Abstract
The aim of this study was to evaluate for the first time in the literature the role of HE4, at primary diagnosis, compared to CA125 as an indicator of endometrial cancer (EC) recurrence. Our study is a retrospective analysis of 252 EC patients treated, between January 2009 and July 2013, at the Division of Gynaecologic Oncology of Campus Bio-Medico University of Rome. Thirty-seven patients experienced recurrence. Median follow-up was 38 months. HE4 and CA125 levels were analyzed at primary diagnosis, during follow-up and either after histological or radiological confirmation of recurrent disease or at last registered visit, when patients returned to our Department with no evidence of recurrent disease. A statistically significant difference was observed between HE4 values at primary diagnosis and at recurrence, respectively, comparing recurrent and non-recurrent patients (p < 0.05), while CA125 values resulted not statistically significant (p = 0.08) at each time point. Considering the poor specificity of HE4 at threshold of 70 pmol/L at primary diagnosis, in our cohort of patients, we found out that HE4 cut-off of 201.3 pmol/L is able to correctly classify patients at high or low risk of EC recurrence, with a sensitivity of 80 % and a specificity of 91 % (PPV = 90.3 % and NPV = 90.8 %). In particular, HE4 performance improves in cases of endometrioid histotype. HE4 levels at primary diagnosis correlate with an increased risk of EC recurrence, particularly in cases of endometrioid histotype, and they may help to recognize patients who may need a more intensive follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Endometrial cancer (EC) represents the most common malignancy of the female genital tract in the USA and the fourth most common cancer among women in Western countries, with more than 280,000 cases occurring annually worldwide [1].
Eighty percent of EC patients present with early stage, low-grade endometrioid EC, commonly referred to as type 1. Approximately 20 % of patients are diagnosed with high-risk histological subtypes (serous, clear cell, or high-grade endometrioid), commonly referred to as type II EC [1].
However, data show that the mortality rate for uterine cancer has been increasing more rapidly than the incidence rate [2], probably due to an increased rate of advanced-stage cancers, high-risk histologies (e.g., serous adenocarcinomas) and patients being diagnosed at an older age.
Therefore, 13–17 % of women will develop recurrent disease, generally within 3 years of primary treatment [3, 4].
Actually, after primary treatment, EC patients are monitored according to different follow-up schedules, considering that, according to international guidelines, there is not any consensus about surveillance strategies and, up to now, the value of intensive surveillance has not been yet demonstrated in this disease [3, 5, 6].
On the other hand, there are no specific tumor markers for endometrial cancer management. Several authors have reported that high CA125 serum levels correlate with the presence of extra uterine disease and advanced cases, and they are often falsely elevated in disease-free EC patients submitted to radiotherapy [7].
In the last years, a growing interest about the role of HE4 in EC has been observed. In fact, HE4 serum levels appear to be increased in EC patients compared to healthy controls with a good level of sensitivity and specificity and to associate to myometrial invasion and poor prognosis [8–18].
Up to now in the literature, there are no papers on HE4’s role at primary diagnosis in predicting EC recurrence. In fact, there is only one study that evaluated HE4 values during clinical follow-up and demonstrated a role of this new marker in identifying recurrent disease, particularly in patients with endometrioid histology [19].
Therefore, the primary aim of this study was to evaluate the possible role of HE4 at primary diagnosis, compared to CA125, as an indicator of recurrence.
Secondary aim was to identify an ideal preoperative cut-off of HE4 able to correctly classify patients at high or low risk of EC recurrence.
Materials and methods
All patients with complete staged EC (according to 2011 National Comprehensive Cancer Network, NCCN, guidelines) and referred to the Division of Gynaecologic Oncology of Campus Bio-Medico University of Rome, were reviewed and considered for study recruitment. Inclusion criteria for enrollment were as follows: (1) aged between 18 and 80 years and (2) Eastern Cooperative Oncology Group performance status 0–2 according to World Health Organization (WHO) criteria.
Exclusion criteria included the following: (1) presence of a secondary malignancy, (2) concomitant benign and/or malignant adnexal pathologies, (3) abnormal renal and/or hepatic functions, and (4) at least 24 months of follow-up.
To classify EC patients, we adopted the 2009-FIGO staging and the WHO histological classification, dividing histotypes into endometrioid adenocarcinoma (variants: with squamous differentiation, villoglandular, secretory, with ciliated cells) and non-endometrioid adenocarcinomas (mucinous carcinoma, serous carcinoma, clear-cell carcinoma, mixed carcinoma, squamous-cell carcinoma, transitional-cell carcinoma, small-cell carcinoma, and undifferentiated carcinoma) [20].
As reported in literature, histological grading (G1, G2, and G3) applied only to endometrioid carcinomas; non-endometrioid carcinomas, instead, were considered as G3 high grade, by definition [20].
Follow-up was performed from the end of primary treatment until last recorded visit or death. Follow-up schedule consisted of a physical and gynecological examination every 3 months for the first 2 years, and then with a 6-month interval until 5 years, according to ESMO guidelines [6]. CA125 and HE4 samples were analyzed as previously reported [11] and were collected at three time points: diagnosis (prior to first surgery), interval, and final. For the interval time point, blood was collected every time that patients returned for clinical follow-up, generally, every 3 months for the first 2 years and then with a 6-month interval until 5 years, according to our internal EC follow-up strategy.
For the final time point, we considered blood samples collected after histological/radiological confirmation of recurrent disease or, in cases in which there was no evidence of recurrent disease, at last recorded follow-up visit.
Disease stage, histology, grade, treatment information, age, date of recurrence and date of last follow-up visit or death, and CA125 and HE4 levels were recorded in all cases.
Recurrent disease was defined as a histopathologically or radiologically [21] documented disease occurred after a disease-free interval of 3 or more months.
HE4 and CA125 levels were compared using Wilcoxon rank test as data were not normally distributed. All calculations were performed using Medcalc© statistical software ver. 12.4.0.0. A p value <0.05 was considered statistically significant. Receiver operator curves (ROC) were used to compare the ability of HE4 and CA125 to identify patients with recurrent disease.
Results
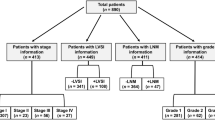
Between January 2009 and July 2013, 282 patients have been reviewed and considered for study recruitment. Thirty patients were lost at follow-up. Therefore, a total of 252 patients were successfully analyzed for our study. Table 1 shows the characteristics of patients.
Patients presented a mean age of 61.2 years and mean BMI was 32.5 kg/m2. During a median follow-up period of 38 months (range 24 to 75 months), 37 patients developed a recurrence and 8 of these patients died. In 12 cases, patients presented distant metastases, in 25 cases a local recurrence.
Characteristics of recurrent and non-recurrent patients were homogenous for stage, grading, performance status, age, and mean follow-up time.
First of all, we evaluated the role of HE4 levels at primary diagnosis compared to CA125 as indicators of recurrence among recurrent and non-recurrent patients.
Among recurrent (n = 37) and non-recurrent (n = 215) patients, we found mean HE4 values of 293.6 and 105.5 pmol/L, respectively.
Therefore, we found a statistically significant difference between HE4 values at primary diagnosis, comparing recurrent and non-recurrent patients (p = 0.002), demonstrating a correlation between this biomarker and recurrence.
Considering CA125 levels, among recurrent (n = 37) and non-recurrent (n = 215) patients, we found mean values of 85.7 and 81.2 UI/mL, respectively, resulting not statistically significant (p > 0.05). All results are reported in Table 2.
At primary diagnosis time point, in our cohort of patients, HE4 levels above 70 pmol/L demonstrated sensitivity of 67 % and a specificity of 53 % in predicting EC recurrence.
Considering the poor specificity of HE4 at threshold of 70 pmol/L, in our cohort of patients, we found out that a HE4 cut-off of 201.3 pmol/L at diagnosis time point is able to correctly classify patients at high or low risk of EC recurrence, with a sensitivity of 80 % and a specificity of 91 % (PPV = 90.3 % and NPV = 90.8 %), as demonstrated by the ROC curve in Fig. 1.
Stratifying patients by histotype, we observed that HE4 performance for the subset of patients with endometrioid histotype (n = 228), considering a cut-off of 70 pmol/L at primary diagnosis, was associated with a sensitivity of 74 % and a specificity of 61 %, when assessing for recurrent disease.
Moreover, using our identified HE4 cut-off of 201.3 pmol/L at primary diagnosis, we observed a sensitivity of 83 % and a specificity of 95 % (PPV = 91.3 % and NPV = 95.1 %).
All related results are summarized in Table 3.
Finally, we tested HE4 and CA125 levels among recurrent and non-recurrent patients at final time point (after histological/radiological confirmation of recurrent disease or at last recorded follow-up visit if they did not have any clinical evidence of recurrent disease).
Among recurrent (n = 37) and non-recurrent (n = 215) patients, we found mean HE4 values of 211.5 and 75.8 pmol/L, respectively.
Therefore, we found a statistically significant difference between HE4 values at final time point, comparing recurrent and non-recurrent patients (p = 0.003), confirming the role for this new marker in EC recurrent disease.
Considering CA125 levels, among recurrent (n = 37) and non-recurrent (n = 215) patients, we found mean values of 38.2 and 30.8 UI/mL, respectively, resulting not statistically significant (p > 0.05). All above results are reported in Table 4.
Discussion
EC recurrence rates among 13–17 % of women, mostly within 3 years of primary treatment [3, 4]. Three-year survival following recurrence is almost 73 % for vaginal recurrence but less than 15 % for pelvic or distant recurrences [22]. Moreover, 60 % of all recurrences occur in “low risk” patients (endometrioid subtype, low grade, and stage) who are not routinely submitted to adjuvant therapy, and 50 % of these cases experience distant recurrences which are characterized by a poor prognosis [4]. Despite the current recurrence rate of EC, surveillance strategies are not standardized yet. Several authors focused their studies on early diagnosis being related to more therapeutic options and better outcomes [4].
Up to now, there is no general consensus on follow-up routines after treatment for gynecological cancers [3, 23–25].
Actually, symptomatic recurrences are characterized by a significantly decreased survival compared to asymptomatic cases [4, 25]. However, despite intensive surveillance, symptomatic recurrence still ranges from 41–83 % [5] and there is very little evidence to support the role of routine vaginal cytology, imaging, or CA125 in post treatment surveillance of EC patients [5, 25].
Therefore, the research is focusing on new prognostic factors that may lead to a better preoperative patients’ selection in order to tailor different follow-up strategies, standard or intensive, according to the risk of recurrence.
Recently, Brennan et al. [19] published the first retrospective analysis of HE4 role in 98 recurrent EC patients. They demonstrated that HE4 levels decreased after initial treatment (p = 0.001) and increased again at recurrence (p = 0.002). HE4 was elevated (>70 pmol/L) in 21 of 26 (81 %) patients at recurrence, in particular in endometrioid histology (n = 69), where serum HE4 was a better indicator of recurrence than CA125. A HE4 level of 70 pmol/L was associated with a sensitivity of 84 %, a specificity of 74 %, and a negative predictive value of 93 % when assessing for recurrent endometrioid EC. Moreover, a higher proportion of patients who remained disease free had a HE4 < 70 pmol/L at the interval time point compared to those who subsequently developed recurrent disease (67 vs. 35 %, p = 0.004), suggesting that normalization of HE4 after initial treatment may be a prognostic factor. The sensitivity values reported in Brenner’s study are significantly higher than those reported in historical studies of CA125 in recurrent EC [26–28]. Despite these promising data on an emerging role for HE4 in the identification of recurrent endometrioid EC, the lack of baseline renal function data, which is now recognized as the most common cause of false positive readings, represents the major pitfall in clinical application [29].
The current study represents the first study in literature to evaluate the suitability of HE4 levels compared to CA125 at primary diagnosis as an indicator of recurrence and the paper with the larger cohort of recurrent EC patients where HE4 concentration was analyzed. In addition, the strength of our study is the exclusion of patients with abnormal renal function.
As previously reported, we found a statistically significant difference between HE4 values at primary diagnosis, comparing recurrent and non-recurrent patients (p = 0.002).
On the contrary, CA125 values, at primary diagnosis, resulted not statistically significant among recurrent and non-recurrent patients (p > 0.05).
In the subset of patients with endometrioid histotype (n = 228), a HE4 level of 70 pmol/L at primary diagnosis was associated with a sensitivity of 74 % and a specificity of 61 %, lower than those reported by Brenner (Brenner). On the other hand, using our identified HE4 cut-off of 201.3 pmol/L at primary diagnosis, we reported a sensitivity of 83 % and a specificity of 95 % (PPV = 91.3 % and NPV = 95.1 %), showing a better accuracy.
In the last part of our study, we tested HE4 and CA125 levels among recurrent and non-recurrent patients at final time point and we found a statistically significant difference between HE4 values comparing recurrent and non-recurrent patients (p = 0.003), confirming the role for this new marker in EC recurrent disease and the findings of Brennan et al. [19].
Considering CA125 levels, among recurrent (n = 37) and non-recurrent (n = 215) patients, we found that CA125 resulted not statistically significant (p > 0.05) at each time point.
A critical issue regarding tumor markers is their role in clinical practice.
In absence of prospective studies concerning the optimal frequency of post-treatment follow-up, in the last years, several authors have focused their researches in the identification of prognostic factors able to stratify patients by risk of EC recurrence, leading to “tailored” treatment and post-treatment programs. The lack of evidence has often led to an excess of clinical and radiological examinations, which has not lead to a benefit in terms of oncologic outcome, with high medical costs.
Our results showed a promising role for HE4 in the identification of “high-risk” patients for recurrence, specially in the subset of endometrioid histology.
Finally, it is important for oncologists to contextualize and translate the value of a tumor marker in clinical practice. Currently, no data suggest that modifying treatment, based on the absolute level or change in the serum level of tumor markers from baseline, impacts on outcome (except, in case of disease progression). Therefore, it would certainly be inappropriate to base a treatment variation solely on laboratory values. In fact, these findings are promising but resulted from a retrospective analysis and surely require a validation in independent prospective cohort studies.
However, they could provide an impulse for the development of predictive algorithms using HE4 alone or in combination with several features, such as age, grading, and stage, and help to recognize patients who need a more intensive post-treatment program in order to modify their surveillance strategy.
References
Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon: International Agency for Research on Cancer; 2013.
Ueda SM, Kapp DS, Cheung MK, et al. Trends in demographic and clinical characteristics in women diagnosed with corpus cancer and their potential impact on the increasing number of deaths. Am J Obstet Gynecol. 2008;198:218.e211–6.
Fung-Kee-Fung M, Dodge J, Elit L, Lukka H, Chambers A, Oliver T, et al. Follow-up after primary therapy for endometrial cancer: a systematic review. Gynecol Oncol. 2006;101(3):520–9.
Smith CJ, Heeren M, Nicklin JL, Perrin LC, Land R, Crandon AJ, et al. Efficacy of routine follow-up in patients with recurrent uterine cancer. Gynecol Oncol. 2007;107(1):124–9.
Salani R, Backes FJ, Fung MF, et al. Posttreatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncologists recommendations. Am J Obstet Gynecol. 2011;204:466–78.
Colombo N, Preti E, Landoni F, Carinelli S, Colombo A, Marini C, Sessa C; ESMO Guidelines Working Group. Endometrial cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013 Oct;24 Suppl 6:vi33–8.
Aggarwal P, Kehoe S. Serum tumour markers in gynaecological cancers. Maturitas. 2010;67:46–53.
Bignotti E, Ragnoli M, Zanotti L, Calza S, Falchetti M, Lonardi S, et al. Diagnostic and prognostic impact of serum HE4 detection in endometrial carcinoma patients. Br J Cancer. 2011;104(9):1418–25.
Moore RG, Brown AK, Miller MC, Badgwell D, Lu Z, Allard WJ, et al. Utility of a novel serum tumor biomarker HE4 in patients with endometrioid adenocarcinoma of the uterus. Gynecol Oncol. 2008;110(2):196–201.
Zanotti L, Bignotti E, Calza S, Bandiera E, Ruggeri G, Galli C, et al. Human epididymis protein 4 as a serum marker for diagnosis of endometrial carcinoma and prediction of clinical outcome. Clin Chem Lab Med. 2012;50(12):2189–98.
Capriglione S, Plotti F, Miranda A, Ricciardi R, Scaletta G, Aloisi A, et al. Utility of tumor marker HE4 as prognostic factor in endometrial cancer: a single-center controlled study. Tumour Biol. 2015;36(6):4151–6.
Angioli R, Miranda A, Aloisi A, Montera R, Capriglione S, De Cicco NC, et al. A critical review on HE4 performance in endometrial cancer: where are we now? Tumour Biol. 2014;35(2):881–7.
Angioli R, Capriglione S, Aloisi A, Luvero D, Cafà EV, Dugo N, et al. REM (risk of endometrial malignancy): a proposal for a new scoring system to evaluate risk of endometrial malignancy. Clin Cancer Res. 2013;19(20):5733–9.
Angioli R, Plotti F, Capriglione S, Montera R, Damiani P, Ricciardi R, et al. The role of novel biomarker HE4 in endometrial cancer: a case control prospective study. Tumour Biol. 2013;34(1):571–6.
Kalogera E, Scholler N, Powless C, Weaver A, Drapkin R, Li J, et al. Correlation of serum HE4 with tumor size and myometrial invasion in endometrial cancer. Gynecol Oncol. 2012;124(2):270–5.
Brennan DJ, Hackethal A, Metcalf AM, Coward J, Ferguson K, Oehler MK, et al. Serum HE4 as a prognostic marker in endometrial cancer—a population based study. Gynecol Oncol. 2014;132(1):159–65.
Moore RG, Miller CM, Brown AK, Robison K, Steinhoff M, Lambert-Messerlian G. Utility of tumor marker HE4 to predict depth of myometrial invasion in endometrioid adenocarcinoma of the uterus. Int J Gynecol Cancer. 2011;21(7):1185–90.
Mutz-Dehbalaie I, Egle D, Fessler S, Hubalek M, Fiegl H, Marth C, et al. HE4 is an independent prognostic marker in endometrial cancer patients. Gynecol Oncol. 2012;126(2):186–91.
Brennan DJ, Hackethal A, Mann KP, Mutz-Dehbalaie I, Fiegl H, Marth C, et al. Serum HE4 detects recurrent endometrial cancer in patients undergoing routine clinical surveillance. BMC Cancer. 2015;15:33.
Amant F, Moerman P, Neven P, Timmerman D, Van Limbergen E, Vergote I. Endometrial cancer. Lancet. 2005;366(9484):491–505.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–16.
Creutzberg CL, van Putten WL, Koper PC, Lybeert ML, Jobsen JJ, Warlam-Rodenhuis CC, et al. Survival after relapse in patients with endometrial cancer: results from a randomized trial. Gynecol Oncol. 2003;89(2):201–9.
Lajer H, Jensen MB, Kilsmark J, Albaek J, Svane D, Mirza MR, et al. The value of gynecologic cancer follow-up: evidence-based ignorance? Int J Gynecol Cancer. 2010;20:1307–20.
Zanagnolo V, Ming L, Gadducci A, Maggino T, Sartori E, Zola P, et al. Surveillance procedures for patients with cervical carcinoma: a review of the literature. Int J Gynecol Cancer. 2009;19:194–201.
Sartori E, Pasinetti B, Chiudinelli F, Gadducci A, Landoni F, Maggino T, et al. Surveillance procedures for patients treated for endometrial cancer: a review of the literature. Int J Gynecol Cancer. 2010;20:985–92.
Lo SS, Khoo US, Cheng DK, Ng TY, Wong LC, Ngan HY. Role of serial tumor markers in the surveillance for recurrence in endometrial cancer. Cancer Detect Prev. 1999;23(5):397–400.
Patsner B, Orr Jr JW, Mann Jr WJ. Use of serum CA 125 measurement in posttreatment surveillance of early-stage endometrial carcinoma. Am J Obstet Gynecol. 1990;162(2):427–9.
Rose PG, Sommers RM, Reale FR, Hunter RE, Fournier L, Nelson BE. Serial serum CA 125 measurements for evaluation of recurrence in patients with endometrial carcinoma. Obstet Gynecol. 1994;84(1):12–6.
Lv YW, Yang L, Zhang M, Jiang LH, Niu JH, Hou J, et al. Increased human epididymis protein 4 in benign gynecological diseases complicated with chronic renal insufficiency patients. Genet Mol Res. 2015;14(1):2156–61.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Angioli, R., Capriglione, S., Scaletta, G. et al. The role of HE4 in endometrial cancer recurrence: how to choose the optimal follow-up program. Tumor Biol. 37, 4973–4978 (2016). https://doi.org/10.1007/s13277-015-4324-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4324-z