Abstract
In asthma and chronic obstructive pulmonary disease (COPD), mucins display disease-related alterations caused by airway mucus obstruction. MUC5AC, MUC5B and MUC8 are known as the major secretory mucins in human airway epithelial cells. Analysis of mucin genes has identified the presence of several features with a variable number of tandem repeats (VNTR; minisatellites) in the central region of each mucin. In our previous study, six minisatellites in the region of the MUC8 gene were identified, and the MUC8-MS5 minisatellite showed the highest heterozygosity among them. In this study, we evaluated the relationship between MUC8-MS5 and susceptibility to asthma and COPD. A case-control study was performed with 229 controls, 123 COPD cases and 77 asthma cases. A significant association (OR 3.96) between short alleles (2/2 repeats) and the occurrence of COPD was observed [95% confidence interval (CI) 1.32–11.88; p = 0.008]. Hence, the increased frequency of 2/2 homo-short alleles were also found in asthma cases (3.11; CI 0.88–11.05; p = 0.066), though this association was not statistically significant. These results revealed a genetic association between MUC8 and COPD, and that the specific short minisatellite alleles (2/2) of MUC8-MS5 may be a risk factor for COPD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Epithelial surfaces of human respiratory, gastrointestinal and reproductive tracts are protected by mucus secretions (Martínez-Antón et al. 2006). Mucus is secreted by the airway surface epithelium and glands, and it plays an important role in protecting the human airway from the external environment. In chronic inflammatory airway diseases, mucins display disease-related alterations in quantity, composition and glycosylation (Hollingsworth and Swanson 2004). There are seven secreted mucins, including five polymeric mucins (MUC2, MUC5AC, MUC5B, MUC6, and MUC19) and two nonpolymeric glycoproteins (MUC7 and MUC8). MUC7 and MUC8 also encode mucin-like glycoproteins lacking a transmembrane domain (Byrd and Bresalier 2004). MUC5AC, MUC5B and MUC8 are the airway mucin genes most prominently involved in chronic respiratory diseases (Yuta et al. 1997; Nadel 2001): MUC5AC and MUC5B are found in higher amounts in asthma and COPD patients. Furthermore, overexpressed MUC8 was also identified in respiratory organs related to chronic inflammatory respiratory organs (Basbaum et al. 1999; Bernacki et al. 1999; Song et al. 2008).
In chronic inflammatory airway diseases, such as asthma and COPD, mucins display alterations in quantity, composition and glycosylation. Asthma and COPD affect more than 500 million people worldwide and are characterized by mucus hypersecretion as a common clinical feature (Ha and Rogers 2016). Despite recent advances and multiple available therapies and interventions, there remains a clinical need to determine the mechanisms that exacerbate airway diseases. The development of clinical biomarkers for use in prognosis will allow easier detection of conflict, and intervention can take place before the disease progresses.
Human minisatellites are located predominantly in the subtelomeric regions of chromosomes (Jeffreys et al. 1985), and MUC8 is located at q24.3 of MUC8 on chromosome 12 (Thornton et al. 2008). In many reports, the length of minisatellites of several genes (HRAS1, hTERT, BORIS, MUC2 and MUC6) has been associated with susceptibility to certain human disorders (Krontiris et al. 1993; Leem et al. 2002; Jeong et al. 2007; Kwon et al. 2010; Yoon et al. 2010). We focused on the major components of mucus, involved in respiratory disorders, which are coded by mucin genes; these genes contain minisatellite regions (Vinall et al. 1998; Thornton et al. 2008). In a previous study, we characterized the genomic features of the MUC8 region, and six novel minisatellites (MUC8-MS1, -MS2, -MS3, -MS4, -MS5, -MS6) were analyzed (Lee et al. 2009). Among those minisatellites, MUC8-MS5 showed the highest heterozygosity (h = 0.662) in the MUC8 region. In several reports, abnormal expression of MUC8 occurred in various diseases and in conditions associated with high risk to respiratory organs (Basbaum et al. 1999; Bernacki et al. 1999; Song et al. 2008). Therefore, we examined the association between MUC8-MS5 and asthma or COPD in this study. Here, we report the allelic variations of MUC8-MS5 and its association with asthma or COPD.
Materials and methods
Study population
We conducted a case-control study for the minisatellites of MUC8-MS5 using the genomic DNA from 229 unrelated controls, 123 COPD cases and 77 asthma cases. Controls were selected from the Department of Preventive Medicine and Internal Medicine of Dong-A University hospitals from 2000 to 2004 (Busan, Korea). A total of 229 controls with no personal history of asthma or COPD completed an interview (Kwon et al. 2010). The control set for analysis included 109 females and 120 males with a median age of 59.5 years. Individuals with COPD demonstrated a post-bronchodilator FEV1/FVC < 0.7, while controls had normal spirometry (Han et al. 2016). The diagnosis of asthma was made using the asthma-specific International Classification of Diseases, Tenth Revision (ICD-10) codes (J459, J450, J460, J461, J469) by Internal Medicine doctors (Kwon et al. 2016). The controls and cases with asthma or COPD were matched according to sex and age, and all participants provided written, informed consent. All samples were obtained from two different hospitals in Korea: Dong-A University Hospital (#IRB-06-10-02 & IRB-07-10-7) and Kangwon National University School of Medicine (#IRB-2012, 06-007).
Genotyping of MUC8-MS5
For genotyping, genomic DNA was isolated from peripheral leukocytes of 400 µL whole blood using a Blood and Cell Culture DNA Mini Kit (Qiagen, CA). We analyzed MUC8-MS5 VNTR polymorphisms by PCR, using the following primers: Forward (19 bp) 5′-ATGTGGGCCGGCAGGAGAC-3′; Reverse (20 bp) 5′-GGCTCCGTTTTGGGGCTGTT-3′ (Lee et al. 2009). PCR reactions (40 µL) were performed in reaction mixes containing 100 ng genomic DNA, 10 µM primers, 2.5 U Go Taq Flexi DNA polymerase (Promega, WI), 50 mM KCl, 10 mM Tris–HCl (pH 9.0), 5.0 mM MgCl2, 0.2 mM dTTP, dCTP, dGTP and dATP. PCR was performed in a 9700 Thermocycler (Perkin-Elmer, CT), and the general thermocycling conditions were as follows: 2 min initial denaturation at 94 °C, followed by 30 cycles of 45 s at 94 °C and 45 s at 68 °C, then a final 7 min extension at 72 °C. PCR products were separated by gel electrophoresis (1 V cm−1) using 2.5% MetaPhor agarose (Lonza, USA) in 1× TAE buffer.
Statistical analyses
For the association of genetic variants with asthma or COPD, we estimated odds ratios (ORs) and their 95% confidence intervals (95% CIs) using logistic regression analysis. ORs were estimated using the natural logarithm and its standard error. All tests were two-sided, with p < 0.05 considered statistically significant. Statistical analysis was performed using MS Excel with CHITEST and R statistical software (v2.5.2, http://www.r-project.org) (Kwon et al. 2010).
Results
Allelic frequency of MUC8-MS5 among the controls, asthma and COPD cases
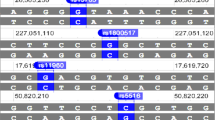
The MUC8 gene is located on chromosome 12q24.3, and we previously analyzed six VNTRs within the MUC8 region (Lee et al. 2009). It has been reported in several studies that differential expression of MUC8 was related to chronic inflammation of respiratory organs (Basbaum et al. 1999; Bernacki et al. 1999; Song et al. 2008). In a previous study, MUC8-MS5 was identified between intron 3 and exon 4, and four alleles were detected (2, 5, 9 and 29 repeats), with eight haplotype-patterns among 200 individuals (Lee et al. 2009). This region showed the highest heterozygosity among the MUC8 VNTR regions in 200 controls.
To examine the association between MUC8-MS5 and respiratory diseases, we analyzed the allelic variations of MUC8-MS5 with 229 controls, 77 asthma and 123 COPD cases (Table 1). Table 1 summarizes the frequency of allelic lengths among controls and cases. There were no significant differences for allelic frequencies between controls and the two cases. The heterozygosity of this region was approximately 0.78 in controls and cases. Compared to our previous study, we detected three additional alleles (10, 28 and 30 repeats) in this region. For further analysis, each allele was grouped into two sets (common and rare alleles) according to their frequency in controls. The expected frequency for rare alleles was set as < 1% (Kwon et al. 2010). We detected seven alleles in this study, which included five common alleles for 2, 5, 9, 29 and 30 repeats and two rare alleles for 10 and 28 repeat copies for MUC8-MS5 in controls (Table 1). The rare allele with 28 repeats was detected only in controls; this seems to depend on the sample size. There are also no significant differences between the frequency of rare alleles of controls and cases. Rare MUC8-MS5 alleles appeared at a frequency of less than 1% (0.87% in controls, 0% in COPD and 0.65% in asthma). Additionally, for each allelic frequency, similar frequency was represented between controls and cases (Table 1).
Genetic susceptibility of the short allelic length of MUC8-MS5 to COPD
Because some minisatellite alleles have been reported to be in association with human diseases (Bailly et al. 1996; Kwon et al. 2010), we analyzed the effects of allelic length of minisatellites on disease. As shown in Table 1, the length of the minisatellites in this region can be divided into short alleles (2, 5, 9 and 10 repeats) and long alleles (28 ~ 30 repeats). According to length, minisatellites can be divided into three categories: L/L, L/S and S/S (Table 2). In the analysis of allelic length, a similar frequency was shown across the three groups (controls vs. COPD, p = 0.4887; controls vs. asthma, p = 0.4699).
For further analysis, we analyzed the frequency of genotype patterns for MUC8-MS5 among control, COPD and asthma groups (Table 3). Interestingly, we found that the frequency of homozygous genotypes with the shortest rare allele (2/2 repeats) showed a significant difference between controls, COPD and asthma: these were 2.18 vs. 8.13 vs. 6.49%, respectively (Table 3). Specifically, the association between COPD cases and the homozygous 2/2 repeats genotype was represented with statistically significant higher frequency [OR 3.96; CI 1.32–11.88; p = 0.0084)]. In the case of asthma, the homozygous 2/2 repeat genotype increased 2.98-fold compared with control (OR 3.11; CI 0.88–11.05; p = 0.0657), but it could not be considered a statistically significant association. The current results show a tendency towards risk, but the sample population appears to be somewhat small for statistical analysis.
Discussion
Excessive airway mucus in asthma and COPD is a powerful inducer of chronic airway inflammation and associated lung disease, even when there is no bacterial infection (Ha and Rogers 2016). Abnormal expression of mucin genes occurs in conditions associated with various diseases and high risk of respiratory organs. Mucus was secreted by the airway surface epithelium and glands, and MUC8, one of the secretory mucins, was found to be overexpressed in diseases associated with chronic inflammatory respiratory infections (Basbaum et al. 1999; Song et al. 2008). Despite recent advances in various therapies for asthma and COPD, the treatment of advanced disease is not easy. If genetic clinical biomarkers are available, treatment interventions can be promoted before disease progression. Therefore, since polymorphic minisatelites are known within the MUC8 region, epidemiological studies are needed to determine whether these polymorphisms are associated with susceptibility to diseases such as asthma or COPD.
In this study, polymorphisms of MUC8 minisatellites were examined to investigate the genetic minisatellite allele, which represents a higher risk for respiratory disease development. As a result, in the case of the homozygous 2/2 repeat genotype, the association of COPD occurrence was significantly increased about four-fold. The number of patients examined in this study seems to be somewhat small, but this results show a link between minisatellites polymorphism and susceptibility to COPD or asthma, suggesting the need for further investigation, such as large-scale epidemiological studies based on this. Such a study could provide a helpful reference for understanding the function and complex genomic properties of mucins.
References
Bailly S, Israel N, Fay M, Gougerot-Pocidano MA, Duff GW (1996) An intronic polymorphic repeat sequence modulates interleukin-alpha gene expression. Mol Immunol 33(11–12):999–1006
Basbaum C, Lemjabar H, Longphre M, Li D, Gensch E, McNamara N (1999) Control of mucin transcription by diverse injury-induced signaling pathways. Am J Respir Crit Care Med 160:S44–S48
Bernacki SH, Nelson AL, Abdullah L, Sheehan JK, Harris A, Davis CW, Randell SH (1999) Mucin gene expression during differentiation of human airway epithelia in vitro: MUC4 and MUC5B are strongly induced. Am J Respir Cell Mol Biol 20:595–604
Byrd JC, Bresalier RS (2004) Mucins and mucin binding proteins in colorectal cancer. Cancer Metastasis Rev 23:77–99
Ha EV, Rogers DF (2016) Novel therapies to inhibit mucus synthesis and secretion in airway hypersecretory disease. Pharmacology 97:84–100
Han SS, Lee WH, Hong Y, Kim WJ, Yang J, Lim MN, Lee SJ, Kwon JW (2016) Comparison of serum biomarkers between patients with asthma and with chronic obstructive pulmonary disease. J Asthma 53(6):583–588
Hollingsworth MA, Swanson BJ (2004) Mucins in cancer: protection and control of the cell surface. Nat Rev Cancer 4:45–60
Jeffreys AJ, Wilson V, Thein SL (1985) Hypervariable ‘minisatellite’ regions in human DNA. Nature 314:67–73
Jeong YH, Kim MC, Ahn EK, Seol SY, Do EJ, Choi HJ, Chu IS, Kim WJ, Kim WJ, Sunwoo YI et al (2007) Rare exonic minisatellite alleles in MUC2 influence susceptibility to gastric carcinoma. PLoS ONE 2:e1163
Krontiris TG, Devlin B, Karp DD, Robert NJ, Risch N (1993) An association between the risk of cancer and mutations in the HRAS1 minisatellite locus. New Engl J Med 329:517–523
Kwon JA, Lee SY, Ahn EK, Seol SY, Kim MC, Kim SJ, Kim SI, Chu IS, Leem SH (2010) Short rare MUC6 minisatellites-5 alleles influence susceptibility to gastric carcinoma by regulating gene. Hum Mutat 31:943–949
Kwon JW, Han YJ, Oh MK, Lee CY, Kim JY, Kim EJ, Kim H, Kim WJ (2016) Emergency department visits for asthma exacerbation due to weather conditions and air pollution in chuncheon, Korea: a case-crossover analysis. Allergy Asthma Immunol Res 8(6):512–521
Lee JS, Kim JY, Ahn EK, Lee SY, Jeong YH, Lee SR, Leem SH (2009) Analysis of the novel VNTR polymorphisms of MUC8 gene. Genes Genom 31(3):235–241
Leem SH, Londoño-Vallejo JA, Kim JH, Bui H, Tubacher E, Solomon G, Park JE, Horikawa I, Kouprina N, Barrett JC et al (2002) The human telomerase gene: complete genomic sequence and analysis of tandem repeat polymorphisms in intronic regions. Oncogene 21:769–777
Martínez-Antón A, Debolós C, Garrido M, Roca-Ferrer J, Barranco C, Alobid I, Xaubet A, Picado C, Mullol J (2006) Mucin genes have different expression patterns in healthy and diseased upper airway mucosa. Clin Exp Allergy 36:448–457
Nadel JA (2001) Role of epidermal growth factor receptor activation in regulating mucin synthesis. Respir Res 2:85–89
Song KS, Kim K, Chung KC, Seol JH, Yoon JH (2008) Interaction of SOCS3 with NonO attenuates IL-1beta-dependent MUC8 gene expression. Biochem Biophys Res Commun 377:946–951
Thornton DJ, Rousseau K, McGuckin MA (2008) Structure and function of the polymeric mucins in airways mucus. Annu Rev Physiol 70:459–486
Vinall LE, Hill AS, Pigny P, Pratt WS, Toribara N, Gum JR, Kim YS, Porchet N, Aubert JP, Swallow DM (1998) Variable number tandem repeat polymorphism of the mucin genes located in the complex on 11p15.5. Hum Genet 102:357–366
Yoon SL, Kim DC, Cho SH, Lee SY, Chu IS, Heo J, Leem SH (2010) Susceptibility for breast cancer in young patients with short rare minisatellite alleles of BORIS. BMB Rep 43:698–703
Yuta A, Ali M, Sabol M, Gaumond E, Baraniuk JN (1997) Mucoglycoprotein hypersecretion in allergic rhinitis and cystic fibrosis. Am J Physiol 273:1203–1207
Acknowledgements
This work was supported by the Dong-A University research fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Se-Ra Lee declares that she does not have conflict of interest. Won-Tae Kim declares that he does not have conflict of interest. Tae Nam Kim declares that he does not have conflict of interest. Jong Kil Nam declares that he does not have conflict of interest. Woo Jin Kim declares that he does not have conflict of interest. Sun-Hee Leem declares that she does not have conflict of interest.
Ethical approval
Dong-A University Hospital (#IRB-06-10-02 & IRB-07-10-7) and Kangwon National University School of Medicine (#IRB-2012, 06–007) approved this study.
Rights and permissions
About this article
Cite this article
Lee, SR., Kim, WT., Kim, T.N. et al. Association between the length of the MUC8-minisatellite 5 region and susceptibility to chronic obstructive pulmonary disease (COPD). Genes Genom 40, 123–127 (2018). https://doi.org/10.1007/s13258-017-0630-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-017-0630-8