Abstract
The increasing incidence of urinary bladder carcinoma is alarming. Approximately seventy percent of these patients are non-muscle invasive bladder cancer (NMIBC). Restage transurethral resection of bladder tumor (TURBT) is the current recommendation for any T1 and or high-grade non muscle invasive bladder cancers (NMIBC) to accurately stage the malignancy. The question whether a second surgery is always required as a restage procedure is still unanswered. The patient’s concern about completeness, morbidity, and financial considerations of a major surgery cannot be overlooked. Moreover, it also puts a strain on the already overburdened healthcare system. To answer this question, whether it is oncologically sound to omit a second resection, the current study evaluated the outcomes of patients undergoing restage TURBT, and analyzed the preoperative factors predicting a change in the staging of this malignancy. The study design was a prospective observational including NMIBC patients from September 2018 to February 2020. A total of 72 patients underwent restage TURBT. Their demographic data, imaging and cystoscopic findings, and histopathological data were recorded. The objective was to study the clinico-pathological correlations and factors predicting recurrence and upstaging of tumor in NMIBC patients undergoing restage TURBT. A total of 101 patients were found eligible for restage TURBT. Eventually, 72 underwent restage TURBT. Twelve (16.7%) patient had recurrence at restage while 3(4.16%) were upstaged to T2. Presence of lower urinary tract symptoms (LUTS) was independently associated with the risk of recurrence of same stage compared to no recurrence (p-0.025, OR-8.793, 95% CI-1.316–98.773). Chemical exposure (p-0.042) was also significantly associated with the same. Presence of lymphadenopathy on CT was independently associated with the risk of upstaging compared to no recurrence (p-0.032, OR-18.25, 95% CI-1.292–257.85). The study concluded that in the presence of a well-performed and adequate initial TURBT, restage TURBT could be skipped for further management. However, in small subgroup of patients with lymphadenopathy on preoperative imaging having a higher risk of tumor recurrence and upstaging, and patients with a history of chemical exposure and previous lower urinary tract symptoms having a high risk of recurrence alone, restage TURBT should still be performed to accurately stage the disease. Further studies with large patient cohort are needed to confirm and reinforce the facts proposed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of urinary bladder cancer is increasing and it is alarming. Urinary bladder cancer is the ninth most common cancer in the world. According to GLOBOCAN 2018, around 550,000 new urinary bladder cancer cases were diagnosed globally with a male to female ratio of approximately 3.5:1 [1]. Seventy percent of these cases are non-muscle invasive [2]. As per Indian cancer registry data, urinary bladder cancer accounts for 3.9% of all cancer cases. The rising trend of bladder cancer cases in India is attributed to increased use of different forms of tobacco, besides the ever increasing menace of air pollution and industrial chemical and fertilizer exposure in our country [3]. This cancer is considered a heterogeneous entity and broadly divided into NMIBC and muscle invasive bladder cancer (MIBC). Transurethral resection of bladder tumor (TURBT) is the preferred treatment for NMIBC. NMIBC represents a wide spectrum of disease with different clinical course, owing to noteworthy risk of progression and possible recurrence. According to existing guideline and evidence, high-grade urothelial cancers or tumors restricted to lamina propria undergo restage to accurately stage the malignancy, whereas MIBC needs a radical cystectomy and chemotherapy due to its high risk of progression and metastasis [4].
Incidence of recurrence and upstaging in NMIBC particularly in T1 and high-grade tumors is 50–70% and 20–30%, respectively [4]. This constitutes the basis of guideline advocating the restage TURBT in all T1 and high-grade carcinoma of urinary bladder. However, few experts have different opinion on this subject and they suggest that restaging TURBT may not be necessary when initial TURBT is properly preformed [5]. The present study attempts to find the utility of restage TURBT and finding the clinical and other factors which predict the probability of upstaging and recurrence of tumor.
Materials and Methods
A prospective observational study with period prevalence was conducted in Department of Urology of our institute in association with Pathology Department from September 2018 to February 2020. Aim of the study was to evaluate the role of restage TURBT in non-muscle invasive bladder cancer (NMIBC) and its clinical implications. Primary objectives were to study the clinico-pathological correlation and factors predicting recurrence and up-staging of tumor in non-muscle invasive bladder cancer (NMIBC) undergoing restage transurethral resection of bladder tumor (TURBT). Secondary objectives were to estimate the incidence of recurrence and up-staging of tumor in non-muscle invasive bladder cancer (NMIBC) undergoing restage TURBT and to find the patients with non-muscle invasive bladder cancer (NMIBC) in whom restage TURBT can be omitted. Patients with tumor size more than 3 cm, multiple tumors on cystoscopy, any T1, any high grade, absence of deep muscle on histopathological examination, and those who were willing to participate in study with informed consent were included. However, those with endoscopically non-manageable disease, TaLG (low grade), muscle invasive bladder cancer (MIBC), and metastatic disease were excluded.
This study was carried out in accordance with the ethical standards laid down by Declaration of Helsinki and ICMR guidelines for biomedical research on human subjects with institutional ethical board approval. All consecutive subjects presenting during the study period fulfilling the criterion were included in the study. A total of 72 patients fulfilled the criterion and underwent restage TURBT during the study period.
Detailed clinical history of patients was taken including history of haematuria, presence of lower urinary tract symptoms, history of smoking, chemical exposure, and previous TURBT if any. Patients were subjected to further evaluation using ultrasound, CT urography prior to surgery, and presence of hydronephrosis and lymphadenopathy on CT was noted. Bladder mapping of initial TURBT was done. Morphological characteristics of tumor-like single/multiple, pedunculated/sessile, and solid/papillary were noted. At our center TURBT was performed using 26 Fr Iglisias continuous flow resectoscope using monopolar or bipolar cautery and irrigation with 3% glycine or normal saline respectively. TURBT was performed either by consultant himself or residents supervised by him. Complete resection was ensured. Patient undergoing complete TURBT and meeting the inclusion criteria were advised to undergo restage TURBT at 2 to 6 weeks of initial TURBT. Findings on restage TURBT like healthy or unhealthy scar, presence of recurrent tumor, site of recurrent tumor (same/distant), and morphology of recurrent tumor were documented. Resection of scar, recurrent tumor, and deep muscle from both previous scar and new growth was ensured. All samples were sent in separate vials for histopathology examination. Examination and reporting were done by a senior pathologist according to WHO/ISUP 2004 classification. Presence of any tumor on histopathology was considered as recurrence whereas upstaging was defined as presence of MIBC at restage TURBT. Major complications like perforation and reintervention to control bleeding in post-operative period were noted.
Statistical Analysis
All statistical analysis was performed using SPSS software (version 26, SPSS Inc., Chicago, IL, USA). Categorical variables were described as frequency and proportion. Continuous variables were described as mean and standard deviation or median with interquartile range as applicable.
Proportions were compared using Chi square test and Fishers exact test. The means in two groups were compared using Student’s t test and Mann–Whitney U test. Paired t test was used for readings of continuous variables at end point of time. Univariate analysis was done to find factors responsible for recurrence at restage TURBT. Multiple logistic regression analysis was used to document independent predictors of desired outcomes.
Results
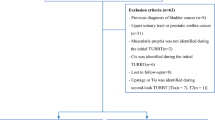
A total 299 patients underwent TURBT during the study period. Forty-four amongst 299 were metastatic on presentation and fifty-four patients underwent incomplete resection. Out of 201 patients undergoing a complete resection, thirty-six turned out to be MIBC whereas fifty-two patients had Ta, LG. One hundred and one patients met the criteria for restage TURBT. Twenty-nine patients did not comply the advice, and finally, seventy two underwent restage TURBT. Amongst these patients, forty-seven had T1HG, 14 had T1LG, 9 had TaHG, 2 had TaLG tumors, and nine did not had deep muscle at initial TURBT. Two TaLG patients were included as they were large (> 3 cm), multiple, sessile, and needed 2 resection for completion and classified as intermediate risk tumors. No patient had CIS on pathology of tumor (Fig. 1).
Demographic profile and patient characteristics of the NMIBC patients who underwent restage are given in Table 1. Mean age of presentation was 55 ± 14.7 years. Approximately 89% of patients were males. Amongst them, 79.2% of patients were smokers while 8(11%) had chemical exposure at their place of work at their place of work like paint, rubber, and petroleum industries. They were involved in various processes of making paint, processing crude rubber to convert into various rubber products like tire, tubes, and processing and segregation of petroleum products. These processes involve handling many different chemicals for industrial work. Detailed description of individual chemical could not be ascertained. They were provided with protective equipment’s like gloves, dresses, and mask. Their utilization and availability were not universal. Painless gross hematuria was the presenting complaint in 67 (93%) of patients. Lower urinary tract symptoms were present in 16 (22.2%) of patients.
Twelve (16.7%) patients had various comorbidities including diabetes, hypertension, and asthma. Nine (12.5%) patients had history of previous surgery for bladder mass. Preoperative CT urography showed hydroureteronephrosis in 9 (12.5%) while significant lymphadenopathy in 8 (11.1%). Mean hemoglobin was 12.29 ± 2.13 (mean ± SD in gm/dl) and blood transfusion was needed in 11 (15.3%) patients due to ongoing hematuria and anemia.
On cystoscopy, 41 (56.9%) had tumors of size < 3 cm, 44 (61.1%) had single tumor, 49 (68.1%) had sessile tumor, while 21 (29.2%) had solid tumor. Complete resection of tumor was ensured during TURBT. Sixty-one (84.7%) had T1 stage while 56 (77.8%) had high-grade histology. Deep muscle was present in 63 (87.5%) of patients at initial resection (Table 2). Six patients received intravesical mitomycin C postoperatively.
The mean interval between initial and restage TURBT was 6.58 weeks with a range of 3 to 14 weeks, although 2 to 6 weeks is ideal time for restage TURBT, due to unavoidable circumstances and multifactorial reasons cannot be done. There was no statistically significant difference in the two at restage TURBT (p-0.545). Healthy scar at restage TURBT was noted in 57 (79.1%) patients while unhealthy scar was noted in 15 (20.9%). Sixty (83.34%) patients did not have any recurrence at restage; however, twelve patients (16.7%) had histologically proven recurrence of tumor either at the same site or a different one. Three patients had recurrence at same site while 9 patients had recurrence at different site. Two had T1HG, 3 had T1LG, 4 had TaLG, while three (4.16%) patients had upmigration of stage to T2 (Fig. 2). Two of these patients did not have deep muscle on initial TURBT, one was upgraded to muscle invasive disease, while other had same stage recurrence. No patient had significant intra- and postoperative complications at restage TURBT needing reintervention.
For analysis of clinico-pathological factor correlation between recurrence at restage, patients were grouped in 2 different groups. Group 1 included patients with no recurrence at restage while group 2 included patients with recurrence at restage. Group 2 was further subcategorised into 2a and 2b with former consisting of patients with same stage (T1 or less) while the later include those who were upstaged to T2.
On univariate analysis, presence of lower urinary tract symptoms and history of occupational chemical exposure (p = 0.057) was significantly associated with risk of tumor positivity when compared between recurrence of same stage vs no recurrence at restage (Table 3). The rest of the variables like age, duration of smoking, size of tumor, focality of tumor, histological grading, initial stage, presence, or absence of deep muscle were not significantly associated with any recurrence at restage (group 1 vs group 2a).
Presence of lymphadenopathy on preoperative CT scan was significantly associated with risk of tumor positivity and upstaging when compared between upstage and no recurrence at restage (p-0.03) (group 1 vs group 2b). No other factors were found statistically significant.
On multivariate analysis, presence of LUTS after adjusting for age, sex, and smoking was found to have a 8.79 times higher chance of same stage recurrence compared to no LUTS (p-0.025, OR-8.793, 95% CI-1.316–98.773). Presence of enlarged lymph nodes on CT scan was associated with 18.25 times higher chance of upstaging as compared with no lymphadenopathy on imaging, when adjusted for age, sex, and smoking status. (p-0.032, OR-18.25, 95% CI-1.292–257.85).
The incidence of recurrence and stage progression at restage was 16.7% and 4.16%, respectively. The factors that emerged to be associated with recurrence or stage migration were history of chemical exposure, history of LUTS, and lymphadenopathy on preoperative CT scan.
Discussion
Risk stratification of superficial bladder cancer is important to facilitate the treatment recommendations. NMIBC is stratified into low-, intermediate-, and high-risk groups and is advocated by all major guidelines. Restage TURBT is indicated for all high risk, T1, high grade, and absence of deep muscle in initial biopsy according to the guidelines of major urological societies [4].
Application of these guidelines involves thorough evaluation and individual practice for managing the patient of carcinoma urinary bladder may vary widely. Tobert et al. in his study found that factor independently associated with compliance for these guidelines was receiving treatment at tertiary academic institute [6]. Gotto et al. highlighted the rate of restage in indicated patients between 26 and 28% in non-Canadian area while in Canada it is mostly unreported [7]. In our study, about a third of the patients did not comply the advice of a restaging procedure. The reason for this could be the patients becoming completely asymptomatic after TURBT and might consider themselves cured of the disease or possibly because of financial limitations. The reasons for this poor compliance are multifactorial, interwoven, and interdependent.
Even if the patient becomes compliant and undergoes restage TURBT, it is not always feasible at desired time. Tseng et al. noted that interval for restage was average of 7.4 week in his study [8]. Manoharan et al. from India also noted delay in restage in their study [9]. In our study, restage was done at an average time interval of 6.58 weeks with a range of 3 to 14 weeks after initial TURBT.
The very need of restage TURBT is probably incompleteness of initial TURBT. It is dependent either on tumor-related factors or surgeon-related factors. Tumor-related factors include large size, difficult location, multiple, obturator jerk, CIS, overt bleeding, quality of specimens provided, and sometimes hardware issues like problem with vision. Pathological analysis also had impact on same. Surgeon-related factors include low experience and expertise in resection. Many studies have shown that limited experience of TURBT, which is generally with resident urologist, is associated with an increased risk of disease recurrence as compared to the consultants [10,11,12]. Mariappan et al. studied 473 NMIBC patient’s specimens and its correlation to the second TUR findings. They showed that lower rate of recurrence of bladder tumor was associated with surgeon experience and the presence of muscle in initial TURBT specimen [13].
To get accurate staging and complete resection on initial TURBT, concept of quality control of initial TURBT has been brought up and many researchers had done work on this. Herr and Donat defined quality of TURBT by asking three questions: (1) Is complete resection done?; (2) deep muscle present or not in pathology; (3) how often tumor recur at site of first TURBT [11].
Over the period in past, second intervention in patient of bladder mass was redefined, refined, and renamed according to its indications. Divrik et al. have proposed many strict criteria for the next level concepts in TURBT. In his terms, repeat TURBT is defined a TUR after incomplete initial resection due to various factors. The term “restaging TUR” (re-TUR) is used whenever a second TUR was done to gather additional histopathologic information from the muscularis propria. And “second TUR” is defined as intervention done after a correct and complete TURBT [14].
There are only few studies from India related to the role of restage. Studies done in the Indian subcontinent on restage TURBT showed rate of recurrence of 28 to 60% and upstaging from 4 to 23% [15,16,17]. Various studies from the foreign countries noted recurrence rate from 16 to 78% and upstaging of 1 to 28% [5, 8, 18]. Current study depicts rate of recurrence as 16.7% which is lower compared to other Indian studies.
A largest retrospective study on restage TURBT by Gontero et al. did not find any survival benefit after second surgery. This study strongly questions the real need for the need of second procedure considering its cost implication to patient and burden on healthcare system 5. Similarly, a recent prospective study questioned the utility of restage in completely resected T1 tumors with muscle included as it did not incur any survival benefit [19].
A systematic review too found a very low risk of upstaging (1–4%), which was attributed to surgeon experience and centralization of procedure [20]. Resident urologist is considered to be having less experience and was associated with recurrence [10]. In our study, all resection was done by faculty or resident always supervised by consultant.
A study from Asian country noted the recurrence rate of 18% and upstaging of 1% [8]. Our study had a recurrence rate of 16.7% and upstaging rate of 4.16% which is comparable to the above study. It could be attributed to the good quality initial TURBT and consultant supervision. There is substantial variability in reported risk factors in different studies. Also, the population characteristics and methodologies used for it were diverse. It might also be affected by complex nature of bladder cancer and heterogeneous surgical technique used [21].
Chemical exposure was found to be significantly associated with the recurrence at restage. Chemicals excreted through the urinary tract lead to the exposure of the bladder urothelium. Prolonged excretion of accumulated carcinogenic chemicals through urinary tract may be the cause of recurrence. Tumorigenic properties of these chemicals are the cause of bladder cancer [22].
Presence of LUTS is significantly associated with risk of same stage recurrence compared to no recurrence (p-0.026) in our study. It has been hypothesized that presence of LUTS might lead to prolonged exposure of bladder mucosa to tobacco products excreted in urine which had tumorigenic properties and increased recurrence [23]. A meta-analysis suggested the decreased risk of recurrence in superficial bladder cancer in patients with BPH and LUTS who underwent simultaneous TURBT and TURP. This highlights the significant association of LUTS with recurrence [24].
Preoperative imaging has important role in clinical staging. Ark et al. found that CT hydronephrosis (p-0.008) and lymphadenopathy (p-0.05) at radical cystectomy are associated with understaging of NMIBC and indicate presence of extravesical disease or higher clinical stage [25]. Dijk et al. emphasized that hydronephrosis and lymph node involvement is not associated with downstaging of bladder cancer [26]. In our study too, lymphadenopathy on CT urogram is significantly and independently associated with stage progression and increased chance of recurrence at restage.
Atta et al. underlined that restage TURBT can be safely omitted in select patients in whom surgeries were done by experienced urologist and teamwork by doubly ensuring the completeness of initial resection [12].
We also propose that in a patient with complete resection by experienced surgeon and teamwork with no risk factors like chemical exposure, LUTS, and lymphadenopathy, restage TURBT can be avoided and patients can be started on intravesical BCG without delay. However, a normal check cystoscopy under local anesthesia at six weeks of initial TURBT would suffice to rule out any recurrence or residual disease before venturing into intravesical BCG. Limitation of the study was the performance of TURBT and restage TURBT by different surgeons and its small sample size.
Conclusion
T1, high-grade disease, tumor > 3 cm, multiple tumors, and CIS are not associated with increased risk of upstaging or recurrence in the setting of well-performed TURBT. However, patients with lymphadenopathy on preoperative imaging are at a higher risk of tumor recurrence and upstaging; and patients with a history of chemical exposure and previous lower urinary tract symptoms were at high risk of recurrence alone. The absence of these factors in the presence of a well-performed and adequate TUTBT may obviate the need for another restaging procedure. Further randomized controlled, multicentre trials with large patient cohort are needed to confirm and reinforce the facts.
References
Richters A, Aben KKH, Kiemeney LALM (2020) The global burden of urinary bladder cancer: an update. World J Urol. 38(8):1895–1904. https://doi.org/10.1007/s00345-019-02984-4
Isharwal S, Konety B (2015) Non-muscle invasive bladder cancer risk stratification. Indian J Urol 31(4):289–296
Yuvaraja TB, Ganesh SW, Bakshi GP (2016) Genitourinary cancers: summary of Indian data. South Asian J Cancer 5(3):122–4
Woldu SL, Bagrodia A, Lotan Y (2017) Guideline of guidelines: non-muscle-invasive bladder cancer. BJU Int 119(3):371–380
Gontero P, Sylvester R, Pisano F, Joniau S, Oderda M, Serretta V et al (2016) The impact of re-transurethral resection on clinical outcomes in a large multicentre cohort of patients with T1 high-grade/Grade 3 bladder cancer treated with bacille Calmette-Guérin. BJU Int 118(1):44–52
Tobert CM, Nepple KG, McDowell BD, Charlton ME, Mott SL, Gruca TS et al (2019 Oct) Compliance With American Urological Association Guidelines for Nonmuscle Invasive Bladder Cancer Remains Poor: Assessing Factors Associated With Noncompliance and Survival in a Rural State. Urology. 132:150–155. https://doi.org/10.1016/j.urology.2019.06.021
Gotto GT, Shea- Budgell MA, Dean Ruether J (2016) Low compliance with guidelines for re-staging in high-grade T1 bladder cancer and the potential impact on patient outcomes in the province of Alberta. Can Urol Assoc J 10(1–2):33–8. https://doi.org/10.5489/cuaj.3143
Tseng WH, Liao A, Shen KH, Chen CH, Liu CL, Hung SH et al (2018) Role of second-look transurethral resection of bladder tumors for newly diagnosed T1 bladder cancer: experience at a single center. Urol Sci 29(2):95–99
Dwivedi US, Kumar A, Das SK, Trivedi S, Kumar M, Sunder S et al (2009) Relook TURBT in superficial bladder cancer: its importance and its correlation with the tumor ploidy. Urol Oncol 5:514–9. https://doi.org/10.1016/j.urolonc.2008.04.015
Rolevich A, Minich A, Nabebina T, Polyakov S, Krasny S, Sukonko O (2016) Surgeon has a major impact on long-term recurrence risk in patients with non-muscle invasive bladder cancer. Cent Eur J Urol 69(2):170–177
Herr HW, Donat SM (2008) Quality control in transurethral resection of bladder tumours. BJU Int 102(9B):1242–1246
Atta MA, Kotb AF, Sharafeldeen M, Elabbady A, Hashad MM. The value of extended good quality transurethral resection of bladder tumour in the treatment of the newly diagnosed bladder cancer. Arab J Urol [Internet]. 2017;15(1):60–3. Available from:https://doi.org/10.1016/j.aju.2016.10.003
Mariappan P, Finney SM, Head E, Somani BK, Zachou A, Smith G et al (2012) Good quality white-light transurethral resection of bladder tumours (GQ-WLTURBT) with experienced surgeons performing complete resections and obtaining detrusor muscle reduces early recurrence in new non-muscle-invasive bladder cancer: Validation across t. BJU Int 109(11):1666–1673
Divrik RT, Yildirim U, Zorlu FOH (2006) The effect of repeat transurethral resection on recurrence and progression rates in patients with T1 tumors of the bladder who received intravesical mitomycin: a prospective, randomized clinical trial. J Urol 175(5):1641–1644
Manoharan V, Mavuduru RS, Kumar S, Kakkar N, Devana SK, Bora GS, Singh SKMA (2018) Utility of restage transurethral resection of bladder tumor. Indian J Urol 34(4):273–277
Katumalla FS, Devasia A, Kumar R, Kumar S, Chacko N, Kekre N (2011) Second transurethral resection in T1G3 bladder tumors-selectively avoidable. Indian J Urol 27(2):176–179
Raj KK, Taneja Y, Ramdev P, Dhaked SK, Singh CK, Shekar AP (2019) A prospective observational study to evaluate the role of restaging transurethral resection of bladder tumour in patients with non-muscle invasive bladder cancer. Int J Res Med Sci 7(11):4134
Herr HW (1999) The value of a second transurethral resection in evaluating patients with bladder tumors. J Urol 162(1):74–76
Calò B, Chirico M, Fortunato F, Sanguedolce F, Carvalho-Dias E, Autorino R et al (2019) Is Repeat Transurethral Resection Always Needed in High-Grade T1 Bladder Cancer? Front Oncol 4(9):465. https://doi.org/10.3389/fonc.2019.00465
Soria F, D’Andrea D, Moschini M, Giordano A, Mazzoli S, Pizzuto G et al (2020) Predictive factors of the absence of residual disease at repeated transurethral resection of the bladder Is there a possibility to avoid it in well-selected patients? Urol Oncol Semin Orig Investig 38(3):77.e1-77.e7
Czech AK, Gronostaj K, Frydrych J, Fronczek J, Przydacz M, Wiatr T et al (2019) Identification of potential prognostic factors for absence of residual disease in the second resection of t1 bladder cancer. Cent Eur J Urol 72(3):252–257
Letašiová S, Medve'ová A, Šovčíková A, Dušinská M, Volkovová K, Mosoiu C, et al. Bladder cancer, a review of the environmental risk factors. Environ Health. 2012;11 Suppl 1(Suppl 1):S11. https://doi.org/10.1186/1476-069X-11-S1-S11
Lunney A, Haynes A, Sharma P (2019) Moderate or severe LUTS is associated with increased recurrence of non - muscle - invasive urothelial carcinoma of the bladder. Int Braz J Urol 45(2):306–314
Luo S, Lin Y, Zhang W (2011) Does simultaneous transurethral resection of bladder tumor and prostate affect the recurrence of bladder tumor? A meta-analysis J Endourol 25(2):291–296
Ark JT, Keegan KA, Barocas DA, Morgan TM, Resnick MJ, You C et al (2014) Incidence and predictors of understaging in patients with clinical T1 urothelial carcinoma undergoing radical cystectomy. BJU Int 113(6):894–899
van Dijk PR, Ploeg M, Aben KKH, Weijerman PC, Karthaus HFM, van Berkel JTH et al (2011) Downstaging of TURBT-based muscle-invasive bladder cancer by radical cystectomy predicts better survival. ISRN Urol 2011:1–6
Acknowledgements
We acknowledge Dr Ajeet Singh Bhadoria, Department of Community and Family Medicine, for his valuable help in doing statistical analysis.
Author information
Authors and Affiliations
Contributions
Conceptualization: Ankur Mittal, Sunil Kumar; methodology: Ankur Mittal, Sunil Kumar, Sanjeev Kishore; acquisition, analysis, or interpretation of data, drafted the work: Deepak Prakash Bhirud, Satish Ranjan; writing original draft: Deepak Prakash Bhirud; review and editing: Sunil Kumar, Tushar Aditya Narain, Vikas Kumar Panwar; all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of AIIMS, Rishikesh, on 29.12.2018 with letter number AIIMS/IEC/18/573.
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Patients’ informed consent regarding publishing their data was obtained.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhirud, D.P., Mittal, A., Kumar, S. et al. When to Avoid a Restaging Procedure for Non-muscle Invasive Bladder Cancer? Inferences from a Tertiary Care Center. Indian J Surg Oncol 13, 604–611 (2022). https://doi.org/10.1007/s13193-022-01516-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-022-01516-8