Abstract
To compare features of clinically defined subtypes of breast cancer on mammography (MG) and ultrasonography (USG). After obtaining approval from the institute ethics committee, a retrospective observational study was performed on biopsy-proven breast cancer patients who underwent baseline MG from 2016 to 2020. MG and USG features were evaluated and the patients were classified based on immunohistochemistry profile into luminal like (LL)-oestrogen receptor (ER)/progesterone receptor (PR) + , Her2neu-; basal like (BL)-ER/PR-, Her2neu-; Her2 like (HL)-Her2neu + . A total of 479 patients (mean age, 51.4 ± 11.7 years; all females) were included: LL—198 (41.3%), BL—121 (25.2%) and HL—160 (33.3%). On MG, round shape (21/115, 18.3%, p < 0.001); circumscribed (16/115, 13.9%, p < 0.001) and microlobulated margins (28/115, 24.4%) were associated with BL tumours. Associated suspicious calcifications (96/160, 60%, p < 0.001) and skin thickening or retraction (75/149, 50.3%, p < 0.001) were more common in HL. On USG, round shape (12/95, 12.8%, p = 0.005); circumscribed (8/94, 8.5%) and microlobulated margins (44/94, 46.8%) and posterior acoustic enhancement (7/95, 7.5%, p = 0.012) were associated with BL. The logistic regression analysis revealed that spiculated margins on MG favoured LL (OR: 8.5, p = 0.001); round shape (OR: 6.8), circumscribed (OR: 10.8) or microlobulated margins (OR: 3.5) (p < 0.001 for each) favoured BL; whereas associated features of calcifications (OR: 3.3) (p = 0.019) and skin retraction or thickening (OR: 1.8) (p < 0.001) favoured HL. On USG, circumscribed (OR: 5.9, p = 0.005) or microlobulated margins (OR: 3, p < 0.001) and posterior acoustic enhancement (OR: 9.5, p = 0.006) favoured BL. Clinically defined subtypes of breast cancer show significant differences in the imaging appearances on mammography and USG. BL tumours may not show the typical imaging features of malignancy, necessitating clinicopathological correlation for accurate diagnosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most common cancer of female population worldwide [1]. Evaluation of breast carcinoma includes clinical, imaging and histopathological examination. Imaging primarily includes mammography (MG) with adjunct modalities like tomosynthesis, ultrasonography (USG) and MRI, aimed at detecting the cancer, followed by cross-sectional imaging to ascertain the stage of the disease. Histopathological examination forms the mainstay for confirmation of the cancer and determination of receptor status which directs the type of treatment the patient will be subjected to. However, histopathologic examination may fail to diagnose carcinoma due to factors like sampling or observer errors [2]. Achievement of radio-pathological concordance thus becomes essential to guide appropriate management. Breast Imaging Reporting and Data System (BI-RADS), which consists of standardised terminology for reporting in breast radiology, is therefore used to determine the probability of malignancy based upon the imaging findings [3].
Breast cancer is a heterogenous disease with multiple molecular subtypes, each having varied features and prognosis. Various intrinsic subtypes have been described based on the gene expression pattern—luminal A, luminal B, Her2neu enriched and triple negative breast cancers [4]. However, since gene expression profiling is not widely available in low and middle countries, receptor status on immunohistochemistry (IHC) is used to classify the tumour into various clinically defined subtypes—luminal like (LL), basal like (BL) and Her2 like (HL) subtypes [5]. This subclassification is crucial in deciding the chemotherapeutic agents to be used for the management. Hormone receptor positive LL cancers benefit from hormonal therapy that cannot be followed in BL cancers as the latter lacks the expression of oestrogen receptor (ER), progesterone receptor (PR) and Her2neu. Although BL tumours have high incidence of pathological complete response after neoadjuvant chemotherapy, the chances of recurrence remain high due to lack of targeted therapy [6, 7]. Overexpression of Her2neu is associated with a greater incidence of axillary lymphadenopathy and a poorer prognosis [4, 8]. Monoclonal antibodies such as trastuzumab, which target Her2neu, have been found to improve survival rates in Her2neu positive breast cancers [9, 10].
The specific imaging features of malignancy are well known and they form the basis of the BI-RADS lexicon [3]. However, imaging appearances of each subtype would vary from each other, owing to the differences in their biologic behaviours. This study has been performed to determine and compare the imaging features of various clinically defined subtypes of breast cancer on mammography and USG, in order to emphasise on the significance of radio-pathological (rad-path) correlation and multidisciplinary approach for treatment of the patients.
Methods
This study was conducted in department of radiology at our cancer centre after obtaining approval from Institute Ethics Committee. In view of the retrospective study design, waiver for informed consent was obtained and patients with biopsy-proven breast cancer who underwent baseline MG between January 2016 and December 2020 in our department were included. The patients whose histopathological reports were not available were excluded from the study.
Imaging
Two-dimensional full-field digital MG with digital breast tomosynthesis was performed on Selenia Dimensions digital mammography system (Hologic, Inc., Massachusetts, USA) in mediolateral oblique and craniocaudal views. Only the baseline imaging of each patient was included in this study. As per BI-RADS lexicon, the descriptors of the mass included number, size, shape, margins, presence of calcifications and associated features including architectural distortion, skin retraction or thickening, nipple retraction and trabecular thickening. Presence of architectural distortion or focal asymmetry was also recorded.
Masses detected on mammography were further evaluated on USG using Acuson S2000 machine (Siemens Healthcare, Erlangens, Germany) as per our institute’s protocol. The features of the mass that were analysed were shape, margins, echogenicity, posterior features and associated features including skin thickening, duct changes and oedema.
The mammographic and USG images of the patients were reviewed by a radiologist having 10 years’ experience in breast radiology. The imaging features that were recorded were then compared with the IHC findings to ascertain the differences between various clinically defined subtypes.
Immunohistochemistry
IHC was performed on either the core biopsy specimen or the surgical specimen to assess the hormone receptor status (ER and PR) and Her2neu expression status. ER and PR status were categorised as positive or negative depending upon the Allred score. Her2neu receptor status was scored from 0 to 3 + , in which scores of 0 and 1 + were taken as negative, while 3 + was considered positive. A score of 2 + implied inconclusive results. In such patients, the results of fluorescent in situ hybridisation (FISH) for detection of Her2neu overexpression were taken into consideration. Among them, those who did not have FISH results were excluded from the analysis. Further, patients were classified into three clinically defined subtypes based upon the receptor status—LL (ER/PR + , Her2neu-), BL (ER-, PR-, Her2neu-) and HL (Her2neu positive cases, irrespective of ER/PR status) [5]. Patients whose IHC reports were not available were excluded from the analysis. The study flowchart is depicted in Fig. 1.
Statistical Analysis
After tabulating the collected data, the analysis was performed using Stata, v. 14.2 (StataCorp LP, Texas, USA). The frequencies of individual variables were evaluated and tabulated. The comparison of the imaging features and histological subtype was performed using Pearson’s chi square test or Fisher’s exact test for qualitative variables and Kruskal–Wallis test for quantitative variables. Further, logistic regression analysis was used to identify the imaging features that favoured a particular subtype. All statistical tests were two-sided and p value less than 0.05 was considered statistically significant.
Results
A total of 520 patients underwent baseline mammogram during the study period, among whom IHC reports were available for 479 patients (mean age, 51.4 ± 11.7 (SD) years; all females) and were included in the study. Of these 479 patients, 198 (41.3%) were LL, 121 (25.2%) were BL and 160 (33.3%) were HL. The mean age and histological subtypes within the three groups are summarised in Table 1.
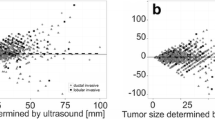
The prevalence of each mammographic feature in the three subtypes is summarised in Table 2. Among the total 479 patients, 449 (93.7%) had one or more masses on mammography. The median size of the largest mass was lowest in LL subtype [median (IQR): 3 (2.4–4) cm, p = 0.009]. Although most of the masses had irregular shape (406/449, 90.4%); BL tumours had significantly high incidence of round shape (21/115, 18.3%) (p < 0.001). On the other hand, HL tumours had lower incidence of round shape (1/149, 0.7%) as compared to others (p < 0.001). In BL subtype, circumscribed (16/115, 13.9%) and microlobulated margins (28/115, 24.4%) were significantly more common, while spiculated margins (41/115, 35.7%) were less common as compared to the other groups (p < 0.001) (Figs. 2a and b, 3a and b). LL subtype had significantly low incidence of circumscribed margins (3/185, 1.6%) (p < 0.001). Suspicious calcifications were more common in HL tumours (96/160, 60%) and less common in BL tumours (30/121, 24.8%) (p < 0.001). While skin thickening and retraction were more common in HL tumours (75/149, 50.3%) (p < 0.001), nipple retraction was significantly less common in BL tumours (9/115, 7.8%) (p = 0.001) (Fig. 4). The associated features that did not show any significant difference between the three groups were architectural distortion and trabecular thickening (p = 0.068 and 0.195, respectively).
Craniocaudal (a) and mediolateral oblique (b) views of right breast mammogram of Her2 positive cancer shows an irregular high density mass in posterior third of central quadrant. The mass (arrows) show spiculated margins and surrounding architectural distortion. Ultrasonography (c) revealed this mass (asterisk) to be irregular and hypoechoic with spiculated margins, dense posterior shadowing and echogenic peripheral rim
Mediolateral oblique (a) and craniocaudal views (b) of left breast mammogram of left breast show a high density mass (asterisk) in lower inner quadrant which is a biopsy-proven basal like tumour. The mass has circumscribed margins with no associated features of architectural distortion, skin thickening or retraction. Ultrasonography image (c) shows the mass to be hypoechoic and irregular with microlobulated margins, internal vascularity and posterior acoustic enhancement
Craniocaudal (a) and mediolateral oblique (b) views of right breast mammogram show an irregular, high density mass (asterisk) in upper outer quadrant with spiculated margins, surrounding architectural distortion, trabecular thickening and coarse heterogeneous calcifications within. There is thickening of skin and subcutaneous tissue along the nipple-areola complex as well (arrows). This is a biopsy-proven Her2 like tumour. On the contrary, biopsy-proven basal like tumour is seen on right breast mammogram (craniocaudal view) as circumscribed equal density mass in retroareolar location (outlined in c) with no associated features
USG data were available for 381 patients. Among them, IHC results were available for 357 patients. There were 157 patients (44%) with LL, 95 (26.6%) with BL and 105 (29.4%) with HL tumours. USG features of the three subtypes are summarised in Table 3. USG revealed mass in 349/357 (97.8%) patients. BL tumours more commonly had round shape (9/94, 9.6%) (p = 0.004), circumscribed (8/94, 8.5%) and microlobulated margins (44/94, 46.8%) with lower incidence of spiculated margins (30/94, 31.9%) (p < 0.001) (Figs. 2c and 3c). Most of the tumours were hypoechoic (301/349, 86.2%), with no significant difference in the echogenicity of the masses between the three groups (p = 0.480). Posterior acoustic enhancement was more common in BL group (7/94, 7.5%) (p = 0.024). No significant difference in the frequency of associated features including skin thickening and duct changes was seen between the three groups (p = 0.090).
On logistic regression analysis, spiculated margins on mammography was a significant predictor of LL subtype (odds ratio, OR: 8.5, p = 0.001). Mammographic features of round shape (OR: 6.8) and circumscribed (OR: 10.8) or microlobulated margins (OR: 3.5) favoured BL subtype as compared to irregular shape and spiculated margins, respectively (p < 0.001 for each). Mammographic features that favoured HL subtype were presence of calcifications (OR: 3.3) (p = 0.019) and skin retraction or thickening (OR: 1.8) (p < 0.001). Similarly, on USG, circumscribed (OR: 5.9) or microlobulated (OR: 3) margins favoured BL subtype as compared to spiculated margins (p = 0.005 and < 0.001, respectively). Posterior acoustic enhancement on USG was a significant predictor of BL subtype (OR: 9.5, p = 0.006).
Discussion
The incidence of breast cancer has been increasing over the past years and management approach includes clinical, radiological and pathological assessment. Combination of the three components has shown to produce a diagnostic accuracy of over 99% [11]. Identification of molecular subtype using immunohistochemical markers has become a standard practice due to its implications on the treatment strategies of breast cancer. Our study showed that LL, BL and HL tumours differ each other in terms of their appearance on mammography and USG that plays a vital role in rad-path correlation and subsequent treatment plan.
LL subtype was the most common among the three subtypes with masses having spiculated margins. A study by Lee et al. had also shown irregular shape and spiculated margins to be the most characteristic MG findings in luminal subtype [12]. Another study by Rashmi et al. found non-circumscribed margins and posterior acoustic shadowing on USG to be significant predictors of luminal subtype [13].
We also observed that round shape and circumscribed or microlobulated margins on MG and USG as predominant features of BL subtype. It also had lower incidence of suspicious calcifications and nipple retraction on mammography and higher incidence of posterior acoustic enhancement on USG. Previous studies have also reported the predominance of indistinct margins in triple negative cancers, in addition to these findings [12,13,14,15,16,17,18,19,20,21,22,23,24,25]. Yang et al. compared the features of triple negative and Her2neu positive cancers on mammography and found that triple negative cancers had oval or round shape in 48% of cases and circumscribed margins in 24% of cases out of a total of 38 patients. They also concluded that the incidence of calcifications and associated features was less in triple negative cancers [19]. A similar study by Lee et al. has shown round or oval shape and circumscribed margins to be characteristic for triple negative tumours on mammography [12]. The high incidence of round shape and circumscribed margins in these tumours can lead to their misinterpretation as benign lesions, which highlights the importance of multidisciplinary approach and clinic-pathological correlation while evaluating a patient with suspected breast cancer. BL tumours often have higher pathological grade and thus show rapid growth, which results in lesser degree of desmoplastic reaction, leading to lower incidence of spiculated margins. The high incidence of posterior acoustic enhancement in BL tumours has been attributed to their high cellularity [26].
The mammography features that favoured Her2neu positivity were spiculated margins, presence of calcifications and skin thickening or retraction, in the present study. This is in concordance with the previous studies that have shown that Her2neu positive tumours tend to have irregular shape and spiculated margins, with higher incidence of calcifications [12,13,14,15,16,17, 19, 22, 23, 27]. A few studies have also shown posterior acoustic shadowing and mixed posterior features to more common in Her2neu positive tumours [13, 17]. LL and HL tumours are of lower grade with slower growth rates and thus show higher degree of desmoplastic reaction, which could explain the spiculated margins and posterior acoustic shadowing compared to the other subtypes [28].
This study highlights the vital role of imaging modalities in characterising the breast cancer subtypes. These imaging features can assist in rad-path correlation in indeterminate settings. However, it is limited by its retrospective study design and the use of IHC for determining the histological subtype rather than gene expression analysis.
To conclude, the clinically defined subtypes of breast cancer show significant differences in the imaging appearances on mammography and USG. BL tumours may not show the typical morphological features of malignancy, thus requiring clinicopathological correlation for accurate diagnosis.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424
Elmore JG, Longton GM, Carney PA et al (2015) Diagnostic concordance among pathologists interpreting breast biopsy specimens. JAMA 313:1122–1132
D’Orsi C, Sickles EA, Mendelson EB, Morris EA (2013) Breast imaging reporting and data system: ACR BI-RADS breast imaging atlas. 5th ed. Reston, VA: American College of Radiology
Parker JS, Mullins M, Cheang MCU et al (2009) Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 27:1160–1167
Hortobagyi GN, Connolly JL, D’Orsi CJ, Edge SB, Mittendorf EA, Rugo HS (2017) Breast. In: Amin M. B, Edge S, Greene F, eds. AJCC Cancer Staging Manual. 8th ed. New York: Springer. pp. 589–636
Goldstein NS, Decker D, Severson D et al (2007) Molecular classification system identifies invasive breast carcinoma patients who are most likely and those who are least likely to achieve a complete pathologic response after neoadjuvant chemotherapy. Cancer 110:1687–1696
Dent R, Trudeau M, Pritchard KI et al (2007) Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res 13:4429–4434
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL (1987) Human breast cancer: correlation of relapse and survival with amplification of the HER2-2/neu oncogene. Science 235:177–182
Slamon DJ, Leyland-Jones B, Shak S et al (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344:783–792
Perez EA, Romond EH, Suman VJ et al (2014) Trastuzumab plus adjuvant chemotherapy for human epidermal growth factor receptor 2–positive breast cancer: planned joint analysis of overall survival from NSABP B-31 and NCCTG N9831. J Clin Oncol 32:3744–3752
Jan M, Mattoo JA, Salroo NA, Ahangar S (2010) Triple assessment in the diagnosis of breast cancer in Kashmir. Indian J Surg 72:97–103
Lee SH, Chang JM, Shin SU et al (2017) Imaging features of breast cancers on digital breast tomosynthesis according to molecular subtype: association with breast cancer detection. Br J Radiol 90:20170470
Rashmi S, Kamala S, Murthy SS, Kotha S, Rao YS, Chaudhary KV (2018) Predicting the molecular subtype of breast cancer based on mammography and ultrasound findings. Indian J Radiol Imaging 28:354–361
Cai S, Yao M, Cai D et al (2019) Association between digital breast tomosynthesis and molecular subtypes of breast cancer. Oncol Lett 17:2669–2676
Sohn Y-M, Han K, Seo M. Immunohistochemical subtypes of breast cancer: correlation with clinicopathological and radiological factors. Iran J Radiol [Internet] 2016; 13:e31386. Cited January 2020. Available from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5116817/
Wu M, Ma J (2017) Association between imaging characteristics and different molecular subtypes of breast cancer. Acad Radiol 24:426–434
Zheng F-Y, Lu Q, Huang B-J et al (2017) Imaging features of automated breast volume scanner: correlation with molecular subtypes of breast cancer. Eur J Radiol 86:267–275
Kojima Y, Tsunoda H (2011) Mammography and ultrasound features of triple-negative breast cancer. Breast Cancer 18:146–151
Yang W-T, Dryden M, Broglio K et al (2008) Mammographic features of triple receptor-negative primary breast cancers in young premenopausal women. Breast Cancer Res Treat 111:405–410
Boisserie-Lacroix M, MacGrogan G, Debled M et al (2013) Triple-negative breast cancers: associations between imaging and pathological findings for triple-negative tumors compared with hormone receptor-positive/human epidermal growth factor receptor-2-negative breast cancers. Oncologist 18:802–811
Wojcinski S, Soliman AA, Schmidt J, Makowski L, Degenhardt F, Hillemanns P (2012) Sonographic features of triple-negative and non-triple-negative breast cancer. J Ultrasound Med 31:1531–1541
Wu T, Li J, Wang D et al (2019) Identification of a correlation between the sonographic appearance and molecular subtype of invasive breast cancer: a review of 311 cases. Clin Imaging 53:179–185
Au FW-F, Ghai S, Lu F-I, Moshonov H, Crystal P. (2017) Histological grade and immunohistochemical biomarkers of breast cancer: correlation to ultrasound features. J Ultrasound Med 36:1883–1894
Gao B, Zhang H, Zhang S-D et al (2014) Mammographic and clinicopathological features of triple-negative breast cancer. Br J Radiol 87:20130496
Kim MY, Choi N (2013) Mammographic and ultrasonographic features of triple-negative breast cancer: a comparison with other breast cancer subtypes. Acta Radiol 54:889–894
Irshad A, Leddy R, Pisano E, Baker N, Lewis M, Ackerman S et al (2013) Assessing the role of ultrasound in predicting the biological behavior of breast cancer. AJR Am J Roentgenol 200:284–290
Seo BK, Pisano ED, Kuzimak CM et al (2006) Correlation of HER-2/neu overexpression with mammography and age distribution in primary breast carcinomas. Acad Radiol 13:1211–1218
Koo MM, von Wagner C, Abel GA, McPhail S, Rubin GP, Lyratzopoulos G (2017) Typical and atypical presenting symptoms of breast cancer and their associations with diagnostic intervals: evidence from a national audit of cancer diagnosis. Cancer Epidemiol 48:140–146
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pulappadi, V.P., Dhamija, E., Baby, A. et al. Imaging Features of Breast Cancer Subtypes on Mammography and Ultrasonography: an Analysis of 479 Patients. Indian J Surg Oncol 13, 931–938 (2022). https://doi.org/10.1007/s13193-022-01606-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-022-01606-7