Abstract
Backgrounds
Although zinc acts as a major regulator of neuronal physiology, its dyshomeostasis may cause neuronal cell death. Hydrogen sulfide (H2S) has been reported to attenuate ischemic brain injury and to suppress Zn2+-induced neuronal cell death, but the underlying mechanisms have not been elucidated.
Methods
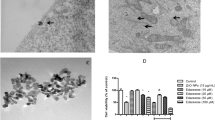
We determined the direct Zn2+-chelating capacity of sodium hydrogen sulfide (NaHS), an H2S donor, using specific Zn2+ fluorescent dyes (Zinpyr-1 and Zinquin) in SH-SY5Y cells.
Results
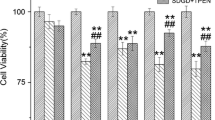
NaHS significantly suppressed the Zn2+-mediated increase in the fluorescence intensities of Zinpyr- 1 and Zinquin in a dose-dependent manner. NaHS significantly inhibited cell death induced by extracellular or intracellular Zn2+ overload. Furthermore, Zn2+-mediated increases in the phosphorylation of glycogen synthase kinase-3β and protein kinase C were highly suppressed by NaHS treatment.
Conclusion
These results demonstrate that NaHS has the capacity to chelate extracellular and intracellular Zn2+, and could therefore be used in the protection against Zn2+ neurotoxicity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kajimura, M., Fukuda, R., Bateman, R. M., Yamamoto, T. & Suematsu, M. Interactions of multiple gas–transducing systems: hallmarks and uncertainties of CO, NO, and H2S gas biology. Antioxid Redox Signal 13, 157–192 (2010).
Ishigami, M. et al. Source of Hydrogen Sulfide and a Mechanism of Its Release in the Brain. Antioxid Redox Signal 11, 205–214 (2008).
Kamoun, P. Endogenous production of hydrogen sulfide in mammals. Amino Acids 26, 243–254 (2004).
Warenycia, M. W. et al. Acute hydrogen sulfide poisoning. Demonstration of selective uptake of sulfide by the brainstem by measurement of brain sulfide levels. Biochem Pharmacol 38, 973–981 (1989).
Zhang, J.–Y. et al. Hydrogen sulfide therapy in brain diseases: from bench to bedside. Med Gas Res 7, 113–119 (2017).
Szabo, C. Hydrogen sulphide and its therapeutic potential. Nat Rev Drug Discov 6, 917–935 (2007).
Qu, K., Lee, S. W., Bian, J. S., Low, C. M. & Wong, P. T. Hydrogen sulfide: neurochemistry and neurobiology. Neurochem Int 52, 155–165 (2008).
Beyersmann, D. & Haase, H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 14, 331–341 (2001).
Noh, S. et al. The direct modulatory activity of zinc toward ion channels. Integr Med Res 4, 142–146 (2015).
Lee, S. R. et al. The Critical Roles of Zinc: Beyond Impact on Myocardial Signaling. Korean J Physiol Pharmacol 19, 389–399 (2015).
Barr, C. A. & Burdette, S. C. The zinc paradigm for metalloneurochemistry. Essays Biochem 61, 225–235 (2017).
Frederickson, C. J. & Moncrieff, D. W. Zinc–containing neurons. Biological Signals 3, 127–139 (1994).
Vasto, S. et al. Inflammation, genes and zinc in Alzheimer’s disease. Brain Res Rev 58, 96–105 (2008).
Zhang, J. et al. Nicotine attenuates beta–amyloid–induced neurotoxicity by regulating metal homeostasis. FASEB 20, 1212–1214 (2006).
McCord, M. C. & Aizenman, E. The role of intracellular zinc release in aging, oxidative stress, and Alzheimer’s disease. Front Aging Neurosci 6, 77 (2014).
Frazzini, V., Rockabrand, E., Mocchegiani, E. & Sensi, S. L. Oxidative stress and brain aging: is zinc the link? Biogerontology 7, 307–314 (2006).
Sensi, S. L. & Jeng, J. M. Rethinking the excitotoxic ionic milieu: the emerging role of Zn(2+) in ischemic neuronal injury. Curr Mol Med 4, 87–111 (2004).
Lee, J. Y. et al. Cytosolic labile zinc accumulation in degenerating dopaminergic neurons of mouse brain after MPTP treatment. Brain Res 1286, 208–214 (2009).
Dexter, D. T. et al. Alterations in the levels of iron, ferritin and other trace metals in Parkinson’s disease and other neurodegenerative diseases affecting the basal ganglia. Brain 114, 1953–1975 (1991).
Shimoji, M., Hara, H., Kamiya, T., Okuda, K. & Adachi, T. Hydrogen sulfide ameliorates zinc–induced cell death in neuroblastoma SH–SY5Y cells. Free Radic Res 51, 978–985 (2017).
Beltowski, J. Hydrogen sulfide in pharmacology and medicine––An update. Pharmacol Rep 67, 647–658 (2015).
Colvin, R. A., Laskowski, M. & Fontaine, C. P. Zinquin identifies subcellular compartmentalization of zinc in cortical neurons. Relation to the trafficking of zinc and the mitochondrial compartment. Brain Res 1085, 1–10 (2006).
Burdette, S. C., Walkup, G. K., Spingler, B., Tsien, R. Y. & Lippard, S. J. Fluorescent sensors for Zn (2+) based on a fluorescein platform: synthesis, properties and intracellular distribution. J Am Chem Soc 123, 7831–7841 (2001).
Lee, S. R. et al. Kobophenol A Inhibits Sodium Nitroprusside–Induced Cardiac H9c2 Cell Death through Suppressing Activation of JNK and Preserving Mitochondrial Anti–apoptotic Bcl–2 and Mcl–1. Chem Pharm Bull 62, 713–718 (2014).
Repetto, G., del Peso, A. & Zurita, J. L. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat Protoc 3, 1125–1131 (2008).
Sheline, C. T., Behrens, M. M. & Choi, D. W. Zinc–induced cortical neuronal death: contribution of energy failure attributable to loss of NAD (+) and inhibition of glycolysis. J Neurosci 20, 3139–3146 (2000).
Gomez Perez, M., Fourcade, L., Mateescu, M. A. & Paquin, J. Neutral Red versus MTT assay of cell viability in the presence of copper compounds. Anal Biochem 535, 43–46 (2017).
Dineley, K. E., Votyakova, T. V. & Reynolds, I. J. Zinc inhibition of cellular energy production: implications for mitochondria and neurodegeneration. J Neurochem 85, 563–570 (2003).
Fukada, T. & Kambe, T. Zinc signlas in cellular functions and Disorders (Springer, Tyoko, 2014).
Lee, S., Chanoit, G., McIntosh, R., Zvara, D. A. & Xu, Z. Molecular mechanism underlying Akt activation in zinc–induced cardioprotection. Am J Physiol Heart Circ Physiol 297, H569–575 (2009).
Aras, M. A., Hara, H., Hartnett, K. A., Kandler, K. & Aizenman, E. Protein kinase C regulation of neuronal zinc signaling mediates survival during preconditioning. J Neurochem 110, 106–117 (2009).
Aras, M. A. & Aizenman, E. Redox Regulation of Intracellular Zinc: Molecular Signaling in the Life and Death of Neurons. Antioxid Redox Signal 15, 2249–2263 (2011).
Paul, B. D. & Snyder, S. H. Gasotransmitter hydrogen sulfide signaling in neuronal health and disease. Biochem Pharmacol 149, 101–109 (2018).
Olas, B. Medical Functions of Hydrogen Sulfide. Adv Clin Chem 74, 195–210 (2016).
Tan, B. H., Wong, P. T. & Bian, J. S. Hydrogen sulfide: a novel signaling molecule in the central nervous system. Neurochem Int 56, 3–10 (2010).
Kimura, H., Nagai, Y., Umemura, K. & Kimura, Y. Physiological roles of hydrogen sulfide: synaptic modulation, neuroprotection, and smooth muscle relaxation. Antioxid Redox Signal 7, 795–803 (2005).
Kimura, H. Hydrogen sulfide: its production, release and functions. Amino Acids 41, 113–121 (2011).
Kimura, H. The physiological role of hydrogen sulfide and beyond. Nitric Oxide 41, 4–10 (2014).
Lee, S. R. Critical Role of Zinc as Either an Antioxidant or a Prooxidant in Cellular Systems. Oxid Med Cell Longev 2018, 9156285 (2018).
Ahn, Y. H., Kim, Y. H., Hong, S. H. & Koh, J. Y. Depletion of intracellular zinc induces protein synthesis–dependent neuronal apoptosis in mouse cortical culture. Exp Neurol 154, 47–56 (1998).
Naganska, E. & Matyja, E. Apoptotic neuronal changes enhanced by zinc chelator–TPEN in organotypic rat hippocampal cultures exposed to anoxia. Folia Neuropathol 44, 125–132 (2006).
Jiang, J. et al. Hydrogen Sulfide–Mechanisms of Toxicity and Development of an Antidote. Sci Rep 6, 20831 (2016).
Mostofa, M. G. et al. Hydrogen sulfide modulates cadmium–induced physiological and biochemical responses to alleviate cadmium toxicity in rice. Sci Rep 5, 14078 (2015).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, S.R. Cellular toxicity of zinc can be attenuated by sodium hydrogen sulfide in neuronal SH-SY5Y cell. Mol. Cell. Toxicol. 14, 425–436 (2018). https://doi.org/10.1007/s13273-018-0047-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-018-0047-8