Abstract
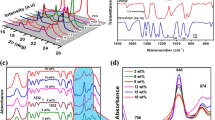
A new series of magnesium-ion conducting solid polymer blend electrolytes based on an optimized blend ratio (90:10) of poly(ethylene oxide) (PEO) and poly(vinylidene fluoride) (PVDF) doped with different concentrations of magnesium bis (trifluoromethane sulfonimide) salt, Mg [(CF3SO2)2N2)] has been prepared by solution casting technique, using dimethylformamide (DMF) as the common solvent. The increase in the amorphous phase with an increase in salt concentration of the prepared blended polymer electrolytes has also been nurtured from the results obtained from X-ray diffraction (XRD) and scanning electron microscopic (SEM) analyses. The electrical transport characteristics were evaluated by means of electrochemical impedance spectroscopy (EIS) and the maximum ionic conductivity obtained at room temperature (298 K) was found to be 1.2×10-5 S cm-1 in the case of the blend containing 15 wt% salt. A detailed investigation concerning the mechanism of magnesium ion transport in the optimized polymer blend electrolyte over the frequency range of 1 MHz to 20 Hz has also been carried out in terms of electrical conductivity spectra, dielectric properties and electrical modulus spectra at room temperature.

Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. Armand and J. M. Tarascon, Nature, 451, 652 (2008).
J. M. Tarascon and M. Armand, Nature, 414, 359 (2001).
W. H. Meyer, Adv. Mater., 10, 439 (1998).
O. Chusid, Y. Gofer, H. Gizbar, Y. Vestfrid, and E. Levi, Adv. Mater., 15, 627 (2003).
S. Jeremias, G. A. Giffin, A. Moretti, and S. Jeong, J. Phys. Chem. C, 118, 28361 (2014).
H. S. Kim, T. S. Arthur, G. D. Allred, and J. Zajicek, Nat. Commun., 2, 427 (2011).
D. Aurbach, A. Schechter, O. Chusid, and H. Gizbar, J. Power Sources, 28, 97 (2001).
A. Magistris and K. Singh, Polym. Int., 28277 (1992).
J. Xia, X. Qiua, J. Lib Tang, and W. Zhu, J. Power Sources, 157, 501 (2006).
G. G. Kumar and N. Munichandraiah, J. Power Sources, 102, 46 (2001).
G. P. Pandey and S. A. Hashmi, J. Power Sources, 187, 627 (2009).
A. R. Polu, R. Kumar, and K. V. Kumar, Adv. Mater. Lett., 3, 406 (2012).
G. G. Kumar and N. Munichandraiah, Electrochim. Acta, 44, 2663 (2001).
G. G. Kumar, and N. Munichandraiah, Electrochim. Acta, 47, 1013 (2002).
M. Morita, N. Yoshimoto, and S. Yakushiji, Electrochem. Solid-State Lett., 4, A177 (2001).
H. D. Yoo, I. Shterenberg, and Y. Gofer, Energy Environ. Sci., 6, 2265 (2013).
R. Mohtadi and F. Mizuno, Beilstein J. Nanotechnol., 5, 1291 (2014).
C. Liebenowa, Electrochim. Acta, 43, 1253 (1998).
D. Aurbach, Z. Lu, A. Schechter, and Y. Gofer, Nature, 407, 724 (2000).
D. Aurbach, I. Weissman, and Y. Gofer, Chem. Rec., 3, 61 (2003).
T. T. Tran, W. M. Lamanna, and M. N. Obrovac, J. Electrochem. Soc., 159, A2005 (2012).
D. Aurbach, A. Schechter, and M. Moshkovich, J. Electrochem. Soc., 148, A1004 (2001).
Y. Shaoa, N. N. Rajput, H. M. Hua, and T. Liu, Nano Energy, 12, 750 (2015).
N. Yoshimoto, S. Yakushiji, and M. Ishikawa, Electrochim. Acta, 48, 2317 (2003).
J. S. Oh, J. M. Ko, and D. W. Kim, Electrochim. Acta, 50, 903 (2004).
N. H. Zainol, Z. Osman, and L. Othman, Adv. Mater. Res., 686, 137 (2013).
A. K. Jonscher, Dielectric Relaxation in Solids, Chelsea Dielectrics Press, London, 1983.
S. A. Suthanthiraraj, R. Kumar, and B. J. Paul, J. Solid State Electrochem., 15, 561 (2011).
V. D. Noto, M. Vittadello, and S. Lavina, J. Phys. Chem. B, 105, 4584 (2001).
M. Piccolo, G. A. Giffin, and K. Vezzu, ChemSusChem, 6, 2157 (2013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rathika, R., Suthanthiraraj, S.A. Ionic Interactions and Dielectric Relaxation of PEO/PVDF-Mg[(CF3SO2)2N2)] Blend Electrolytes for Magnesium Ion Rechargeable Batteries. Macromol. Res. 24, 422–428 (2016). https://doi.org/10.1007/s13233-016-4053-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-016-4053-1