Abstract
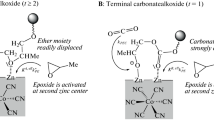
A (Salen)Co(III) complex tethering four quaternary ammonium salts with covalent bonds is one of the most highly active catalysts for CO2/epoxide copolymerization. In this work, we aimed to prepare similar (Salen)Co(III) complexes to which quaternary ammonium salts are linked via ionic interactions. Thus, multiple ammonium salts containing 2–5 quaternary ammonium salt units and (Salen)Co(III) complexes containing one or two -SO3−[PhNH3]+ moieties were prepared. A binary catalytic system composed of the prepared multiple ammonium salts and the prepared (Salen)Co(III) complex containing -SO3−[(nBu)4N]+ moieties showed high activity (TOF, 1500-4500 h−1) for CO2/propylene oxide (PO) copolymerization, whereas a combination of the multiple ammonium salts and the conventional (Salen)Co(III) complex not containing -SO3−[(nBu)4N]+ moieties was inactive under the identical polymerization conditions of [PO]/[Co] = 20000. However, a substantial amount of cyclic carbonate was concomitantly generated, especially when a (Salen)Co(III) complex containing two -SO3−[(nBu)4N]+ moieties was used (25–30%). This side-reaction could be mitigated by ca. 50% by employing a (Salen)Co(III) complex containing one -SO3−[(nBu)4N]+ moiety. Cyclic carbonate generation can reasonably be ascribed to ammonium salts ([(nBu)4N]+[carbonate]−) not linked to the (Salen)Co(III) complex formed in the binary catalytic system.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. H. Lamparelli, and C. Capacchione, Catalysts, 11, (2021).
Y. Y. Zhang, G. P. Wu, and D. J. Darensbourg, Trends Chem., 2, 750 (2020).
D. J. Darensbourg, Green Chem., 21, 2214 (2019).
C. M. Kozak, K. Ambrose, and T. S. Anderson, Coord. Chem. Rev., 376, 565 (2018).
L. Guo, K. J. Lamb, and M. North, Green Chem., 23, 77 (2021).
Y. Kim, K. Hyun, D. Ahn, R. Kim, M. H. Park, and Y. Kim, ChemSusChem., 12, 4211 (2019).
M. Hong, Y. Kim, H. Kim, H. J. Cho, M. H. Baik, and Y. Kim, J. Org. Chem., 83, 9370 (2018).
M. H. Kim, T. Song, U. R. Seo, J. E. Park, K. Cho, S. M. Lee, H. J. Kim, Y. J. Ko, Y. K. Chung, and S. U. Son, J. Mater. Chem. A, 5, 23612 (2017).
S. Inoue, H. Koinuma, and T. Tsuruta, J. Polym. Sci. B Polym. Phys., 7, 287 (1969).
J. Marbach, T. Höfer, N. Bornholdt, and G. A. Luinstra, ChemistryOpen, 8, 828 (2019).
S. Padmanaban, M. Kim, and S. Yoon, J. Ind. Eng. Chem., 71, 336 (2019).
P. Sudakar, D. Sivanesan, and S. Yoon, Macromol. Rapid Commun., 37, 788 (2016).
S. Padmanaban, and S. Yoon, Catalysts, 9, 892 (2019).
M. Ree, Y. Hwang, J. S. Kim, H. Kim, G. Kim, and H. Kim, Catal. Today, 115, 134 (2006).
S. F. Stahl, and G. A. Luinstra, Catalysts, 10, 1 (2020).
J. K. Varghese, D. S. Park, J. Y. Jeon, and B. Y. Lee, J. Polym. Sci. A Polym. Chem., 51, 4811 (2013).
C. H. Tran, S. A. Kim, Y. Moon, Y. Lee, H. M. Ryu, J. H. Baik, S. C. Hong, and I. Kim, Catal. Today, 375, 335 (2021).
Y. J. Huang, G. R. Qi, and Y. H. Wang, J. Polym. Sci. A Polym. Chem., 40, 1142 (2002).
G. W. Coates, and D. R. Moore, Angew. Chem. Int. Ed., 43, 6618 (2004).
X. B. Lu, W. M. Ren, and G. P. Wu, Acc. Chem. Res., 45, 1721 (2012).
D. J. Darensbourg, and S. J. Wilson, Green Chem., 14, 2665 (2012).
M. R. Kember, A. Buchard, and C. K. Williams, Che. Commun., 47, 141 (2011).
E. K. Noh, S. J. Na, S. S., S. W. Kim, and B. Y. Lee, J. Am. Chem. Soc., 129, 8082 (2007).
S. S., J. K. Min, J. E. Seong, S. J. Na, and B. Y. Lee, Angew. Chem. Int. Ed., 47, 7306 (2008).
S. J. Na, S. Sujith, A. Cyriac, B. E. Kim, J. Yoo, Y. K. Kang, S. J. Han, C. Lee, and B. Y. Lee, Inorg. Chem., 48, 10455 (2009).
K. Dyduch, M. Srebro-Hooper, B. Y. Lee, and A. Michalak, J. Comput. Chem., 39, 1854 (2018).
H. J. Lee, J. W. Baek, Y.H. Seo, H. C. Lee, S. M. Jeong, J. Lee, C. G. Lee, and B. Y. Lee, Molecules, 26, 2827 (2021).
J. Y. Jeon, J. K. Varghese, J. H. Park, S. H. Lee, and B. Y. Lee, Eur. J. Org. Chem., 2021, 3566 (2012).
J. Min, J. E. Seong, S. J. Na, A. Cyriac, and B. Y. Lee, Bull. Korean Chem. Soc., 30, 745 (2009).
A. Cyriac, S. H. Lee, J. K. Varghese, E. S. Park, J. H. Park, and B. Y. Lee, Macromolecules, 43, 7398 (2010).
A. J. Plajer and C. K. Williams, Angew. Chem. Int. Ed., 60, 13372 (2021).
A. C. Deacy, E. Moreby, A. Phanopoulos, and C. K. Williams, J. Am. Chem. Soc., 142, 19150 (2020).
J. Deng, M. Ratanasak, Y. Sako, H. Tokuda, C. Maeda, J. Y. Hasegawa, K. Nozaki, and T. Ema, Chem. Sci., 11, 5669 (2020).
H. Asaba, T. Iwasaki, M. Hatazawa, J. Deng, H. Nagae, K. Mashima, and K. Nozaki, Inorg. Chem., 59, 7928 (2020).
G. W. Yang, Y. Y. Zhang, R. Xie, and G. P. Wu, J. Am. Chem. Soc., 142, 12245 (2020).
G. W. Yang, C. K. Xu, R. Xie, Y. Y. Zhang, X. F. Zhu, and G. P. Wu, J. Am. Chem. Soc., 143, 3455 (2021).
C. J. Zhang, S. Q. Wu, S. Boopathi, X. H. Zhang, X. Hong, Y. Gnanou, and X. S. Feng, ACS Sustain. Chem. Eng., 8, 13056 (2020).
M. A. Mikhailov, K. A. Brylev, A. V. Virovets, M. R. Gallyamov, I. Novozhilov, and M. N. Sokolov, New J. Chem., 40, 1162 (2016).
A. Cyriac, J. Y. Jeon, J. K. Varghese, J. H. Park, S. Y. Choi, Y. K. Chung, and B. Y. Lee, Dalton Trans., 41, 1444 (2012).
E. B. Hager, B. C. E. Makhubela, and G. S. Smith, Dalton Trans., 41, 13927 (2012).
M. Holbach, X. Zheng, C. Burd, C. W. Jones, and M. Weck, J. Org. Chem., 71, 2903 (2006).
X. B. Lu, L. Shi, Y. M. Wang, R. Zhang, Y. J. Zhang, X. J. Peng, Z. C. Zhang, and B. Li, J. Am. Chem. Soc., 128, 1664 (2006).
J. Yoo, S. J. Na, H. C. Park, A. Cyriac, and B. Y. Lee, Dalton Trans., 39, 2622 (2010).
A. Roznowska, K. Dyduch, B. Y. Lee, and A. Michalak, J. Mol. Model., 26, 113 (2020).
Acknowledgment
This research was supported by the Carbon to X Program of the Ministry of Science and ICT (grant number 2020M3H7A1098281) and by the Priority Research Centers Program (2019R1A6A1A11051471) through the National Research Foundation of Korea (NRF).
Author information
Authors and Affiliations
Corresponding author
Additional information
The image from this article is used as the cover image of the Volume 29, Issue 12.
Supporting information
Information is available regarding 1H NMR and 13C NMR spectra of new compounds via the Internet at http://www.springer.com/13233.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supporting Information
Rights and permissions
About this article
Cite this article
Seo, Y.H., Hyun, Y.B., Lee, H.J. et al. CO2/Propylene Oxide Copolymerization with a Bifunctional Catalytic System Composed of Multiple Ammonium Salts and a Salen Cobalt Complex Containing Sulfonate Anions. Macromol. Res. 29, 855–863 (2021). https://doi.org/10.1007/s13233-021-9094-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-021-9094-4