Abstract
Several studies investigating the association between heat shock protein beta-1 (HSPB1) polymorphisms and radiation-induced damage in lung cancer patients administrated with radiotherapy have derived conflicting results. This meta-analysis aimed to assess the association between the HSPB1 genes’ (rs2868370 and rs2868371) polymorphisms and the risk of radiation-induced damage in lung cancer patients. After an electronic literature search, four articles including six studies were found to be eligible for this meta-analysis. No association was observed between rs2868370 genotypes and radiation-induced damage risk. However, rs2868371 showed a statistically increased risk of radiation-induced damage under CC vs. CG/GG model (OR = 1.59, 95 % CI = 1.10–2.29). Subgroup analysis by ethnicity showed that the genotypes of rs2868371 were also associated with a significantly increased risk of radiation-induced damage in CC vs. CG/GG model (OR = 1.86, 95 % CI = 1.21–2.83) among mixed ethnicities which are mainly comprised of white people. When the data was stratified by organ-damaged, a significant association was only observed in the esophagus group (OR = 2.94, 95 % CI = 1.35–6.37, for CC vs. CG/GG model). In conclusion, the present study demonstrated that the rs2868371 genotypes of HSPB1 might be associated with radiation-induced esophagus damage risk, especially in Caucasians but not in the Asian population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lung cancer has been one of the most prevalent cancers and the primary cause of cancer-related death in the world [1–3]. To improve survival of lung cancer patients, various treatment options are applied on them. Radiotherapy, with the progress in technique, attributes to better treatment outcome and higher local control rate for stage I nonsmall cell lung cancer [4]. For the extensive-stage small cell lung cancer, radiotherapy was proved to effectively improve long-term survival [5]. Palliative radiotherapy is used widely to relieve symptoms causing from tumors in the patients [6]. Although radiotherapy is effective to control lung cancer, notable toxicity induced by radiation could not be ignored. Lung and esophagus are two common organs easily damaged by the radiation, limiting the wide administration of radiotherapy [7, 8]. Radiation-induced lung damage includes two main stages, radiation pneumonitis as the early stage and radiation fibrosis as the advanced stage [9]. Currently, the risk assessment of radiation-induced pneumonitis relies on some factors including usage of chemotherapy, gross tumor volume, radiation fraction schedule, radiation dose, and patients’ status [10, 11]. During radiotherapy for nonsmall cell lung cancer, radiation dose distributed to the esophagus and the esophagus volume exposed to radiation are both risk factors associated with radiation-induced esophageal toxicity [12–14]. However, lung cancer patients with similar chemotherapy plan, tumor volume, radiation dose and schedule and body status always result in huge differences in radiation-induced damage in clinical practice. So, it is possible that some individual biology characteristics may play a critical role in patients’ response to radiation.
Expression of some cytokines is always associated with radiotherapy. Tests have been done to identify specific cytokines as predicting biomarkers for radiation-induced damage risk. Heat shock protein (HSP) 27, encoded by heat shock protein beta-1(HSPB1), is an adenosine triphosphate-independent molecular chaperone which can facilitate the repair or degradation of damaged proteins to protect against protein aggregation in stressed cells [15, 16]. Furthermore, HSP27 can relieve the toxic effects of oxidized proteins and enhance the antioxidant defense capacity of cells [7, 17]. The characteristic of HSP27 may be of particular importance in the process of radiotherapy, because reactive oxygen species are important in the induction of apoptosis of cells exposed to radiation [18]. It has been suggested that HSP27 works as a radioresistant protein and may be involved in radioresistance in nasopharyngeal cancer [19]. As HSP27 expression is under control of HSPB1, located on chromosome 7 at q11.23, containing three exons and two introns, HSPB1 genotypes may be connected with the function of HSP27 during the radiation-induced damage [20].
Some studies have been performed to investigate the relationship between the risk of radiation-induced damage and HSPB1 genetic polymorphisms among patients with lung cancer, but the results are conflicting [7, 20–22]. Patients from different ethnicities were analyzed, and in particular, some of the studies had a small sample size, so some results could not be replicated. Therefore, we, using all published data, take a meta-analysis to increase the statistical power and confirm whether the HSPB1 rs2868370 or rs2868371 genotypes increase the risk of radiation-induced damage.
Materials and methods
Literature search strategy
A systematic electronic literature search of the PubMed, EMBASE, Web of Science, BIOSIS Previews databases, China National Knowledge Infrastructure (CNKI), and Chinese Biomedical database (CBM) for articles published until 18 August 2015 was performed to identify all of the relevant studies. The search strategy included the following keywords: (“hspb1” or “Heat shock protein”) and (rs2868370 or rs2868371 or “single nucleotide polymorphism” OR “SNP” OR “genetic variation” OR “genetic polymorphism”) AND (“lung cancer” OR “lung neoplasms” OR “lung tumor”). Additionally, references of relevant articles we retrieved were also checked to identify other potential eligible publications. If the same case series were used in more than one article, we only select one of them. The languages were limited to English and Chinese.
Selection criteria
The inclusion criteria were as follows: (1) studies exploring the HSPB1 rs2868370 or rs2868371 polymorphisms and the risk of radiation-induced damage in lung cancer patients; (2) using the case-control design; and (3) being able for examining an odds ratio (OR) and 95 % confidence interval (CI). The exclusion criteria were as follows: (1) some essential information was not reported; (2) the reviews, abstracts, and comments; and (3) same cases were reported in two or more papers.
Data extraction
Data extraction from all the eligible publications were conducted by two investigators independently according to the selection criteria listed above, the consensus was achieved for all the data. The following information was extracted: first author’s name, year of publication, country, ethnicity, organ-damaged, and total number of cases and controls with the HSPB1 rs2868370 or rs2868371 polymorphisms.
Statistical analysis
The strength of the correlation between the HSPB1 polymorphisms and the risk of radiation-induced damage was measured by ORs with 95 % CIs. To evaluate the association of rs2868370 and radiation-induced damage, pooled ORs of GG vs. AG/AA contrast model was calculated. For rs2868371, the CC vs. GG, CC vs. CG/GG, GG vs. CG/CC, and CG vs. CC models were performed. Subgroup analysis was conducted according to ethnicity or organ-damaged. The Hardy-Weinberg equilibrium (HWE) in control groups were tested using the chi-squared (χ 2) test when available. Heterogeneity was evaluated by the χ 2 test or Fisher exact test. When P < 0.10 or I 2 > 50 % indicated an obvious of the between-study heterogeneity, the random-effects model was used. Otherwise, the fixed-effects model was used. Begg’s funnel plot and Egger’s test were used to assess the publication bias, P < 0.05 was considered statistically significant. All analyses were performed using the STATA package version 11.0 program (Stata Corporation, College Station, TX, USA).
Results
One hundred twenty-five articles were identified at the initial search. Through screening the titles, abstracts, reading the full articles, and removing the replications, four eligible articles (three in English and one in Chinese) were included in this meta-analysis. The study selection process is described in Fig. 1. Because two articles [7, 21] contained two studies respectively, and each study has different samples and different radiation technique compared with another, so the total studies in the meta-analysis is 6. Details of each study estimated are summarized in Tables 1 and 2.
Four case-control studies [7, 21] with 150 cases and 410 controls were evaluated for the association between rs2868370 polymorphism and radiation-induced damage risk. The summary OR for GG vs. AG/AA was 0.87 (95 % CI 0.56–1.33), which suggested that no association was existed (Fig. 2). As the heterogeneity was not significant among these studies (P > 0.05), so the fix-effects model was used.
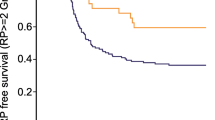
As for the rs2868371 polymorphism, six studies [7, 20–22] including 214 cases and 672 controls were analyzed with fix-effects model. The pooled ORs yielded for the contrast models were 1.59 (95 % CI 1.10–2.29) for CC vs. CG/GG, 0.86 (95 % CI 0.37–1.98) for CC vs. GG, 1.17 (95 % CI 0.68–2.03) for GG vs. CC/CG, and 0.99 (95 % CI 0.43–2.29) for CG vs. CC. These results showed that lung cancer patients with the CC genotype of rs2868371 were associated with a higher risk of suffering from radiation-induced damage compared with patients having CG or GG genotype. However, when stratified by ethnicity, the significant association was not found in Asians, but found in the mixed ethnicities which were mostly comprised of Caucasians [7, 21] (CC vs. CG/GG: OR = 1.86, 95 % CI = 1.21–2.83, Fig. 3a). Subgroup analysis by organ-damaged showed that the significant association existed in esophagus group (CC vs. CG/GG: OR = 2.94, 95 % CI = 1.35–6.37, Fig. 3b). The controls were consistent with HWE in two studies [20, 22] (P > 0.05).
The publication bias was assessed by Begg’s funnel plot and Egger’s test for GG vs. AG/AA (rs2868370) and CC vs. CG/GG (rs2868371) models. As shown in Figs. 4 and 5, the funnel plots appear to be basically symmetric. Additionally, Egger’s test showed no publication bias existing (P > 0.05).
Discussion
Radiotherapy plays an important role in controlling lung cancer progression. However, the adverse effects, some of which are acute or dangerous, existed [9, 23, 24]. Thus, it is necessary finding biomarkers to identify people who are more easily damaged by radiation among lung cancer patients. HSP27 is an important cytokine that can enhance cellular resistance to heat shock, oxidative damage, and inflammatory mediators [25–27]. And, it was also reported that HSP27 participated in the process of radioresistance [28]. Recently, some researchers focused on HSPB1, the gene of HSP27, and found a potential relationship between HSPB1 and radiation-induced damage susceptibility. Thus, as the results remain conflicting, we conducted this meta-analysis. To our knowledge, this is the first meta-analysis investigating the association of HSPB1 and risk of radiation-induced damage.
In this analysis, though no association between rs2868370 gene polymorphism of HSPB1 and the risk of radiation-induced damage was found, the rs2868371 genotypes were proved to be associated with an increased radiation-induced damage (OR = 1.59, 95 % CI = 1.10–2.29 for CC vs. CG/GG). Stratified by ethnicity, we found that the association only existed in the mixed ethnicities in which the white people accounted for more than 78 %. It suggested that the association mainly existed in the Caucasians. Subgrouped by organ-damaged, the significant association was only found in esophagus group. These results imply that HSPB1 genotypes may play a pivotal role in influencing patient susceptibility to radiation-induced damage.
The distribution of single nucleotide polymorphisms (SNP) is well known to vary among different races. It was reported that the most common genotype of the rs2868371 SNP in Caucasian populations was CC [29]; however, it was CG in Chinese [30]. This different distribution of rs2868371 SNP also existed in this meta-analysis. Thus, differences in the genetic backgrounds of various races may explain, at least in part, why the association was only appearing in the mixed ethnicities. One recent study demonstrated that the HSPB1 rs2868371 CC was associated with a poorer overall survival than other genotypes of HSPB1 rs2868371 in NSCLC patients treated with radiotherapy possibly by upregulating the expression of Hsp27 protein [29]. Hsp27 can play antiapoptotic roles by interacting with cytochrome c, Bax, Akt, and other key apoptotic cytokines [31] and stabilize the structure of the cytoskeleton through interaction with tubulin and microfilaments [32] to help cells survive. It was demonstrated that radiotherapy could cause Hsp27 downregulation [33] and low Hsp27 expression increased cell sensitivity to radiation reducing DNA repair capacity [30]. Thus, we hypothesize that the esophagus cells of patients with CC genotype of rs2868371 may have more Hsp27 downregulation than those carrying CG/GG genotype after radiotherapy, and therefore, low level expression of Hsp27 attribute to radiation-induced esophagus damage easily.
Some limitations of this meta-analysis exist. Firstly, though searching from many databases online, relevant studies are not enough and only four articles including six studies are eligible; thus, the bias cannot be ignored. Secondly, some detail information limited in original studies prevents us to do an in-depth and comprehensive analysis. Because of the case numbers of several genotypes are not shown, the HWE in control groups of some studies [7, 21] was not tested and some analysis models were not performed. Thirdly, patients may have more than one organs injured during radiotherapy, but we could not find the clear information in the original studies. All the limitations require us to interpret the results with caution.
Conclusions
Despite the existing limitations, a meta-analysis of the relationship of HSPB1 polymorphisms with the risk of radiation-induced damage is more powerful than any single study. In conclusion, the present study demonstrated that the rs2868371 genotypes of HSPB1 may be associated with radiation-induced damage of esophagus, especially in the Caucasians but not in the Asian population. Future studies investigating the association between the HSPB1 polymorphisms and the susceptibility for radiation-induced damage need to be performed.
References
Herbst RS, Heymach JV, Lippman SM. Lung cancer. N Engl J Med. 2008;359:1367–80.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96.
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90.
Ricardi U, Frezza G, Filippi AR, Badellino S, Levis M, Navarria P, et al. Stereotactic ablative radiotherapy for stage I histologically proven non-small cell lung cancer: an Italian multicenter observational study. Lung Cancer. 2014;84:248–53.
Slotman BJ, van Tinteren H, Praag JO, Knegjens JL, El Sharouni SY, Hatton M, et al. Use of thoracic radiotherapy for extensive stage small-cell lung cancer: a phase 3 randomised controlled trial. Lancet. 2015;385:36–42.
Spencer K, Morris E, Dugdale E, Newsham A, Sebag-Montefiore D, Turner R, et al. 30 day mortality in adult palliative radiotherapy—a retrospective population based study of 14,972 treatment episodes. Radiother Oncol. 2015;115:264–71.
Lopez Guerra JL, Wei Q, Yuan X, Gomez D, Liu Z, Zhuang Y, et al. Functional promoter rs2868371 variant of HSPB1 associates with radiation-induced esophageal toxicity in patients with non-small-cell lung cancer treated with radio(chemo)therapy. Radiother Oncol. 2011;101:271–7.
Madani I, De Ruyck K, Goeminne H, De Neve W, Thierens H, Van Meerbeeck J. Predicting risk of radiation-induced lung injury. J Thorac Oncol. 2007;2:864–74.
Zhang XJ, Sun JG, Sun J, Ming H, Wang XX, Wu L, et al. Prediction of radiation pneumonitis in lung cancer patients: a systematic review. J Cancer Res Clin Oncol. 2012;138:2103–16.
Das SK, Zhou S, Zhang J, Yin FF, Dewhirst MW, Marks LB. Predicting lung radiotherapy-induced pneumonitis using a model combining parametric Lyman probit with nonparametric decision trees. Int J Radiat Oncol Biol Phys. 2007;68:1212–21.
Tsoutsou PG, Koukourakis MI. Radiation pneumonitis and fibrosis: mechanisms underlying its pathogenesis and implications for future research. Int J Radiat Oncol Biol Phys. 2006;66:1281–93.
Wei X, Liu HH, Tucker SL, Liao Z, Hu C, Mohan R, et al. Risk factors for acute esophagitis in non-small-cell lung cancer patients treated with concurrent chemotherapy and three-dimensional conformal radiotherapy. Int J Radiat Oncol Biol Phys. 2006;66:100–7.
Caglar HB, Othus M, Allen AM. Esophagus in-field: a new predictor for esophagitis. Radiother Oncol. 2010;97:48–53.
Ahn SJ, Kahn D, Zhou S, Yu X, Hollis D, Shafman TD, et al. Dosimetric and clinical predictors for radiation-induced esophageal injury. Int J Radiat Oncol Biol Phys. 2005;61:335–47.
Haslbeck M. sHsps and their role in the chaperone network. Cell Mol Life Sci. 2002;59:1649–57.
Parcellier A, Gurbuxani S, Schmitt E, Solary E, Garrido C. Heat shock proteins, cellular chaperones that modulate mitochondrial cell death pathways. Biochem Biophys Res Commun. 2003;304:505–12.
Arrigo AP, Virot S, Chaufour S, Firdaus W, Kretz-Remy C, Diaz-Latoud C. Hsp27 consolidates intracellular redox homeostasis by upholding glutathione in its reduced form and by decreasing iron intracellular levels. Antioxid Redox Signal. 2005;7:414–22.
Pinthus JH, Bryskin I, Trachtenberg J, Lu JP, Singh G, Fridman E, et al. Androgen induces adaptation to oxidative stress in prostate cancer: implications for treatment with radiation therapy. Neoplasia. 2007;9:68–80.
Zhang B, Qu JQ, Xiao L, Yi H, Zhang PF, Li MY, et al. Identification of heat shock protein 27 as a radioresistance-related protein in nasopharyngeal carcinoma cells. J Cancer Res Clin Oncol. 2012;138:2117–25.
Xu F, Han JC, Zhang YJ, Zhang YJ, Liu XC, Qi GB, et al. Associations of LIG4 and HSPB1 genetic polymorphisms with risk of radiation-induced lung injury in lung cancer patients treated with radiotherapy. Biomed Res Int. 2015;2015:860373.
Pang Q, Wei Q, Xu T, Yuan X, Lopez Guerra JL, Levy LB, et al. Functional promoter variant rs2868371 of HSPB1 is associated with risk of radiation pneumonitis after chemoradiation for non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 2013;85:1332–9.
Liu D, Sun CY, Zhao YX, Bai L. Relationship between genetic single-nucleotide polymorphisms of LIG4 and HSPB1 and the radiation-induced lung injury in lung cancer patients. J Radiat Res Radiat Proc. 2015;33:25–30.
Shaikh T, Turaka A. Predictors and management of chest wall toxicity after lung stereotactic body radiotherapy. Cancer Treat Rev. 2014;40:1215–20.
Eldabaje R, Le DL, Huang W, Yang LX. Radiation-associated cardiac injury. Anticancer Res. 2015;35:2487–92.
Charette SJ, Lavoie JN, Lambert H, Landry J, et al. Inhibition of Daxx-mediated apoptosis by heat shock protein 27. Mol Cell Biol. 2000;20:7602–12.
Wu R, Zhao YH, Plopper CG, Chang MM, Chmiel K, Cross JJ, et al. Differential expression of stress proteins in nonhuman primate lung and conducting airway after ozone exposure. Am J Physiol. 1999;277(3 Pt 1):L511–22.
Merendino AM, Paul C, Vignola AM, Costa MA, Melis M, Chiappara G, et al. Heat shock protein-27 protects human bronchial epithelial cells against oxidative stress-mediated apoptosis: possible implication in asthma. Cell Stress Chaperones. 2002;7:269–80.
Choi SH, Lee YJ, Seo WD, Lee HJ, Nam JW, Lee YJ, et al. Altered cross-linking of HSP27 by zerumbone as a novel strategy for overcoming HSP27-mediated radioresistance. Int J Radiat Oncol Biol Phys. 2011;79:1196–205.
Xu T, Wei Q, Lopez Guerra JL, Wang LE, Liu Z, Gomez D, et al. HSPB1 gene polymorphisms predict risk of mortality for US patients after radio(chemo)therapy for non-small cell lung cancer. Int J Radiat Oncol Biol Phys. 2012;84(2):e229–35.
Guo H, Bai Y, Xu P, Hu Z, Liu L, Wang F, et al. Functional promoter -1271G > C variant of HSPB1 predicts lung cancer risk and survival. J Clin Oncol. 2010;28:1928–35.
Concannon CG, Gorman AM, Samali A. On the role of Hsp27 in regulating apoptosis. Apoptosis. 2003;8:61–70.
Pivovarova AV, Chebotareva NA, Chernik IS, Gusev NB, Levitsky DI. Small heat shock protein Hsp27 prevents heat-induced aggregation of F-actin by forming soluble complexes with denatured actin. FEBS J. 2007;274:5937–48.
Guisasola MC, Calvo F, Marcos P, Simon I, Villanueva FJ, Andres E, et al. Peripheral leukocyte response to oncological radiotherapy: expression of heat shock proteins. Int J Radiat Biol. 2006;82:171–9.
Acknowledgments
This work was supported in part by financial support (to Dr. J. Shu) with the Grant for the General Item from the Natural Science Foundation of Anhui Province, China (1308085MH141), the Grant for the Key Item of Provincial Research of Natural Science in Anhui Universities from the Department of Education of Anhui Province, China (KJ2011A173), the Grant from Anhui Medical University (2010xkj115), and the Research Grant for Outstanding Talents from the Fourth Affiliated Hospital of Anhui Medical University, China (2009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Li, X., Xu, S., Cheng, Y. et al. HSPB1 polymorphisms might be associated with radiation-induced damage risk in lung cancer patients treated with radiotherapy. Tumor Biol. 37, 5743–5749 (2016). https://doi.org/10.1007/s13277-016-4959-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-4959-4