Abstract
Introduction
Intrauterine insemination (IUI) is one of the most commonly performed procedure of assisted reproductive technology, for the treatment of infertility. Controlled ovarian hyperstimulation is an important first step while performing IUI. This study aims at establishing a relationship between stimulation protocol and pregnancy outcome following IUI.
Methods
This is a retrospective study of 1001 cycles of IUI in which the patients were divided into two groups: Group A Clomiphene citrate (CC only) and Group B Clomiphene citrate and Gonadotropin or Gonadotropin alone(CC+GN OR GN alone). The primary outcome assessed was clinical pregnancy rates (CPRs), and the secondary outcomes were miscarriage rate (MR), multiple pregnancy rates (MPRs), follicle numbers and endometrial thickness (ET).
Results
Significantly, higher CPR was observed in Group B in comparison with Group A (14.55 vs. 7.82%; p = 0.05). MR was much higher in Group A in comparison with Group B, (14.29 vs. 5.43%; p = 0.94), but it was non-significant. The follicle number and the ET of the Group A versus Group B are (1.54 ± 0.69 vs. 1.90 ± 1.04; p = 0.0003) and (8.56 ± 1.33 vs. 8.39 ± 1.29; p = 0.1784), respectively; and for subgroups, Group B1 and Group B2 are 1.92 ± 0.99 versus 1.65 ± 0.92; p = 0.0008 and 8.32 ± 1.27 vs. 8.69 ± 1.24; p = 0.0004, respectively.
Conclusion
GN, either alone or the combination with CC, gives a higher CPR and a lower abortion rate following IUI, thus increasing the multiple pregnancy rate.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Infertility, a worldwide health issue, is the inability of a couple to conceive, despite unprotected intercourse for the period of 1 year. Most common causes for infertility include mild endometriosis, male factor, unexplained infertility, ovulatory dysfunction and ageing [1, 2]. Intrauterine insemination (IUI) is the most performed treatment in women with infertility owing to mild male factor infertility, anovulation, endometriosis with at least one patent tube and unexplained infertility [3,4,5].
Simplicity, easy management, low cost and absence of potentially serious complications make IUI a leading routinely used technique for infertility [3, 6, 7]. Further, development in the type of stimulation protocols, gonadotropins, ultrasound monitoring and sperm preparation techniques has enhanced the success rate of IUI.
IUI may be performed in natural cycle as well as in combination with controlled ovarian hyperstimulation (COH) [6]. A higher CPR was observed in the latter cases in some studies [8], thus leading to use of potential ovarian stimulants such as clomiphene citrate (CC). Higher pregnancy rates are observed in patients who were given CC [5, 9]. Bae and co-workers observed a higher rate of pregnancy in patients administered with 3-day treatment of CC than those given 5-day treatment with CC. This was believed to be due to an adequate endometrial growth by CC in the 3-day treatment [9]. Gonadotropins (GN) such as human menopausal gonadotropin (hMG), follicle stimulating hormone (FSH) have also been used as an alternative source for COH [6, 10,11,12]. GNs although more responsive in COH than CC are expensive and have certain associated adverse effects (AE) [ovarian hyperstimulation syndrome (OHSS) and high-order multiple pregnancies]. Gonadotropins being highly responsive and effective are used in COH [5]. Using a combination of CC and GN, COH and IUI can be made a cost-effective and efficient process.
In this study, we assessed the efficacy and safety of CC, GN alone and in combination with CC for COH in infertile female patients undergoing IUI. The study aimed at establishing a standard ovulation stimulation protocol including the type and dose of the ovarian stimulants (including CC and GNs) to be used combined with IUI to treat infertility.
Materials and Methods
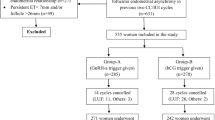
This was a retrospective, observational study conducted from 1 November 2014 to 29 February 2016 under good clinical practices as per the International Conference on Harmonization (ICMR, 2006) guidelines and the Declaration of Helsinki [13]. Women unable to conceive due to ovulatory dysfunction, mild male factors, unexplained infertility, mild endometriosis, etc. were included in the study. Following the physical examination and medical history collection, baseline scan for the female patients (willing to participate) was performed on Day 2 or Day 3. The patients were divided into two groups: Group A received CC and Group B received GN [human menopausal gonadotropin (hMG)]. The patients in Group B were subdivided into two subgroups: Group B1 received GNs (hMG) plus CC and Group B2 received GN only. Figure 1 represents the patient disposition. The dosage details are given in Fig. 2.
Post-semen collection by masturbation and liquefaction (at room temperature), it was analysed for pre-wash count and motility under microscope. The concentrate of motile sperms was obtained using either swim-up or density gradient technique and was re-analysed for post-wash count. A final volume of 0.5 ml was used for insemination.
IUI was performed 38 h after administration of injection hCG (Injection Ovitrelle® 250 µg, Merck Sereno) using the treated semen samples. Urine pregnancy test was performed to confirm pregnancy if the patients did not have their periods within 2 weeks, and if positive, sonography was performed 1 week later. The CPR was the primary outcome, while miscarriage rate (MR), multiple pregnancy rate (MPR), endometrial thickness and follicle number were the secondary outcomes. These were compared among different patient groups using SPSS version 19.0 (IBM Corporation, USA).
Results
A total of 1001 patients were distributed into three different groups depending on the type of the treatment given. Table 1 gives the number of patients with different etiologies for infertility among the two patient groups, which was found to be non-significant (p = 0.1161).
CPR varied significantly between Group A (n = 9) and Group B (n = 129). It was lower in Group A versus Group B (7.82% vs. 14.55%); (p = 0.05). The subgroup analysis of Group B1 (n = 101) and Group B2 (n = 28) showed comparable CPR, i.e. (14.44% vs. 14.97%, respectively); (p = 0.85) (Table 2).
MR was higher in Group A patients (n = 1) in comparison with Group B (n = 7) (14.29 vs. 5.43%; p = 0.94); but the difference was not significant. The test of significance could not be performed in the groups B1 (6/129, 4.65%) and B2 (1/129, 0.077%) due to a small sample size.
While no multiple pregnancies were observed in Group A, the MPR was 13.17% in Group B. Among a total 17 patients exhibiting multiple pregnancy, 11 and six patients belonged to Group B1 and B2, respectively, which was non-significant (10.89 vs. 21.42%; p = 0.14). Thus, MPR was higher in patients receiving GN alone.
Group A had a lower number of follicles than Group B (1.54 ± 0.69 vs. 1.90 ± 1.04; p = 0.0003) which was found to be statistically significant, while Group B1 had a higher number of follicles than Group B2 (1.92 ± 0.99 vs. 1.65 ± 0.92; p = 0.0008). The difference in endometrial thickness was non-significant between groups (Group A: 8.56 ± 1.33 vs. Group B: 8.39 ± 1.29; p = 0.17). The comparison between the subgroups showed that the follicle number (1.92 ± 0.99 vs. 1.65 ± 0.92; p = 0.0008) was significantly higher in Group B1 than in Group B2. While, endometrial thickness (8.32 ± 1.27 vs. 8.69 ± 1.24; p = 0.0004) was significantly higher in Group B2 (Table 3).
Discussion
The effectiveness of different protocols for COH that can be used in combination with IUI was assessed in the current study in terms of primary (CPR) and secondary outcomes (MR, MPR, follicle numbers and endometrial thickness).
In the present study, CPR was significantly higher in Group B patients who received GN alone or in combination with CC, in comparison with Group A patients, i.e. 14.55% and 7.82% (p = 0.05), who received CC alone. CPR was comparable between the groups (Group B1 vs. Group B2) receiving GN + CC or GN alone (14.44 vs. 14.97%; p = 0.86).
Previous studies have reported varying outcomes on following use of CC and hMG alone and in combination. Deshpande et al. [7] reported 13.89%, 15.58% and 7.43% CPR, in patient groups treated with hMG + CC, hMG only and CC only, respectively. The pregnancy rate was higher when low dose of hMG (75 IU) was administered either alone or in combination with CC (15.58% and 13.89%, respectively). Similar conclusion was drawn by Kamath et al. [10].
The per cycle pregnancy rates for CC + IUI, FSH + IUI and IVF were 7.6%, 9.8% and 30.7%, respectively, obtained during the Fast Track and Standard Treatment Trial (FASTT) study [14]. Recently, Karadag et al. [15] observed a higher CPR in the GN group than in the CC group. The higher pregnancy rates after using GN could be due to a better follicular development, development of multiple follicles and good endometrial thickness.
Recently, in a study conducted by Diamond et al. [16] in women with unexplained infertility aged 18–40 years, the CPRs in the patient groups given GN alone versus CC alone were 35.5% versus 28.3%, respectively. The multiple gestation rates were higher in GN only group (31.8%; p = 0.006) than that in CC only group (9%; p = 0.44). Mostly, twins (4, 1.3%) were found to be born in CC only group, while there were 25 twins (8.3%) and 6 (2%) triplets in GN only treatment group [16]. This could possibly be due to multiple follicular development and multiple ovulation. The present study also has demonstrated multiple follicles with the use of GNs.
Mukherjee and co-workers conducted a prospective, randomized study comparing the effect of the single dose of uFSH + CC with only CC. The CPR observed was 17 versus 8.3% (p = 0.0001) in patient groups administered with a combination of uFSH + CC versus CC only, respectively. The difference in MR was observed (p = 0.99). MR was higher in Group A patients (n = 1; 14.29%) than Group B (n = 7/129; 5.43%); but the difference was not significant (p = 0.94). The test of significance could not be performed in the groups B1 (6/129; 4.65%) and B2 (1/129; 0.077%) due to a small sample size. Higher MR in CC group can be due to inappropriate endometrial thickness due to its anti-oestrogenic nature.
In an open labelled, randomized study by Peeraer et al. [17], CPR in hMG only versus CC only patient groups was 14.4% (48/334) versus 9% (29/323). The same study reported 8.5 versus 7.5 mm endometrial thickness in hMG versus CC patients groups, respectively (p < 0.001). The CPR and endometrial thickness in our study were 14.97 versus 7.82% and 8.69 ± 1.24 versus 8.56 ± 1.33 mm in the GN (hMG) only (Group B2) versus CC (Group A) only groups, respectively. Thus, the results were in agreement with the study by Peeraer and co-workers.
Denkart and co-workers compared the CPR, MPR and live births in patient groups (with unexplained infertility and male subfertility) given CC only and FSH only. The respective outcome rates were 38% and 34.3%, 7.4% and 4.3% and 70.4 and 73.9%, in CC only and FSH only groups, respectively [18]. The dose given in the current study was almost same, for all the treatment groups, as in the above two studies, viz., GN alone group was given 75 IU daily from Day 2/3 of natural cycle; CC + GN group was administered with 50 mg CC from Day 2 to Day 6 and 75 IU GN on alternate days as Day 3, Day 5 and Day 7. The comparable rates for the outcomes assessed were observed in the above two studies and the present study.
Karadag et al. [15] observed no multiple pregnancies in both the patient groups. However, in the present study, no cases of multiple pregnancies were observed in CC alone patient group, while a higher rate was seen in hMG alone group (21.42%) than in CC + hMG group (10.89%).
However, in a study by Abdelazim and co-workers showed contrasting results to the present study wherein CPR was higher in CC + hMG versus hMG only group, 26.7% versus 6.7%, respectively. Although, no significant differences were found in the outcomes such as endometrial thickness and number of follicles [19]. Whereas, in the present study significant differences were observed in the endometrial thickness (8.35 ± 1.44 mm vs. 8.63 ± 1.43 mm in CC + hMG vs. hMG only group, respectively) and number of follicles (2.01 ± 0.99 vs. 1.69 ± 0.94 in CC + hMG vs. hMG only group, respectively) for the concerned groups.
Thus, based on the findings of present study and the past research, we could state that the protocol followed in our study could be used as an effective, standard protocol for COH with IUI. The stepwise protocol given in Fig. 2 should be followed.
Conclusion
GN, either alone or in combination with CC, gives a higher CPR and a lower MR following IUI, however, increasing the MPR. Thus, GN or the combination of GN and CC can be used for an effective COH in combination with IUI for improved CPRs.
References
Kumar S, Mohsen N, Malini S. Association of obesity with male infertility among infertile couples is not significant in Mysore, South India. Adv Stud Biol. 2013;5:319–25.
Shadap A. Causes of infertility among married women—a review. SMU Med J. 2015; 2: 347–56.
Soria M, Pradillo G, García J, et al. Pregnancy predictors after intrauterine insemination: analysis of 3012 cycles in 1201 couples. J Reprod Infertil. 2012;13:158–66.
Merviel P, Heraud MH, Grenier N, et al. Predictive factors for pregnancy after intrauterine insemination (IUI): an analysis of 1038 cycles and a review of the literature. Fertil Steril. 2010;93:79–88.
Chaudhary K, Suri V, Dhaliwal LK, et al. Comparison of the efficacy of letrozole and low-dose gonadotropin combination with clomiphene and low-dose gonadotropin combination as a controlled ovarian stimulation regime prior to intrauterine insemination in patients with unexplained infertility. Fertil Sci Res. 2014;1:98.
Koli P, Anil M, Ramya N, et al. Intrauterine insemination: a retrospective review on determinants of success. Int J Reprod Contracept Obstet Gynecol. 2013;2:311–4.
Deshpande SA, Deshpande AB. Intra uterine insemination an experience in rural population. IJMRHS. 2013;2:341–9.
Liu J, Chian R, Ma X, et al. The pregnancy outcomes comparison on natural or controlled ovarian stimulation cycles in intrauterine insemination treatment: an analysis of 8,893 cycles. La Prensa Med 2016;2016: 1–6.
Bae S-A, Joo J-K, Choi J-R, et al. Clinical outcomes of three-or five-day treatment with clomiphene citrate combined with gonadotropins and a timed intercourse cycle in polycystic ovary syndrome patients. CERM. 2015;42:106–10.
Kamath MS, Bhave P, Aleyamma T, et al. Predictive factors for pregnancy after intrauterine insemination: a prospective study of factors affecting outcome. J Hum Reprod Sci. 2010;3:129.
Kaser DJ, Goldman MB, Fung JL, et al. When is clomiphene or gonadotropin intrauterine insemination futile? Results of the Fast Track and Standard Treatment Trial and the Forty and Over Treatment Trial, two prospective randomized controlled trials. Fertil Steril. 2014;102(1331–1337):e1331.
Rashidi BH, Tanha FD, Rahmanpour H, et al. Luteal phase support in the intrauterine insemination (IUI) cycles: a randomized double blind, placebo controlled study. J Family Reprod Health. 2014;8:149.
Hurst SA. Declaration of Helsinki and protection for vulnerable research participants. JAMA. 2014;311:1252.
Reindollar RH, Regan MM, Neumann PJ, et al. A randomized clinical trial to evaluate optimal treatment for unexplained infertility: the Fast Track and Standard Treatment (FASTT) trial. Fertil Steril. 2010;94:888–99.
Karadag B, Dilbaz B, Karcaaltincaba D, et al. The effect of luteal-phase support with vaginal progesterone on pregnancy rates in gonadotropin and clomiphene citrate/intra-uterine insemination cycles in unexplained infertility: a prospective randomised study. J Obstet Gynaecol. 2016;36:794–9.
Diamond MP, Legro RS, Coutifaris C, et al. Letrozole, gonadotropin, or clomiphene for unexplained infertility. N Engl J Med. 2015;373:1230–40.
Peeraer K, Debrock S, De Loecker P, Tomassetti C, Laenen A, Welkenhuysen M, Meeuwis L, Pelckmans S, Mol BW, Spiessens C, De Neubourg D, D'Hooghe T. M. Low-dose human menopausal gonadotrophin versus clomiphene citrate in subfertile couples treated with intrauterine insemination: a randomized controlled trial. Human Reprod. 2015;30(5):1079–88.
Dankert T, Kremer J, Cohlen B, et al. A randomized clinical trial of clomiphene citrate versus low dose recombinant FSH for ovarian hyperstimulation in intrauterine insemination cycles for unexplained and male subfertility. Hum Reprod. 2007;22:792–7.
Abdelazim IA, Makhlouf HH. Sequential clomiphene citrate/hMG versus hMG for ovulation induction in clomiphene citrate-resistant women. Arch Gynecol Obstet. 2013;287:591–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Manish Banker, Director, in the Nova IVI Fertility Clinic, Ahmedabad, India; Azadeh Patel, Associate consultant, in the Nova IVI Fertility Clinic, Ahmedabad, India; Ashwini Deshmukh, Former Fellow, in the Nova IVI Fertility Clinic, Ahmedabad, India; Sandeep Shah, Senior consultant, in the Nova IVI Fertility Clinic, Ahmedabad, India.
Rights and permissions
About this article
Cite this article
Banker, M., Patel, A., Deshmukh, A. et al. Comparison of Effectiveness of Different Protocols Used for Controlled Ovarian Hyperstimulation in Intrauterine Insemination Cycle. J Obstet Gynecol India 68, 65–69 (2018). https://doi.org/10.1007/s13224-017-1054-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-017-1054-8