Abstract
The present study was aimed at producing enhanced and sustained bioelectricity from distillery wastewater in a double chamber microbial fuel cell (MFC) by changing inter-electrode distance, inoculum and reactor volume. Using double chamber MFC with 1 L working volume, when the distance between the electrodes was kept shorter (1 cm), it generated power density of 1.74 W/m3, which was 42.5% higher than that of MFC with electrode spacing of 10 cm (1 W/m3). Using inoculum from different sources viz. garden soil (MFC-GS), wetland sediment (MFC-WS) and sludge from wastewater treatment plant (MFC-S), the highest open circuit voltage (OCV) of 0.84 V and power density of 2.74 W/m3 were produced by MFC-WS, which also showed sustained electricity production (1.68 W/m3) from the wastewater during a 10-day experiment. Relatively lower power density was generated from MFC-S (1.42 W/m3), while that from MFC-GS was the lowest (0.94 W/m3). Bioelectricity generation and overall performance were then assessed using a smaller reactor size. Smaller working volume of MFC (250 ml) favoured greater production of power density (3.2 W/m3) than that with 1 L working volume (2.96 W/m3) with electrode distance of 1 cm. The present study was novel in selecting a suitable mixed-microbial inoculum out of the diverse sources screened and reducing resistance by sharply narrowing down inter-electrode distance and reactor volume, which led to significantly enhanced and sustained electricity generation from double chamber MFC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Microbial fuel cells (MFCs) have emerged as a relatively newer option in the recent years performing the dual function of wastewater treatment and electricity production (Logan 2005). High organic load in various types of wastewaters has been a major water pollution issue leading to development of different types of treatment technologies, mediated by different aerobic and anaerobic microbes. These conventional technologies, though effective, are costly and energy demanding, and mostly end up with toxic sludge generation. The MFCs, on the other hand, treat the organic substrate in the wastewaters through bio-electrochemical processes mediated by microorganisms which is cheaper and produce negligible sludge. Though MFCs show great promise for varied applications, yet there are certain bottlenecks and there is an urgent need to improve their performance for enhanced power density and pollutant removal from wastewaters to make it a commercially viable technology. For exploiting the MFCs for their maximum performance, optimization of operational parameters has been found to be important (Anam et al. 2020). Earlier researchers have focussed on MFC performance in relation to various operational parameters like pH (Kaushik and Chetal 2013; Singh and Kaushik 2020) temperature (Ren et al. 2017), conductivity (Karthikeyan et al. 2016), electrode material (Hamed et al. 2020), proton exchange membrane (Lee et al. 2015), inoculum type (Vázquez-Larios et al. 2011), catholyte (Wei et al. 2012), substrate type and concentration (Ullah and Zeshan 2020) and shown that power generation and COD removal from wastewater were enhanced by manipulating the parameters to optimal levels.
Microorganisms growing in the wastewater in the MFC feed on the organic substrate and in the process release electrons, which are utilized further in producing electricity. The type of microbial population in the biofilm plays a crucial role in both substrate degradation and power generation (Saratale et al. 2017). Microbial communities capable of transferring their electrons have been successfully established from different sources of microbial inocula such as blended wastewater (Elakkiya and Niju 2021), sludge (Salar-Garcia et al. 2020), fresh sediment (Armato et al. 2019), dye processing wastewater (Karuppiah et al. 2018), marine sediment (De Sá et al. 2017), manure (Pasupuleti et al. 2016) and compost (Cercado et al. 2013). Since, the type of microbial population in the biofilm plays a crucial role in both substrate degradation and power generation, the selection and suitability testing of inoculum source used in the MFC becomes important. Pure cultures of electrogens may be useful in high bioelectricity production in MFCs, but mixed consortia of microbes are more efficient practically as these are easier to obtain and handle. Using inoculum from different sources Mathuriya (2013) found mixed electrogenic inocula to be more effective in producing higher current. Proper selection of inoculum has been found to play a key role in improving performance of MFC as stressed by (Logan 2008) and Srikanth et al. (2018). However, the presence of electrogenic microbes in mixed consortium have been seen to be highly affected over time due to growth of some non-electrogenic microbes, particularly methanogenic microbes. Selective treatment of inoculum has been found to suppress the methanogens using 2-bromoethane sulfonic acid (BESA) (Pasupuleti et al., 2015), aluminium dosing (Bagchi and Behera 2019) and several others, as critically reviewed by Jadhav et al. (2019).
The present study was aimed at enhancing the power output by altering some architectural features of MFC, such as distance between electrodes and reactor volume and selecting a suitable source of mix-microbe inoculum screened from diverse sources. Performance of a mediator-less and membrane-less microbial fuel cell was earlier reported to be improved when the electrodes were placed 20 cm apart (Ghangrekar and Shinde 2007), while Sangeetha and Muthukumar (2013) reported 10 cm distance to favour better MFC performance when electrodes of 10–15 cm were used. The present study was designed for further narrowing down the electrode distance to a minimal of 1 cm with an aim to improve the performance of the MFC by minimizing restrictions in proton transfer that are known to increase internal resistance. Reactor volume of MFC is also reported to influence resistivity (Clauwaert et al. 2008) in MFC. Power density was reported to decrease 2–4 orders of magnitude in larger (litre scale) MFCs as compared to smaller (ml scale) MFCs (Fan et al. 2007a, b). For single cell MFCs, Vázquez-Larios et al. (2011) demonstrated reduction of ohmic resistance when electrode distance and working volume were reduced.
In the present study, effect of inter-electrode distance was studied in double chamber MFC (1L capacity) fed with distillery wastewater, which was followed by experiments using three mixed-microbial inoculum sources viz. wetland sediment, garden soil and sludge from wastewater treatment plant, and finally the wetland sediment inoculated MFCs of 1 L and 250 ml reactor sizes with minimal inter-electrode distance of 1 cm were compared for bioelectricity production. The novel approach in the present study involved screening out a suitable mix-microbe inoculum and decreasing the inter-electrode distance and reactor volume for reducing the resistance while providing sustained and significantly enhanced bioelectricity from double chamber MFC.
Materials and methods
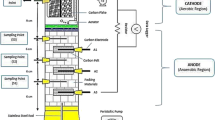
MFC configuration and working
Two sizes of double chamber H-shaped rectangular MFCs were fabricated viz. larger working volume (1 L) and smaller working volume (250 ml). The larger MFC with shorter spacing distance gave significantly improved output and therefore, for the smaller MFC, the electrode spacing distance was kept 1 cm only. The MFCs were equipped with graphite brush electrodes supported on aluminium-based alloy wire and separated using a proton exchange membrane (Nafion-117). Dimensions of the brush electrodes used in larger MFCs were 15 cm (L) × 9 cm (B) in the 1 L MFC, while that in small MFCs were 6 cm (L) × 6 cm (B) in 250 ml MFC. Electrodes were connected through a decade type external load (20–2000 Ω) as described by Singh and Kaushik (2019). Molasses based distillery wastewater collected from a local distillery plant was fed as substrate in anode chamber of the MFCs. The original wastewater was acidic (pH 4.7) and chemical oxygen demand (COD) of 67,000 mg/L. The 50% strength of distillery wastewater was used for conducting the experiments as it was the optimised strength already validated by this research group (Singh and Kaushik 2020). Potassium permanganate (3 g/L) was used as catholyte in the cathode chamber. Operating temperature of the MFCs was maintained at ambient conditions of 26–32 °C.
Experiment 1: electrode spacing
Electrode spacing in the 1 L MFCs were varied by keeping the length of the connecting arm between the two chambers as 10 cm (MFC-LS) and 1 cm (MFC-SS). No inoculum was added to the anode chamber in this experiment and distillery wastewater (50% strength) was fed as feedstock in anode chamber.
Experiment II: different inoculum sources
Three sources of inoculum were used: (i) Wetland sediment: sediment collected from Nazafgarh wetland, Haryana, India (28°30.303'N; 76°56.709'E) (depth: 15–20 cm); (ii) Garden soil: soil was collected from the university campus (depth: 0–5 cm); (iii) Wastewater sludge: it was collected from university sewage treatment plant. These were brought to the laboratory, air-dried and stored in refrigerator at 4 °C until used for the experiments. To compare the performance of these differently inoculated MFCs, 10 days’ batch experiments were run, one inoculated with wetland sediment (MFC-WS); other inoculated with garden soil (MFC-GS) and the last one inoculated with wastewater sludge (MFC-S) with 50% strength distillery wastewater fed as feedstock in anode chamber of each MFCs. The rest of the parameters such as anolyte, catholyte, pH was similar in all the MFCs.
Experiment III: working volume
MFC reactors with two different working volumes (1 L, 250 ml) were used, which had inter-electrode distance of 1 cm and the anode chamber was fed with the same distillery wastewater (50% strength) and the MFCs were inoculated with wetland sediment.
Bioelectricity measurement
Various output parameters (voltage (V), current (I), power (P) current density (CD) and power density (PD)) were measured in all the three experiments at regular time intervals. Multimeter (Model FLUKE 17B +) was used to record the voltage from the MFCs. Polarisation experiments were performed at different external resistances (R) (20–2000 Ω) to find out the internal resistance (IR) of MFC from slope of the graph plotted between voltage and current density. Power density and current density were calculated by dividing the power and current, respectively, by the working volume (v) of anode chamber. Following equations (1–4) were used for calculating current, power, power density and current density.
After initial start-up for 24 h, when stable voltage from the MFC was obtained, experiments were conducted.
Results and discussion
Effect of electrode spacing on power generation
The electrode spacing between electrodes had a remarkable impact on power generation. Power density of 1.74 W/m3 was produced in MFC with electrode spacing of 1 cm, which was 1.7 times that of MFC with electrode spacing of 10 cm (Fig. 1). The power density differences encountered between MFC-SS and MFC-LS, when tested through t-test were also statistically significant (p < 0.01). A recent study by Papillon et al. (2021) reported power improvement by 21% when electrode spacing was reduced from 6 to 4 cm and by 28% when spacing reduced from 4 to 2 cm. In the present study, on reducing the electrode distance from 10 to 1 cm, there was 42.5% improvement in power generation. When the inter-electrode distance is narrowed down, the protons produced in the anode chamber have to travel less to reach to the cathode, the reaction is faster and therefore, higher power density is produced. Cheng et al. (2006) also found that power generation was enhanced in single cell MFC when electrode spacing was reduced. Reducing electrode space from 100 to 50 cm in sediment MFCs resulted in better COD removal and higher power density (Sajana et al., 2013). High power output was also reported in a study by Khamis et al. (2020) with shorter distance between electrodes.
The voltage generation in open circuit mode was monitored in these MFCs up to 5 days in batch mode. The voltage generation was higher in MFC-SS (0.74 V) than MFC-LS (0.65 V). Proton transfer in the cathode chamber is a limiting step if proton permeability is poor. Poor permeability results in slow cathode reaction and hence could result in low electricity generation (Gil et al. 2003). In MFC-LS, there is lower electricity generation because of the larger distance between anode and cathode since protons would have taken longer time to reach the cathode, which will limit the proton availability and lowered efficiency.
With the help of polarization behaviour, internal resistance was also calculated in these MFCs (Fig. 1). It was found to be the least in MFC-SS (183.4 Ω) i.e. 34% lower than MFC-LS (277.9 Ω). This shows that larger distance between anode and cathode is creating increased resistance in the reactor and hence it is resulting in low power generation. The high internal resistance in larger spaced electrode is due to increased ohmic losses and mass transfer resistance. Mass transfer resistance is a limiting step in MFCs with larger spacing between electrodes. Harimawan et al. (2018) found least mass transfer resistance when the anode and cathode were in closer contact with the membrane, whereas when the distance increased, mass transfer resistance also increased and hence power density declined. Though, there are several studies suggesting the positive role of reduced electrode spacing in improving power generation, but in the present study, highly reduced electrode spacing of just 1 cm is taken, which yields superior MFC output. This shows that the distance between electrodes should be kept at minimum for exploiting the full potential of MFC.
Effect of inoculum sources on MFC output
Open circuit voltage (OCV) generation
Sustained electricity generation from three MFCs carrying distillery wastewater as substrate was found to be influenced significantly by the inoculum source. Voltage generation in the open circuit condition by three differently inoculated MFCs i.e., MFC-WS, MFC-GS and MFC-S are shown in Fig. 2. Though there was instantaneous voltage generation in all the three MFCs, but the same could not be sustained equally for a 10-day run. At 3 h, voltage generation by MFC-GS was maximum (0.8 V), followed by MFC-S (0.78 V), while MFC-WS produced least voltage of all (0.77 V). However, the trend was reversed with advancing run of the MFCs. The MFC-WS started producing maximum OCV (0.84 V) that remained stable over 10-day operation. However, voltage generation from MFC-GS as well as the MFC-S tended to sharply decline over time. It seems that microbial diversity in MFC-WS have rightfully exploited the substrate compared to that of MFC-GS and MFC-S. Though, the initial voltage was low in MFC-WS but with time due to better acclimatization to the anodic environment, the voltage stabilised and started increasing while in the other two cases, voltage declined with time. Mixed communities have different varieties of microorganisms that could be electrogenic or non-electrogenic or both. While MFC can operate with just one electrogenic microbe (Cao et al. 2019), presence of non-electrogenic microbes like methanogenic microbes can compete for food and space (Jadhav et al. 2019). In MFC-WS, it may be possible that electrogenic microbes outcompete the non-electrogenic microbes that facilitates higher electricity generation with time.
Polarization behaviour and output of power density
After acclimatization, all the MFCs were compared in terms of their polarization behaviour and production of power density. Since all the MFCs were able to produce quite high voltage in open circuit condition, it was important to study their behaviour on application of different resistances (closed circuit condition).
Polarization was performed to calculate the internal resistance of the system and to identify electron losses during transfer of the same from the microbes to the anode and ultimately to the cathode. Ohmic losses in all these MFCs with different inoculum were studied based on their polarization behaviour, which varied over the passage of operation time (Fig. 3a–d). Initially, internal resistance was minimum for MFC-GS (131.9 Ω) and the maximum internal resistance was shown by MFC-WS (140.4 Ω). However, on 4th day, the trend changed showing high internal resistance for MFC-GS (144.9 Ω) and lower internal resistance for MFC-WS (120 Ω). After 4 days of run, internal resistance of all the MFCs started increasing, with least increase in the MFC-WS. Thus, inoculum type had an influence on internal resistivity of MFCs and consequently on power production. Varied impact of different inoculum types on MFC resistance was shown by Vazques-Larios et al. (2011). Internal resistance in the MFC inoculated with aerobic consortium was much higher than that inoculated with methanogenic microbes and sulphate reducing consortium. Since wetland sediment is derived from anaerobic habitat, the microbial consortium seems to be well adapted to the anode environment favouring less resistance.
The trend of power density is represented in Fig. 3a–d. Highest power density was produced by MFC-GS on 1st day (2.63 W/m3) followed by that from MFC-WS (2.24 W/m3) and the least by MFC-S (1.63 W/m3). Power density however, tended to decline for the MFC-GS with time, and on 10th day it was the lowest (0.94 W/m3). The MFC-S maintained a higher power density (1.42 W/m3). The MFC-WS not only gave maximum power density of 2.74 W/m3, but also maintained a higher power density of 1.68 W/m3 till the end of the experiment. Thus, MFC-WS gave more stabilised power density for 10 days. The decline in power density by MFC-GS may be due to the presence of some non-electrogenic strains that quickly use up all the substrates and electricity generation declines. MFC-WS gave sustained power density, suggesting the involvement of more electrogenic strains which help produce stabilised power.
The source of inoculum in MFCs thus plays a major role in increasing the output of MFC and suggested to be a key parameter in MFC design (Cercado-Quezada et al. 2010). Microorganisms on the anode surface being responsible for degradation of contaminants and electricity production simultaneously (Logan and Regan 2006) are the most vital in MFC. The activity of the bacteria on the surface of anode is therefore particularly important for electricity production. Related to the present study, Ishii et al. (2017) also found differences in electrochemical features of MFCs while using different inoculum. Change in inoculum sources have recently been found to show significant changes in electricity generation by Marks et al. (2020). When inoculum source was replaced from anaerobic sludge to cow dung, it led to increase in voltage production due to change in microbial composition. Relationship between bacterial composition and originating inoculum was shown recently by Salar-Garcia et al. (2020) who observed changes in power density with changing microbial composition over a period of time. Ahmed et al. (2016) also stressed on the role of inoculum in MFC and found higher COD removal and power density using anaerobic sludge as inoculum compared to using a mixed-microbial solution.
The OCV and power density generated from the differently inoculated MFCs in the present study clearly show that wetland sediment used as a source of inoculum in the MFC gives the highest and sustained output. The wetland sediment being anaerobic got adapted to the anaerobic anode environment, and effectively formed a stable biofilm on the anode surface in the presence of distillery wastewater. Considering a major role played by microbes in electricity production, enrichment of sulphate reducing consortium was done with micronutrients like Fe (III) and Mn (IV) that gave very good results (Vázques-Larios et al. 2015). Another study reported betterment in MFC performance by mixing different types of inoculums (Sun et al. 2009). Thus, there is a scope for further improving MFC output by modifying the microbial inoculum.
Effect of working volume of MFC
As the reactor working volume of the MFC was reduced to 250 ml, there was a sharp increase in maximum power density value (3.2 W/m3) higher than that of larger MFC (1 L) in which maximum power density was 2.96 W/m3 (Fig. 4). The results of power density generation in both the MFCs were compared statistically through t-test. Significant difference (p < 0.01) was observed in the power density generation by these two MFCs. The resistance calculated from the polarization behaviour (Fig. 4) shows least internal resistance (67.2 Ω) generated in small size MFCs, and higher internal resistance (123.7 Ω) in larger sized MFCs. Clauwaert et al. (2008) analysed the volume based resistivity in MFCs and suggested that internal resistance is mainly created due to increased volume of reactor. This resistivity includes all the components of MFCs i.e., anode and cathode overpotential, overpotential created due to concentration, membrane and solution resistance. Resistance in small sized MFCs is thus found to be reduced. Studies have reported that with increase in MFC reactor size, the volumetric power density decreases (Ieropoulos et al. 2008). To increase the overall wastewater treatment effectivity, stacking of smaller units of MFCs is suggested to get better removal of COD and increased energy recovery from MFC. Jadhav et al. (2020) have critically discussed the challenges of large scale MFCs for practical applicability. However, some large capacity MFCs such as 720-L hexagonal MFC (Das et al. 2020), submerged 255 L prototype MFC (Hiegemann et al. 2019), 648 L four chamber concrete MFC (Akatah et al. 2019), 1500 L bioelectric toilet (Das et al. 2018) have been operated. Most of these studies conducted on large capacity MFCs are effective in sanitation wastewater treatment with considerable amount of electricity production.
Conclusion
The study shows that inoculum from different habitats influence the performance of MFCs and in the present study, the wetland sediment inoculum, which was adapted to anaerobic environment was the most suitable, developing a good biofilm on anode surface and producing remarkably higher and sustained power density from distillery wastewater. Narrowing down the inter-electrode distance to as low as 1 cm significantly reduced resistance and improved OCV and power density. Reducing the reactor volume also proved effective in improving MFC output. Thus, using MFC with 250 ml reactor volume, short inter-electrode spacing (1 cm) inoculated with wetland sediment inoculum led to least internal resistance (67.2 Ω) and enhanced and sustained power density (3.2 W/m3).
Data availability
All the data generated and analysed are reported in the present manuscript.
References
Ahmed S, Rozaik E, Abdel-Halim H (2016) Performance of single-chamber microbial fuel cells using different carbohydrate-rich wastewaters and different inocula. Pol J Environ Stud 25(2):503–510
Akatah BM, Kalagbor IA, Gwarah LS (2019) Electricity generation from septic waste water using septic tank as microbial fuel cell. Sustain Energy 7(1):1–5
Anam M, Yousaf S, Ali N (2020) Comparative assessment of microbial-fuel-cell operating parameters. J Environ Eng 146(9):04020093
Armato C, Ahmed D, Agostino V, Traversi D, Degan R, Tommasi T, Saracco G (2019) Anodic microbial community analysis of microbial fuel cells based on enriched inoculum from freshwater sediment. Bioprocess Biosyst Eng 42:697–709
Bagchi S, Behera M (2019) Methanogenesis suppression in microbial fuel cell by aluminium dosing. Bioelectrochemistry 129:206–210
Cao Y, Mu H, Liu W, Zhang R, Guo J, Xian M, Liu H (2019) Electricigens in the anode of microbial fuel cells: pure cultures versus mixed communities. Microb Cell Fact 18(1):1–14
Cercado B, Byrne N, Bertrand M, Pocaznoi D, Rimboud M, Achouak W, Bergel A (2013) Garden compost inoculum leads to microbial bioanodes with potential-independent characteristics. Biores Technol 134:276–284
Cercado-Quezada B, Delia ML, Bergel A (2010) Testing various food-industry wastes for electricity production in microbial fuel cell. Biores Technol 101(8):2748–2754
Cheng S, Liu H, Logan BE (2006) Increased power generation in a continuous flow MFC with advective flow through the porous anode and reduced electrode spacing. Environmen Sci Technol 40(7):2426–2432
Clauwaert P, Aelterman P, Pham TH, De Schamphelaire L, Carballa M, Rabaey K, Verstraete W (2008) Minimizing losses in bio-electrochemical systems: the road to applications. Appl Microbiol Biotechnol 79:901–913
Das I, Ghangrekar MM, Satyakam R, Srivastava P, Khan S, Pandey HN (2020) On-site sanitary wastewater treatment system using 720-L stacked microbial fuel cell: case study. J Hazard Toxic Radioact Waste 24(3):04020025
Das I, Jadhav DA, Ghangrekar MM (2018) Scaling up of microbial fuel cell for treatment of human waste to develop bioelectric toilet. In: International Conference on Sustainable Technologies for Intelligent Water Management, IIT Roorkee, India
De Sá JS, Mezzomo H, Fraga MF, Ogrodowski CS, Santana FB (2017) Anode air exposure during microbial fuel cell operation inoculated with marine sediment. J Environ Chem Eng 5(2):1821–1827
Elakkiya E, Niju S (2021) Bioelectrochemical treatment of real-field bagasse-based paper mill wastewater in dual-chambered microbial fuel cell. 3 Biotech 11(2):1–10
Fan Y, Hu H, Liu H (2007a) Enhanced coulombic efficiency and power density of air-cathode microbial fuel cells with an improved cell configuration. J Power Sources 171(2):348–354
Fan Y, Hu H, Liu H (2007b) Sustainable power generation in microbial fuel cells using bicarbonate buffer and proton transfer mechanisms. Environ Sci Technol 41(23):8154–8158
Ghangrekar MM, Shinde VB (2007) Performance of membrane-less microbial fuel cell treating wastewater and effect of electrode distance and area on electricity production. Biores Technol 98(15):2879–2885
Gil GC, Chang IS, Kim BH, Kim M, Jang JK, Park HS, Kim HJ (2003) Operational parameters affecting the performance of a mediator-less microbial fuel cell. Biosens Bioelectron 18:327–334
Hamed MS, Majdi HS, Hasan BO (2020) Effect of electrode material and hydrodynamics on the produced current in double chamber microbial fuel cells. ACS Omega 5(18):10339–10348
Harimawan A, Devianto H, Al-Aziz RHR, Shofinita D, Setiadi T (2018) Influence of electrode distance on electrical energy production of microbial fuel cell using tapioca wastewater. J Eng Technol Sci 50(6):841–855
Hiegemann H, Littfinski T, Krimmler S, Lübken M, Klein D, Schmelz KG, Wichern M (2019) Performance and inorganic fouling of a submergible 255 L prototype microbial fuel cell module during continuous long-term operation with real municipal wastewater under practical conditions. Bioresour Technol 294:122227
Ieropoulos I, Greenman J, Melhuish C (2008) Microbial fuel cells based on carbon veil electrodes: stack configuration and scalability. Int J Energy Res 32(13):1228–1240
Ishii S, Suzuki S, Yamanaka Y, Wu A, Nealson KH, Bretschger O (2017) Population dynamics of electrogenic microbial communities in microbial fuel cells started with three different inoculum sources. Bioelectrochemistry 117:74–82
Jadhav DA, Chendake AD, Schievano A, Pant D (2019) Suppressing methanogens and enriching electrogens in bioelectrochemical systems. Biores Technol 277:148–156
Jadhav DA, Das I, Ghangrekar MM, Pant D (2020) Moving towards practical applications of microbial fuel cells for sanitation and resource recovery. J Water Process Eng 38:101566
Karthikeyan R, Selvam A, Cheng KY, Wong JWC (2016) Influence of ionic conductivity in bioelectricity production from saline domestic sewage sludge in microbial fuel cells. Biores Technol 200:845–852
Karuppiah T, Pugazhendi A, Subramanian S, Jamal MT, Jeyakumar RB (2018) Deriving electricity from dye processing wastewater using single chamber microbial fuel cell with carbon brush anode and platinum nano coated air cathode. 3 Biotech 8(10):1–9
Kaushik A, Chetal A (2013) Power generation in microbial fuel cell fed with post methanation distillery effluent as a function of pH microenvironment. Biores Technol 147:77–83
Khamis A, Nordin N, Mokhtar MH (2020) Performance of double chamber microbial fuel cell: effect of wastewater, electrode thickness and distance. Indones J Electr Eng Comput Sci 20(2):619–626
Lee YY, Kim TG, Cho KS (2015) Effects of proton exchange membrane on the performance and microbial community composition of air-cathode microbial fuel cells. J Biotechnol 211:130–137
Logan BE (2005) Simultaneous wastewater treatment and biological electricity generation. Water Sci Technol 52(1–2):31–37
Logan BE, Regan JM (2006) Electricity-producing bacterial communities in microbial fuel cells. Trends Microbiol 14(12):512–518
Logan BE (2008) Microbial fuel cells. John Wiley & Sons, New Jersey
Marks J, Kirkel J, Sekoai P, Enweremadu C, Daramola M (2020) Effect of combining different substrates and inoculum sources on bioelectricity generation and cod removal in a two-chambered microbial fuel cell: a preliminary investigation. Environ Clim Technol 24(2):67–78
Mathuriya AS (2013) Inoculum selection to enhance performance of a microbial fuel cell for electricity generation during wastewater treatment. Environ Technol 34(13–14):1957–1964
Papillon J, Ondel O, Maire É (2021) Scale up of single-chamber microbial fuel cells with stainless steel 3D anode: Effect of electrode surface areas and electrode spacing. Bioresour Technol Reports 13:100632
Pasupuleti SB, Srikanth S, Dominguez-Benetton X, Mohan SV, Pant D (2016) Dual gas diffusion cathode design for microbial fuel cell (MFC): optimizing the suitable mode of operation in terms of bioelectrochemical and bioelectro-kinetic evaluation. J Chem Technol Biotechnol 91(3):624–639
Pasupuleti SB, Srikanth S, Mohan SV, Pant D (2015) Development of exoelectrogenic bioanode and study on feasibility of hydrogen production using abiotic VITO-CoRE TM and VITO-CASE TM electrodes in a single chamber microbial electrolysis cell (MEC) at low current densities. Bioresour Technol 195:131–138
Ren H, Jiang C, Chae J (2017) Effect of temperature on a miniaturized microbial fuel cell (MFC). Micro Nano Syst Lett 5(1):1–7
Sajana TK, Ghangrekar MM, Mitra A (2013) Effect of pH and distance between electrodes on the performance of a sediment microbial fuel cell. Water Sci Technol 68(3):537–543
Salar-Garcia MJ, Obata O, Kurt H, Chandran K, Greenman J, Ieropoulos IA (2020) Impact of inoculum type on the microbial community and power performance of urine-fed microbial fuel cells. Microorganisms 8(12):1921
Sangeetha T, Muthukumar M (2013) Influence of electrode material and electrode distance on bioelectricity production from sago-processing wastewater using microbial fuel cell. Environ Prog Sustain Energy 32(2):390–395
Saratale GD, Saratale RG, Shahid MK, Zhen G, Kumar G, Shin HS, Kim SH (2017) A comprehensive overview on electro-active biofilms, role of exo-electrogens and their microbial niches in microbial fuel cells (MFCs). Chemosphere 178:534–547
Singh A, Kaushik A (2019) Anode modification for increased power generation and COD removal in microbial fuel cell. Asian J Microbiol Biotechnol Environ Exp Sci 21(1):181–186
Singh A, Kaushik A (2020) Suitability of wetland microbial consortium for enhanced and sustained power generation from distillery effluent in microbial fuel cell. Energy Sour Part A Recover Util Environ Eff. https://doi.org/10.1080/15567036.2020.1864515
Srikanth S, Singh D, Vanbroekhoven K, Pant D, Kumar M, Puri SK, Ramakumar SSV (2018) Electro-biocatalytic conversion of carbon dioxide to alcohols using gas diffusion electrode. Biores Technol 265:45–51
Sun J, Hu Y, Bi Z, Cao Y (2009) Improved performance of air-cathode single-chamber microbial fuel cell for wastewater treatment using microfiltration membranes and multiple sludge inoculation. J Power Sour 187(2):471–479
Ullah Z, Zeshan S (2020) Effect of substrate type and concentration on the performance of a double chamber microbial fuel cell. Water Sci Technol 81(7):1336–1344
Vázquez-Larios AL, Poggi-Varaldo HM, Solorza-Feria O, Rinderknecht-Seijas N (2015) Effect of type of inoculum on microbial fuel cell performance that used RuxMoySez as cathodic catalyst. Int J Hydrogen Energy 40(48):17402–17412
Vazquez-Larios AL, Solorza-Feria O, Vazquez-Huerta G, Esparza-García F, Rinderknecht-Seijas N, Poggi-Varaldo HM (2011) Effects of architectural changes and inoculum type on internal resistance of a microbial fuel cell designed for the treatment of leachates from the dark hydrogenogenic fermentation of organic solid wastes. Inter J Hydrog Energy 36(10):6199–6209
Wei L, Han H, Shen J (2012) Effects of cathodic electron acceptors and potassium ferricyanide concentrations on the performance of microbial fuel cell. Int J Hydrog Energy 37(17):12980–12986
Acknowledgements
Financial assistance in the form of Indraprastha Research Fellowship (IPRF) to AS from Guru Gobind Singh Indraprastha University is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conceptualization and design. Material preparation, data collection and analysis were performed by AS. Review and editing were done by AK. The first draft of the manuscript was written by AS under supervision of AK. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interest or personal relationships that could have appeared to influence the work reported in this paper.
Rights and permissions
About this article
Cite this article
Singh, A., Kaushik, A. Sustained energy production from wastewater in microbial fuel cell: effect of inoculum sources, electrode spacing and working volume. 3 Biotech 11, 344 (2021). https://doi.org/10.1007/s13205-021-02886-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02886-6