Abstract
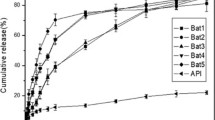
Patients at risk for hypertension with comorbidities such as diabetes and metabolic syndrome often require two or more antihypertensive drugs to lower their blood pressure. Telmisartan and amlodipine are widely known drugs to treat hypertension. However, telmisartan suffer of poor solubility in water that makes necessary to increase its dosage to reach a sufficient therapeutic concentration. In this study, a solid dispersion containing a water-soluble polymer was prepared to make the absorption rate of telmisartan similar to that of amlodipine, a water soluble drug. As water-soluble polymers, polyvinylpyrrolidone K30, polyethylene glycol 6000, and poloxamer 407 were used. The encapsulation of the solid dispersion was analyzed by differential scanning calorimetry, Xray diffraction, and high-performance liquid chromatography. The reduced flowability of the solid dispersion was improved by a wet granulation. Scanning Electron Microscopy was used to study the morphology and the flowability was verified by measuring the angle of repose, bulk density, and tap density, and expressed by Hausner ratio and Carr index. The release behavior was confirmed by dissolution test and high-performance liquid chromatography. As result we proved that telmisartan a higher release rate when encapsulated with PVP K30. Finally, we designed a drug formulation in which amlodipine and telmisartan could be absorbed at a similar rate.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
R. P. Ames and P. B. Hill, Am. J., 61, 748 (1976).
P. Ernsberger, R. J. Koletsky, L. A. Collins, and D. Bedol, Cardiovasc. Drugs Ther., 10, 275 (1996).
M. M. Ibrahim, J. Hum. Hypertens., 20, 101 (2006).
A. K. Halperin and L. X. Cubeddu, Am. Heart J., 111, 363 (1986).
Y. H. Kim, H. S. Lim, S. H. Cho, J. L. Ghim, S. Choe, J. A. Jung, and K. S. Bae, J. Korean. Soc. Clin. Pharmacol. Ther., 21, 95 (2013).
W. Liu, W. Wang, S. W. Song, X. F. Gu, X. J. Ma, F. Y. Su, H. Zhang, A. J. Liu, and D. F. Su, J. Cardiovasc. Pharmacol., 57, 308 (2011).
M. D. Moen, Am. J. Cardiovasc. Drugs, 10, 401 (2010).
D. R. Abernethy, Am. Heart J., 118, 1100 (1989).
A. Dubey, A. A. Kharia, and D. P. Chatterjee, Int. J. Pharm. Sci., 5, 4478 (2014).
P. A. A. Borba, M. Pinotti, C. E. Campos, B. R. Pezzini, and H. K. Stulzer, Carbohydr. Polym., 137, 350 (2016).
A. A. Faruqui, J. Indian. Med. Assoc, 106, 612 (2008).
M. C. Eswaraiah, and S. Jaya, Res. J. Pharm. Technol., 13, 2217 (2020).
J. Park, W. Cho, K. H. Cha, J. Ahn, K. Han, and S. J. Hwang, Int. J. Pharm., 441, 50 (2013).
B. Patel, R. H. Parikh, and D. Swarnkar, J. Pharm. Bioallied. Sci., 5, 4478 (2014).
J. S. Chae, B. R. Chae, D. J. Shin, Y. T. Goo, E. S. Lee, H. Y. Yoon, C. H. Kim, and Y. W. Choi, AAPS PharmSciTech, 19, 2990 (2018).
W. K. Kim, J. S. Kim, M. E. Shin, J. S. Baek, D. Kim, A. Bucciarelli, J. E. Song, and G. Khang, Macromol. Res., 28, 553 (2020).
W. K. Kim, H. H. Cho, G. W. Lee, Y. W. Jeong, J. S. Kim, A. Bucciarelli, J. E. Song, and G. Khan., Macromol. Res., 28, 417 (2020).
E. Y. Shin, J. H. Park, M. E. Shin, J. E. Song, and G. Khang, Polym., 43, 274 (2019).
V. Dhiman, G. Jain, V. Jagtap, and R. V. Sheorey, Int. J. Pharm. Pharm. Res., 3 (2012).
S. Kiortsis, K. Kachrimanis, Th. Broussali, and S. Malamataris, Eur. J. Pharm. Biopharm., 59, 73 (2005).
E. Erica, O. Jasmine, P. Todd, S. Jitendra, and Z. Joe, Powder Technol., 189, 409 (2009).
A. Crouter and L. Briens, AAPS PharmSciTech, 15, 65 (2014).
T. Hao, RSC Adv., 5, 57212 (2015).
L. R. Bhat, R. K. Godge, A. T. Vora, and M. C. Damle, J. Liq. Chromatogr. Relat. Technol., 30, 3059 (2007).
A. Saxena, K. Sachin, H. B. Bohidar, and A. Verma, Colloids Surf. B, 45, 42 (2005).
X. Xu, M. A. Khan, and D. J. Burgess, Int. J. Pharm., 423, 410 (2012).
D. Train, J. Pharm. Pharmacol., 10, 127T (1958).
A. Saker, M. G. C. Pacheco, P. Marchal, and V. Falk, Powder Technol., 354, 52 (2019).
A. Mohammadi, N. Rezanour, M. A. Dogaheh, F. G. Bidkorbeh, M. Hashem, and R. B. Walker, J. Chromatogr. B: Biomed. Appl., 846, 215 (2007).
D. Patil, A. Ahmed, M. Awais, S. Akhtar, and V. Bairagi, Pharma Sci. Monit., 9, 171 (2018).
S. B. Tan and J. M. Newton, Int. J. Pharm., 61, 145 (1990).
R. L. Carr, Chem. Eng., 18, 163 (1965).
P. Jain, A. Goel, S. Sharma, and M. Parmar, Int. J. Pharm. Sci. Res., 1, 34 (2010).
K. D. Ertel and J. T. Carstensen, J. Pharm. Sci., 77, 625 (1988).
S. Paul and C. C. Sun, Powder Technol., 309, 126 (2017).
Acknowledgments
This research was supported by Basic Science Research Program (2020R1A2C2103089) and the bilateral cooperation Program (2019K2A9A1A06098563) of the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT & Future Planning.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Been, S., Choi, J., Kim, P.Y. et al. Release Behavior of Telmisartan/Amlodipine Combination Drug According to Polymer Type. Macromol. Res. 29, 217–223 (2021). https://doi.org/10.1007/s13233-021-9029-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-021-9029-0