Abstract
Lysosome-associated protein transmembrane 4ß-35 (LAPTM4B-35) is overexpressed in several solid malignancies. This study determines the expression level of LAPTM4B-35 in the cervical cancer during tumor development and progression. The present study investigated the clinicopathological significance of the coexpression of LAPTM4B-35 and VEGF in patients with cervical cancer. Immunohistochemistry was used to evaluate the expression of LAPTM4B-35 and VEGF in 62 cervical intraepithelial neoplasia (CIN) and 226 cervical carcinoma in comparison with 45 normal cervical specimens. The correlation of combined LAPTM4B-35 and VEGF with clinicopathologic characteristics was analyzed using a chi-squared test. Patient survival was determined using Kaplan–Meier method and log-rank test. A Cox regression analysis was performed to determine the prognostic significance of the factors. Combined LAPTM4B-35 and VEGF expression was significantly associated with FIGO stage (P = 0.014), tumor histologic grade (P = 0.033), lymph node metastasis (P = 0.045), and recurrence (P = 0.010). Kaplan–Meier survival analysis showed that patients with cervical cancer expressing both LAPTM4B-35 and VEGF exhibited both poor overall survival (OS) and disease-free survival (DFS) (P = 0.015 and P = 0.016, respectively). Cox analysis demonstrated that combined LAPTM4B-35 and VEGF expression was an independent factor for both OS and DFS (P = 0.015 and P = 0.016, respectively). Overexpression of LAPTM4B-35combined with positive VEGF expression may serve as a new biological marker to predict the prognosis of cervical carcinoma patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical cancer (CC) is the fourth most common cancer in women worldwide, with the highest prevalence in developing countries where CC is the leading cause of death [1, 2]. Persistent infection of high-risk human papillomavirus (HPV) is considered the main cause for developing CC. HPV vaccines are effectively used to prevent type 16 and 18 HPV infections. However, the natural history of the disease indicates that an effect on cervical cancer incidence will not occur for decades because approximately 30–60 % of women who are infected with genital HPV do not develop cancer [3, 4]. Therefore, identifying valuable prognostic factors is necessary to improve procedures for early detection and treatment of CC.
Recently, lysosomal protein transmembrane 4 beta (LAPTM4B), a novel cancer-related gene, was successfully cloned [5]. The LAPTM4B gene has been demonstrated to encode the LAPTM4B-35 protein with molecular weight of 35 kDa. Studies have shown that LAPTM4B-35 is upregulated in several human cancers [6–13]. Our previous studies also indicated that the overexpression of LAPTM4B is associated with poor prognosis in a large portion of CC tissues [14] and contributes to cellular proliferation, transformation, tumorgenesis, metastatic progression, and angiogenesis in CC cell line HeLa [15].
Angiogenesis is critical for cancer growth, invasion, and metastasis [16]. Vascular endothelial growth factor (VEGF) is a potent vascular active molecule that is important in regulating physiological and pathological angiogenesis [17]. An increasing amount of evidence suggests that VEGF has been widely assessed in many types of tumors, including CC [18–20]. Our previous study revealed that the downregulation of LAPTM4B by RNA interference significantly reduced VEGF protein expression in the CC cell line HeLa [15]. Therefore, further investigation is needed to better define the combined predictive values of these two potential prognostic factors in CC.
The present study investigated the potential correlation of the coexpression of LAPTM4B-35 and VEGF in CC tissues using clinical findings and patient outcome. Our results indicate that the combined LAPTM4B-35 and VEGF expression may be used as an independent marker for prognosis of CC in patients.
Materials and methods
The specimens (n = 333) were obtained from patients at the Department of Gynecology of Harbin Medical University’s affiliated tumor hospital between September 2006 and March 2008. Of the 333 patients, 45 had normal cervical epithelia, 62 had cervical intraepithelial neoplasia (CIN), and 226 had cervical carcinoma. The patients with cervical carcinoma underwent radical hysterectomy and pelvic lymphadenectomy. The tumor stages were evaluated following the International Federation of Gynecology and Obstetrics (FIGO) staging system. The clinicopathologic features in these patients included gender, age, clinical hepatic status, viral infection status, and tumor size. All patients with CC were monitored for survival analysis until 31 March 2013 (median, 57 months; range, 14–78 months). Table 1 summarizes detailed information of the tumor characteristics.
The approval from the Medical Ethical Committee of Harbin Medical University’s affiliated tumor hospital was obtained before the research began.
Immunohistochemistry
After the collection of paraffin-embedded cervical specimens, all tissue blocks were cut to 4-μm-thick slices using a microtome and affixed onto slides. Tissue sections were dewaxed in xylene and rehydrated by graded alcohol concentrations according to standard procedures. The sections were immersed in 0.3 % hydrogen peroxide (H2O2) for 15 min to block the activity of endogenous peroxidase. Then, LAPTM4B-35 antigen retrieval was performed by heating the sections in a microwave for 15 min in 0.01 mol/L citrate buffer (pH 6.0). The slide was incubated at room temperature for 2 h with anti-LAPTM4B-35 antibody (Abcam, CA, USA) or anti-VEGF antibody (Santa Cruz, CA, USA) at a dilution of 1:100. The sections were incubated with a second biotinylated antibody, followed by horseradish peroxidase-conjugated streptavidin for 30 min. Then, 3,3′-diaminobenzidine tetrahydrochloride (Dako, Germany) was used to visualize the reaction. The slides were counterstained with Meyer’s hematoxylin, and negative control slides were stained with normal rabbit IgG instead of primary antibodies.
Staining evaluation
LAPTM4B-35 and VEGF expression levels were classified semi-quantitatively based on the total combined scores of positive-staining tumor cell percentage and staining intensity. The percentage of positive cells was scored as “0” if <5 % of the tumor cells were stained positive, “1” if 5–50 % were stained positive, and “2” if >50 % of the cells were stained positive. The staining intensity was scored as follows: “0” in cases of no or only weak staining, “1” in cases of moderate staining, and “2” in cases of strong staining. The staining intensity score plus the percentage of positive staining was used to evaluate expression levels, where 0–2 indicated low expression and 3–4 indicated high expression.
Statistical analysis
The χ 2 test was used to show differences in categorical variables. Patient survival and follow-up data were analyzed using the Kaplan–Meier method and log-rank test, respectively. P values of less than 0.05 were considered statistically significant. All analyses were carried out using the statistical software package SPSS 13.0 software (SPSS, Chicago, IL, USA).
Results
Expression of LAPTM4B-35 and VEGF in patients with CC
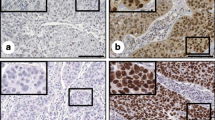
Statistical analysis showed that LAPTM4B-35 and VEGF expression was significantly elevated in CINs and CC compared with the normal controls (P < 0.05, data not shown). Representative images of LAPTM4B-35 and VEGF expression are shown in Fig. 1. The expression levels of LAPTM4B-35 and VEGF in CC patients are summarized in Table 1. Based on the scoring system used in the present study, 156 (69.0 %) cases were both LAPTM4B-35(+) and VEGF (+), 56 (24.8 %) cases were both LAPTM4B-35(−) and VEGF(−), 9 (4.0 %) cases were LAPTM4B-35(+) and VEGF(−), and 5 (2.2 %) cases were LAPTM4B-35(−) and VEGF(+). According to the chi-squared test, the LAPTM4B-35 expression was significantly associated with VEGF expression (P = 0.05, Tables 2 and 3).
Representative immunohistochemical staining of LAPTM4B-35 and VEGF in cervical specimens: a positive expression of LAPTM4B-35 in a cervical carcinoma; b negative expression of LAPTM4B-35 in a cervical carcinoma; c positive expression of VEGF in a cervical carcinoma; d negative expression of VEGF in a cervical carcinoma
Correlation of clinicopathologic parameters with LAPTM4B-35 and VEGF expression in CC
As shown in Table 1, the coexpression of LAPTM4B-35 and VEGF in CC was associated with FIGO stage, histological grade, lymph node metastasis, and tumor recurrence (P < 0.05), but not with age or histological type (P > 0.05).
Prognostic significance of LAPTM4B-35 and VEGF expression in CC
Univariate survival analysis revealed that CC with high expression of both LAPTM4B-35 and VEGF were correlated with shorter overall survival (OS) or disease-free survival (DFS) of patients (Fig. 2 and Table 3; both P < 0.001).
Kaplan–Meier analysis of overall survival and disease-free survival related to the expression of LAPTM4B-35 and VEGF. a Overall survival curves of the cervical carcinoma patients; b disease-free survival curves of the cervical carcinoma patients; LAPTM4B-35(−) and VEGF(−), blue line; LAPTM4B-35(−) and VEGF(+), yellow line; LAPTM4B-35(+) and VEGF(−), green line; LAPTM4B-35(+) and VEGF(+), purple line
Multivariate analysis showed that combined LAPTM4B-35 and VEGF expression was an independent prognostic marker for both OS and DFS of patients with CC (Table 4; P = 0.001 and P < 0.001, respectively).
Discussion
CC remains a serious health problem for women worldwide, and good prognostic factors are urgently needed [21]. In the present study, we analyzed the combined expression of LAPTM4B-35 and VEGF with the clinicopathologic characteristics in CC tissues. The results of this study indicate that the expression of both LAPTM4B-35 and VEGF were more common in CC tissues than in CINs and normal tissues. Moreover, we observed that the coexpression of LAPTM4B-35 and VEGF is associated with portal vein invasion, tumor recurrence, and patient prognosis. Our results therefore suggest that the coexpression of VEGF and LAPTM4B-35 was widely activated in CC, making it a possible pivotal marker for progression and prognosis in CC patients.
Angiogenesis plays a key role in tumor invasion and metastasis [22]. Angiogenesis regulators have been found owing to the increase in angiogenesis studies since the early 1990s. Of these regulators, VEGF is recognized as an active member of one of the main signaling pathways in angiogenesis. This regulator is involved in tumor angiogenesis and in the invasion of malignant tumor cells [23, 24]. VEGF is significantly overexpressed in a large number of human tumor types, such as gastric cancer [25], colorectal cancer [26], lung cancer [27], breast carcinoma [28], and prostate carcinoma [29]. Several studies have demonstrated that high VEGF protein expression is a valuable predictor of cervical cancer prognosis [30–32]. Consistent with these researches, the VEGF has clinical outcomes both in terms of overall survival and disease-free survival (Fig. 2a, b and Table 3; both P < 0.001).
LAPTM4B-35 is significantly overexpressed in several malignant tumors. Current studies revealed that LAPTM4B-35 might be important for tumor development, progression, and metastasis through the activation of some proto-oncogenes, such as c-myc, c-jun, and c-fos [7]. Moreover, recent data indicate that LAPTM4B is involved in signal transduction pathways, thus enhancing tumor cell malignant transformation [33, 34]. These findings provide important evidence to explicate the mechanism by which LAPTM4B-35 contributes to oncogenesis and tumor development. Tang et al. evaluated the expression of LAPTM4B and CD34 proteins in CC tissues through immunohistochemistry, and the results revealed that LAPTM4B protein could be used in predicting patient prognosis and in angiogenesis [35]. Our previous study indicated that downregulated LAPTM4B also exhibited decreased migration and invasion activity, as well as significantly reduced VEGF expression [15]. LAPTM4B-35 expression, accompanied by the overexpression of VEGF, promotes growth and proliferation of CC cell HeLa. Therefore, LAPTM4B-35 and VEGF both exist in the CC. With this, we hypothesized that coexpressing LAPTM4B-35 and VEGF may lead to CC progression, angiogenesis, and poor prognosis. In accordance with this concept, our results demonstrated that patients with CC that coexpress LAPTM4B-35 and VEGF have poor clinical outcomes for OS and DFS. In addition, combined LAPTM4B-35 and VEGF expression may serve as an independent marker for the prognosis of patients with CC. This is the first study to investigate the association between LAPTM4B-35 and VEGF coexpression and patient prognosis using CC tissues.
In conclusion, this study indicated that combined LAPTM4B-35 and VEGF expression may provide important information for predicting CC prognosis and individualizing therapy for patients with CC.
References
de Sanjose S, Serrano B, Castellsague X, Brotons M, Munoz J, Bruni L, et al. Human papillomavirus (HPV) and related cancers in the global alliance for vaccines and immunization (GAVI) countries. A WHO/ICO HPV information centre report. Vaccine. 2012;30 Suppl 4:D1–D83. vi.
Hwang SJ, Shroyer KR. Biomarkers of cervical dysplasia and carcinoma. J Oncol. 2012;2012:507286.
Crosbie EJ, Einstein MH, Franceschi S, Kitchener HC. Human papillomavirus and cervical cancer. Lancet. 2013;382(9895):889–99.
Romanowski B. Long term protection against cervical infection with the human papillomavirus: review of currently available vaccines. Human vaccies. 2011;7(2):161–9.
Shao GZ, Zhou RL, Zhang QY, Zhang Y, Liu JJ, Rui JA, et al. Molecular cloning and characterization of LAPTM4B, a novel gene upregulated in hepatocellular carcinoma. Oncogene. 2003;22(32):5060–9.
Yang H, Xiong F, Wei X, Yang Y, McNutt MA, Zhou R. Overexpression of LAPTM4B-35 promotes growth and metastasis of hepatocellular carcinoma in vitro and in vivo. Cancer Lett. 2010;294(2):236–44.
Zhou L, He XD, Yu JC, Zhou RL, Yang H, Qu Q, et al. Overexpression of LAPTM4B promotes growth of gallbladder carcinoma cells in vitro. Am J Surg. 2010;199(4):515–21.
Liu X, Xiong F, Wei X, Yang H, Zhou R. LAPTM4B-35, a novel tetratransmembrane protein and its PPRP motif play critical roles in proliferation and metastatic potential of hepatocellular carcinoma cells. Cancer Sci. 2009;100(12):2335–40.
Yang H, Xiong F, Qi R, Liu Z, Lin M, Rui J, et al. LAPTM4B-35 is a novel prognostic factor of hepatocellular carcinoma. J Surg Oncol. 2010;101(5):363–9.
Zhang H, Tian B, Yu H, Yao H, Gao Z. LAPTM4B-35 protein as a potential therapeutic target in gastric cancer. Tumour Biol. 2014;35(12):12737–42.
Zhou L, He XD, Yu JC, Zhou RL, Shan Y, Rui JA. Overexpression of LAPTM4B-35 attenuates epirubucin-induced apoptosis of gallbladder carcinoma GBC-SD cells. Surgery. 2011;150(1):25–31.
Zhou L, He XD, Cui QC, Zhou WX, Qu Q, Zhou RL, et al. Expression of LAPTM4B-35: a novel marker of progression, invasiveness and poor prognosis of extrahepatic cholangiocarcinoma. Cancer Lett. 2008;264(2):209–17.
Kang Y, Yin M, Jiang W, Zhang H, Xia B, Xue Y, et al. Overexpression of LAPTM4B-35 is associated with poor prognosis in colorectal carcinoma. Am J Surg. 2012;204(5):677–83.
Meng F, Luo C, Hu Y, Yin M, Lin M, Lou G, et al. Overexpression of LAPTM4B-35 in cervical carcinoma: a clinicopathologic study. Int J Gynecol Pathol. 2010;29(6):587–93.
Meng F, Chen X, Song H, Lou G. LAPTM4B down regulation inhibits the proliferation, invasion and angiogenesis of HeLa cells in vitro. Cell Physiol Biochem. 2015;37(3):890–900.
Folkman J, Kalluri R. Cancer without disease. Nature. 2004;427(6977):787.
Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9(6):669–76.
Wang Y, Huang L, Wu S, Jia Y, Yang Y, Luo L, et al. Bioinformatics analyses of the role of vascular endothelial growth factor in patients with non-small cell lung cancer. PLoS One. 2015;10(9):e0139285.
Eswarappa SM, Fox PL. Antiangiogenic VEGF-Ax: a new participant in tumor angiogenesis. Cancer Res. 2015;75(14):2765–9.
Abdel-Rahman O. Targeting vascular endothelial growth factor (VEGF) pathway in iodine-refractory differentiated thyroid carcinoma (DTC): from bench to bedside. Crit Rev Oncol Hematol. 2015;94(1):45–54.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA: a Cancer J Clin. 2015;65(1):5–29.
Gadducci A, Lanfredini N, Sergiampietri C. Antiangiogenic agents in gynecological cancer: State of art and perspectives of clinical research. Crit Rev Oncol Hematol. 2015;96(1):113–28.
Kerbel R, Folkman J. Clinical translation of angiogenesis inhibitors. Nat Rev Cancer. 2002;2(10):727–39.
Rigiracciolo DC, Scarpelli A, Lappano R, Pisano A, Santolla MF, De Marco P, Cirillo F, Cappello AR, Dolce V, Belfiore A et al.: Copper activates HIF-1alpha/GPER/VEGF signalling in cancer cells. Oncotarget 2015
Scartozzi M, Loretelli C, Galizia E, Mandolesi A, Pistelli M, Bittoni A, et al. Role of vascular endothelial growth factor (VEGF) and VEGF-R genotyping in guiding the metastatic process in pT4a resected gastric cancer patients. PLoS One. 2012;7(7):e38192.
Gremonprez F, Descamps B, Izmer A, Vanhove C, Vanhaecke F, Wever O, Ceelen W: Pretreatment with VEGF(R)-inhibitors reduces interstitial fluid pressure, increases intraperitoneal chemotherapy drug penetration, and impedes tumor growth in a mouse colorectal carcinomatosis model. Oncotarget 2015
Zhang Y, Yu LK, Lu GJ, Xia N, Xie HY, Hu W, et al. Prognostic values of VEGF and endostatin with malignant pleural effusions in patients with lung cancer. Asian Pac J Cancer Prev. 2014;15(19):8435–40.
Koutras A, Kotoula V, Fountzilas G. Prognostic and predictive role of vascular endothelial growth factor polymorphisms in breast cancer. Pharmacogenomics. 2015;16(1):79–94.
Huang S, He P, Peng X, Li J, Xu D, Tang Y. Pristimerin inhibits prostate cancer bone metastasis by targeting PC-3 stem cell characteristics and VEGF-induced vasculogenesis of BM-EPCs. Cell Physiol Biochem. 2015;37(1):253–68.
Saijo Y, Furumoto H, Yoshida K, Nishimura M, Irahara M. Clinical significance of vascular endothelial growth factor expression and microvessel density in invasive cervical cancer. J Med Invest. 2015;62(3–4):154–60.
Dong J, Cheng M, Sun H. Function of inducible nitric oxide synthase in the regulation of cervical cancer cell proliferation and the expression of vascular endothelial growth factor. Mol Med Rep. 2014;9(2):583–9.
Chen L, Wu YY, Liu P, Wang J, Wang G, Qin J, et al. Down-regulation of HPV18 E6, E7, or VEGF expression attenuates malignant biological behavior of human cervical cancer cells. Med Oncol. 2011;28 Suppl 1:S528–39.
Huygens C, Lienart S, Dedobbeleer O, Stockis J, Gauthy E, Coulie PG, et al. Lysosomal-associated transmembrane protein 4B (LAPTM4B) decreases transforming growth factor beta1 (TGF-beta1) production in human regulatory T cells. J Biol Chem. 2015;290(33):20105–16.
Milkereit R, Persaud A, Vanoaica L, Guetg A, Verrey F, Rotin D. LAPTM4b recruits the LAT1-4F2hc Leu transporter to lysosomes and promotes mTORC1 activation. Nat Commun. 2015;6:7250.
Tang H, Tian H, Yue W, Li L, Li S, Gao C, et al. Overexpression of LAPTM4B is correlated with tumor angiogenesis and poor prognosis in non-small cell lung cancer. Med Oncol. 2014;31(6):974.
Acknowledgments
We express our thanks to Dr. H-T Song for the evaluation procedures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Grant support
This work was supported by grants of the National Natural Science Foundation of China (81201613), the Specialized Research Fund for the Doctoral Program of Higher Education (20122307120027), the Postdoctoral Foundation of Heilongjiang Province of China (LBH-Z11067), the scientific research project of Health Department of Heilongjiang Province (663), and the Haiyan Foundation of the Affiliated Tumor Hospital of Harbin Medical University/the Foundation of the Affiliated Tumor Hospital of Harbin Medical University (JJZ2011-04). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional information
Fanling Meng and Shu Tan contributed equally to this work.
Rights and permissions
About this article
Cite this article
Meng, F., Tan, S., Liu, T. et al. Predictive significance of combined LAPTM4B and VEGF expression in patients with cervical cancer. Tumor Biol. 37, 4849–4855 (2016). https://doi.org/10.1007/s13277-015-4319-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4319-9