the index date (date of the first prescription of iGlarLixi, T0) for each patient were analyzed.
Baseline (T0) patient characteristics included: age, gender, diabetes duration, HbA1c, FBG, weight/body mass index (BMI), glucose-lowering drugs, number of insulin injections, blood pressure, lipid profile, estimated glomerular filtration rate (eGFR), albuminuria, and cardiovascular complications. Cardiovascular complications were classified according to ICD-9-CM codes.
Treatment schemes and type of drugs adopted before, during, and after treatment with iGlarLixi were described, as well as the discontinuation rate. Type of pen (10–40 U, peach pen) or (30–60 U, olive pen) at first prescription was also recorded.
The following effectiveness endpoints were considered: changes at 6 months (T6) in HbA1c (primary endpoint), FBG, body weight, and iGlarLixi dose (continuous endpoints). Furthermore, the proportion of patients with HbA1c < 7% (53 mmol/mol) and < 8% (64 mmol/mol) at T0 and T6 (categorical endpoints) was assessed.
The following safety endpoints were considered: severe hypoglycemia (as reported in EMRs) during 6 months, and episodes of hypoglycemia ≤ 70 or < 54 mg/dL [as derived from self-monitoring blood glucose tests (SMBG) downloaded in EMRs] [13].
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. The study protocol was approved by all local ethics committees of the participating centers (Supplementary Materials, ECs list). Data were collected, and processed analyzed anonymously, in compliance with the local and European code on protection of personal data (General Data Protection Regulation).
Furthermore, each site filled in a specific questionnaire on the main reasons for initiating iGlarLixi in their patients with T2D. Specifically, for each site the physician was asked to report the three main reasons (i.e., better control, better adherence, hypoglycemia concern, efficacy, weight gain, simplification of therapy, less variability, easier titration, frequency of administration, adverse events, cost, other) for initiating iGlarLixi.
Statistical Analyses
Sample size estimation was based on the precision of the estimate of reduction in HbA1c levels after 6 months from initiation of iGlarLixi. A sample size of 200 allowed the estimation of a two-sided 95% confidence interval (95% CI) with a distance from the mean paired difference to the limits equal to 0.18 when the estimated standard deviation (SD) of the paired differences is 1.3. As an example, a reduction in HbA1c levels of 0.5% would produce a 95% CI of 0.32–0.68.
Statistical methods were similar to those applied to previous multicenter, retrospective, cohort studies based on EMRs [14,15,16]. Descriptive data were summarized as mean and standard deviation (continuous, normally distributed variables), median and interquartile range (continuous, not normally distributed variables), or frequency and proportion (categorical variables).
Changes in HbA1c, FBG, body weight, and iGlarLixi dose were assessed using mixed models for repeated measurements. Results were expressed as estimated mean and estimated mean difference from T0 with their 95% CI. Paired t-test derived from linear mixed models for repeated measurements was applied for within-group comparisons.
As categorical secondary outcomes, the proportions of patients with HbA1c < 7.0% (< 53 mmol/mol) and < 8.0% (< 64 mmol/mol) at T0 and T6 were evaluated. Within-group statistical comparisons (McNemar test for change versus baseline) were applied.
Incidence rates for hypoglycemic events were calculated and expressed as numbers of events per patient-month with their 95% CI. Incidence of hypoglycemic events was compared between visits using Poisson regression model with correction for overdispersion.
Different analysis populations were identified:
-
Intention-to-treat population (ITT) included all patients with at least one iGlarLixi prescription. This population was used to describe the clinical profile of patients receiving the treatment and assess the glucose-lowering drugs used before and in association with iGlarLixi and after iGlarLixi discontinuation. Since the proportion of ITT patients with an available follow-up HbA1c value was low [the coronavirus disease 2019 (COVID-19) pandemic limited the access to the visits and laboratory tests], ITT population was not used for the evaluation of the primary endpoint.
-
Efficacy population (EP) represented the subset of ITT patients having valid values at baseline (T0) and after 6 months (T6) for the primary endpoint (HbA1c). Primary effectiveness analysis was performed on EP.
-
Per-protocol population (PP) was composed of the subset of EP treated according to the summary of product characteristics (SmPC). This population included patients not treated with short-acting insulin and off-label oral hypoglycemic agents (OHAs) in association with iGlarLixi, i.e., no OHA other than metformin and sodium–glucose cotransporter-2 inhibitors (SGLT2i). The PP analysis represented a secondary effectiveness analysis.
-
Safety population (SP) was represented by all ITT patients for the evaluation of severe hypoglycemia (events recorded in EMRs). For the evaluation of glycemic values ≤ 70 and < 54 mg/dL, the subsample of the SP having at least one SMBG value available was considered.
Results
Overall, 25 diabetes clinics were involved and 675 eligible patients (ITT) were identified in EMRs. The study flow chart is shown in Fig. 1.
The complete clinical profile of ITT patients treated with iGlarLixi is reported in Table 1. At the first prescription of iGlarLixi, patients (54.2% men) had a mean age of 66.4 ± 10.1 years and diabetes duration of 15.5 ± 11.5 years. Mean baseline levels of HbA1c and FBG were 8.6 ± 1.4% and 167.8 ± 58.9 mg/dL, respectively.
Treatment Approaches in Patients Prescribed iGlarLixi
Patients initiating iGlarLixi were previously treated with basal insulin ± OHAs (39.6%) or basal + short-acting insulin ± OHAs (21.9%); among the latter, patients were treated mainly with three injections (62.1%) (Table 1). Only 6.4% and 4.4% of patients were treated respectively with OHAs alone or GLP1-RA ± OHAs before initiating iGlarLixi. Free combinations of basal insulin + GLP-1 RA (± OHAs) were used in 5.5% of patients (Table 1). Classes of drugs prescribed before, during, and after iGlarLixi initiation are reported in Supplementary materials—table S1.
Drugs associated with iGlarLixi were not only metformin and SGLT2i, as defined in the SmPC. iGlarLixi in combination with one OHA (metformin) was the most frequent treatment scheme (54.5%) (Table 1), while an SGLT2i was used in a small minority of patients (0.9% overall; 0.4% in combination with metformin) (Table 1 and Supplementary Materials—Table S1). Alternative (off-label) combinations were found in 32.4% of patients (Table 1) (21.9% sulfonylureas). Furthermore, 5.8% of patients used short-acting insulin in combination with iGlarLixi (64.1% of them maintaining the three daily injections) (Table 1, Supplementary Materials—Table S1).
Table 1 reports type of pen and doses of iGlarLixi at first prescription. Furthermore, among patients already treated with basal insulin before starting iGlarLixi (67%), 84.6% were prescribed the 10–40 U (peach) pen, and 15.4% the 30–60 U (olive) pen. In these patients, starting doses of iGlarLixi slightly differed from doses of basal insulin used before: 17.2 ± 8.4 U of basal insulin versus 18.3 ± 6.3 U of iGlarLixi in peach pen; 26.6 ± 14.3 U of basal insulin versus 28.7 ± 11.7 U of iGlarLixi in olive pen.
Effectiveness Analysis
Patients selected for the EP (N = 184) had a follow-up of 6.6 ± 1.7 months and were seen at diabetes clinics 2.0 ± 0.9 times during the study period.
EP patients (52.7% men) had a mean age of 66.1 ± 9.7 years and diabetes duration of 18.0 ± 14.3 years. Mean baseline levels of HbA1c and FBG were 8.8 ± 1.5% (72.7 ± 16.4 mmol/mol) and 175.3 ± 65.9 mg/dL. Therefore, diabetes duration and HbA1c and FBG levels were higher in EP as compared with ITT population (Supplementary Materials—Table S2).
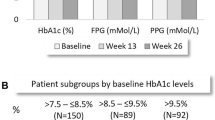
A statistically significant reduction in HbA1c levels from baseline to 6 months of −0.77% (95% CI −1.00, −0.54) was obtained [from 8.82% (95% CI 8.61, 9.03) to 8.04% (95% CI 7.85, 8.23); p < 0.0001] (primary endpoint) (Fig. 2).
The proportion of patients with HbA1c < 7.0% (< 53 mmol/mol) increased from 5.4% at T0 to 16.3% at T6 (p = 0.0001), while the proportion of patients with HbA1c < 8.0% (< 64 mmol/mol) increased from 25.5% at T0 to 53.8% at T6 (p < 0.0001) (Table 2).
After 6 months, levels of FBG were significantly reduced by −17.7 mg/dL (95% CI −28.3, −7.2) [from 175.3 mg/dL (95% CI 165.6, 185.1) to 157.6 mg/dL (95% CI 149.0, 166.2); p = 0.001], without significant changes in body weight [−0.48 kg (95% CI −1.11, 0.15); p = 0.14]. The iGlarLixi dose significantly increased by 5.14 U (95% CI 3.95, 6.33) during 6 months [from 18.73 (95% CI 17.58, 19.88) to 23.86 U (95% CI 22.41, 25.31); p < 0.0001] (Table 3).
Per-Protocol Analysis
Patients selected for the PP (N = 117) had a follow-up of 6.7 ± 1.7 months and were seen at diabetes clinics 1.9 ± 0.8 times during the study period. Baseline characteristics of PP population were very similar to those of EP (Supplementary Materials—Table S2).
In PP, HbA1c levels were reduced by −0.92% (95% CI −1.22, −0.62) (Fig. 2).
Proportions of patients achieving HbA1c levels < 7.0% (< 53 mmol/mol) and < 8.0% (< 64 mmol/mol) were similar in EP and PP, with PP showing larger improvements (Table 3).
In PP, levels of FBG were significantly reduced by −28.4 mg/dL (95% CI −41.2, −15.7) [from 177.1 mg/dL (95% CI 163.8, 190.3) to 148.6 mg/dL (95% CI 140.7, 156.5); p < 0.0001] with a statistically significant body weight decrease [−1.21 kg (95% CI −1.95, −0.47)]. The iGlarLixi dose significantly increased by 4.40 U (95% CI 3.18, 5.62) during 6 months [from 17.73 (95% CI 16.49, 18.97) to 22.12 U (95% CI 20.35, 23.89)] (Table 3).
Safety Analysis
A total of 171 patients had at least one SMBG test downloaded in EMRs in the study period. SP patients (53.2% men) had mean age of 67.1 ± 9.2 years and diabetes duration of 15.3 ± 12.0 years. Mean baseline levels of HbA1c and FBG were 8.4 ± 1.2% and 163.8 ± 53.5 mg/dL, respectively. Therefore, baseline characteristics of SP were very similar to those of ITT (Supplementary Materials—Table S2).
Overall, 25,878 SMBG tests were available for 152 subjects in the 6 months preceding the first prescription of iGlarLixi, and 25,033 SMBG tests were available for 171 subjects during 6 months of follow-up.
Incidence rates of BG ≤ 70 and < 54 mg/mL are presented in Table 4. A 75% risk reduction with respect to the 6 months before iGlarLixi initiation was documented both for BG events ≤ 70 (IRR 0.25; 95% CI 0.06, 0.97) and < 54 (IRR 0.23; 95% CI 0.05, 0.98) mg/mL.
No severe hypoglycemic episodes were reported in EMRs.
Discontinuations
Patients discontinuing iGlarLixi during 6 months were 122 (18.1%) (Table 1).
After discontinuation, basal insulin was prescribed to 108 (88.5%) patients according to the following schemes: basal oral therapy in 34.4% of cases, basal bolus (± OHAs) in 41.8%—mainly with three daily injections (76.4% of treated) (Table 1 and Supplementary Materials—Table S1).
Overall, GLP1-RA was maintained in 29 (23.8%) patients; among them, 19.7% in free or fixed combination with basal insulin and 4.1% in association with OHAs only (Table 1 and Supplementary Materials—Table S1).
Site Questionnaire
The three main reasons for initiating iGlarLixi resulted in better control (70.8%), simplification of therapy (66.7%), and better adherence (50%) (Supplementary Materials—Fig. S1).
Discussion
Main Findings
This real-world study documented that initiating iGlarLixi in adult patients with T2DM was associated with an average reduction in HbA1c of −0.7%; however, a reduction of −0.9% was observed when the drug was used in combination with metformin ± SGLT2i. iGlarLixi was proven to increase the proportion of patients with HbA1c < 7% (< 53 mmol/mol) and < 8% (< 64 mmol/mol), without weight gain and with a significant reduction in the rate of hypoglycemia as compared with the 6-month period before starting iGlarLixi, with no severe episodes observed during an average of 6 months follow-up. This was also due to a group of patients (21.9%) treated with basal bolus before initiation of a FRC. However, the study also highlighted the need for improving treatment appropriateness and the issue of the insufficient dose escalation. In fact, 32.4% of patients used drugs other than metformin and SLGT2i in combination with iGlarLixi, and iGlarLixi was only partially titrated at T6.
Comparisons with Existing Knowledge
RCTs clearly documented benefits of iGlarLixi on metabolic control and secondary endpoints [7,8,9, 12, 17,18,19]. Particularly, in the LixiLan-L trial, iGlarLixi was compared with iGlar in patients with T2D inadequately controlled on basal insulin with or without up to two OHAs. iGlarLixi showed greater reductions in HbA1c from baseline (−1.1%) compared with IGlar alone. HbA1c < 7.0% (53 mmol/mol) was achieved in 55% of patients on iGlarLixi. Mean body weight decreased (−0.7 kg) with iGlarLixi, while it increased with IGlar. Rates of documented symptomatic hypoglycemia (≤ 70 mg/dL) were comparable between groups, whereas mild gastrointestinal adverse effects were very low but more frequent with iGlarLixi [8]. The LixiLan-G trial compared switching to iGlarLixi versus continuing prior GLP-1 RA. iGlarLixi reduced HbA1c (−1.1% versus −0.4%) more than continued GLP1-RA therapy, and a higher proportion of patients on iGlarLixi achieved HbA1c < 7% (62% versus 26%) [10].
A post-hoc analysis from the LixiLan-L trial showed that residual hyperglycemia, which was defined as HbA1c ≥ 7.0% despite fasting plasma glucose of < 140 mg/dL, can be better addressed with iGlarLixi than iGlar alone. In fact, after 30 weeks, the proportion of patients with residual hyperglycemia declined to 23.8% in the iGlarLixi versus 47.1% in the iGlar arm (p < 0.0001) [19]. Furthermore, a subanalysis of LixiLan-L documented that iGlarLixi lowered HbA1c more than iGlar, regardless of T2D duration, with benefit retained even among patients with the longest T2D duration who can be challenging to treat because of progressive loss of β-cell function [18]. A recent post-hoc, propensity-score-matched analysis from the LixiLan-L trial data showed that, compared with basal-bolus regimen, treatment with iGlarLixi was associated with statistically significant greater improvements in HbA1c (−1.02% versus −0.74%; p < 0.0001), body weight (−0.62 kg versus + 0.70 kg; p < 0.0001) and fewer episodes of overall (0.68 versus 1.94 events per patient-year) and nocturnal (0.13 versus 0.57 events per patient-year) hypoglycemia [19].
On the other hand, the Solimix study showed that intensifying BOT through iGlarLixi versus twice-daily premix insulin was associated with better glycemic control, fewer daily injections, weight benefit, and less hypoglycemia [11, 12].
Finally, the randomized, pragmatic, pilot BEYOND study provided evidence that it is possible and safe to switch from a basal-bolus regimen to a FRC. In fact, after 6 months, a similar HbA1c reduction (−0.6%) was obtained in patients intensifying previous basal-bolus therapy versus those switching to FRC (iGlarLixi or iDegLira), with lower insulin doses (62 U versus 27 U), fewer daily injections, and less hypoglycemia (17.8% versus 7.8% of patients with at least one episode) in the FRC group than in the basal-bolus group [20].
Existing literature also showed that iGlarLixi is effective irrespective of age, diabetes duration, and ethnicity [18,19,20], and that iGlarLixi has an enhanced gastrointestinal tolerability compared with the single‐agent GLP1-RA [18, 19, 21,22,23,24].
In general, data from RCTs document higher HbA1c reduction and number of patients at target compared with ENSURE data, possibly owing to undertitration of the FRC and/or inappropriate use of concomitant OHAs. The BEYOND pragmatic trial instead seems to show comparable effectiveness [20].
Implications for Research and Clinical Practice
Effectiveness and safety of iGlarLixi were clearly documented in real-world T2D patients, but to achieve results comparable to those of RCTs, there is a need to improve treatment appropriateness.
Similar to the real-world RESTORE program on basal insulin [14,15,16], a message that can be drawn from the results of this study is that the achievement of metabolic targets does not depend only on the initiation of a new drug, but also on its appropriate use and titration. In fact, there is an urgent need to limit association of inadequate OHAs such as sulfonylureas or short-acting insulin and reduce clinical inertia by optimizing iGlarLixi dosing.
In line with the findings from the BEYOND trial, the ENSURE study, where a subgroup of patients was previously treated with complex regimens such as basal bolus schemes, suggests that a simplification of intensive insulin treatment is possible and even advisable for patients with T2D [20]. FRC can reduce hypoglycemia risk and number of daily injections, thus improving medication adherence and quality of life [24, 25].
Changes in time-in-range and risk of diabetes-related complications associated with the use of iGlarLixi represent relevant endpoints to be increasingly investigated in future studies [26, 27].
Finally, from a methodological standpoint, this study confirms the importance of the secondary use of preexisting data for clinical research purposes. In this respect, the Italian network of diabetes centers adopting the same EMR system represents a unique opportunity to conduct large, RWE studies. However, educational activities should be implemented to increase the culture of the download and systematic revision of SMBG data in EMRs to improve patient care and increase the robustness of safety endpoints.
Strengths and Limitations
The study has strengths and limitations. This is one of the first RWE studies available on effectiveness and safety of iGlarLixi in T2D. Another strength is the generalizability of the results (large sample of patients with T2D routinely cared for by centers located in different areas of Italy) and the efficient use of EMR data for research purposes. Finally, in this study the rate of hypoglycemic episodes was based on documented events, with the caveat that only a subset of patients had their SMBG downloaded in EMR.
The main limitation of this retrospective analysis was the lack of a control group; nevertheless, the study had descriptive purposes regarding patient profile and real-world outcomes of iGlarLixi therapy. Moreover, the low proportion of ITT patients with a follow-up HbA1c value (partly due to the COVID-19 pandemic that limited access to visits and laboratory tests) represented another limitation; therefore, EP, not ITT, population was used for the evaluation of the primary endpoint. Furthermore, study populations showed some differences in baseline characteristics in terms of diabetes duration, metabolic control, and use of previous and concomitant drugs that could have had an impact on effectiveness analyses. Another limitation was the lack of information on SMBG tests for a large proportion of patients, although the performed analysis is robust owing to the large number of SMBG tests considered. Finally, no information on adverse events or reasons for iGlarLixi discontinuation could be derived from EMRs.
Conclusions
The study documented the effectiveness and safety of iGlarLixi in adults with T2D mainly pretreated with basal or basal-bolus regimens. Intensifying or de-intensifying previous insulin regimens by initiating iGlarLixi was associated with significant improvements in glycemic control, without weight gain and with a significant reduction in the rate of hypoglycemia, with no severe episodes observed during an average of 6 months follow-up. This simplification strategy with FRC may work, in terms of significant and clinically relevant reduction of HbA1c, in many patients with T2D who maintain their results for at least 6 months.
However, there is a need to improve treatment appropriateness. The study highlighted some inappropriate use of this FRC related to the type of the combined OHAs. The insufficient dose escalation is a known and common feature associated with the use of insulin in Italy. This undertitration in real-life conditions concurred to partly limit the effectiveness of the product, especially in patients who were switched from other basal insulin to iGlarLixi. Further research of the effect of irGlarLixi in clinical practice compared with other intensification approaches is warranted.
References
Castellana M, Cignarelli A, Brescia F, Laviola L, Giorgino F. GLP-1 receptor agonist added to insulin versus basal-plus or basal-bolus insulin therapy in type 2 diabetes: a systematic review and meta-analysis. Diabetes Metab Res Rev. 2019;35: e3082. https://doi.org/10.1002/dmrr.3082.
Blonde L, Anderson JE, Chava P, Dendy JA. Rationale for a titratable fixed-ratio co-formulation of a basal insulin analog and a glucagon-like peptide 1 receptor agonist in patients with type 2 diabetes. Curr Med Res Opin. 2019;35:793–804. https://doi.org/10.1080/03007995.2018.1541790.
Gerstein HC, Bosch J, Dagenais GR, et al. Basal insulin and cardiovascular and other outcomes in dysglycemia. N Engl J Med. 2012;367:319–28. https://doi.org/10.1056/NEJMoa1203858.
Becker RH, Stechl J, Steinstraesser A, Golor G, Pellissier F. Lixisenatide reduces postprandial hyperglycaemia via gastrostatic and insulinotropic effects. Diabetes Metab Res Rev. 2015;31:610–8. https://doi.org/10.1002/dmrr.2647.
Gautier T, Umpierrez G, Renard E, Kovatchev B. The differential and combined action of insulin glargine and lixisenatide on the fasting and postprandial components of glucose control. J Diabetes Sci Technol. 2021;15:371–6. https://doi.org/10.1177/1932296819891170.
Aronson R, Umpierrez G, Stager W, Kovatchev B. Insulin glargine/lixisenatide fixed-ratio combination improves glycaemic variability and control without increasing hypoglycaemia. Diabetes Obes Metab. 2019;21:726–31. https://doi.org/10.1111/dom.13580 (Epub 2018 Dec 10).
Aroda VR, Rosenstock J, Wysham C, et al. Efficacy and safety of LixiLan, a titratable fixed-ratio combination of insulin glargine plus lixisenatide in type 2 diabetes inadequately controlled on basal insulin and metformin: the LixiLan-L randomized trial. Diabetes Care. 2017;40:809. https://doi.org/10.2337/dc17-er06d.
Rosenstock J, Aronson R, Grunberger G, et al. Benefits of LixiLan, a titratable fixed-ratio combination of insulin glargine plus lixisenatide, versus insulin glargine and lixisenatide monocomponents in type 2 diabetes inadequately controlled on oral agents: the LixiLan-O randomized trial. Diabetes Care. 2016;39:2026–35. https://doi.org/10.2337/dc16-0917.
Blonde L, Rosenstock J, Del Prato S, et al. Switching to iGlarLixi versus continuing daily or weekly GLP-1 RA in type 2 diabetes inadequately controlled by GLP-1 RA and oral antihyperglycemic therapy: the LixiLan-G randomized clinical trial. Diabetes Care. 2019;42:2108–16. https://doi.org/10.2337/dc19-1357 (Epub 2019 Sep 17 PMID: 31530665).
Trujillo JM, Roberts M, Dex T, Chao J, White J, LaSalle J. Low incidence of gastrointestinal adverse events over time with a fixed-ratio combination of insulin glargine and lixisenatide versus lixisenatide alone. Diabetes Obes Metab. 2018;20:2690–4. https://doi.org/10.1111/dom.13444.
McCrimmon RJ, Al Sifri S, Emral R, et al. Advancing therapy with iGlarLixi versus premix BIAsp 30 in basal insulin-treated type 2 diabetes: design and baseline characteristics of the SoliMix randomized controlled trial. Diabetes Obes Metab. 2021;23:1221–31. https://doi.org/10.1111/dom.14354.
Rosenstock J, Emral R, Sauque-Reyna L, et al. Advancing therapy in suboptimally controlled basal insulin-treated type 2 diabetes: clinical outcomes with iGlarLixi versus premix BIAsp 30 in the SoliMix randomized controlled trial. Diabetes Care. 2021;44:2361–70. https://doi.org/10.2337/dc21-0393.
American Diabetes Association. 6. Glycemic targets: standards of medical care in diabetes-2021. Diabetes Care 2021;44:S73–S84. https://doi.org/10.2337/dc21-S006.
Laviola L, Porcellati F, Bruttomesso D, Larosa M, Rossi MC, Nicolucci A, RESTORE-1 Study Group. Comparative effectiveness of switching from first-generation basal insulin to glargine 300 U/ml or degludec 100 U/ml in type 1 diabetes: the RESTORE-1 study. Diabetes Ther. 2021;12:509–25. https://doi.org/10.1007/s13300-020-00982-z.
Fadini GP, Buzzetti R, Nicolucci A, Larosa M, Rossi MC, Cucinotta D, RESTORE-2 Study Group. Comparative effectiveness and safety of glargine 300 U/mL versus degludec 100 U/mL in insulin-naïve patients with type 2 diabetes. A multicenter retrospective real-world study (RESTORE-2 NAIVE STUDY). Acta Diabetol. 2022;59(10):1317–30. https://doi.org/10.1007/s00592-022-01925-9.
Buzzetti R, Fadini GP, Nicolucci A, Larosa M, Rossi MC, Cucinotta D, RESTORE-2 Study Group. Comparative effectiveness of glargine 300 U/mL vs. degludec 100 U/mL in patients with type 2 diabetes switching from 1° generation basal insulins. Nutr Metab Cardiovasc Dis. 2022;32(9):2255–63. https://doi.org/10.1016/j.numecd.2022.06.003.
Davies MJ, Russell-Jones D, Barber TM, et al. Glycaemic benefit of iGlarLixi in insulin-naive type 2 diabetes patients with high HbA1c or those with inadequate glycaemic control on two oral antihyperglycaemic drugs in the LixiLan-O randomized trial. Diabetes Obes Metab. 2019;21:1967–72. https://doi.org/10.1111/dom.13791.
Blonde L, Berard L, Saremi A, Huang Y, Aroda VR, Raccah D. Fixed-ratio combination of insulin and GLP-1 RA in patients with longstanding type 2 diabetes: a subanalysis of LixiLan-L. Diabetes Ther. 2020;11:1007–15. https://doi.org/10.1007/s13300-020-00797-y.
Morea N, Retnakaran R, Vidal J, et al. iGlarLixi effectively reduces residual hyperglycaemia in patients with type 2 diabetes on basal insulin: a post hoc analysis from the LixiLan-L study. Diabetes Obes Metab. 2020;22:1683–9. https://doi.org/10.1111/dom.14077.
Giugliano D, Longo M, Caruso P, et al. Feasibility of simplification from a basal-bolus insulin regimen to a fixed-ratio formulation of basal insulin plus a GLP-1RA or to basal insulin plus an SGLT2 inhibitor: BEYOND, a randomized, pragmatic trial. Diabetes Care. 2021;44:1353–60. https://doi.org/10.2337/dc20-2623.
Dailey G, Bajaj HS, Dex T, Groleau M, Stager W, Vinik A. Post hoc efficacy and safety analysis of insulin glargine/lixisenatide fixed-ratio combination in North American patients compared with the rest of world. BMJ Open Diabetes Res Care. 2019;7: e000581. https://doi.org/10.1136/bmjdrc-2018-000581 (PMID: 31114694; PMCID: PMC6501856).
Del Prato S, Frias JP, Blonde L, et al. Impact of disease duration and β-cell reserve on the efficacy of switching to iGlarLixi in adults with type 2 diabetes on glucagon-like peptide-1 receptor agonist therapy: exploratory analyses from the LixiLan-G trial. Diabetes Obes Metab. 2020;22:1567–76. https://doi.org/10.1111/dom.14068 (Epub 2020 May 28. PMID: 32323437; PMCID: PMC7754453).
Handelsman Y, Chovanes C, Dex T, et al. Efficacy and safety of insulin glargine/lixisenatide (iGlarLixi) fixed-ratio combination in older adults with type 2 diabetes. J Diabetes Complications. 2019;33:236–42. https://doi.org/10.1016/j.jdiacomp.2018.11.009 (Epub 2018 Nov 30 PMID: 30600136).
Rayner CK, Wu T, Aroda VR, et al. Gastrointestinal adverse events with insulin glargine/lixisenatide fixed-ratio combination versus glucagon-like peptide-1 receptor agonists in people with type 2 diabetes mellitus: a network meta-analysis. Diabetes Obes Metab. 2021;23:136–46. https://doi.org/10.1111/dom.14202.
Jude EB, Malecki MT, Gomez Huelgas R, et al. Expert panel guidance and narrative review of treatment simplification of complex insulin regimens to improve outcomes in type 2 diabetes. Diabetes Ther. 2022;13:619–34. https://doi.org/10.1007/s13300-022-01222-2.
Bajaj HS, Chu L, Bansal N, et al. Randomized comparison of initiating the fixed-ratio combination of iGlarLixi or biosimilar insulin glargine together with gliclazide in South Asian origin participants with type 2 diabetes: VARIATION 2 SA Trial. Can J Diabetes. 2022;S1499–2671(22):00015–6. https://doi.org/10.1016/j.jcjd.2022.02.003.
Shao H, Kianmehr H, Guo J, Li P, Fonseca V, Shi L. Efficacy of iGlarLixi on 5-year risk of diabetes-related complications: a simulation study. J Diabetes Complications. 2022;36: 108132. https://doi.org/10.1016/j.jdiacomp.2022.108132.
Acknowledgements
The authors thank the participating centers and all the collaborators from SANOFI, CORESEARCH and OPIS involved in the study. ENSURE Study group: Participating centers (in alphabetical order by town): Enrico Gabellieri, Alessandria; Valeria Montani, Atri (TE); Alberto Marangoni, Bassano del Grappa (VI), Giovanni Saitta, Capo d'Orlando (ME), Andrea Del Buono, Cellole (CE); Stefano Ettori, Chiari (BS); Anita Minnucci, Chieti; Roberto Norgiolini, Città di Castello (PG), Pasquale Maiellaro, Foggia; Graziano Di Cianni, Silvia Del Guerra, Livorno; Gabriele Brandoni, Macerata; Giovanna Gregori, Massa Carrara; Di Benedetto Antonino, Messina; Piermarco Piatti, Milano; Annamaria Terracciano, Maria Popolizio, Mondragone (CE); Paola Del Sindaco, Chiara Di Loreto, Perugia; Roberto Anichini, Pistoia; D'Angelo Paola, Silvia Carletti, Roma; Carmela Vinci, San Donà Piave (VE); Francesco Calcaterra, Schio (VI); Vincenzo Fiore, Tivoli (RM); Riccardo Candido, Trieste; Monica Modugno, Triggiano/Gioia del Colle (BA); Paola Sarnelli, Viterbo. Additional Assistance: CORESEARCH (Pescara, Italy) was the Clinical Research Organization involved in the data management (Giuseppe Prosperini, Michele Sacco), statistical analysis (Giuseppe Lucisano, Antonio Nicolucci), and medical writing (Maria Chiara Rossi, Antonio Nicolucci) of the study. Medical writing was funded by Sanofi S.r.l. Meteda (San Benedetto, Italy) developed the software for the data extraction. OPIS (Milano, Italy) was the Clinical Research Organization involved in regulatory activities of the study.
Funding
The study was funded by Sanofi S.r.l., Milan, Italy. Rapid Service Fee and Open Access fee were supported by Sanofi S.r.l.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authors’ Contributions
Riccardo Candido, Monica Modugno, Enrico Gabellieri, Monica Larosa, and Antonio Nicolucci made substantial contributions to the conception and design of the work. Riccardo Candido, Monica Modugno, and Enrico Gabellieri contributed to the data collection. Maria Chiara Rossi and Antonio Nicolucci conducted the statistical analyses and drafted the article. All authors revised the article critically for important intellectual content. All authors approved the final version to be published. All authors agreed all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article.
Disclosures
Riccardo Candido has received consultancy fees from Boehringer Ingelheim, Eli-Lilly, Novo Nordisk, Astra-Zeneca, Sanofi-Aventis, Roche Diabetes Care; speaking fees from Astra Zeneca, Boehringer Ingelheim, Eli-Lilly, Novo Nordisk, Sanofi-Aventis, Mundipharma Pharmaceutical, Abbott, MSD, Neopharmed Gentili, Menarini, Essex Italia, Ascensia Diabetes, Monica Modugno has received consultancy fees from Novo Nordisk, Astra-Zeneca, Sanofi-Aventis, and speaking fees from Boehringer Ingelheim, Eli-Lilly, Novo Nordisk, Sanofi-Aventis, Mundipharma Pharmaceutical, Neopharmed Gentili. Monica Larosa is an employee of Sanofi and may hold shares and/or stock options in the company. Maria Chiara Rossi and Antonio Nicolucci have received funding for research from Sanofi, NovoNordisk, Alfasigma, Artsana, AstraZeneca, Johnson&Johnson, Medtronic, Shionogi, SOBI, Meteda and Theras. Enrico Gabellieri has received consultancy fees from Novo Nordisk, Sanofi-Aventis; speaking fees from Astra Zeneca, Boehringer Ingelheim, Eli-Lilly, Novo Nordisk, Sanofi-Aventis, Abbott, Neopharmed Gentili.
Compliance with Ethics Guideline
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. The study protocol was approved by all local ethics committees of the participating centers (Supplementary Materials—ECs list). Informed consent was obtained from all patients for being included in the study.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Qualified researchers may request access to patient level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient level data will be anonymized and study documents will be redacted to protect the privacy of our trial participants. Further details on Sanofi data sharing criteria, eligible studies, and process for requesting access can be found at: https://www.vivli.org/.
Author information
Authors and Affiliations
Consortia
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Candido, R., Modugno, M., Larosa, M. et al. Effectiveness, Safety, and Appropriateness in the Use of the Fixed-Ratio Combination of Insulin Glargine and Lixisenatide in Type 2 Diabetes: The ENSURE Retrospective Real-World Study. Diabetes Ther 14, 77–92 (2023). https://doi.org/10.1007/s13300-022-01328-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-022-01328-7