Abstract
Coffee being the beverage consumed worldwide is also a very competitive commodity. Consequently, producers seek ways of attracting consumers by proposing e.g. novel ingredient combinations usually without evaluating their health quality. In this study, variations in health-promoting determinants for five special purpose coffee brews were characterized. The major bioactive components - chlorogenic acids (CAs) - detected by HPLC-DAD-MS included caffeoyl-, feruloyl- and dicaffeoylquinic acids. Roasting declined CAs content by 50 %, while caffeine content increased by about 30 % during this process. In roasted coffee brews studied, CAs content varied from 14.1 to 26.6 mg/g; smaller variations were seen in antioxidant potential (determined by spectrophotometric and cyclic voltammetry assays). Profiles generated by HPLC with post-column on-line antioxidant detection revealed that caffeoylquinic acids were the most abundant antioxidants in samples studied. The highest antioxidative potential exhibited coffee enriched with green coffee extract confirming the soundness of such an approach to obtain healthier products.
Highlights
• Special purpose coffees are equivalent to regular ones as regards antioxidant potential.
• Caffeoylquinic acids are the most abundant antioxidants in coffee samples studied.
• Roasting process causes some degradation of chlorogenic acids but not caffeine.
• Special purpose coffees retain health benefits while being more attractive to consumers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coffee infusions prepared from roasted coffee beans belong to the most widely consumed beverages in the world. This consumption is so high not only because of their pleasant flavour and aroma, but also stimulatory properties associated with caffeine content. Over the centuries, both anecdotical and scientific opinions about impact of coffee on human health were undergoing significant fluctuations, from totally negative to extremely positive. Very recent studies have revealed the previously unknown molecular mechanisms by which coffee consumption may influence the functions of human organism.
Among bioactive compounds present in coffee infusions, the best known is caffeine belonging to the most important psychoactive alkaloids. This compound enhances psychomotor performance, improves memory and cognitive function (Corley et al., 2010; Desbrow et al., 2012). In recent human studies, high blood caffeine levels (above 1.2 mg/L) entirely prevented the progression from mild cognitive impairment to dementia over 2–4 years period of patients’ monitoring (Cao et al., 2012). In addition, caffeine prevents non-alcoholic fatty liver disease (NAFLD), the hepatic manifestation of metabolic syndrome, which is the most common cause of primary liver disease (Birerdinc et al., 2012). It follows that currently caffeine is no longer seen only as a stimulant. Moreover, it is also becoming recognized that caffeine may have anticarcinogenic potential, e.g. in a large prospective study involving health professionals, the association between caffeine consumption and decreased skin cancer risk was found (Song et al., 2012).
The demonstrated beneficial effects of coffee consumption on health promotion and/or disease prevention such as improvement of gonad and liver function, combating metabolic (diabetes, liver cirrhosis, gallstones) and neurological disorders (Alzheimer’s and Parkinson’s diseases, infant hyperactivity), or psychoactive responses (alertness and mood change) (Dorea and da Costa, 2005; Higdon and Frei, 2006) justify some researchers’ proposition to place coffee among important functional foods. Even the issued in 2008 American Heart Association suggestion that coffee drinking may increase heart failure risk was contradicted by the results of subsequent epidemiological studies that found the inverse statistically significant association between this beverage consumption and heart failure risk, especially in the case of 4 servings per day (Mostofsky et al., 2012). Though it is too early to make firm conclusions, there are appearing reports describing so convincingly anticarcinogenic properties of coffee, that American Institute for Cancer Research (AICR) have recently decided to include this beverage into the list of “Foods that Fight Cancer”. It is important to note however, that none of these recommendations are directed to pregnant women, as maternal coffee consumption may not only retard fetus development, but it seems to increase the risk of childhood acute leukaemia according to the latest meta-analysis (Cheng et al., 2014).
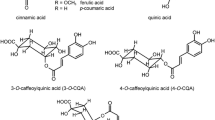
At least some of the above mentioned chemopreventive properties of coffee result from the fact that it stands out as a dietary source of antioxidants and free radical scavengers, in particular such its components as chlorogenic, caffeic and ferulic acids. It has been estimated that coffee drinks constitute 65 % of the daily intake of antioxidants in the human diet, leaving behind fruits, tea, wine and vegetables (Svilaas et al., 2004). Though roasting of coffee (light, medium, dark) causes decomposition of chlorogenic acids, it stimulates generation of Maillard reaction products including melanoidins or other low and high molecular mass compounds possessing antioxidant activity (Vignoli et al., 2014).
Though researchers concentrate on chemopreventive properties of coffee bioactive components, for producers most important is the fact that coffee is a very competitive commodity. To grasp consumers’ attention, the producers seek various ways of making their products more attractive, e.g. by proposing novel combinations of ingredients. However, it is not recognized how these marketing activities influence health-promoting properties of coffee brews. In this study, we compared content and composition of bioactive phytochemicals in five special purpose roasted coffee brands: low-irritating with diminished content of 5-hydroxytryptamides (green and roasted), supplemented with green coffee extract, organically grown, aromatized and with thin layer of dry mucilage (green and roasted). Phenolic acids and caffeine regarded as major determinants of bioactive properties, were identified and quantified by reverse phase HPLC with photodiode array and MS detection. The total antioxidant activity was determined by spectrophotometric (ABTS, DPPH, FC) and cyclic voltammetry assays. The profiles of antioxidants were generated using an HPLC system with post-column on-line antioxidant detection based on ABTS and Folin-Ciocalteu reagent (FCR) derivatization. The influence of roasting process on these parameters defining healthiness of coffee brews was also evaluated.
Materials and methods
Chemicals
Caffeine was purchased from Fluka, 5-O-caffeoylquinic acid (5-CQA), Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid), ABTS (2,2’azinobis-3-ethyl benzothiazoline-6-sulphonic acid) and DPPH (2,2-diphenyl-1-picrylhydrazyl) from Sigma-Aldrich (Germany), Folin-Ciocalteau’s reagent, HPLC grade methanol and formic acid from Merck (Germany). High purity water was produced in-house using a Milli-Q System (18.2 MΩ/cm). All other reagents were of analytical grade.
Coffee samples
Astra® low-irritant 100 % arabica, green (G1) and roasted with decreased level of carboxylic acids 5-hydroxytryptamides (R1) coffees derived from the same batch were obtained from the producer. Coffees Super® 100 % arabica, green (G2) and light roasted with thin layer of dry mucilage (R2) originating from the same batch were obtained from small local coffee roaster in Sopot, Poland. Aromatised, ethnic, delicately roasted 100 % arabica Sati® Splendid Moment from Costa Rica and Guatemala (R3), Sati® Response enriched with 0.7 % extract of green coffee containing 30 % more antioxidants compared to regular coffee as declared on the label (R4) and dark roasted, organically grown Sati-Bio® 100 % arabica (R5) were purchased in a local shop.
Preparation of coffee
Each sample of coffee beans was ground to powder. The samples of green and roasted coffees were brewed with boiling water in a ratio of 7 g of coffee and 100 mL of boiling water. After 8 min of brewing, coffee aqueous extracts were cooled and filtered through Chromafil Xtra MV-45/25 filters (Millipore). To prepare methanolic extracts, 1 g of green or roasted coffee was mixed with 5 mL of methanol and sonicated for 10 min. The extraction was repeated 3 times and the extract portions pooled together and filtered through paper filters. Both aqueous and methanolic filtrates were stored frozen at −20 °C until used for analyses.
Spectrophotometric determination of total antioxidant activity
Antioxidant activity of green and roasted coffee extracts was estimated using ABTS, DPPH and FCR tests as described previously (Kusznierewicz et al., 2012). Briefly, ABTS radical cation was produced in a solution containing 7 mmol/L ABTS in 2.45 mmol/L potassium persulphate. After 12–16 h in the dark at room temperature, the solution of ABTS radical cation formed was diluted with methanol to display the absorbance of 0.7 ± 0.02 at λ =734 nm. The DPPH solution freshly prepared in methanol was diluted until the absorbance reached 1.0 ± 0.02 at λ =515 nm. Commercial Folin-Ciocalteau’s reagent (FCR) solution was diluted with water (1:9 v/v). The determinations of antioxidant activity of coffee extracts were carried out in 48-well plates at room temperature. In each case, a Trolox solution was used to generate the calibration curve (concentration range 0–1 mg/mL). ABTS, DPPH or FCR solutions were aliquoted to plates in the amount of 1 mL per well. Then, the coffee extracts were added as follows: 10 μL, 30 μL or 100 μL in the case of ABTS, DPPH or FCR test, respectively. The absorbance of reaction mixtures was measured with UV–VIS Tecan Infinite M200 spectrophotometer (Tecan Group Ltd, Switzerland). If necessary, coffee extracts were diluted so as absorbance of reaction mixtures stayed within the linear range of calibration curve.
Cyclic voltammetric determination of total antioxidant activity
A potentiostat/galvanostat G 750 (Gamry Ins., USA) was used for cyclic voltammetry (CV) measurements carried out according to Cosio et al., (2006). The conventional three-electrode system consisting of a 3 mm diameter glassy carbon-working electrode (BAS MF-2012), an Ag/AgCl electrode as a reference one, and a platinum electrode as a counter electrode, was used. In order to avoid the decline of sensitivity, the working electrode was carefully polished with 0.05 μm alumina paste (Polishing alumina, BAS) and ultrasonically rinsed in deionized water at the end of each cycle. After washing with methanol, the electrode was ready for further tests.
The coffee extracts (100 μL) were transferred to voltammetric apparatus cell and mixed with 0.2 mol/L sodium acetate-acetic buffer (pH 4.5) as supporting electrolyte (100 μL). The cyclic voltammograms were recorded at room temperature by scanning the potential from −100 to +1200 mV at a scanning rate of 100 mV/s. The total charge below anodic wave curve of the voltammogram was calculated within 100 – 1200 mV based on the correlation between the total charge below the anodic wave of cyclic voltammograms represented by the area under the curve (AUC) and the antioxidant capacity of the sample. The 80 % methanol or water solutions of Trolox within the concentration range of 0.10 – 2.5 mmol/L served as reference. The total charge under anodic wave of the background signal (solvent + supporting electrolyte) was subtracted from the total charge under anodic wave obtained for each standard or coffee extract.
Chromatographic determination of bioactive compounds
Chlorogenic acids and caffeine were analysed by Agilent Technologies 1200 Series HPLC-DAD-MS system (Agilent Technologies, USA) equipped with Zorbax XDB C18 (150 × 4.60 mm, 5 μm) column. The mobile phase was combined of 5 % (v/v) formic acid in water (solvent A) and methanol (solvent B). During analysis the gradient of solvent B linearly increased from 10 to 65 % over 50 min. The flow rate of mobile phase was set at 0.7 mL/min, and the injection volume of all samples at 2 μL. Absorbance spectra were recorded between 190 and 700 nm while the chromatograms were monitored at 325 and 280 nm for chlorogenic acids and caffeine, respectively. MS analyses were performed using a spectrometer (quadrupole analyser, ESI, Agilent) for the following parameters: capillary voltage, 3000 V; fragmentor, 120 V; drying gas temperature, 350 °C; gas flow (N2), 12 L/min; nebulizer pressure, 35 psig. The instrument was operated in a negative ion mode, scanning from m/z 100 to 800. Individual compounds were identified by comparing their retention times with those of standards or on the basis of available literature data and mass spectra (Clifford, Johnston, Knight, & Kuhnert, 2003; Fujioka & Shibamoto, 2008). For quantitative determination of analytes, the calibration curves were generated by the integration of the areas of absorption peaks determined during chromatographic analysis of serial dilutions of caffeine or 5-CQA.
On-line profiling of antioxidants
For on-line profiling of antioxidants, the HPLC-DAD system (Agilent Technologies, USA) was connected with Pinnacle PCX Derivatisation Instrument (Pickering Laboratories, Inc., USA) and UV–vis detector (MWD Agilent Technologies, USA). The conditions of chromatographic separation of bioactive compounds present in coffee extracts were identical with those described above. The post-column derivatisation procedure was done according to Kusznierewicz et al., (2011). In all experiments, the 0.5 mL (PTFE, 0.25 mm, 10 m) reaction coil heated to 130 °C was used. The derivatisation reagents were prepared as follows: ABTS was dissolved in aqueous sodium persulphate (2.45 mmol/L) to obtain concentration of 7 mmol/L. The mixture was stored in the dark at room temperature for 12 h and then diluted with methanol to the concentration of 30 % (v/v). Commercially available FCR was diluted with water to a concentration of 40 % (v/v). ABTS and FCR solutions were fed into the system at the flow rate of 0.1 mL/min. Chromatograms after derivatisation with either ABTS or FCR were registered at 734 and 750 nm, respectively.
Results and discussion
Selection of solvents for extraction
Methanol is the most frequently used solvent to extract bioactive compounds from plant material. Also in the case of coffee beans, methanol turned out to give higher yield of antioxidants than hexane, chloroform or acetone (Ramalakshmi et al., 2008). Therefore, this solvent was initially selected to quantitatively isolate phytochemicals from coffee beans. On the other hand, coffee is drunk in a form of water brew, so it was essential to compare the recovery of phytochemicals achieved with methanol to that with boiling water. Ideally, both these methods should give comparable results confirming that methanolic extracts indeed reflect the composition of what is being consumed. This assumption was verified by evaluating the antioxidant potential of water and methanol extractable phytochemicals with ABTS, DPPH, FCR and CV tests. For the comparisons, three roasted coffees were selected: R3 - aromatised, R4 - enriched with green coffee extract and R5 - organically grown coffee, as in their case the substantial differences in composition could be expected, hence diversified solubility of bioactive compounds in two solvents verified.
The results of measurements for all tests applied indicated profound discrepancy in the content of antioxidant compounds between methanolic and water extracts. Firstly, the water extracts were characterised by stronger colour intensity that suggested higher content of Maillard reaction products such as brown pigments. Secondly, the shapes of cyclic voltammograms presented in Fig. 1 varied substantially depending on the solvent used. Maximum currents for water and methanolic coffee extracts were observed at 0.8 and 1.2 V, respectively. Such a shift of peaks suggests that different groups of electroactive compounds dominated in each type of extract. Oxidative potentials of strong antioxidants, such as chlorogenic acids, ferulic acid, caffeic acid and gallic acid are in the range from 0.4 to 0.6 V. Thus, based on the shape of voltammograms, it can be concluded that these phenolic acids were underrepresented in methanolic extracts.
The representative cyclic voltammograms of (a) - water extracts (70 mg/mL) or (b) - methanolic extracts (76 mg/mL) of different modified coffees: R3 - aromatized, R4 - enriched with green coffee extract and R5 - organically grown. Voltammograms were recorded from −100 to +1200 mV at the scan rate of 100 mV/s
The summary of antioxidant activities measured for extracts by spectrophotometric and electroanalytical methods is presented in Table 1. Water extracts displayed approximately 20 %, 40 %, 60 % and 80 % higher antioxidant activity than methanolic ones in CV, FCR, ABTS and DPPH tests, respectively. This discrepancy between determinations is caused by different chemical specificity of the assays. Based on the results presented, it can be presumed that the methanolic extracts were deprived especially of hydrophilic compounds detected with the highest sensitivity by DPPH test. Altogether, these data suggest that the use of methanol provides no advantage as regards the isolation of phytochemicals from coffee beans and further experiments were performed only for brews of ground beans with boiling tap water.
Influence of coffee processing on the content of bioactive compounds
In brewed coffee, caffeine and antioxidants, such as chlorogenic acids, are the most valuable compounds that may have an influence on human health. However, it is well known that roasting process transforms the chemical and physical properties of coffee bringing about changes in colour, taste, smell and even size of coffee beans (Franca et al., 2009). This process on one hand leads to the formation of Maillard reaction products, on the other to the decomposition of chlorogenic acids and consequently may change antioxidant activity of coffee (Liu and Kitts, 2011; Vignoli et al., 2014). Therefore, it was essential to study the influence of roasting and also additional processing on the health quality of the ready for sale special purpose coffees. For this comparison, two coffees processed in a different way were chosen. Coffee G1 was roasted and additionally treated so as to diminish the content of stomach-irritating C5-HT in the final R1 roasted coffee. Coffee G2 contained a thin layer of dry mucilage. Mild processing conditions according to the producer allowed to obtain light roasted and full aroma coffee R2.
The results of spectrophotometric and electroanalytical tests presented in Fig. 2 indicate that roasting and additional processing of G1 coffee had negative influence on the total antioxidant activity of coffee beans. ABTS, FCR, DPPH and CV tests showed that G1 coffee during this process lost about 13 %, 26 %, 31 % and 67 % of antioxidants, respectively. Theoretically, the content of antioxidants in coffee depends on roasting conditions, so it should be possible to pinpoint whether additional processing needed to remove irritants influenced the abundance of antioxidants in the final coffee product. Unfortunately, the results of previous studies regarding the relationship between the antioxidant activities and degree of roasting are inconsistent. For example Perrone et al. (2012) observed the highest antioxidant activity for green coffee, while Duarte et al., (2005) indicated that degree of roasting is inversely correlated with antioxidant activity and observed the highest values for light roasted coffee. Different results were obtained by Del Castillo et al. (2002) and Napolitano et al. (2007), who found that light and medium roasted coffee displayed greater antioxidant activity than green coffee. Moreover, the results presented by Vignoli et al. (2014) indicated that antioxidant activity of coffee may be constant during roasting and is dependent on the balance of the compounds formed during this process. Therefore, based on the available data, it is hard to decide if the elimination of C5-HT from coffee is connected with the loss of antioxidants.
The representative cyclic voltammograms of water extracts (70 mg/mL) of green (G1, G2) and corresponding roasted (R1, R2) coffees, recorded from −100 to +1200 mV at the scan rate of 100 mV/s (a) and the total antioxidant activity determined by ABTS, FCR, DPPH and CV method (b). Antioxidant capacity is expressed as mg Trolox equivalents per g of coffee beans and represents means ± SD of three independent measurements
Technology of G2 coffee roasting contributed to the reduction of degradation of antioxidants and in the case of some of them even caused the increase in concentration in this coffee brews as was indicated by ABTS test (Fig. 2). Positive effect may be attributed to the mild roasting conditions or to protective effect of the layer of mucilage, which might slow the rate of beans heating and consequently, reduced the degradation of thermally labile components.
Based on the chemical specificity of the assays used, it can be concluded that during roasting of green coffees studied, hydrophilic antioxidants were degraded to a greater extent. Moreover, the amount of reducing agents, especially strong antioxidants that exhibit low redox potential, decreased during processing, as indicated by the voltammograms presented in Fig. 2. Results of CV test were confirmed by more detailed analysis of the chemical composition by HPLC (Fig. 3). After roasting, both green coffees lost thermally labile CAs that play an important role in the final cup quality and health benefits of coffee. Our determinations and those reported by others showed that light roasting conditions, additional processing or presence of mucilage as well as its inner layer of silverskin, which contain similar CAs content to roasted coffee (Bresciani et al., 2014) did not prevent the decomposition of these important chemopreventive phenolics. The content of CAs declined with the degree of roasting (Vignoli et al. 2014) and the rate of reduction amounted to 15.9 % for light, 57.6 % for medium and 84.2 % for dark roasted arabica coffee (Budryn et al., 2009). In our study, the total CAs content ranged from 53.6 mg/g to 59.6 mg/g for G2 and G1 coffees, respectively. The decline of CAs level during processing of G1 to R1 coffee was about 50 %, while for G2 to R2 about 58 %. These results classify beans studied as medium roasted. Degradation of CAs in the latter green coffee was higher than expected for light roasting declared by the producer and could be caused by specific roasting conditions or different method of sample preparation.
The sample HPLC-DAD chromatograms obtained for water extracts of green (G1, G2) or corresponding roasted (R1, R2) coffees derived from two producers. The peak numbers in top of chromatograms (a) refer to: chlorogenic acids (peaks 1–3 and 5–8 at 325 nm) and caffeine (peak 4 at 270 nm). Top HPLC-DAD chromatograms in each series - (a) are assembled with profiles of antioxidants detected on-line for each coffee extract with either FCR - middle chromatograms monitored at 750 nm (b) or ABTS - bottom chromatograms monitored at 734 nm (c). For identity of peaks see Table 2
During roasting process, the concentration of CAs, especially 5-CQA, decreased, but the overall number of peaks attributed to antioxidants increased (Fig. 3). As a result, antioxidant activity of the coffees during processing did not change as much as CAs concentration. It has been shown that CAs may be incorporated into high molecular weight melanoidins that result from caramelization of carbohydrates or combination of sugars and amino acids through the Maillard reactions (Bekedam et al., 2008). These new compounds often exhibit antioxidant activity and may be a source of new antioxidants in human diet (Liu and Kitts, 2011; Vignoli et al., 2014). Nonetheless, it is important to emphasize that health benefits of melanoidins and chlorogenic acids differ. Although, both classes of compounds exhibit anticarcinogenic activities (Vitaglione et al., 2012), the in vivo experiments conducted in mice on high-fat diet suggested that chlorogenic acids may additionally improve body weight control, lipid metabolism and cholesterol levels (Cho et al., 2010). It is not known, whether such activity is also exhibited by melanoidins.
Comparison of composition of bioactive compounds in roasted special purpose coffees
Four of tested roasted coffees (R1, R2, R4, R5) are promoted as products, which may have positive impact on human health, while coffee R3 as a product with enhanced taste and aroma. Flavour of coffee is determined by the shifts in the concentration of aroma substances developed during roasting (Buffo and Cardelli-Freire, 2004; Vignoli et al. 2014), while the health-beneficial properties are attributed to antioxidants considered as important biologically active ingredients of food. To compensate for the losses of some compounds that may have beneficial effect, roasted coffee may be enriched with green coffee phytochemicals obtained by the extraction with alcohol (Thom, 2007) or hot water (Upadhyay et al., 2012). Such extracts contain secondary metabolites of green coffee which are known to influence reduction of body mass and body fat (Shimoda et al., 2006; Thom, 2007) to exhibit antibacterial activity (Antonio et al., 2011; Arora et al., 2009) or to lower blood pressure (Watanabe et al., 2006). The most abundant beneficial non-nutrients found in green coffee extracts are CAs (Esquivel and Jimenez, 2012). Our results indicated, that during roasting of coffee beans, the content of these compounds decreased (Fig. 4), so the idea of reintroducing them into roasted coffee seems to be justified. Coffee R4 enriched with green coffee extract contained more CAs than other samples from the same producer (R3, R5), but less than R1 and R2 coffees. The latter indicates that both quality of beans and the roasting technology are important in ensuring healthiness of coffee brews. Although, the addition of green coffee extract may improve health benefits associated with coffee drinking, it also changes taste and aroma of the beverage. As a result, the dose of extract added is usually limited. Such a limitation may be overcome by designing specific coffee blends like the one proposed by Ribeiro et al. (2014) that includes 3 % cocoa powder, 2 % coffee silverskin and 1 % golden (minimally processed) coffee.
Comparison of the content of chlorogenic acids in water extracts of special purpose coffees and their antioxidant activity measured by post-column derivatisation with ABTS or FC. The coffees studied included: G1 and R1 Astra® low-irritant; G2 and R2 Super light roasted®; R3 Sati® Splendid moment; R4 Sati® Response and R5 Sati® Bio. The bars correspond to means of three independent measurements. Standard deviations did not exceed 5 % of the means
There are studies suggesting that coffee plants exposed to environmental stress increase production of some primary (Bento dos Santos et al., 2011), as well as secondary metabolites (Simkin et al., 2008). In the light of these data, it could be expected that the content of CAs in organically grown coffee will be elevated. As presented in table 2, coffee R5 contains less 3-CQA, 5-CQA and 4,5-diCQA than other samples. This finding however not necessarily contradicts the above expectations. High content of melanoidins confirmed by spectrophotometric analysis (λ = 420 nm; data not shown) may suggest that the aggressive roasting conditions were applied and partial incorporation of CAs into melanoidins took place. In terms of health benefits associated with the presence of CAs, coffees R1 and R2 seem to represent the best sources of these compounds. Furthermore, these coffees are also a good source of caffeine, the main alkaloid of coffee, which is well-known for raising blood circulation and stimulation of the central nervous system (Cao et al., 2012; Terai et al., 2012).
The brews of green coffees G1 and G2 contained less caffeine than their counterparts R1 and R2 (Table 2). This phenomenon was also observed by other laboratories (Budryn et al., 2009) and can be explained by the lower efficiency of extraction of caffeine trapped in green plant tissue or by weight loss of coffee beans during roasting. Caffeine is regarded as a good scavenger of · OH radicals, but is inert to ABTS and DPPH radicals (Brezova et al., 2009). Data presented in Table 2 are consistent with this previous study and also indicate that caffeine does not interfere with typical coffee antioxidants when FCR reagent is used.
Conclusion
The special purpose coffees examined in our study did not differ from typical products of that kind and the level of antioxidants such as CAs and caffeine seemed to depend on roasting process or coffee variety not additional treatment they were submitted. As expected, roasting process caused degradation of CAs and formation of new antioxidants, whose health benefits are not always well recognized. Especially, roasted organically grown coffee contained lower level of phenolics, but high dose of melanoidins, which may also exhibit antioxidant activity and are regarded by some researchers as compounds with health promoting activity. The addition of green coffee extract into roasted coffee beans elevated the content of CAs and caffeine. In conclusion, the additional treatments such as addition of aromatic substances or removal of stomach irritants did not seem to alter the composition of desirable phytochemicals, thus may provide consumers with more attractive coffee products without compromising chemopreventive properties of this beverage.
References
Antonio AG, Iorio NL, Pierro VS, Candreva MS, Farah A, dos Santos KR, Maia LC (2011) Inhibitory properties of Coffea canephora extract against oral bacteria and its effect on demineralisation of deciduous teeth. Arch Oral Biol 56(6):556–564. doi:10.1016/j.archoralbio.2010.12.001
Arora DS, Kaur GJ, Kaur H (2009) Antibacterial Activity of Tea and Coffee: Their Extracts and Preparations. Int J Food Prop 12(2):286–294. doi:10.1080/10942910701675928
Bekedam EK, Schols HA, Van Boekel MA, Smit G (2008) Incorporation of chlorogenic acids in coffee brew melanoidins. J Agric Food Chem 56(6):2055–2063. doi:10.1021/jf073157k
Bento dos Santos T, Budzinski IGF, Marur CJ, Petkowicz CLO, Pereira LFP, Vieira LGE (2011) Expression of three galactinol synthase isoforms in Coffea arabica L. and accumulation of raffinose and stachyose in response to abiotic stresses. Plant Physiol Biochem 49(4):441–448. doi:10.1016/J.Plaphy.2011.01.023
Birerdinc A, Stepanova M, Pawloski L, Younossi ZM (2012) Caffeine is protective in patients with non-alcoholic fatty liver disease. Aliment Pharmacol Ther 35(1):76–82. doi:10.1111/J.1365-2036.2011.04916.X
Bresciani L, Calani L, Bruni R, Brighenti F, Del Rio D (2014) Phenolic composition, caffeine content and antioxidant capacity of coffee silverskin. Food Res Int 61:196–201. doi:10.1016/j.foodres.2013.10.047
Brezova V, Slebodova A, Stasko A (2009) Coffee as a source of antioxidants: An EPR study. Food Chem 114(3):859–868. doi:10.1016/J.Foodchem.2008.10.025
Budryn G, Nebesny ED, Podsędek A, Żyżelewicz DJ, Materska M, Jankowski S, Janda B (2009) Effect of different extraction methods on the recovery of chlorogenic acids, caffeine and Maillard reaction products in coffee beans. Eur Food Res Technol 228(6):913–922. doi:10.1007/s00217-008-1004-x
Buffo RA, Cardelli-Freire C (2004) Coffee flavour: an overview. Flavour Frag J 19(2):99–104. doi:10.1002/Ffj.1325
Cao C, Loewenstein DA, Lin X, Zhang C, Wang L, Duara R, Wu Y, Giannini A, Bai G, Cai J, Greig M, Schofield E, Ashok R, Small B, Potter H, Arendash GW (2012) High Blood caffeine levels in MCI linked to lack of progression to dementia. J Alzheimer Dis 30(3):559–572. doi:10.3233/JAD-2012-111781
Cheng J, Su H, Zhu R, Wang X, Peng M, Song J, Fan D (2014) Maternal coffee consumption during pregnancy and risk of childhood acute leukemia: a metaanalysis. Am J Obstet Gynecol 210 (2). doi: 10.1016/j.ajog.2013.09.026
Cho AS, Jeon SM, Kim MJ, Yeo J, Seo KI, Choi MS, Lee MK (2010) Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem Toxicol 48(3):937–943. doi:10.1016/j.fct.2010.01.003
Clifford MN, Johnston KL, Knight S, Kuhnert N (2003) Hierarchical scheme for LC-MSn identification of chlorogenic acids. J Agric Food Chem 51(10):2900–2911. doi:10.1021/jf026187q
Corley J, Jia XL, Kyle JAM, Gow AJ, Brett CE, Starr JM, McNeill G, Deary IJ (2010) Caffeine consumption and cognitive function at Age 70: the Lothian Birth Cohort 1936 study. Psychosom Med 72(2):206–214. doi:10.1097/Psy.0b013e3181c92a9c
Cosio MS, Buratti S, Mannino S, Benedetti S (2006) Use of an electrochemical method to evaluate the antioxidant activity of herb extracts from the Labiatae family. Food Chem 97(4):725–731. doi:10.1016/J.Foodchem.2005.05.043
Del Castillo MD, Ames JM, Gordon MH (2002) Effect of roasting on the antioxidant activity of coffee brews. J Agric Food Chem 50(13):3698–3703
Desbrow B, Biddulph C, Devlin B, Grant GD, Anoopkumar-Dukie S, Leveritt MD (2012) The effects of different doses of caffeine on endurance cycling time trial performance. J Sports Sci 30(2):115–120. doi:10.1080/02640414.2011.632431
Dorea JG, da Costa TH (2005) Is coffee a functional food? Br J Nutr 93(6):773–782
Duarte SMS, Abreu CMP, Menezes HC, Santos MH, Gouvêa CMCP (2005) Effect of processing and roasting on the antioxidant activity of coffee brews. Cienc Tecnol Aliment 25:387–393
Esquivel P, Jimenez VM (2012) Functional properties of coffee and coffee by-products. Food Res Int 46(2):488–495. doi:10.1016/J.Foodres.2011.05.028
Franca AS, Oliveira LS, Oliveira RCS, Agresti PCM, Augusti R (2009) A preliminary evaluation of the effect of processing temperature on coffee roasting degree assessment. J Food Eng 92(3):345–352. doi:10.1016/j.jfoodeng.2008.12.012
Fujioka K, Shibamoto T (2008) Chlorogenic acid and caffeine contents in various commercial brewed coffees. Food Chem 106(1):217–221. doi:10.1016/J.Foodchem.2007.05.091
Higdon JV, Frei B (2006) Coffee and health: a review of recent human research. Crit Rev Food Sci Nutr 46(2):101–123. doi:10.1080/10408390500400009
Kusznierewicz B, Piasek A, Bartoszek A, Namiesnik J (2011) The optimisation of analytical parameters for routine profiling of antioxidants in complex mixtures by HPLC coupled post-column derivatisation. Phytochem Anal 22(5):392–402. doi:10.1002/pca.1294
Kusznierewicz B, Piekarska A, Mrugalska B, Konieczka P, Namiesnik J, Bartoszek A (2012) Phenolic composition and antioxidant properties of Polish blue-berried honeysuckle genotypes by HPLC-DAD-MS, HPLC postcolumn derivatization with ABTS or FC, and TLC with DPPH visualization. J Agric Food Chem 60(7):1755–1763. doi:10.1021/jf2039839
Liu YZ, Kitts DD (2011) Confirmation that the Maillard reaction is the principle contributor to the antioxidant capacity of coffee brews. Food Res Int 44(8):2418–2424. doi:10.1016/J.Foodres.2010.12.037
Mostofsky E, Rice MS, Levitan EB, Mittleman MA (2012) Habitual Coffee Consumption and Risk of Heart Failure A Dose–response Meta-Analysis. Circ-Heart Fail 5(4):401–405. doi:10.1161/Circheartfailure.112.967299
Napolitano A, Fogliano V, Tafuri A, Ritieni A (2007) Natural occurrence of ochratoxin A and antioxidant activities of green and roasted coffees and corresponding byproducts. J Agric Food Chem 55(25):10499–10504. doi:10.1021/jf071959+
Perrone D, Farah A, Donangelo CM (2012) Influence of coffee roasting on the incorporation of phenolic compounds into melanoidins and their relationship with antioxidant activity of the brew. J Agric Food Chem 60(17):4265–4275. doi:10.1021/jf205388x
Ramalakshmi K, Kubra IR, Rao LJM (2008) Antioxidant potential of low-grade coffee beans. Food Res Int 41(1):96–103. doi:10.1016/J.Foodres.2007.10.003
Ribeiro VS, Leitão AE, Ramalho JC, Lidon FC (2014) Chemical characterization and antioxidant properties of a new coffee blend with cocoa, coffee silverskin and green coffee minimally processed. Food Res Int 61:39–47. doi:10.1016/j.foodres.2014.05.003
Shimoda H, Seki E, Aitani M (2006) Inhibitory effect of green coffee bean extract on fat accumulation and body weight gain in mice. BMC Complement Altern Med 6:9. doi:10.1186/1472-6882-6-9
Simkin AJ, Moreau H, Kuntz M, Pagny G, Lin CW, Tanksley S, McCarthy J (2008) An investigation of carotenoid biosynthesis in Coffea canephora and Coffea arabica. J Plant Physiol 165(10):1087–1106. doi:10.1016/J.Jplph.2007.06.016
Song FJ, Qureshi AA, Han JL (2012) Increased Caffeine Intake Is Associated with Reduced Risk of Basal Cell Carcinoma of the Skin. Cancer Res 72(13):3282–3289. doi:10.1158/0008-5472.Can-11-3511
Svilaas A, Sakhi AK, Andersen LF, Svilaas T, Strom EC, Jacobs DR Jr, Ose L, Blomhoff R (2004) Intakes of antioxidants in coffee, wine, and vegetables are correlated with plasma carotenoids in humans. J Nutr 134(3):562–567
Terai N, Spoerl E, Pillunat LE, Stodtmeister R (2012) The effect of caffeine on retinal vessel diameter in young healthy subjects. Acta Ophthalmol (Copenh) 90(7):E524–E528. doi:10.1111/J.1755-3768.2012.02486.X
Thom E (2007) The effect of chlorogenic acid enriched coffee on glucose absorption in healthy volunteers and its effect on body mass when used long-term in overweight and obese people. J Int Med Res 35(6):900–908
Upadhyay R, Ramalakshmi K, Rao LJM (2012) Microwave-assisted extraction of chlorogenic acids from green coffee beans. Food Chem 130(1):184–188. doi:10.1016/J.Foodchem.2011.06.057
Vignoli JA, Viegas MC, Bassoli DG, Benassi MT (2014) Roasting process affects differently the bioactive compounds and the antioxidant activity of arabica and robusta coffees. Food Res Int 61:279–285. doi:10.1016/j.foodres.2013.06.006
Vitaglione P, Fogliano V, Pellegrini N (2012) Coffee, colon function and colorectal cancer. Food Funct 3(9):916–922. doi:10.1039/c2fo30037k
Watanabe T, Arai Y, Mitsui Y, Kusaura T, Okawa W, Kajihara Y, Saito I (2006) The blood pressure-lowering effect and safety of chlorogenic acid from green coffee bean extract in essential hypertension. Clin Exp Hypertens 28(5):439–449. doi:10.1080/10641960600798655
Acknowledgments
We acknowledge the financial support of the Polish Ministry of Science and Higher Education (MNiSzW) in the form of research grant (N N312 323039).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pilipczuk, T., Kusznierewicz, B., Zielińska, D. et al. The influence of roasting and additional processing on the content of bioactive components in special purpose coffees. J Food Sci Technol 52, 5736–5744 (2015). https://doi.org/10.1007/s13197-014-1646-6
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-014-1646-6