Abstract
Neoadjuvant chemoradiation followed by surgery has been the standard of care for locally advanced carcinoma esophagus. We present our experience and inference of various factors associated with the same treatment and the prognostic influence of the same. A retrospective analysis of a cohort of 132 squamous cell carcinoma esophagus patients post neoadjuvant chemoradiation operated with curative intent was carried out. The 2-year overall survival rate was 64.5%. A pathological complete response was achieved in 32.5% of patients and was the only factor that significantly determined overall survival (p = 0.048). Neoadjuvant chemoradiation before surgery for locally advanced squamous cell cancer of the esophagus remains the standard of care with a pathological complete response being a significant factor in predicting overall survival. More prospective randomized studies are necessary to analyze factors affecting and predicting a pathological complete response which would help organ preservation in patients with a complete response.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide esophageal cancer ranks seventh in terms of incidence and sixth in mortality [1]. India has an age-standardized incidence rate of 6.5 per 100,000 population for males and 4.2 per 100,000 population for females. This translates into approximately 47,000 new cases each year and 42,000 deaths [2].
Squamous cell carcinoma (SCC) accounts for approximately 80% of esophageal cancer in India [3, 4]. Adenocarcinoma is the major histological subtype in the western world [5].
Treatment options for esophageal cancer vary based on the stage and also the location of the tumor [6].
Multimodality treatment in the form of neoadjuvant chemoradiotherapy (NACTRT) followed by surgery has become the standard of care for advanced non cervical esophageal cancer [7,8,9].
Multiple studies have shown that patients who achieve a pathological complete response (PCR) have better disease-free and even overall survival with a greater benefit for SCC than for adenocarcinoma [7, 10,11,12].
There are a lot of international publications on esophageal cancer but there is a dearth of the same in an Indian setting. Our aim was to present our real-world experience from a tertiary cancer center of patients undergoing surgery post NACTRT for SCC of the esophagus. The objective was to study the survival of the above-mentioned patients and assess the factors which affect it in detail. In addition, we have studies for factors associated with PCR for SCC.
Material and Methods
This study is a retrospective study. After approval from the scientific review committee, patient data and follow-up were collected from the hospital database and telephonic communication.
Patient Population and Data Collection
From June 2016 to October 2020, 132 patients with histologically proven SCC of the esophagus, who received NACTRT and underwent surgery, were enrolled in this study. All patients were diagnosed by esophagoscopic biopsy and subject to a chest and upper abdominal computed tomography (CT) scan pre and post NACTRT. Bronchoscopy and fluorodeoxyglucose–positron emission tomography/computed tomography (FDG–PET/CT) scan were done on an individual basis.
Inclusion criteria were patients with SCC of the esophagus who underwent NACTRT followed by surgery. Patients were included if they were under the age of 75 and had a performance status of Eastern Cooperative Oncology Group (ECOG) 0 or 1.
Patients who were inoperable, had a follow-up less than 6 months, or who had a dual malignancy were excluded.
Treatment
Neoadjuvant Chemoradiotherapy
Our institute protocol was to administer NACTRT as per the CROSS protocol with a different radiation dose (7). Forty-five gray given in 25 fractions was the dosage of radiotherapy administered over 5 weeks. Concurrent dual-agent (platinum and taxane combination) chemotherapy was given weekly and was administered intravenously. Patients unable to withstand dual-agent chemotherapy were given only single-agent platinum chemotherapy.
Surgery
Surgery was performed 4 to 8 weeks after completion of NACTRT. Surgical procedures were performed either by an open or a video-assisted thoracoscopic surgical (VATS) approach. Based on the location and surgeon preference, a McKeown, Ivor Lewis, or transhiatal esophagectomy (THE) was done. A 2-field or extended 2-field lymphadenectomy was done in transthoracic approach and only single field (abdominal) was done in THE. Reconstruction was done in all cases with a gastric tube.
Pathological Analysis
Pathological examination included histology type, tumor extension, lymph node, and resection margin, perineural invasion, and lymphovascular invasion. The treatment response was assessed using the Modified Ryan Scoring system [13].
Follow-Up and Survival
Patients were followed up every 2 to 3 months for the first two of years and then at 6 monthly intervals. They underwent a history and physical examination on each follow-up. A contrast-enhanced CT scan of the thorax and upper abdomen was done at 6 months post-treatment and then yearly for the first 3 years. Endoscopy was done as clinically indicated.
Statistical Analysis
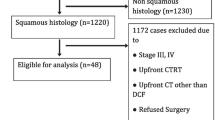
Statistical analysis was performed with the use of IBM SPSS software, version 25.0 (SPSS). Average and frequency distribution were analyzed by descriptive statistics. The Kaplan-Meier method was used to analyze survival. OS was calculated from the date of histological diagnosis to the date of death and censored at last follow-up. RFS was calculated from the date of surgery to the date of relapse and censored at death or last follow-up. Comparisons of factors affecting survival were performed with the log-rank test and using Cox regression. Comparisons of factors affecting PCR was calculated using binary logistic regression. A p value less than 0.05 was considered statistically significant (Fig. 1).
Results
Median age of the patients was 52 (range 28 to 71), and 61.36% (81) of the patients were male. More than half (55.3%) of tumors were located in the lower third of the esophagus including the gastroesophageal junction.
Dual-agent chemotherapy was given to 67.4% (89) of the patients. The majority, i.e., 56.8% of the patients, underwent VATS-guided surgery, 38.6% of the patients underwent open surgery, and 4.5% patients underwent THE. Of all these patients, a majority 88.6% underwent a McKeown esophagectomy. 81.1% of the patients underwent an extended 2 field lymphadenectomy.
A 30-day post-surgical mortality occurred in 7.5% (10) patients. Four patients expired due to ARDS, three had a cardiac event, two patients with thoracic anastomoses expired due to an anastomotic leak and one patient expired due to a tracheal fistula.
The median lymph node yield was 17 (interquartile range (IQR) − 11 to 23). Positive lymph nodes were identified in 34% (45) patients. An R0 resection was achieved in 98% of patients. PCR was observed in 32.5% (43) patients.
The median follow-up time of alive patients was 18 months (IQR − 12 to 25). The 2-year OS was 64.5% and the 2-year RFS was 75.6%. Of the various factors analyzed affecting overall survival, PCR was the only significant factor (p 0.048) (Table 1 and Table 2; Fig. 2).
As PCR was the only factor affecting survival on log rank test and had a trend towards affecting survival on cox regression, we conducted an analysis of various factors affecting PCR (Table 3). However, no significant factor could be identified.
Discussion
NACTRT followed by surgery is an acceptable practice for treatment of locally advanced cancer of the esophagus. The chemotherapy we administer is as per the CROSS protocol in most cases. However, the radiotherapy administered is 45 Gy [16].
Median age of the patients was 52 in our study which was almost a decade younger than what was seen in the CROSS trial [7]. This leads to more years of life lost than in the Western world, the cause of which needs to be further studied.
Initially 93% of our patients were SCC (prior to final inclusion) contrasting with the CROSS study in which only 23% of the patients had SCC [7]. Other studies from India by Anap et al. [17], Krishnamurthy et al. [18], and Bhattacharya et al. [19] also had all or majority of their patients as SCC. This underlines the much greater incidence of SCC, still, in India compared to the Western world (Table 4). Probable causes for the same would be the lower socio-economic status and lesser burden of obesity.
The Western world has a preponderance of cancer in the lower third as seen in the CROSS trial. In comparison, studies from India including ours have at least 40% of patients with cancer in the middle third (Table 4). This is most likely due to the majority of cases being of the squamous subtype in India.
67.4% (89) of the patients received double agent chemotherapy and 32.6% (43) of the patients received single-agent chemotherapy. Single-agent chemotherapy was given to those patients unable to withstand double-agent chemotherapy after a discussion in the institutional tumor board. There was however no difference in OS or RFS, which gives rise to the question of whether a single-agent chemotherapy is adequate to be given concurrently with radiation. This would have to be addressed in a proper randomized trial.
Positive lymph nodes were identified in 34% of patients which was similar to the 31% lymph node positivity in the neoadjuvant subgroup in the CROSS trial. Having at least a third of patients with positive lymph nodes underscores the need for an adequate lymphadenectomy even after chemoradiation.
The 2-year OS at a median follow-up time of 18 months (IQR 12 to 23 months) in our study was 64.5%, and the 2-year RFS was 75.6%. The CROSS trial had a 1-year and 2-year OS of 85% and 73% respectively for SCC patients. The study by Bhattacharya et al. had a 1-year OS of 84.4% and 2-year OS of 76.3% at a median follow-up of 16 months. Lower survival in our data could be due to advanced stage, poor nutrition, and post-op mortality occurring in 10 (7.57%) patients as ours is real-world data in contrast to the CROSS trial.
Of the various factors analyzed affecting OS in our study, PCR was the only significant factor (p 0.048). The 2-year OS was 81.4% and 64% for the PCR and nonPCR groups, respectively. A study by Berger et al. showed a significant OS for those with a PCR [10] (p = 0.015). However, a large majority of the patients in this study were adenocarcinoma. Similarly, a study by Meredith et al. showed PCR to be a significant factor for OS in patients with adenocarcinoma esophagus [11]. A study from Taiwan showed a significant survival difference for esophageal SCC patients with a PCR [20].
A PCR is an important prognostic factor in our and in various other studies as mentioned above. Hence, we studied factors affecting PCR which can be divided as patient, tumor, or treatment factors. In addition, there is a need to identify factors that can predict PCR.
Patient factors are age, sex, smoking status, and co-morbidities. Tumor or biological factors are the location of the tumor, histology, length, thickness, genetic makeup, and the activity on an FDG/PET scan. Treatment factors would be the dose and type of chemoradiation administered.
Huang et al. showed age, smoking, and the length of the tumor to be significant predictors of patients undergoing a PCR [21].
Li et al. recently published a paper that showed that the response to neoadjuvant treatment as assessed by a PET scan is a reasonable method to assess response [22].
A recent study compared proton versus photon radiation therapy and found photon beam to be equivalent to the former in getting a complete response [23].
MD Anderson devised a nomogram to predict PCR and another study used multimodal imaging (diffusion-weighted MRI and activity on an FDG/PET C) to predict a PCR [24, 25].
In our study, no specific factor was however found to determine which patient would undergo a pathological complete response. We did not study factors predicting PCR.
PCR being a significant factor that affects OS allows us to consider the possibility of organ preservation with an intensive follow-up schedule to facilitate surgery for those patients’ showing signs of disease.
Our study lacks in being a retrospective study with its associated biases. The median follow-up was only 18 months. A detailed comprehensive staging was not done for all patients. Chemoradiation administered was not standard for all patients. All the factors affecting and predicting PCR that are mentioned above were not studied.
In conclusion, NACTRT prior to surgery for locally advanced SCC esophagus remains the standard of care with PCR being a significant factor in predicting overall survival as per our real-world data from a tertiary cancer center in Western India. Additional prospective randomized studies are necessary to analyze factors affecting and predicting PCR which would help in organ preservation.
References
Sung H, Ferlay J, Siegel R, Laversanne M, Soerjomataram I, Jemal A et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249
[Internet]. Main.icmr.nic.in. 2022 [cited 14 February 2022]. Available from: https://main.icmr.nic.in/sites/default/files/guidelines/Esophagus%20final%20ICMR2014_0.pdf
Samarasam I (2017) Esophageal cancer in India: current status and future perspectives. Int J Adv Med Health Res 4(1):5
Pandey V, Choksi D, Kolhe K, Ingle M, Rathi C, Khairnar H et al (2020) Esophageal carcinoma: an epidemiological analysis and study of the time trends over the last 20 years from a single center in India. J Fam Med Prim Care 9(3):1695
Hur C, Miller M, Kong C, Dowling E, Nattinger K, Dunn M et al (2012) Trends in esophageal adenocarcinoma incidence and mortality. Cancer 119(6):1149–1158
Mawhinney MR, Glasgow RE (2012) Current treatment options for the management of esophageal cancer [Internet]. U.S. National Library of Medicine. [cited 2023 Oct 5] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3496368/
van Hagen P, Hulshof M, van Lanschot J, Steyerberg E, Henegouwen M, Wijnhoven B et al (2012) Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 366(22):2074–2084
Gebski V, Burmeister B, Smithers B, Foo K, Zalcberg J, Simes J (2007) Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol 8(3):226–234
AJCC Cancer Staging Manual [Internet]. SpringerLink. 2022 [cited 14 August 2022]. Available from: https://springerlink.bibliotecabuap.elogim.com/book/9783319406176
Berger A, Farma J, Scott W, Freedman G, Weiner L, Cheng J et al (2005) Complete response to neoadjuvant chemoradiotherapy in esophageal carcinoma is associated with significantly improved survival. J Clin Oncol 23(19):4330–4337
Meredith K, Weber J, Turaga K, Siegel E, McLoughlin J, Hoffe S et al (2010) Pathologic response after neoadjuvant therapy is the major determinant of survival in patients with esophageal cancer. Ann Surg Oncol 17(4):1159–1167
Donahue J, Nichols F, Li Z, Schomas D, Allen M, Cassivi S et al (2009) Complete pathologic response after neoadjuvant chemoradiotherapy for esophageal cancer is associated with enhanced survival. Ann Thorac Surg 87(2):392–399
Shi C, Berlin J, Branton PA et al (2017) In: available at http://www.cap.org (ed) Protocol for the examination of specimens from patients with carcinoma of the esophagus. College of American Pathologists Cancer Protocols, pp 1–17
Li S, Rau K, Lu H, Wang Y, Tien W, Liang J et al (2012) Pre-treatment maximal oesophageal wall thickness is independently associated with response to chemoradiotherapy in patients with T3-4 oesophageal squamous cell carcinoma. Eur J Cardiothorac Surg 42(6):958–964
Wu Y, Li J (2021) Change in maximal esophageal wall thickness provides prediction of survival and recurrence in patients with esophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy and surgery. Cancer Manag Res 13:2433–2445
Buckstein M, Rhome R, Ru M, Moshier E (2017) Neoadjuvant chemoradiation radiation dose levels for surgically resectable esophageal cancer: predictors of use and outcomes. Dis Esophagus 31(5):dox148
Anap Y, Tanawade P, Mathankar M, Mane-Patil A, Bagul K, Pawar R et al (2020) Preoperative chemoradiation in locally-advanced resectable carcinoma of the esophagus in a single rural cancer hospital in Western India. South Asian J Cancer 09(03):158–162
Krishnamurthy A, Mohanraj N, Radhakrishnan V, John A, Selvaluxmy G (2017) Neoadjuvant chemoradiation for locally advanced resectable carcinoma of the esophagus: a single-center experience from India with a brief review of the literature. Indian J Cancer 54(4):646
Bhattacharyya T, Arunsingh M, Chakraborty S, Harilal V, Sasidharan R, Saha S et al (2021) Can the CROSS protocol be safely implemented in real world scenario with broader eligibility criteria? Experience from a tertiary care centre in India. Ecancermedicalscience 15:1291.
Lin J, Hsu C, Yeh H, Chuang C, Lin C (2018) The impact of pathological complete response after neoadjuvant chemoradiotherapy in locally advanced squamous cell carcinoma of esophagus. J Chin Med Assoc 81(1):18–24
Huang R, Chao Y, Wen Y, Chang H, Tseng C, Chan S et al (2014) Predictors of pathological complete response to neoadjuvant chemoradiotherapy for esophageal squamous cell carcinoma. World J Surg Onc 12(1). https://doi.org/10.1186/1477-7819-12-170
Li C, Lin J, Yeh H, Chuang C, Chen C. Good prediction of treatment responses to neoadjuvant chemoradiotherapy for esophageal cancer based on preoperative inflammatory status and tumor glucose metabolism. Sci Rep 11(1):11626. https://doi.org/10.1038/s41598-021-90753-y
DeCesaris C, Berger M, Choi J, Carr S, Burrows W, Regine W et al (2020) Pathologic complete response (pCR) rates and outcomes after neoadjuvant chemoradiotherapy with proton or photon radiation for adenocarcinomas of the esophagus and gastroesophageal junction. J Gastrointestinal Oncol 11(4):663–673
Lin SH, Wang J, Allen PK, Correa AM, Maru DM, Swisher SG et al (2015) A nomogram that predicts pathologic complete response to neoadjuvant chemoradiation also predicts survival outcomes after definitive chemoradiation for esophageal cancer [Internet]. U.S. National Library of Medicine. [cited 2023 Oct 5] Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4294819/
Fang P, Musall B, Son J, Moreno A, Hobbs B, Carter B et al (2018) Multimodal imaging of pathologic response to chemoradiation in esophageal cancer. Int J Radiat Oncol *Biology*Phys 102(4):996–1001
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent
Written informed consent was obtained from all patients regarding publishing data.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mohamed Taher Mithi is the principal (first) author.
Mohit Sharma is the second author.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mithi, M.T., Sharma, M., Puj, K. et al. Neoadjuvant Chemoradiation Followed by Surgery for Locally Advanced Squamous Cell Carcinoma Esophagus: Demographics and Evaluation of Prognostic Factors at a Tertiary Care Center in India. Indian J Surg Oncol 15, 129–135 (2024). https://doi.org/10.1007/s13193-023-01828-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-023-01828-3