Abstract
The aim of this study was to determine the plant growth-promoting potential of the nodule endophytic Pseudomonas brassicacearum strain Zy-2-1 when used as a co-inoculant of Medicago lupulina with Sinorhizobium meliloti under copper (Cu) stress conditions. Strain Zy-2-1 was capable of producing ACC deaminase activity, IAA and siderophores, and was able to grow in the presence of Cu2+ up to 2.0 mmol/L. Co-inoculation of S. meliloti with Zy-2-1 enhanced M. lupulina root fresh weight, total plant dry weight, number of nodules, nodule fresh weight and nitrogen content in the presence of 100 or 300 mg/kg Cu2+. In the presence of 500 mg/kg Cu2+, co-inoculation with S. meliloti and strain Zy-2-1 increased plant height, number of nodules, nodule fresh weight and nitrogen content in comparison to S. meliloti inoculation alone. Furthermore, a higher amount of Cu accumulation in both shoots and roots and a higher level of Cu translocation to shoots were observed in co-inoculated plants. These results demonstrate that co-inoculation of M. lupulina with S. meliloti and P. brassicacearum Zy-2-1 improves plant growth, nitrogen nutrition and metal extraction potential. This can be of practical importance in the remediation of heavy metal-contaminated soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Copper is an essential redox-active micronutrient for normal growth and development of plants, as it is directly involved in a variety of metabolic activities, including photosynthesis, respiration, protein synthesis, cell wall lignification and oxidative stress protection. Indeed, these properties make copper ions indispensable for the life of plants; however, they are also the reason that the copper ion could be strongly toxic for plants when it is present at even slightly higher than optimal levels. Over the centuries, as a result of industrial production, sewage irrigation and extensive use of feed additives, organic fertilizer, fungicides and urban sewage-sludge compost, copper pollution of both soil and water has become a major environmental problem, as it poses a significant direct toxicity threat to plants, which in turn impacts negatively on both human and environmental health (Figueira et al. 2002; Lu et al. 2009; Manusadžianas et al. 2012; Srinivasa Gowd et al. 2010). For example, the East China Sea and Pearl River estuary were subjected to heavy copper pollution as a result of the rapid development of information technology (IT). In Jiangxi Province, up to 163 hm2 of farmland along the Le’an River has been contaminated by wastewater from copper mining, resulting in severe crop losses (Tang 2006).
Phytoremediation, as a cost-effective and environmentally friendly biotechnological approach for remediation of heavy metal contamination of soil, has been highly touted (Ali et al. 2013; Brígido and Glick 2015; Ma et al. 2016). However, many of the plants used in phytoremediation are characterized by slow growth rates and/or low biomass production, thus reducing their remediation potential and restricting their practical use in this technology (Baker et al. 1994; Komárek et al. 2007). Plant growth-promoting bacteria (PGPB) can act as adjuncts in heavy metal phytoremediation and significantly facilitate the growth of plants in the presence of otherwise inhibitory levels of metals (Glick 2010; Gamalero and Glick 2011; Kong et al. 2015b). The association of PGPB with plants may confer a number of advantages upon host plants, including the production of the phytohormone indole-3-acetic acid (IAA), solubilization of phosphate, secretion of siderophores to mobilize iron, and synthesis of the enzyme ACC deaminase to lower stress ethylene levels in plants.
Legumes are well known for their ability to form nodules on roots and stems with compatible rhizobial strains, within which atmospheric nitrogen is reduced to ammonia. The legume–rhizobia symbiosis is of great environmental and agricultural importance, and has been studied extensively (Hao et al. 2014; Naveed et al. 2015). However, environmental constraints such as drought, freezing, high temperature, salinity and toxic metals can reduce or restrict the expected beneficial effects of rhizobial symbiosis on plant growth, as both nodulation and nitrogen fixation processes can be impaired (Tejera et al. 2005; Wani et al. 2008; Sánchez-Pardo et al. 2013). Enhancement of legume nitrogen fixation by inoculation with both rhizobia and a PGPB is a way to overcome these environmental limitations and improve plant growth (Yadegari et al. 2010; Fox et al. 2011; Hungria et al. 2013). In this regard, recent studies have reported the use of legumes inoculated with rhizobia and metal-resistant PGPBs for metal phytoremediation (Dary et al. 2010; Fatnassi et al. 2013). Although most previous studies dealing with co-inoculation in legumes have reported plant growth promotion and enhancement of symbiotic parameters, contradictory results have also been observed, suggesting that co-inoculation might impair rhizobial colonization and interfere with the nodulation process (Lucas García et al. 2004a; Lucas García et al. 2004b; Berggren et al. 2005; Estévez et al. 2009). For example, Estévez et al. (2009) reported that soybean plants co-inoculated with Chryseobacterium balustinum and rhizobia did not always show better symbiotic performance under moderate saline conditions. These contrary results indicate that in order to optimize the phytoremediation potential of the system under particular environmental conditions, close attention must be paid to the selection of the appropriate co-inoculation partners and the traits that they encode.
According to a field survey, we found that M. lupulina was a dominant plant growing in lead-zinc mine tailings in Northwest China (Wei and Ma 2010). The Cu-resistant strain Sinorhizobium meliloti CCNWSX0020 was isolated from the root nodules of M. lupulina, and this symbiosis was found to display potential for use in Cu phytostabilization (Kong et al. 2015a, b). The aim of the present study was the characterization of a plant growth-promoting bacterium, Pseudomonas brassicacearum strain Zy-2-1, and its effects when co-inoculated with Sinorhizobium meliloti on the symbiotic performance and metal uptake of Medicago lupulina plants under copper stress.
Materials and methods
Bacterial strains and cultures
Pseudomonas brassicacearum strain Zy-2-1 was originally isolated from the root nodules of the leguminous weed Sphaerophysa salsula growing on the Loess Plateau in China (Deng et al. 2011). This strain was deposited in the Agricultural Culture Collection of China and named ACCC19944. Strain P. brassicacearum Zy-2-1 inoculants were grown for 2 days at 30 °C with shaking at 150 rpm in tryptic soybean broth (TSB) medium (BD Difco, Detroit, MI, USA).
Sinorhizobium meliloti strain CCNWSX0020, which is resistant to 1.4 mmol/L Cu2+, was isolated from Medicago lupulina plants growing in lead-zinc mine tailings in China (Fan et al. 2010). This strain was deposited in the Agricultural Culture Collection of China and named ACCC19736. The S. meliloti CCNWSX0020 inoculant was grown for 2 days at 28 °C, with shaking at 150 rpm in tryptone yeast extract (TY) liquid medium (5 g tryptone, 3 g yeast extract, and 0.7 g CaCl2 · 2H2O per liter; pH 7.2).
Pseudomonas putida UW4 was originally isolated from the rhizosphere of common reeds, based on its ability to utilize ACC as a sole source of nitrogen (Glick et al. 1995; Duan et al. 2013). Pseudomonas fluorescens 17400, obtained from the American Type Culture Collection, was previously reported to have no plant growth-promoting activity (Shah et al. 1998). The Pseudomonas spp. strains, growing in TSB medium at 30 °C, were used as the positive and negative controls, respectively, for the measurement of plant growth-promoting characteristics.
Presence and activity of ACC deaminase
The presence of the ACC deaminase structural gene (acdS) in P. brassicacearum Zy-2-1 was tested by polymerase chain reaction (PCR). Genomic DNA from P. brassicacearum Zy-2-1 was extracted according to the method described by Terefework et al. (2001). The acdS DNA fragment was amplified from the genomic DNA of strain Zy-2-1 by PCR using the oligonucleotides 5’-GGCAAGGTCGACATCTATGC-3’ and 5’-GGCTTGCCATTCAGCTATG-3’ as primers. The PCR product was electrophoresed at 100 V for 40 min in 1 % w/v agarose gel, and the band corresponding to the expected size (approximately 1 kb) was excised from the gel, purified and sequenced directly. The sequence that was obtained was deposited in the GenBank database (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and was aligned with related sequences.
ACC deaminase activity was determined by spectrophotometrically measuring the production of α-ketobutyrate as described by Penrose and Glick (2003), with a standard curve of α-ketobutyrate from 0.05 to 0.5 μ moles. The protein concentration of the disrupted cell suspension was determined according Bradford (1976) using the Bio-Rad protein reagent (Bio-Rad Laboratories, Hercules, CA, USA), according to the manufacturer’s instructions.
Indoleacetic acid (IAA) production
IAA production was measured as described by Patten and Glick (2002), with minor modifications. Aliquots of 20 μl of overnight bacterial cultures were used to inoculate 5 mL TSB medium without and with tryptophan (100, 250 and 500 μg/mL; Sigma-Aldrich, St. Louis, MO, USA) and incubated at 30 °C for 24 h. When the cell cultures reached stationary phase, they were centrifuged (5,500×g, 10 min), and 1 mL of supernatant was mixed with 4 mL Salkowski’s reagent (150 mL of concentrated H2SO4, 250 mL distilled H2O, 7.5 mL of 0.5 mol/L FeCl3 · 6H2O) and incubated for 20 min at room temperature, after which the absorbance was measured at 535 nm. The concentration of IAA in each culture was determined by comparison with a standard curve of pure IAA (Sigma-Aldrich) from 0.1 to 40.0 μg/mL.
Siderophore production
Siderophore levels in the bacterial culture were assayed according to the universal chemical assay of Schwyn and Neilands (1987). A 5-μL of aliquot of overnight bacterial culture in King’s B medium was spotted onto a chrome azurol S (CAS) agar plate (Alexander and Zuberer 1991) in triplicate, and incubated at 30 °C for 48 hours.
Tolerance of bacteria to copper
Pseudomonas strains were grown overnight at 30 °C in TSB medium at pH 7.3 ± 0.2, and 10 μL of each culture was then spotted onto solid seawater yeast extract (SWYE) medium (Nieto et al. 1987). In this way, 20 cultures could be tested per plate. The medium was supplemented with a filter-sterilized CuSO4 solution at concentrations of 0.005, 0.01, 0.02, 0.05, 0.1, 0.2, 0.5, 1.0, 2.0 and 5.0 mmol/L to determine the minimum inhibitory concentration (MIC) of each bacterial strain. The MIC was defined as the lowest concentration of metal that prevented bacterial growth. Duplicate plates were prepared for each Cu2+ concentration and incubated at 28 °C for 10 days. The agar plates without CuSO4 were used as controls. For the purpose of defining copper resistance, a strain that could grow in the presence of 1 mmol/L Cu2+ was considered to be resistant (Nieto et al. 1987).
Plant growth and treatments
Medicago lupulina seeds (provided by Gansu Agricultural University, China) were surface-sterilized by treatment with 75 % v/v ethanol for 2 min, followed by 10 min in 20 % v/v NaClO (containing 8 % available chlorine). After the seeds were thoroughly rinsed with several changes of sterile distilled water, they were germinated on moist sterile filter paper in the dark at 25 °C for 3 days. The 3-day-old sterilized seedlings were planted in plastic pots (10 cm diameter) filled with 100 g of a sterilized perlite–vermiculite (1:1) mixture. The seedling medium was supplemented with copper in the form of CuSO4. Copper stock solution (0.1 mol/L, pH 3.97) was made in distilled water, sterilized by filtration through a 0.22-μm-pore membrane filter. The seedling medium was thoroughly watered by properly diluting this stock solution with distilled water to produce Cu (II) concentrations of 100 mg/kg (lightly polluted), 300 mg/kg (moderately polluted) and 500 mg/kg (heavily polluted). The range of copper concentrations was determined in preliminary experiments (data not shown). After it was thoroughly mixed with the copper solution, the soil medium was packed into the plastic pots and allowed to equilibrate for 1 week. The seedlings were then maintained in a plant growth incubator at 25 °C at 200 μmol/(m2/s) light for 16 h, and 21 °C in the dark for 8 h. Fåhraeus nitrogen-free mineral nutrient solution (Fåhraeus 1957) was used to water the plants when necessary (approximately 150 mL every 5–6 days). Six seedlings were planted in each pot, and four replicates were conducted for each treatment. After 5 days, seedlings were inoculated with cell suspensions of S. meliloti CCNWSX0020 or a co-inoculant cell suspension of S. meliloti CCNWSX0020 with P. brassicacearum Zy-2-1, respectively. The bacterial cultures were standardized to an optical density of 0.8 at 600 nm, and 1 mL of the bacterial cell suspension was inoculated onto each seedling.
Plant growth, nodulation, N content and Cu content
Plants were harvested after 50 days, separated into above-ground plant tissues and roots, carefully rinsed with distilled water and dried at 65 °C for 48 hours before determining the dry weight. The fresh weight, dry weight, plant height, nodule fresh weight and the number of effective nodules (pink-red colour) were recorded. The pink-red colour, because of the presence of leghemoglobin, was considered as an index of potential N fixation (Ott et al. 2005; Reichman 2007).
The N content of plant tissue samples was determined based on the Kjeldahl method using an automatic Kjeltec™ 8400 analyzer unit (FOSS A/S, Hilleroed, Denmark). Nitrogenase activity in nodules was measured by a acetylene reduction assay as described by Weaver and Danso (1994). Acetylene and ethylene were quantified through an HP-AL/M column (30 m, I.D. 0.53 nm, 15 μm; J&W Scientific/Agilent Technologies, Folsom, CA, USA) using a Shimadzu GC-17A gas chromatograph (Shimadzu Corporation, Kyoto, Japan) and a flame ionization detector. Helium was used as the carrier gas, with a flow rate set at 6 mL/min and 36 kPa total pressure. The injector, column and detector temperatures were 120 °C, 100 °C and 150 °C, respectively. Ethylene elutes after 1.9 min, and acetylene elutes after 3.0 min. The amount of ethylene produced by each nodule sample (0.20 g) was calculated using a standard curve of ethylene.
The above-ground plant tissues and roots were separated and rinsed three times with sterilized deionized distilled water (ddH2O) to remove any loosely bound Cu2+, and then dried at 65 °C for 48 hours. Aliquots of precisely 0.2 g powdered plant tissue samples were digested with an acid mixture (HNO3:HClO4 = 3:1), and the copper content was determined by atomic absorption spectrophotometry (Z-5000; Hitachi, Tokyo, Japan). To evaluate the transport behavior of Cu from plant roots to shoots under excess Cu conditions, the translocation factor (Singh and Agrawal 2007) was calculated using the following formula:
where Cu s and Cu r are Cu content in shoots and roots, respectively.
Statistical analyses
All statistical analyses were performed with SPSS for Windows, version 16.0 (SPSS Inc., Chicago, IL, USA), statistical software. Data were analyzed by one-way analysis of variance (ANOVA), followed by Duncan’s test (p < 0.05). All data were analyzed using OriginPro v8.0 (OriginLab Corporation, Northampton, MA, USA) to create figures.
Results
Isolation and characterization of ACC deaminase gene
The expected amplification product of approximately 1 kb representing the ACC deaminase structural gene (acdS) was observed following PCR amplification of the genomic DNA of P. brassicacearum Zy-2-1 (Supplemental, Fig. S1). The DNA sequence of this gene was deposited in the GenBank database under accession number JN624298. Based on sequence alignments, the acdS gene of P. brassicacearum Zy-2-1 had a high degree of similarity, 94.18 % and 94.65 %, to the acdS genes from P. brassicacearum strain Am3 (AY604528) and P. fluorescens strain FY32 (FJ465155), respectively.
Copper tolerance and plant growth-promoting characteristics of P. brassicacearum Zy-2-1
The MIC of Cu2+ for P. brassicacearum Zy-2-1 was 2.0 mmol/L, which was higher than either P. putida UW4 or P. fluorescens 17400 (1.0 and 0.005 mmol/L, respectively) (Table 1). Therefore, P. brassicacearum Zy-2-1 was considered to be resistant to CuSO4.
P. brassicacearum Zy-2-1 was capable of producing ACC deaminase, IAA and siderophores, all to a greater extent than either P. putida UW4 or P. fluorescens 17400 (Table 1).
Measurement of plant growth, nodulation and N content
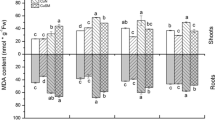
The total fresh and dry weights of plants decreased significantly with an increased amount of Cu in the medium. No significant differences were observed in plant biomass between the Sinorhizobium inoculation alone or in combination with Zy-2-1 under control conditions (Fig. 1). However, in the presence of 100 or 300 mg/kg Cu2+, co-inoculation of plants with Zy-2-1 and Sinorhizobium produced a significantly greater plant biomass than the single inoculation with Sinorhizobium. For the aerial parts, although the differences were not statistically significant for the fresh weight (Fig. 1a), the dry weight of co-inoculated plants increased by 53.04 % and 78.3 % in the presence of 100 and 300 mg/kg Cu2+, respectively, compared with the plants inoculated with Sinorhizobium alone (Fig. 1b). Similarly, the fresh weight of roots of co-inoculated plants increased by 50.42 % and 65.99 % in the presence of 100 and 300 mg/kg Cu2+, respectively, and the dry weight of roots was 39.97 % and 73.02 % greater in the co-inoculated plants than in the plants with Sinorhizobium inoculation alone. The plant height was significantly increased, by 30.79 % and 46 %, in co-inoculated plants compared to the plants inoculated with Sinorhizobium alone in the 300 and 500 mg/kg Cu2+ treatments, respectively (Fig. 2).
Fresh weight (a) and dry weight (b) of shoots (white bar) and roots (grey bar) of Medicago lupulina plants with increasing concentrations of Cu. SM = Inoculated with Sinorhizobium meliloti CCNWSX0020; SM + Zy-2-1 = Co-inoculated with S. meliloti CCNWSX0020 and P. brassicacearum Zy-2-1. The values indicate the mean ± SE of four replicates. Bars with different letters are significantly different at p < 0.05 by the Duncan test
Plant height of Medicago lupulina plants with increasing concentrations of Cu. SM = Inoculated with Sinorhizobium meliloti CCNWSX0020; SM + Zy-2-1 = Co-inoculated with S. meliloti CCNWSX0020 and P. brassicacearum Zy-2-1. The values indicate the mean ± SE of four replicates. Bars with different letters are significantly different at p < 0.05 by the Duncan test
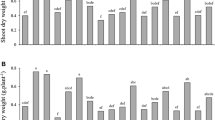
The pink-red colour of nodules could be observed, indicting the establishment of effective symbioses. The number of effective nodules, nodule fresh weight and nitrogenase activity were significantly reduced by treatment with 300 or 500 mg/kg Cu2+ (Fig. 3). Sinorhizobium inoculation alone or in combination with Zy-2-1 had little effect on the number of effective nodules, nodule fresh weight or nitrogenase activity under control conditions. However, dual inoculation of Sinorhizobium with Zy-2-1 produced 50 %, 100 % and 100 % more effective nodules per plant in the presence of 100, 300 and 500 mg/kg Cu2+, respectively, compared with Sinorhizobium inoculation alone (Fig. 3a). Similarly, nodule fresh weight of co-inoculated plants also showed significant increases in the presence of 100, 300 and 500 mg/kg Cu2+, in comparison to the single inoculation with Sinorhizobium (Fig. 3b). No significant alterations were observed in the nitrogenase activity of co-inoculated plants in either the absence or presence of Cu compared with the plants inoculated with Sinorhizobium alone under the same conditions (Fig. 3c). However, significant positive effects on N content were observed in both shoots and roots of co-inoculated plants under Cu stress conditions (Fig. 4).
Number of effective nodules (a), nodule fresh weight (b) and nitrogenase activity (c) of Medicago lupulina plants with increasing concentrations of Cu. SM = Inoculated with Sinorhizobium meliloti CCNWSX0020; SM + Zy-2-1 = Co-inoculated with S. meliloti CCNWSX0020 and P. brassicacearum Zy-2-1. The values indicate the mean ± SE of four replicates. Bars with different letters are significantly different at p < 0.05 by the Duncan test
N content of shoots (white bar) and roots (grey bar) of Medicago lupulina plants with increasing concentrations of Cu. SM = Inoculated with Sinorhizobium meliloti CCNWSX0020; SM + Zy-2-1 = Co-inoculated with S. meliloti CCNWSX0020 and P. brassicacearum Zy-2-1. The values indicate the mean ± SE of four replicates. Bars with different letters are significantly different at p < 0.05 by the Duncan test
Cu content
The Cu content in both shoots and roots of plants inoculated with either Sinorhizobium or Sinorhizobium + Zy-2-1 was significantly elevated with the increased level of Cu in the medium, an effect that was more pronounced in roots than in shoots (Fig. 5a). Furthermore, co-inoculation with Zy-2-1 dramatically increased the Cu content in both shoots and roots in the presence of excess Cu. The Cu content increased by 145.99 %, 209.56 % and 289.08 % in shoots, and by 108.29 %, 102.35 % and 89.40 % in roots of the co-inoculated plants in the presence of 100, 300 and 500 mg/kg Cu2+, respectively, compared with the plants inoculated with Sinorhizobium alone. Interestingly, co-inoculation with Zy-2-1 also increased the Cu content in both shoots and roots under control (with no Cu2+ in the medium) conditions. Plant dry weight and Cu content were calculated to obtain the total amount of Cu uptake in each plant. The total Cu uptake in both shoots and roots of co-inoculated plants were significantly increased in comparison to the single-inoculated plants in the presence of 100 or 300 mg/kg Cu2+, while a significant increase occurred only in the shoots of co-inoculated plants in the presence of 500 mg/kg Cu2+ (Fig. 5b). With respect to the transport behavior of Cu from the roots to shoots of plants, the translocation factor of co-inoculated plants increased in the presence of 300 and 500 mg/kg Cu2+, although the difference was not statistically significant for the co-inoculated plants treated with 100 mg/kg Cu2+ (Fig. 5c). A significant increase was similarly observed in the translocation factor of co-inoculated plants under control conditions.
Cu content (a), total Cu uptake (= dry weight × Cu content) (b) and translocation factor (c) of shoots (white bar) and roots (grey bar) of Medicago lupulina plants with increasing concentrations of Cu. SM = Inoculated with Sinorhizobium meliloti CCNWSX0020; SM + Zy-2-1 = Co-inoculated with S. meliloti CCNWSX0020 and P. brassicacearum Zy-2-1. The values indicate the mean ± SE of four replicates. Bars with different letters are significantly different at p < 0.05 by the Duncan test
Discussion
Most of the bacterial endophytes that have been isolated from various plants are also capable of living outside plant tissues as rhizospheric bacteria, and thus the beneficial effects on their host plants appear to occur through mechanisms similar to those described for other PGPB (Zablotowicz et al. 1991; Höflich et al. 1994; Di Fiore and Del Gallo 1995). P. brassicacearum Zy-2-1 was isolated as an endophytic bacterium from root nodules of the wild legume Sphaerophysa salsula growing on the Loess Plateau in China, which has a dry monsoonal climate with sandy loam and saline/alkaline soil. The results presented here show that P. brassicacearum Zy-2-1, which has a greater level of ACC deaminase activity than the well-studied plant growth-promoting bacterium P. putida UW4, also displayed a higher level of IAA production and positive siderophore activity. This can be understood in terms of the observation that stressful growth conditions may select for plant-associated bacteria that possess an increased level of ACC deaminase, thereby protecting plants and facilitating both bacterial and plant survival (Belimov et al. 2001; Timmusk et al. 2011). In this regard, both siderophores and IAA produced by bacteria in the rhizosphere enhance plant growth and mineral uptake, resulting in increased plant nutrition (Wani et al. 2007; Nimnoi et al. 2014). Moreover, the IAA released by PGPB may be involved at different levels in plant–bacteria interactions. In particular, root development and nodulation are both significantly influenced by IAA (Wani et al. 2007). The present study also revealed that P. brassicacearum strain Zy-2-1 has a high level of copper resistance, thereby facilitating the use of this bacterium for co-inoculation with Cu2+-resistant rhizobia in contaminated environments.
The present study shows that plant biomass and nodulation parameters under light or moderate Cu stress conditions were enhanced in co-inoculated plants compared to plants inoculated with Sinorhizobium alone. Even though in some cases there were no significant differences between co-inoculated and single-inoculated plants under severe Cu stress conditions, co-inoculation of Sinorhizobium and P. brassicacearum Zy-2-1 significantly increased plant height, number of effective nodules and nodule fresh weight compared to single inoculation. Similarly, many previous studies working on co-inoculation with PGPBs in legumes have reported increased plant growth and enhanced symbiotic performance (Hungria et al. 2013; Nimnoi et al. 2014; Sánchez et al. 2014). On the other hand, plant growth and nodulation parameters in the absence of Cu2+ showed no significant differences whether plants were inoculated with Sinorhizobium or with the co-inoculation of Sinorhizobium and P. brassicacearum Zy-2-1. This may be due to the presence of the endogenous plant growth-promoting characteristics of S. meliloti strain CCNWSX0020, including positive siderophore activity, a high level of IAA production and a moderate level of ACC deaminase activity (Kong et al. 2015a). Although an increase was found in the effective nodule number and nodule fresh weight, the co-inoculation pattern did not promote nitrogenase activity of Medicago plants under either control or Cu stress conditions. However, significant increases were found in the N content of both shoots and roots of co-inoculated plants under Cu stress conditions. This means that the copper-resistant strains P. brassicacearum Zy-2-1 and S. meliloti CCNWSX0020 are able to survive under the Cu concentrations used in this study, and thus the co-inoculation pattern promotes an increased level of plant nitrogen. Moreover, the co-inoculated plants had a healthy green colour, indicting the establishment of effective symbioses, as supported by the observation of pink-red colour of nodules. The observed benefits to Medicago plants from the combined inoculation of Sinorhizobium and P. brassicacearum Zy-2-1 may be due to improved N nutrition in addition to growth-promoting substances. Similar positive effects of bacterial co-inoculation on the N content of associated legumes have been reported in previous studies (Tilak et al. 2006; Fox et al. 2011; Nimnoi et al. 2014). These findings indicate that multiple plant growth-promoting properties, such as nitrogen fixation, siderophore activity and IAA biosynthesis, together with ACC deaminase activity, are responsible for the observed plant growth promotion and yield increases (Sarathambal et al. 2015). The positive results on specific plant growth-promoting properties of P. brassicacearum Zy-2-1 in this study suggest that this particular bacterium can promote plant growth under Cu stress conditions by more than one mechanism, and that these properties could be better exploited as co-inoculant.
It was previously observed that an increase or decrease in the amount of metal taken up by plant tissues is a consequence of the particular host plant involved, the bacterium, and the metal species and its concentration (Rajkumar et al. 2009). In this study, metal accumulation was considerably higher in roots than in shoots in the presence of excess Cu. The value of the translocation factor was considerably less than 1 for both single- and double-inoculated plants, suggesting that Cu accumulated mainly in roots, with a very low level of Cu translocation to shoots. This may be explained by the fact that metal cations such as Cu2+ bind quite tightly to organic ligands within the root cell walls (Kochian 1991; Garau et al. 2015). These results indicate the potential use of M. lupulina for phytostabilization of copper-contaminated soils. The co-inoculation with Sinorhizobium and P. brassicacearum Zy-2-1 significantly increased the Cu content in both shoots and roots with increasing concentrations of Cu in the medium. Compared to the plants inoculated with Sinorhizobium alone, the higher translocation factor for the co-inoculated plants under Cu stress conditions suggests that the co-inoculation pattern improves the translocation behavior of Cu in Medicago plants from roots to shoots. These results are consistent with those of previous studies in which inoculation with PGPB increased metal uptake by plant organs compared with non-inoculated plants (Mastretta et al. 2009; Prapagdee et al. 2013). In addition to plant growth-promoting activities, certain metal-resistant soil microorganisms have been shown to possess other traits that can alter heavy metal mobility and availability to the plants. For instance, the inoculation of canola (Brassica napus) with Pb-resistant endophytic bacteria enhanced the availability of Pb to B. napus by bacterial siderophores or by solubilization of Pb (Sheng et al. 2008). Although an increased heavy metal concentration in plant tissues has toxic effects on plants, increased levels are valuable for effective phytoremediation of soils (Fan et al. 2010). These results demonstrate that the application of selected metal-resistant PGPBs can improve the metal extraction potential of plants, which can be useful for the remediation of heavy metal-contaminated soils. Similarly, a higher translocation factor was also observed for the co-inoculated plants under control conditions. Since Cu is also an essential micronutrient for plants when it is present at optimal level, it is assumed that plants grown under the growth-limiting conditions used in the experiments might eventually take advantage (i.e. a more efficient micronutrition uptake) of the root apparatus in the presence of bacterial plant growth-promoting activities.
In summary, our results suggest positive effects (i.e. increased plant growth, nodulation and metal uptake) from co-inoculation of M. lupulina with Sinorhizobium and PGPB P. brassicacearum Zy-2-1, which can be exploited for the phytoremediation of copper-contaminated soils. Nevertheless, these results need to be tested under field conditions, and further work is required to determine the precise role played by the PGPB P. brassicacearum Zy-2-1 in plant copper uptake.
References
Alexander DB, Zuberer DA (1991) Use of chrome azurol S reagents to evaluate siderophore production by rhizosphere bacteria. Biol Fertil Soils 12:39–45. doi:10.1007/BF00369386
Ali H, Khan E, Sajad MA (2013) Phytoremediation of heavy metals-concepts and applications. Chemosphere 91:869–881. doi:10.1016/j.chemosphere.2013.01.075
Baker AJM, McGrath SP, Sidoli CMD, Reeves RD (1994) The possibility of in situ heavy metal decontamination of polluted soils using crops of metal-accumulating plants. Resour Conserv Recycl 11:41–49. doi:10.1016/0921-3449(94)90077-9
Belimov AA et al (2001) Characterization of plant growth promoting rhizobacteria isolated from polluted soils and containing 1-aminocyclopropane-1-carboxylate deaminase. Can J Microbiol 47:642–652
Berggren I, Alström S, Van Vuurde J, Mårtensson AM (2005) Rhizoplane colonisation of peas by Rhizobium leguminosarum bv. viceae and a deleterious Pseudomonas putida. FEMS Microbiol Ecol 52:71–78
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Brígido C, Glick BR (2015) Phytoremediation using rhizobia. In: Ansari AA, Gill SS, Gill R, Lanza GR, Newman L (eds) Phytoremediation: management of environmental contaminants. Springer, New York, pp 95–114
Dary M, Chamber-Pérez M, Palomares A, Pajuelo E (2010) “In situ” phytostabilisation of heavy metal polluted soils using Lupinus luteus inoculated with metal resistant plant-growth promoting rhizobacteria. J Hazard Mater 177:323–330
Deng ZS, Zhao LF, Kong ZY, Yang WQ, Lindström K, Wang ET, Wei GH (2011) Diversity of endophytic bacteria within nodules of the Sphaerophysa salsula in different regions of Loess Plateau in China. FEMS Microbiol Ecol 76:463–475. doi:10.1111/j.1574-6941.2011.01063.x
Di Fiore S, Del Gallo M (1995) Endophytic bacteria: their possible role in the host plant. In: Fendrik I, del Gallo M, Vanderleyden J, de Zamaroczy M (eds) Azospirillum VI and related microorganisms, vol 37. Springer, Berlin Heidelberg, pp 169–187. doi:10.1007/978-3-642-79906-8_18, NATO ASI Series
Duan J, Jiang W, Cheng Z, Heikkila JJ, Glick BR (2013) The complete genome sequence of the plant growth-promoting bacterium Pseudomonas sp. UW4. PLoS ONE 8:e58640. doi:10.1371/journal.pone.0058640
Estévez J, Dardanelli MS, Megías M, Rodríguez-Navarro DN (2009) Symbiotic performance of common bean and soybean co-inoculated with rhizobia and Chryseobacterium balustinum Aur9 under moderate saline conditions. Symbiosis 49:29–36. doi:10.1007/s13199-009-0008-z
Fåhraeus G (1957) The infection of clover root hairs by nodule bacteria studied by a simple glass slide technique. J Gen Microbiol 16:374–381. doi:10.1099/00221287-16-2-374
Fan LM, Ma ZQ, Liang JQ, Li HF, Wang ET, Wei GH (2010) Characterization of a copper-resistant symbiotic bacterium isolated from Medicago lupulina growing in mine tailings. Bioresour Technol 102:703–709. doi:10.1016/j.biortech.2010.08.046
Fatnassi IC, Chiboub M, Saadani O, Jebara M, Jebara SH (2013) Phytostabilization of moderate copper contaminated soils using co-inoculation of Vicia faba with plant growth promoting bacteria. J Basic Microbiol 55:303–311
Figueira R, Sérgio C, Sousa AJ (2002) Distribution of trace metals in moss biomonitors and assessment of contamination sources in Portugal. Environ Pollut 118:153–163. doi:10.1016/S0269-7491(01)00203-2
Fox SL, O’Hara GW, Bräu L (2011) Enhanced nodulation and symbiotic effectiveness of Medicago truncatula when co-inoculated with Pseudomonas fluorescens WSM3457 and Ensifer (Sinorhizobium) medicae WSM419. Plant Soil 348:245–254
Gamalero E, Glick BR (2011) Mechanisms used by plant growth-promoting bacteria. In: Maheshwari DK (ed) Bacteria in agrobiology: plant nutrient management. Springer, Berlin Heidelberg, pp 17–46. doi:10.1007/978-3-642-21061-7_2
Garau G, Mele E, Castaldi P, Lauro GP, Deiana S (2015) Role of polygalacturonic acid and the cooperative effect of caffeic and malic acids on the toxicity of Cu (II) towards triticale plants (× Triticosecale, Wittm). Biol Fertil Soils 51(5):535–544. doi:10.1007/s00374-015-0999-y
Glick BR (2010) Using soil bacteria to facilitate phytoremediation. Biotechnol Adv 28:367–374. doi:10.1016/j.biotechadv.2010.02.001
Glick BR, Karaturovíc DM, Newell PC (1995) A novel procedure for rapid isolation of plant growth promoting pseudomonads. Can J Microbiol 41:533–536. doi:10.1139/m95-070
Hao X, Taghavi S, Xie P, Orbach MJ, Alwathnani H, Rensing C, Wei G (2014) Phytoremediation of heavy and transition metals aided by legume-rhizobia symbiosis. Int J Phytoremediation 16:179–202
Höflich G, Wiehe W, Kühn G (1994) Plant growth stimulation by inoculation with symbiotic and associative rhizosphere microorganisms. Experientia 50:897–905. doi:10.1007/BF01923476
Hungria M, Nogueira M, Araujo R (2013) Co-inoculation of soybeans and common beans with rhizobia and azospirilla: strategies to improve sustainability. Biol Fertil Soils 49:791–801. doi:10.1007/s00374-012-0771-5
Kochian LV (1991) Mechanisms of micronutrient uptake and translocation in plants. In: Mortvedt JJ (ed) Micronutrients in agriculture. SSSA Book Series. Soil Science Society of America, Fitchburg, pp 229–296. doi:10.2136/sssabookser4.2ed.c8
Komárek M, Tlustoš P, Száková J, Chrastný V, Ettler V (2007) The use of maize and poplar in chelant-enhanced phytoextraction of lead from contaminated agricultural soils. Chemosphere 67:640–651. doi:10.1016/j.chemosphere.2006.11.010
Kong Z, Glick BR, Duan J, Ding S, Tian J, McConkey BJ, Wei G (2015a) Effects of 1-aminocyclopropane-1-carboxylate (ACC) deaminase-overproducing Sinorhizobium meliloti on plant growth and copper tolerance of Medicago lupulina. Plant Soil 391:383–398
Kong Z, Mohamad OA, Deng Z, Liu X, Glick BR, Wei G (2015b) Rhizobial symbiosis effect on the growth, metal uptake, and antioxidant responses of Medicago lupulina under copper stress. Environ Sci Pollut Res 22:12479–12489
Lu X, Wang L, Lei K, Huang J, Zhai Y (2009) Contamination assessment of copper, lead, zinc, manganese and nickel in street dust of Baoji, NW China. J Hazard Mater 161:1058–1062. doi:10.1016/j.jhazmat.2008.04.052
Lucas García JA, Probanza A, Ramos B, Barriuso J, Gutierrez Mañero FJ (2004a) Effects of inoculation with plant growth promoting rhizobacteria (PGPRs) and Sinorhizobium fredii on biological nitrogen fixation, nodulation and growth of Glycine max cv. Osumi. Plant Soil 267:143–153. doi:10.1007/s11104-005-4885-5
Lucas García JA, Probanza A, Ramos B, Colón Flores JJ, Gutiérrez Mañero FJ (2004b) Effects of plant growth promoting rhizobacteria (PGPRs) on the biological nitrogen fixation, nodulation, and growth of Lupinus albus L. cv. Multolupa. Eng Life Sci 4:71–77. doi:10.1002/elsc.200400013
Ma Y, Rajkumar M, Zhang C, Freitas H (2016) Beneficial role of bacterial endophytes in heavy metal phytoremediation. J Environ Manag 174:14–25
Manusadžianas L et al (2012) Toxicity of copper oxide nanoparticle suspensions to aquatic biota. Environ Toxicol Chem 31:108–114. doi:10.1002/etc.715
Mastretta C et al (2009) Endophytic bacteria from seeds of Nicotiana tabacum can reduce cadmium phytotoxicity. Int J Phytoremediation 11:251–267
Naveed M, Mehboob I, Hussain MB, Zahir ZA (2015) Perspectives of rhizobial inoculation for sustainable crop production. In: Plant microbes symbiosis: applied facets. Springer, pp 209–239
Nieto JJ, Ventosa A, Ruiz-Berraquero F (1987) Susceptibility of halobacteria to heavy metals. Appl Environ Microbiol 53:1199–1202
Nimnoi P, Pongsilp N, Lumyong S (2014) Co-inoculation of soybean (Glycine max) with actinomycetes and Bradyrhizobium japonicum enhances plant growth, nitrogenase activity and plant nutrition. J Plant Nutr 37:432–446
Ott T et al (2005) Symbiotic leghemoglobins are crucial for nitrogen fixation in legume root nodules but not for general plant growth and development. Curr Biol 15:531–535. doi:10.1016/j.cub.2005.01.042
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801. doi:10.1128/aem.68.8.3795-3801.2002
Penrose DM, Glick BR (2003) Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol Plant 118:10–15. doi:10.1034/j.1399-3054.2003.00086.x
Prapagdee B, Chanprasert M, Mongkolsuk S (2013) Bioaugmentation with cadmium-resistant plant growth-promoting rhizobacteria to assist cadmium phytoextraction by Helianthus annuus. Chemosphere 92:659–666. doi:10.1016/j.chemosphere.2013.01.082
Rajkumar M, Ae N, Freitas H (2009) Endophytic bacteria and their potential to enhance heavy metal phytoextraction. Chemosphere 77:153–160. doi:10.1016/j.chemosphere.2009.06.047
Reichman SM (2007) The potential use of the legume-Rhizobium symbiosis for the remediation of arsenic contaminated sites. Soil Biol Biochem 39:2587–2593. doi:10.1016/j.soilbio.2007.04.030
Sánchez AC, Gutiérrez RT, Santana RC, Urrutia AR, Fauvart M, Michiels J, Vanderleyden J (2014) Effects of co-inoculation of native Rhizobium and Pseudomonas strains on growth parameters and yield of two contrasting Phaseolus vulgaris L. genotypes under Cuban soil conditions. Eur J Soil Biol 62:105–112. doi:10.1016/j.ejsobi.2014.03.004
Sánchez-Pardo B, Carpena RO, Zornoza P (2013) Cadmium in white lupin nodules: impact on nitrogen and carbon metabolism. J Plant Physiol 170:265–271
Sarathambal C, Ilamurugu K, Balachandar D, Chinnadurai C, Gharde Y (2015) Characterization and crop production efficiency of diazotrophic isolates from the rhizosphere of semi-arid tropical grasses of India. Appl Soil Ecol 87:1–10. doi:10.1016/j.apsoil.2014.11.004
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56. doi:10.1016/0003-2697(87)90612-9
Shah S, Li J, Moffatt BA, Glick BR (1998) Isolation and characterization of ACC deaminase genes from two different plant growth-promoting rhizobacteria. Can J Microbiol 44:833–843. doi:10.1139/w98-074
Sheng XF, Xia JJ, Jiang CY, He LY, Qian M (2008) Characterization of heavy metal-resistant endophytic bacteria from rape (Brassica napus) roots and their potential in promoting the growth and lead accumulation of rape. Environ Pollut 156:1164–1170. doi:10.1016/j.envpol.2008.04.007
Singh RP, Agrawal M (2007) Effects of sewage sludge amendment on heavy metal accumulation and consequent responses of Beta vulgaris plants. Chemosphere 67:2229–2240. doi:10.1016/j.chemosphere.2006.12.019
Srinivasa Gowd S, Ramakrishna Reddy M, Govil PK (2010) Assessment of heavy metal contamination in soils at Jajmau (Kanpur) and Unnao industrial areas of the Ganga Plain, Uttar Pradesh, India. J Hazard Mater 174:113–121. doi:10.1016/j.jhazmat.2009.09.024
Tang S (2006) The principle and methods of phytoremediation of contaminated environment. Science Press, Beijing
Tejera N, Campos R, Sanjuan J, Lluch C (2005) Effect of sodium chloride on growth, nutrient accumulation, and nitrogen fixation of common bean plants in symbiosis with isogenic strains. J Plant Nutr 28:1907–1921
Terefework Z, Kaijalainen S, Lindström K (2001) AFLP fingerprinting as a tool to study the genetic diversity of Rhizobium galegae isolated from Galega orientalis and Galega officinalis. J Biotechnol 91:169–180. doi:10.1016/S0168-1656(01)00338-8
Tilak KVBR, Ranganayaki N, Manoharachari C (2006) Synergistic effects of plant-growth promoting rhizobacteria and Rhizobium on nodulation and nitrogen fixation by pigeonpea (Cajanus cajan). Eur J Soil Sci 57:67–71. doi:10.1111/j.1365-2389.2006.00771.x
Timmusk S, Paalme V, Pavlicek T, Bergquist J, Vangala A, Danilas T, Nevo E (2011) Bacterial distribution in the rhizosphere of wild barley under contrasting microclimates. PLoS One 6:e17968
Wani PA, Khan MS, Zaidi A (2007) Effect of metal tolerant plant growth promoting Bradyrhizobium sp. (vigna) on growth, symbiosis, seed yield and metal uptake by greengram plants. Chemosphere 70:36–45. doi:10.1016/j.chemosphere.2007.07.028
Wani PA, Khan MS, Zaidi A (2008) Effects of heavy metal toxicity on growth, symbiosis, seed yield and metal uptake in pea grown in metal amended soil. Bull Environ Contam Toxicol 81:152–158. doi:10.1007/s00128-008-9383-z
Weaver RW, Danso SK (1994) Dinitrogen fixation. In: Weaver RW, Angle JS, Bottomley PS (eds) Methods of soil analysis: part 2-microbiological and biochemical properties. Soil Science Society of America, Madison, pp 1019–1045
Wei GH, Ma ZQ (2010) Application of rhizobia-legume symbiosis for remediation of heavy-metal contaminated soils. Acta Microbiol Sin 50(11):1421–1430 (In Chinese)
Yadegari M, Rahmani HA, Noormohammadi G, Ayneband A (2010) Plant growth promoting rhizobacteria increase growth, yield and nitrogen fixation in Phaseolus vulgaris. J Plant Nutr 33:1733–1743
Zablotowicz R, Tipping E, Lifshitz R, Kloepper J (1991) Plant growth promotion mediated by bacterial rhizosphere colonizers. In: Keister D, Cregan P (eds) The rhizosphere and plant growth, vol 14. Springer, Netherlands, pp 315–326. doi:10.1007/978-94-011-3336-4_70, Beltsville Symposia in Agricultural Research
Acknowledgments
This work was supported by funds from the 863 Project of China (2012AA101402) and National Science Foundation of China (41601337, 31125007 and 31370142).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PPTX 138 kb)
Rights and permissions
About this article
Cite this article
Kong, Z., Deng, Z., Glick, B.R. et al. A nodule endophytic plant growth-promoting Pseudomonas and its effects on growth, nodulation and metal uptake in Medicago lupulina under copper stress. Ann Microbiol 67, 49–58 (2017). https://doi.org/10.1007/s13213-016-1235-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-016-1235-1