Abstract
Purpose
Present study aims to examine the relationship of the bone mineral density (BMD) with body mass index (BMI) in an apparently healthy premenopausal and postmenopausal north Indian female population.
Methods
Two hundred and fifty-five apparently healthy women were evaluated by a detailed questionnaire including all possible factors that could affect the BMD. The height and weight of all the subjects were recorded and BMI was calculated. Bone mineral density was measured by DXA (Dual Electron X-ray Absorptiometry) at L1-L4 Lumbar spine (LS) and femur neck (FN) by using the Prodigy DXA system (software version:8.50) manufactured by GE medical systems LUNAR.
Results
The population was divided into pre and postmenopausal groups. The BMD at all sites increased with BMI in both groups. In a multiple regression model, the attributability of BMI and age to variation in LSBMD and FNBMD was 10.3 and 13.9 %, respectively. In postmenopausal women, age, BMI, and a previous history of fracture together attributed to a variance in BMI of 40.1 and 27.6 % at lumbar spine and femur neck, respectively. BMI was found to be significantly associated with low BMD at both sites in premenopausal women, while the association was lost in postmenopausal females.
Conclusion
BMI is an important determinant of BMD in Indian females. However, the association is not present in postmenopausal women with osteopenia. In addition, the effect of increase in BMI on BMD has a ceiling effect, and moderate to morbid obesity might not actually be a preventive factor for osteopenia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is defined as a systemic skeletal disease characterized by low bone mass and micro-architectural deterioration with a consequent increase in bone fragility with susceptibility to fracture [1]. Osteoporosis is a major problem of health care delivery services in both developed and developing countries. The first normative reference database of bone mineral density (BMD) in the Indian women and men was established using digital x-ray radiometry. Further analysis of this database revealed that 29.9 % of women and 24.3 % of men between the age of 20 and 79 years had low bone mass. About 50 % women and 36 % of men over 50 years of age had low bone mass, suggesting a higher prevalence of low bone mass in the Indian population compared to the west [2].
Several risk factors affect the BMD including age, sex, diet, menopausal status, etc. However, the effect of body mass index (BMI) on BMD is variable in different ethnic groups. Currently, there is a paucity of data on the relationship between the two in the Indian population. The present study aims to examine the relationship of the BMD with a change in BMI in an apparently healthy premenopausal and postmenopausal north Indian population.
Materials and Methods
The two hundred and seventy-three subjects in this study were the same cohort of healthy normal women who participated in a previous cross-sectional study [3].
Subjects with history of current or past chronic diseases like Cushing’s disease, hyperparathyroidism, thyrotoxicosis, and renal disease or those with history of drug intake like anticonvulsants, corticosteroids, thiazides, or thyroxine were excluded. In addition, the women using oral contraceptives, postmenopausal hormone replacement therapy, other drugs to prevent or treat osteoporosis and pregnant, and lactating women were excluded. Menopause was defined as previous natural cessation of menstruation for more than 12 months [4].
All women who gave their consent to participate were evaluated by a questionnaire. It dealt with the detailed demographic, menstrual, and obstetric history and other factors that could affect the BMD. The institutional ethics committee approved the study.
The height and weight of all the subjects were recorded. The woman’s standing height was measured by using a wall mounted stadiometer in centimeters to the nearest 1 mm. The weight was recorded in kilograms to the nearest 0.5 kg (with clothes, without shoes). The same observer took all the measurements. BMI in kg/m2 was calculated by dividing the baseline weight in kilograms by height in meters squared.
The subjects were classified into four groups according to their BMI [5]: underweight (U), when BMI was less <18 kg/m2; normal (N), when BMI was between 18 and 22.9 kg/m2; overweight (OW) with BMI between 23 and 24.9 kg/m2; and obese, when BMI was more than 25 kg/m2.
Bone mineral density was measured by DXA (Dual Electron X-ray Absorptiometry) at L1-L4 Lumbar spine (LS) and femur neck (FN) by using the Prodigy DXA system (software version:8.50) manufactured by GE medical systems LUNAR.
In this system, NHANES/USA population aged 20–40 years was used for anteroposterior (AP) spine and femur reference BMD and 68 % of repeat scans fall within ISD (±0.010 g/cm2) for AP spine L1–L4 and ±0.012 g/cm2 for total femur). Those subjects with lumbar osteophytes or deformities were excluded.
BMD values were classified according to WHO criteria: BMD within 1 standard deviation (SD) of reference mean based on normal values for a young adult was considered normal, more than 1 SD below mean was taken as low BMD (including both osteopenia and osteoporosis) [6].
Statistical Analysis
The subjects selected were divided into 2 different cohorts, first of young menstruating females and second cohort of postmenopausal females. Student’s t test was applied to compare continuous data between the two groups. Qualitative data were compared using Chi- square or Fisher’s exact test wherever applicable. Bonferroni’s post-hoc comparison tests were used at the second stage of the analysis of variance (ANOVA) if the null hypothesis was rejected. The question of interest at this stage was which subgroups significantly differed from others with respect to the mean. This quantified the difference between individual subcategories of BMI. The relationship between continuous variables like BMD and BMI with respect to other risk factors was analyzed by applying multiple regression analysis. The population was further subdivided into normal and low BMD and the relationship with BMI was analyzed using logistic regression analysis. P < 0.05 was considered significant.
Results
Of the population, screened 14 patients had hypothyroidism, two were on hormone replacement therapy, and two were on anticonvulsants. The data of 160 premenopausal and 95 postmenopausal women were analyzed.
The mean BMI of the study population was 24.2 ± 4.7 kg/m2. Of these, 21(8.23 %)) were underweight, 139(54.51 %) had a normal BMI, 64(25.09 %) of subjects were overweight, and 31(12.15 %) belonged to the obese category.
The descriptive features of the participants have been shown in Table 1.
The association between BMD and the various sub categories of BMI was studied in the overall population, premenopausal, and postmenopausal women, at femur neck and lumbar spine. Furthermore, the post-hoc test was applied to study the difference in BMD between the various sub categories of BMI. It was found that, BMD at both sites increased with BMI in the study population but the association was not statistically significant at lumbar spine in premenopausal women (Table 2).
Multiple regression analysis was applied to assess the individual affect of BMI on BMD in the presence of other factors like age and previous history of fracture which were also affecting the BMD significantly. It was found that the attributability of BMI and age to variation in LSBMD and FNBMD was 10.3 and 13.9 %, respectively. In postmenopausal women, age, BMI, and a previous history of fracture together attributed to a variance in BMI of 40.1 and 27.6 % at lumbar spine and femur neck, respectively (Table 3).
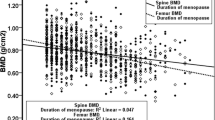
Table 4 depicts the analysis done on the population with low BMD. When the study population was divided into either a low BMD or a normal BMD and then the logistic regression analysis was applied to determine the association, it was found that BMI was an important determinant of low BMD in the premenopausal age group along with age. However, in the postmenopausal age group, the significant determinants of low BMI were a previous history of fracture and the years since menopause. BMI did not seem to be associated with a low BMD.
Discussion
There are a number of studies on the effect of BMI on BMD. However, as ethnicity may modify the relation between the two, this is the first study on the relationship between BMI and BMD in the Indian population.
On univariate analysis, BMD increased with BMI at both lumbar spine and femur neck. On multiple regression, BMD along with other factors like age and a previous history of fracture was an important determinant of BMD. However, when a targeted population of only low BMD was taken, the association with BMD was lost in the postmenopausal group. This implies that the relationship between the two is much more complex than can be easily assumed in the Indian population.
Preventing osteoporosis, a multifactorial disease, requires not only recognizing its risk factors, but also identifying the potentially modifiable determinants of bone mineral density (BMD) [7]. Among postmenopausal women of Caucasian descent, there is sufficient evidence to suggest that moderate obesity is a protective factor in osteoporosis [8]. In this high-risk group, BMI has been shown to be positively correlated with BMD [8–10]. As BMI increases, BMD increases while the rate of bone loss decreases [11]. It is unclear as to what extent ethnicity plays a role in modifying the effect of BMI on BMD. The National and regional surveys in the United States demonstrate significant ethnic differences with higher incidence of hip fracture in Caucasian women compared with African American and Mexican–American women [12, 13]. In another study, with each unit increase in BMI, BMD increases for White women, while a slight but significant decrease in BMD occurs in African American women [14]. We found that the BMD was positively correlated to the BMI in a representative subpopulation of Indian women.
The pathogenesis of postmenopausal osteoporosis is complex and incompletely understood. In a study by Lim et al. [15], BMI was not a determinant of postmenopausal BMD in multiple regression analysis in a population of Korean women. They found that age, years since menopause and possibly a genetically determined alteration of hydroxylation of estrogen could be important for determination of BMD in postmenopausal osteopenia. The loss of association between BMI and BMD when the specific subgroup with low BMD was taken in Indian population in our study further substantiates this hypothesis.
Another important observation that can be made from observing the trend of post-hoc tests of significant subcategories is that the difference between BMD of overweight and obese category was not significant in any of the groups. This implies that the effect of increase in BMI on BMD has a ceiling effect, and moderate to morbid obesity might not actually be a preventive factor for osteopenia and osteoporosis as has been hitherto believed. Greco et al. [16] also observed that when the study population was divided into subgroups based on different BMI, it was noted that overweight (BMI > 25 < 29.9) was neutral or protective for BMD, whereas obesity (BMI > 30) was associated with a low bone mass, compatible with a diagnosis of osteoporosis.
To conclude, BMI is an important determinant of BMD in Indian females. However, the association is not present in postmenopausal women with osteopenia. In addition, the effect of increase in BMI on BMD has a ceiling effect, and moderate to morbid obesity might not actually be a preventive factor for osteopenia.
The limitation of the present study firstly lies in the fact that this study involves the participants who visited the hospital for their patients. The demography of the participants was comparable in terms of age and BMI to that of the Indian female population [17]. Secondly, the Lunar USA normal database supplied by the manufacture was used to define low BMD (reference range 20–40 years) as the nomograms for BMD values in different age groups for Indian female population are not available.
References
Consensus Development Conference V. Diagnosis, prophylaxis, and treatment of osteoporosis. Am J Med. 1994;90:646–50.
Pande KC. Prevalence of low bone mass in healthy Indian population. J Ind Med Assoc. 2002;100(10):598–600.
Kumar A, Mittal S, Orito S, et al. Impact of dietary intake, education, and physical activity on bone mineral density among north Indian women. J Bone Miner Metab. 2010;28:192–201.
World health organization. Report of a WHO Scientific Group: Research on menopause, World health organization, Geneva. WHO Technical Report Series No. 670. 1981.
Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. The Lancet. 2005;365:1415–28.
World Health Organization. Assessment of fracture risk and its application to screening postmenopausal osteoporosis: report of a WHO study group. World Health Organ Tech Rep Ser. 1994;843:1–129.
Melton LJ. Epidemiology of osteoporosis: predicting who is at risk. Ann N Y Acad Sci. 1990;592:295–306.
Siris ES, Miller PD, Barrett-Connor E, et al. Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women: results from the National Osteoporosis Risk Assessment. JAMA. 2001;286:2815–22.
Albala C, Yanez M, Devoto E, et al. Obesity as a protective factor for postmenopausal osteoporosis. Int J Obes Relat Metab Disord. 1996;20:1027–32.
Ribot C, Tremollieres F, Pouilles JM, et al. Obesity and postmenopausal bone loss: the influence of obesity on vertebral density and bone turnover in postmenopausal women. Bone. 1987;8(6):327–31.
Ribot C, Tremollieres F, Pouilles JM. The effect of obesity on postmenopausal bone loss and the risk of osteoporosis. Adv Nutr Res. 1994;9:257–71.
Anderson JJ, Pollitzer WS. Ethnic and genetic differences in susceptibility to osteoporotic fractures. Adv Nutr Res. 1994;9:129–49.
Silverman SL, Madison RE. Decreased incidence of hip fracture in Hispanics, Asians, and blacks: California Hospital Discharge data. Am J Public Health. 1988;78:1482–3.
Castro PJ, Joseph LA, Shin JJ, et al. Differential effect of obesity on bone mineral density in White, Hispanic and African American women: a cross sectional study. Nutr Metab. 2005;2:9.
Lim SK, Won JY, Lee JH, et al. Altered hydroxylation of estrogen in patients with postmenopausal osteopenia. J Clin Endocrinol Metab. 1997;82:1001–6.
Greco EA, Fornari R, Rossi F, et al. Is obesity protective for osteoporosis? Evaluation of bone mineral density in individuals with high body mass index. Int J Clin Pract. 2010;64(6):817–20.
International Institute for population sciences (IIPS) and macrointernational. 2007. National Family Health Survey (NFHS-3) 2005-06, India: volumeII. Mumbai: IIPS.
Compliance with ethical requirements and conflict of interest
No conflicts of interest or disclosures to be made. The study was approved by the institute ethics committee.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, A., Sharma, A.K., Mittal, S. et al. The Relationship Between Body Mass Index and Bone Mineral Density in Premenopausal and Postmenopausal North Indian Women. J Obstet Gynecol India 66, 52–56 (2016). https://doi.org/10.1007/s13224-014-0629-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-014-0629-x