Abstract
More than 100 tidal marsh creation projects were constructed throughout the Fraser River Estuary, British Columbia, Canada from the 1980s to present. Past studies described and evaluated many of these projects and found varied success, but the underlying factors that determine project outcomes remain uncertain. Combining field sampling, spatial analysis, and statistical modeling of plant communities, we aim to address this knowledge gap by asking what factors influence the resilience of created marshes, as measured by (1) persistence of marsh vegetation, (2) native species dominance, and (3) species richness. We observed marsh recession in 40 of the 78 projects visited, representing 23,666 m2 (9.3%) of the 254,357 m2 of created marsh surveyed. Increases in mean site elevation had a negative effect on percent recessed area, while sites in the north branch of the river and sites further upriver were more prone to recession. From field observations and data interpretation we suggest that wake erosion and Canada Goose (Branta canadensis) herbivory may be drivers behind these losses and warrant further investigation. Dominance of native species declined with distance upriver, though invasive cattail (Typha angustifolia, T. × glauca) defied this trend, dominating outer estuary sites, particularly closed embayments, when present. Native and non-native richness shared similar patterns and were comparable between reference and created marshes, increasing on average with elevation and distance upriver. These findings offer insight into how site design and location influence the outcome of marsh creation projects, and the challenges presented by stressors and environmental change in estuaries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human settlement has occurred in estuaries for millennia, as they are abundant in natural resources and are in close proximity to the ocean (Small and Nichols 2003; Day et al. 2012). The result, particularly in recent centuries as human populations have exponentially increased, has been the escalated alteration, fragmentation, and loss of estuarine habitats around the world. These losses have led to declines in the function, services, and resilience of these ecosystems in an age when threats such as climate change and species invasions abound (Barbier et al. 2011; O’Meara et al. 2017). To this day, habitat loss remains one of the major threats to global estuaries (Kennish 2002).
Estuaries along the west coast of North America have not been immune to habitat loss. An estimated 85% of tidal wetlands have disappeared in estuaries along the contiguous U.S. Pacific Coast since European settlement, with the greatest losses occurring in major river deltas (Brophy et al. 2019). The Fraser River Estuary (FRE), the largest estuary on Canada’s Pacific Coast, has seen similar declines. An estimated 70 – 90% of wetlands have been lost due to extensive diking and development (Kistritz and Scott 1992; Boyle 1997), and the FRE now contains the largest port and third largest metropolitan area in Canada. These losses are detrimental to the many species that depend on estuarine habitats, including declining North American Pacific salmon, particularly Chinook (Oncorhynchus tshawytscha) and chum (O. keta; Levy and Northcote 1982; Chalifour et al. 2019), and numerous species at risk (Kehoe et al. 2021). The north–south network of estuaries along the Pacific Coast also provides critical stopover points for migratory bird species travelling along the Pacific Flyway, and productive foraging, resting, and roosting habitat for migratory and resident waterfowl, shorebirds, songbirds, raptors, and gulls (Butler and Campbell 1987).
As understanding of the impacts of human activities in the estuaries have increased, so too have efforts to mitigate them. In the FRE, an abundance of research was conducted in the late 1970s and early 1980s to better understand the ecology of local tidal marshes (e.g., Levy and Northcote 1982), and to research and test tidal marsh creation as a means of mitigating habitat loss (Adams and Williams 2004). Building upon this knowledge, the Department of Fisheries and Oceans Canada (DFO) introduced the Policy for the Management of Fish Habitat (1986), which included “no net loss” guidelines aimed to maintain the productive capacity of fish habitats in Canada by using habitat replacement, primarily compensation, to balance unavoidable fish habitat losses. Though fisheries policies have been updated since 1986, marsh habitat creation remains a common practice for mitigating habitat loss, and has resulted in over 100 compensation and offsetting projects throughout the FRE, representing nearly all marshes constructed to date.
A small number of studies have documented and evaluated marsh creation projects in the FRE, and have reported varied outcomes. Kistritz et al. (1992) documented the degradation of some projects by erosion and debris, likely due to ineffective shear booms or erosion protection. Based on area, Kistritz (1995) calculated a net gain of marsh habitat in the FRE from 1983 – 1992 due to compensation activities, but recommended follow-up remediation at multiple projects. A rigorous research program conducted in the early 1990s evaluated the ecological functioning of created marshes (see Levings and Nishimura 1996). Through this program it was found that the average percent cover of Lyngbye’s sedge (Carex lyngbyei Hornem.), the target plant species of nearly all projects due to its high productivity, detrital value (Kistritz et al. 1983) and dominance in mid-elevation brackish marshes throughout the northwest coast of North America, was lower in created marshes than adjacent reference marshes (Moody 1993; Stronach 1995). Benthic invertebrate abundance was also equal or higher in created and reference marshes than disrupted, unvegetated sites (Stronach 1995). No differences were found in Chinook salmon and chum salmon abundance between created, reference, and unvegetated sites, however the results should be interpreted with caution due to the small number of sample sites and replicates (Scott and Susanto 1993). In more recent years, Adams and Williams (2004) provided an extensive summary of marsh creation projects prior to 1997, and noted that early projects were more prone to failure due to inappropriate species selection and poor quality assurance during implementation. Lievesley et al. (2016) evaluated 54 projects and highlighted the challenges of marsh creation in the estuary, as 65% of projects achieved their intended marsh area, 50% had comparable or higher dominance of native plants than nearby reference sites, and 33% achieved both criteria. Despite the relatively high success rate, it is not enough to support the goal of achieving net zero habitat loss through marsh creation.
Vegetative cover is a commonly used metric to evaluate created tidal marshes (Kentula 2000; Broome et al. 2019). Functioning tidal marshes support high net primary production, which contributes to the accumulation of soil organic matter. This organic surface soil is an integral part of the detritus-based food web of estuaries. Due to this, and the refuge offered by standing biomass, vegetative cover has historically been used as a proxy for high-quality fish habitat in the region (Bradford et al. 2017). In addition to providing food and refuge for numerous other species, tidal marshes support a multitude of ecosystem services, including soil stabilisation, water quality maintenance, wave attenuation, carbon sequestration, sediment accretion, and nutrient cycling (e.g., Peterson et al. 2008; Mudd et al. 2010; Broome et al. 2019; Arias-Ortiz et al. 2021; Correa et al. 2021). Conversely, marsh recession occurs when vegetation cover declines, resulting in an unvegetated tidal mudflat or sandflat. This phenomenon has been observed in both natural and created marshes of the FRE, including at least 160 ha of natural marsh that disappeared from the foreshore of one deltaic island between 1989 and 2011 (Balke 2017; Marijnissen 2017), and in several degraded marsh creation projects described by Lievesley et al. (2016).
The abundance of native species is also a metric frequently used to monitor and assess projects, as dominant non-native or invasive species can alter the function and services of an ecosystem (Zedler and Kercher 2004; Jessop et al. 2015; Alldred and Baines 2016). For example, decomposition rates of invasive purple loosestrife (Lythrum salicaria L.), which likely established in the FRE in the mid-twentieth century, are significantly faster than sympatric native C. lyngbyei, potentially altering the timing of detritus supply and prey availability for juvenile salmon (Grout et al. 1997). Non-native cattail, especially hybrid Typha × glauca Godron has established in more recent decades, and now occupies an estimated 4% or 50 ha of tidal marsh habitats in the FRE, forming near-monocultures where present (Stewart et al. 2023). These invaded areas are less floristically diverse, contain fewer chironomids, and fewer benthic invertebrates than nearby sedge meadows, representing a major threat to biodiversity and food web interactions (Lee 2021; Stewart 2021). In general, the effects of non-native plant invasions are understudied in many wetland ecosystems, including the FRE. In the absence of realised or quantified effects, managers often justify the preservation of native species through a precautionary lens, based on the potential negative effects of invasion (Blossey et al. 2001).
Plant species richness may offer insight into the resilience of tidal marsh ecosystems when faced with environmental change. Biodiversity can stabilize the functioning of ecosystems through insurance mechanisms, such as asynchronous responses to environmental fluctuations, variable response speeds to disturbance, reduced strength of competition, and complementarity. These mechanisms can insure that biomass production, productivity, nutrient retention, and other ecosystem functions are maintained or enhanced in the wake of environmental stress, disturbance, and change (Yachi and Loreau 1999; Engelhardt and Ritchie 2002; Bruno et al. 2003; Loreau and de Mazancourt 2013).
The objective of this study was to advance our understanding of marsh habitat creation and management by learning from the successes and failures of over 40 years of projects in the FRE. This is motivated by a recent surge of interest among rightsholders and stakeholders in the estuary to build new habitats and enhance past projects. To achieve this, we used a combination of field sampling, spatial analysis, and modeling of plant communities to investigate key factors that contribute to the outcome of tidal marsh creation projects. Specifically, we asked (1) which factors are associated with marsh recession in created tidal marshes? (2) which factors influence the dominance of native species in created and natural marshes? and (3) which factors influence plant community diversity in created and natural tidal marshes?
Methods
Field Sites
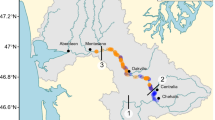
This study includes data from 78 marsh creation projects constructed between 1982 – 2015, and 16 reference marshes located in the FRE, southwest British Columbia (Fig. 1). All study sites are tidally influenced, with a mixed semidiurnal tidal cycle and tidal ranges varying from 3 m – 5 m. Among these are 51 projects and 7 reference sites surveyed in 2015 by Lievesley et al. (2016), in addition to 27 projects and 9 reference sites surveyed between June – August 2021. Apart from 12 embayments (enclosed marshes connected to the river via channels – see examples in Online Resource 1), all marsh creation sites were located along the riverward side of a rip rap dike. Created marshes included in this study are adjacent to industrial areas (50%), residential areas (18%), developed parks (17%), natural upland (9%), natural wetland (3%), agriculture (3%), and commercial areas (1%). Wetland reference sites may vary in selection criteria depending on study objectives (Kentula 2000). For the purposes of this study, we define reference sites as natural tidal marshes that, to our knowledge, were not created or significantly disrupted by human activity based on available satellite imagery and orthophotos dating to the 1950s (see detailed reference site descriptions in Online Resource 2). Reference sites were adjacent to natural uplands (40%), industrial areas (33%), agriculture (13%), natural wetlands (7%), and developed parks (7%). Apart from a small number of sites owned by the federal government (2), local municipalities (9), and private landowners (7), all study sites were located on Provincial Crown land (i.e., land owned by the provincial government but accessible to the public).
Created tidal marshes were located using a combination of desk- and field-based reconnaissance, correlating project descriptions and historical photographs provided in the Fraser River Estuary Management Program Atlas (CMN 2021) with field observations and 10 cm georeferenced aerial imagery. The location of randomized sample plots (1 × 1 m quadrats) were generated prior to site visits using the University of New Hampshire KML Tool Project for 2015 points (KML Tools Project 2023) and QGIS for 2021 points (v3.20, QGIS 2021) with spacing ranging from 2 m – 88 m. We targeted an optimum minimum sample size of 20 plots per site (James-Pirri et al. 2007), though occasionally fewer were sampled due to small marsh size or tidal constraints, and more were sampled in larger, heterogeneous sites (with spacing ≥ 2 m between plots). Quadrats were oriented perpendicular to the nearest major channel, typically the Fraser River. Surveyors recorded the aerial percent cover of all living macrophytes originating from within the quadrat, as well as exposed substrates (i.e., litter, mud, rock, wood debris). We focussed on macrophyte cover and invasive species abundance to align with metrics used by regulators, though other metrics, such as microbial communities (Santini et al. 2019), could be beneficial to add to monitoring data in the future. An overlapping percent cover method was employed to account for vegetative layering, frequently resulting in a total percent cover of > 100%. Each species was then classified into one of three origin classes: native, introduced, and unknown (see Online Resource 3 for a list of all species observed, and their respective origin classes). Plant identification and nomenclature was based on Hitchcock et al. (2018) and nativity was based on the B.C. Conservation Data Centre. On rare occasions plants were classified as unknown if no traits were present to differentiate between native and non-native species, and included the genera Lycopus, Alisma, Persicaria, Cardamine, and various immature grasses.
In addition to vegetation sampling, we mapped the boundary of each site using a combination of handheld GPS units (Garmin GPSMap® 64 s) and Apple iPad mini (5th generation) with Avenza Maps mapping software (v3.14.1; Avenza Systems Inc. 2021). Vegetated areas and unvegetated substrates > 5 m2 were mapped using the same approach, and the presence of other site features were noted, such as shear booms, docks, and other offshore structures.
Spatial Data
Spatial analyses were used to describe the condition and environmental context of plots and sites. Project area was calculated using site polygons that were mapped in the field, and was defined as the created marsh boundary of a project. Where available, original project descriptions, design schematics, historical imagery, and historical photographs were also used to optimize marsh boundary accuracy. We calculated the percent of recessed marsh in each project by dividing the area of recessed marsh, also mapped in the field, by the total project area. For the purposes of this study, recessed marsh was defined as areas > 5 m2 within tidal marsh creation projects that were designed for emergent macrophytes (e.g., Carex spp., Juncus spp.) but contained less than an estimated 5% cover in 2015 or 2021 surveys (see Online Resource 4 for visual examples). When present, vegetation in recessed areas was sporadic and primarily restricted to a small number of low-lying mudflat specialists < 10 cm in height (e.g., Callitriche stagnalis Scop., Eleocharis parvula Roem. & Schult., Limosella aquatica L.).
Using the Measurement Tool in QGIS, we quantified the percentage of edge habitat for each site by calculating the area of marsh located within 5 m of the river channel, which we then divided by the total project area. For each sample plot, proximity to the river channel was calculated in QGIS using the GRASS Toolbox (v7.8.6; GRASS Development Team 2012). Each site and plot were assigned a distance upriver, which was calculated as the channel-distance to a standardized line across the river mouth. In cases where multiple pathways to the river mouth were possible, distances were based on those of the largest, and therefore most influential, channel.
Elevation data were acquired from a digital elevation model (DEM) derived from a 2016 Province of British Columbia dataset (vertical accuracy ± 15 cm RMSE, horizontal accuracy ± 65 cm RMSE). Other studies have shown that LiDAR can overestimate marsh elevation, particularly when acquired during the growing season, depending on the height and density of vegetation (Hood 2007; Hladik et al. 2013). To investigate potential biases in LiDAR error we compared the discrepancies between survey-grade real-time kinematic (RTK)- and DEM-derived elevation at 211 points to test the effects of various dominant species on DEM error (Online Resource 5). We found that low-lying and sparsely-arranged species had little effect on DEM accuracy, while LiDAR overestimated ground elevation in areas dominated by tall, dense species by approximately 20 cm. To mitigate this error, we determined the mean site elevation by calculating the DEM-derived elevation of unvegetated areas and vegetated areas separately. Elevation data from unvegetated areas remained uncorrected, while vegetated areas were lowered by 20 cm, unless they were dominated by sparse or low emergent species, in which case they remained uncorrected. Based on area, a weighted mean site elevation was then calculated. For plot data, elevation was also corrected by looking at the dominant cover type of each plot. Plots were lowered by 20 cm if they contained > 40% cover of tall emergent species, while all other plots remained uncorrected. In some cases, plots or sites were dominated by a species not included in our error analysis. When this occurred, we used the correction factor of similar species as a proxy, based on morphological traits such as height and density.
Statistical Analysis
Marsh Recession Model
We used a zero-inflated beta regression model in R to estimate the effects of factors on recession in created marshes (‘gamlss’ package in R v4.0.5; R Core Team 2020, Rigby and Stasinopoulos 2005). Only created marshes were included in this model, as the complexity of measuring recession in reference marshes, which lack the clear boundaries of created sites, was beyond the scope of this study. We modelled the proportion of recessed marsh as a function of project age (years), project size (m2), distance upriver (km), percent edge habitat, mean project elevation (metres), river arm (Main or North) and the presence of offshore erosion protection (shear boom, log storage boom, or dock structure, see Online Resource 6 for more details). We also included an interaction between distance upriver and mean project elevation, as we predicted that elevational effects would be dependent on proximity to the marine environment, which influences both tidal range and salinity. We scaled numeric variables to a mean of 0 and a standard deviation of 1 before model fitting.
Dominance & Richness Models
To investigate factors influencing the dominance of native species and species richness in FRE marshes we used generalized linear mixed-effects models (‘lme4’ package in R; Bates et al. 2015). Unlike the site-based data of the marsh recession model, we used sample plot data from both created and reference marshes for this analysis. Dependent variables entailed relative cover of native species (defined as the cover of native species as a percentage of the total plant cover in a plot) for the native dominance model, native species richness per plot and non-native species richness per plot for the richness models. Independent variables for these models were selected based on data availability and their impact on plant species distributions in estuaries, as suggested in the literature. These included distance upriver (a proxy for environmental stressors including salinity and inundation), elevation, and distance to the nearest channel (Sanderson et al. 2000; Crain et al. 2004; Engels and Jensen 2009; Watson and Byrne 2009). We also tested for differences between created and natural marshes. Other categorical variables included river arm, closed embayments, and sample year, which was added to account for sampling differences between recording teams and differences in growing conditions between years. Sites were included as random effects to account for site-to-site variation. An interaction term between plot distance upriver and plot elevation was included, with the same rationale as our marsh recession model. Fit for the native dominance and richness models were evaluated using marginal and conditional R2 values (Nakagawa et al. 2017) using the ‘MuMIn’ package in R (Bartoń 2020).
Results
Marsh Recession
We sampled 78 created tidal marshes that varied in size from 20 – 59,309 m2 and were generally small (mean = 3261 ± 859.9 m2, median = 1115.7 m2). Recessed marsh ranged from 0 – 100% across the 78 created tidal marshes, averaging 13.6% (SD = 21.7%). This equates to approximately 23,666 m2 or 9.3% of the 254,357 m2 of created tidal marsh sampled. Two sites (3%) were fully recessed, and 40 (51%) contained recessed areas. Sites varied considerably in their numeric variable ranges: distance upriver (0.4 – 46.9 km), age (7 – 40 years), size (20 – 59,309 m2), mean elevation (-0.04 – 1.84 m) and proportion of edge habitat (0.0 – 100%). There were 29 sites (37%) with offshore erosion control present, and 35 sites (45%) were in the North Arm versus the Main Arm (Online Resource 7).
Sites further upriver were more likely to have marsh recession (βν = -1.43 ± 0.45, t = -3.17, p = 0.002; βμ = 0.70 ± 0.24, t = 2.97, p = 0.004). The model-predicted proportion of marsh recession increased from 0.20 at 0.44 km upriver to 0.89 at 46.7 km upriver (Fig. 2a). Sites with higher mean elevations were less likely to have marsh recession (βν = 1.30 ± 0.39, t = 3.36, p = 0.001; βμ = -0.32 ± 0.21, t = -1.56, p = 0.12; Fig. 2b). The model-predicted proportion of marsh recession decreased from 0.66 at an elevation of 0 m to 0.18 at an elevation of 1.84 m (Fig. 2b). Sites in the North Arm of the channel had higher proportions of recessed marsh than the Main Arm (βν = 1.41 ± 0.69, t = -2.03, p = 0.05; βμ = 0.97 ± 0.38, t = 2.57, p = 0.01; Fig. 2c). Project age (Fig. 2d), size, percent of edge habitat, or erosion protection did not affect the proportion of recessed marsh (Online Resource 8).
Plots visualizing the distribution of distance upriver (a), elevation (b), and river arm (c), and project age (d) in relation to marsh recession (proportion). Scatterplot lines visualize the model predictions for each variable while all other model variables are held fixed at the mean value of samples. Violin plot means are displayed with black dots, with error bars visualising standard error
Native Species Dominance
A total of 1,716 sample plots were included in the native species dominance model, with 1,244 originating from 78 created marshes (72%). Of these 1,244 plots, 850 were sampled at 51 sites in 2015, and 394 were sampled at 28 sites in 2021. The remaining 472 plots (28%) originated from 16 reference marshes, with 292 sampled in 2015 and 180 sampled in 2021. Numeric plot data encompassed a wide range of marsh conditions: channel proximity (0 – 201 m), distance upriver (0.4 – 46.9 km), and elevation (-0.77 – 2.25 m). Among categorical variables, 651 (38%) of plots were in the North Arm versus Main Arm, and 273 (16%) were in closed embayments versus exposed sites (Online Resource 9).
Dominance of native species ranged from 0 – 100% in the sample plots, averaging 61.7% (SD = 35.9%). Plots located further upriver were less likely to be dominated by native species (β = -0.44 ± 0.16, z = -2.82, p = 0.01; marginal R2 = 0.07, conditional R2 = 0.33, Fig. 3a; Online Resource 10). Native species dominance did not differ between plots located in closed embayments versus exposed sites (β = -0.73 ± 0.42, z = -1.74, p = 0.08; Fig. 3b). Elevation, river arm, reference site, sample year, and channel proximity did not affect native dominance per plot. Notably, native species dominance per plot also did not differ significantly between reference marshes (65.8%, SD = 35.9%) versus created marshes (60.2%, SD = 35.8%; Fig. 3c).
Plots visualizing the relationship between distance upriver (a), closed embayment designs (b), and reference marshes (c) on native dominance. Scatterplot line displays the model predictions for the variable while other model variables are held fixed. Violin plot mean values are shown by black dots, with accompanying lines displaying standard errors. ‘Yes’ and ‘No’ violin plot values (b) indicate whether a plot occurred inside a closed embayment or exposed marsh
Native Species Richness
Native richness ranged from 0 – 13 species/plot, averaging 3.7 (SD = 2.4) across all plots. A total of 107 native plant species were observed in plots (Online Resource 3). A significant interaction was observed between elevation and distance upriver, suggesting that the effects of elevation on native richness/plot is dependent on proximity to the river mouth (β = -0.42 ± 0.20, z = -2.11, p = 0.03, delta marginal R2 = 0.17, conditional R2 = 0.42). Plots that were further upriver (β = 0.12 ± 0.04, z = 2.73, p = 0.006, Fig. 4a) and higher in elevation (β = 0.89 ± 0.19, z = 4.75, p < 0.001, Fig. 4b), and were more likely to have higher native richness/plot. Conversely plots located in closed embayments (β = -0.56 ± 0.12, z = -4.48, p < 0.001, Fig. 4c) and those sampled in 2021 (β = -0.28 ± 0.85, z = -3.30, p = 0.001) had fewer native species/plot. There were no differences in species richness between plots in reference and created marshes (Fig. 4d), nor between the North Arm and Main Arm (Online Resource 11). There was no unequivocal evidence that the distance from a channel affected native species richness (β = 0.05 ± 0.03, z = 1.71, p = 0.09).
Plots visualizing the relationship between distance upriver, elevation, closed embayments, and reference sites on native richness (top row) and non-native richness (bottom row). Coloured scatterplot lines display the model predictions for the variable while other model variables are held fixed at the lower (Q1), mid (Q2), and upper (Q3) percentile values of an interacting effect, which includes elevation (plots a, e) and distance upriver (plots b, f). Violin plot mean values are shown by black dots, with accompanying lines displaying standard errors. Violin plot ‘Yes’ and ‘No’ values indicate whether a plot occurred inside a closed embayment or exposed marsh (e, g) and inside a reference site or natural marsh (d, h) respectively
Non-native Species Richness
Non-native species richness ranged from 0 – 12 species/plot, averaging 2.5 (SD = 1.9) across all plots. A total of 74 non-native plant species were observed in plots (Online Resource 3). As with native richness, there was a significant interaction between elevation and distance upriver, indicating the effects of elevation on non-native species richness/plot is dependent on proximity to the river mouth (β = -0.15 ± 0.02, z = -6.22, p < 0.001, delta marginal R2 = 0.27, conditional R2 = 0.56). Plots that were further upriver (βν = 0.40 ± 0.06, z = 6.60, p < 0.001; Fig. 4e), higher in elevation (β = 0.22 ± 0.02, z = 9.54, p < 0.001, Fig. 4f), and further from the nearest major channel (β = 0.07 ± 0.03, z = 2.38, p = 0.02) were likely to have more non-native species/plot, while plots sampled in 2021 were less likely to have fewer non-native species (β = -0.31 ± 0.12, z = -2.59, p = 0.01). There were no differences in the number of non-native plant species between plots located inside versus outside closed embayments (Fig. 4g), in reference versus created marshes (Fig. 4h), nor between the North and Main Arms (Online Resource 12).
Discussion
Marsh Recession
We found that marsh recession is frequent in created marshes of the FRE, occurring in 40 (51%) of the 78 projects included in this study, representing an estimated 23,666 m2 of created marsh lost. Our results suggest several contributing and interacting factors are likely involved, as found by others (Balke 2017; Marijnissen 2017). Possible factors include erosion from vessel wakes and river currents (Houser 2010; Bilkovic et al. 2019; El Safty and Marsooli 2020), nutrient enrichment (Deegan et al. 2012), sediment deficiency (Williams and Hamilton 1995; Atkins et al. 2016), poor project design and implementation (Kistritz 1995; Adams and Williams 2004), Canada Goose (Branta canadensis) herbivory (Dawe et al. 2000; Lievesley et al. 2016; Janus 2022), and relative sea-level rise (rSLR; Kirwan and Murray 2008; Russell et al 2023). This study found that marsh recession was more likely in North Arm versus Main Arm sites, which points towards vessel wakes and herbivory as possible drivers behind these losses. Wake energy is influenced by factors that vary within the estuary, such as channel morphology (depth and width), vessel characteristics (frequency, length, depth), and vessel speed, which to our knowledge is not regulated (Glamore 2008; Bilkovic et al 2019; El Safty and Marsooli 2020). Based on these factors, the North Arm appears to be more vulnerable to wake erosion for two reasons. First, our field observations suggest that boat activity and wake frequency is higher in the North Arm than the Main Arm. North Arm vessels are primarily recreational boats and tugs, the latter of which are more abundant in the North Arm due to extensive log booming tenures, and can produce wave heights that are well-beyond the threshold for shoreline sediment erosion (see Coops et al. 1996; Bilkovic et al. 2017). Second, the North Arm is substantially narrower than the Main Arm, limiting the ability of wake energy to dissipate before it reaches the shore (Bilkovic et al. 2019). In addition to wake erosion, the positive relationship between marsh recession, North Arm sites, and distance upriver may point towards Canada Goose herbivory as a driver behind marsh loss. Canada Geese were historically a migratory species in the FRE, and an infrequent winter visitor. However in the late 1960s and early 1970s several new resident genotypes were introduced, interbred, and have rapidly grown in number (Smith 2000). Janus (2022) detected over 4,000 Canada Geese in our study area in July 2022, and found that goose abundance and grazing impacts were more concentrated in the North Arm and upriver portions of the Main Arm, aligning with our marsh recession model predictions. Marsh habitat is less abundant in the North Arm and upriver reaches, possibly leading to the overexploitation of plant communities due to a lack of neighbouring habitat to dissipate grazing impacts (Kondoh 2003).
Our model predicted that created marshes with higher mean elevations were less likely to have marsh recession than lower elevation sites. To a degree, this may reflect the environmental limits of tidal marsh species, as emergent plants cease to grow at certain elevation thresholds (Cronk and Fennessy 2001). However, low elevation tidal marshes may also represent a “canary in a coal mine” for larger environmental issues, as these plant communities are at the front line of environmental change, stress, and disturbance. Low marsh plant species depend on specialized tissues and structures to survive the environmental stresses of prolonged inundation, elevated salinity, and anoxic conditions. Disturbances that inhibit these adaptations could result in lowered plant fitness or marsh dieback, reducing the resilience of these ecosystems to further degradation (Baldwin and Mendelssohn 1998; van Belzen et al. 2017; Rolando et al. 2023). For example, grazing could limit the effectiveness of aerenchyma (air pockets within the plant) to transport gases between the atmosphere and roots, resulting in anoxic stress or mortality (Barclay and Crawford 1982). Photosynthetic losses from grazing can result in lower belowground carbohydrate storage, which enables plants to survive anoxia (Barclay and Crawford 1982; Albrecht and Biemelt 1998). Belowground plant biomass may also decline in response to eutrophication, a common issue in coastal ecosystems worldwide (Deegan et al. 2012). These losses in above and belowground biomass could have biophysical ramifications, by amplifying erosional losses and reducing the potential for sediment accretion, thus limiting the ability of these habitats to resist erosion and self-adjust to rising sea levels, which are estimated to regionally increase by 1 m by the year 2100 (Coops et al. 1996; Morris et al. 2002; Bornhold et al. 2008; Gedan et al. 2011; Mitchell and Bilkovic 2019).
Unexpectedly, we found that project size and proportion of edge habitat did not have a significant effect on marsh recession. This fails to support the prevailing view that bigger projects are more resilient than smaller projects (Yan et al. 2021), but does not discount their value. Larger tidal marsh creation projects are typically more cost-effective, support more natural processes, are more heterogenous, and likely support more species as a result (Larkin et al. 2008; Varty and Zedler 2008; Diefenderfer et al. 2018; Hood 2020); therefore, larger created marshes are more likely to maintain ecosystem functioning than smaller projects in the presence of marsh recession.
Species Dominance
We found that dominance of native species declined with distance upriver, a trend that aligns with our frequency data for invasive plants (Online Resource 13). The high invasion resilience of marshes near the delta front can likely be attributed to elevated salinity and tidal stress (Engels and Jensen 2009; Borde et al. 2020), which excludes most non-native species and facilitates the dominance of a small number of native halophytes, such as three-square bulrush (Schoenoplectus pungens Vahl) and Lyngbye’s sedge (Cronk and Fennessy 2001; Crain et al. 2004). A larger pool of non-native species can establish as salinity and inundation stress diminishes, as evidenced by the positive relationship between non-native richness with distance upriver and elevation. Ongoing anthropogenic and natural riverine disturbances (e.g., anthropogenic log debris, eutrophication) also continue to facilitate the colonization of uninvaded areas (Zedler and Kercher 2004).
Invasive species that can defy common estuary-wide patterns and successfully establish in the delta front should be of particular concern to managers, as they may be able to exploit the low competitive ability of sympatric natives (Crain et al. 2004). These species are currently English cordgrass (Spartina anglica C.E. Hubb.) a highly-invasive species that has undergone significant management upon its detection in 2003 (Williams et al. 2004) and was not detected in this study, and narrow-leaved and hybrid cattail (T. angustifolia L, T. × glauca), which currently occupy approximately 50 ha of tidal marsh in the FRE, the majority of which is concentrated in the lower 10 km of the estuary (Stewart et al. 2023; Online Resource 13). Invasive cattail was fairly localized in our study sites, occurring in only 17 (22%) of created tidal marsh sites, while other invasive species such as yellow flag iris (Iris pseudacorus L.), reed canarygrass (Phalaris arundinacea L.), and purple loosestrife were found in 48 (62%), 65 (83%) sites and 73 (94%), respectively. Though most were located close to the delta front, many cattail-invaded sites defied estuary–wide patterns and had low native dominance. This may be attributed to the high displacement ability of cattail (Wilcox et al. 2008; Stewart 2021), and the comparatively low competitive ability of sympatric species, as evidenced by the averaged relative percent cover of 68.8 (SD = 37.2%) for cattail when present, higher than any other invasive species in this study (Online Resource 13).
The observed positive correlation between non-native dominance and richness with distance upriver may be useful for managers and practitioners as they plan for invasive species in the design and maintenance of created tidal marshes. Sites constructed further upriver may require a multi-faceted approach to monitoring, managing, and mitigating invasives, as they appear more vulnerable to a wider pool of non-native species. Near the estuary mouth, managers may need to direct their attention towards cattail. Site designs across the estuary could be adapted to face these unique threats. For example, practitioners in the Lower Columbia River Estuary have mitigated the threat of P. arundinacea by targeting elevations that are either too low to support it but can still support emergent vegetation, or are high enough that woody plants can grow and compete with it (Sinks et al. 2021). Similarly, the low elevation range of C. lyngbyei has been targeted by practitioners at some projects in the FRE as a means of mitigating L. salicaria, which is typically most abundant in mid to high elevation C. lyngbyei marshes (Adams and Williams 2004). Stewart et al. (2023) found that created tidal marshes in the FRE were disproportionately more invaded and vulnerable to cattail invasions than natural marshes, and suggested that design, including factors such as elevation, proximity to neighbouring infestations, and connectivity to the river, can influence their vulnerability. Our findings support the role of design in invasability, as of the 17 created marshes where cattail was present, 9 (60%) are closed embayments, representing 75% of all such sites in this study. Embayed marshes occur in isolated basins that are connected to adjacent river channels via a small number of engineered outlets (Online Resource 1). Poor soil drainage and flushing results (Mossman et al. 2012), which favours cattail establishment by increasing saturation stress (Farrer and Goldberg 2009), nutrient availability (Woo and Zedler 2002), and the accumulation of leaf litter (Vaccaro et al. 2009; Stewart 2021).
Project Trajectories
Contrary to our expectations, project age did not have a significant effect on the proportion of recession, nor the dominance of native species in created marshes. This suggests that (1) modern projects are not guaranteed to have positive outcomes, as they remain exposed to unaddressed systemic stressors (see 4.1) and (2) project designs, location, and maintenance activities that mitigate stressors are more important than age in predicting whether created tidal marshes are resilient over time. We and others (Kistritz 1995) have observed evidence of this in the FRE, where several projects of varying ages failed to support marsh vegetation upon project completion or were degraded soon after, and have remained in mudflat alternative stable states ever since. Similar observations have been made in other coastal systems, for example Rolando et al. (2023) noted that many salt marshes in their study did not naturally recover after dieback events, while supplemental planting was highly successful at restoring and improving the resilience of degraded areas, without any environmental amendments. Handa and Jefferies (2000) similarly found that marshes damaged by grazing were unlikely to recover without assisted planting due to secondary feedbacks in the soil environment. If project success or failure is predictable soon after completion, this could potentially validate the ≤ 5 year monitoring period prescribed to most projects in the region historically (Harper and Quigley 2005). We advise caution in this interpretation. Environmental changes and disturbances are predicted to increase in frequency and intensity with climate change, with possible adverse and unpredicted effects on created marsh health (Alber et al. 2008; Stagg et al. 2021; Rolando et al. 2023). Long-term monitoring and adaptive management may be critical tools for ensuring the functioning and resilience of projects in perpetuity (Levings 2000). For example, debris supply can temporarily increase during extreme flood events, accumulate on marsh platforms during high tide or river flows, and smother marsh vegetation if it remains in situ. By our estimation wood debris now smothers approximately 2,650 m2 or 1% of created marsh area in the FRE and is a widespread regional issue (Hood 2022). Protective shear booms and debris fences were constructed in some projects to mitigate wood debris, however no long-term monitoring, maintenance, or remedial clauses were required by regulators (Kistritz 1995; Levings 2000; Adams and Williams 2004). As a result, many of these structures are non-functional or altogether absent due to aging materials, and the sites remain vulnerable to and degraded by debris (Lievesley et al. 2016). In some instances new and unpredicted stressors may arise, such as Canada Geese, whose regional impact and population size have increased in recent decades (Dawe et al. 2000; Janus 2022). Sites that were unimpacted or resilient to herbivory at the time of their construction may undergo degradation in current conditions, thus requiring modern intervention (Dawe et al. 2011).
Our findings suggest that the plant communities of created marshes resemble natural marshes in their species composition, as measured by species richness, within decades. This is surprising given most sites were planted with almost exclusively C. lyngbyei, and even multi-species plantings rarely exceeded five species (Adams and Williams 2004). These findings align with the 15-year observations of Mitsch et al. (2012), who found that 90–95% of their final species richness was achieved within five years of marsh construction. The comparable richness between created and reference marsh plots may be a result of several factors, including dispersal from upstream or adjacent source populations, despite extensive habitat fragmentation that has widened dispersal distances in the estuary. The small size of most projects may also increase their susceptibility to colonization. Donor plugs (golf hole-sized plugs extracted from nearby marshes) used to vegetate many of the projects may have possessed viable and diverse seed assemblages. One final explanation is that the act of disturbance required to create novel marsh habitats may support colonization by novel species, resulting in elevated richness. Nevertheless these findings show that created marsh plant communities in the FRE can possess a rich diversity of species within a short period of time, which is important for their ecological function and resilience in the context of global environmental change (Zedler et al. 2001; Steudel et al. 2011; Fitzgerald et al. 2021; Hong et al. 2022).
Future Studies
Although this study points towards boat wakes and herbivory as possible drivers of created marsh recession in the FRE, mechanistic studies are required to further understand their impacts. To our knowledge no estuary-wide boat wake study has occurred to date, and dedicated Canada Goose research only commenced recently. Interestingly, both of these stressors have been accounted for in several past project designs, through features such as offshore sheer booms, piling and timber walls, and rock sills to mitigate wake erosion, or exclosure fencing and planting of less-palatable species to reduce herbivory (Kistritz et al. 1992; Adams and Williams 2004). Despite these efforts, the findings of this and other studies indicate these mitigation approaches may be temporary “band-aid” solutions, and that research-informed regional management may be required to produce meaningful change (Kistritz 1995; Lievesley et al. 2016).
We quantified losses in created marshes, but the status of nearby reference marshes remains unknown and requires further investigation. Reference sites were not included in the recession analysis, largely because they have no permanent foreshore boundary (e.g., rock sill) to refer to for easily documenting historical change. As a result, we are unable to evaluate whether the recession we observed was an unique issue to created marshes, suggesting their compromised resilience, or whether this is reflective of a larger recession issue occurring throughout the estuary (Kistritz et al. 1992). Future research should investigate this further, using historical imagery and other knowledge sources (e.g., records, maps, photos), to quantify large-scale changes in marsh foreshores of the FRE, and shed further light on the health and resilience of these ecosystems.
In addition to serving as a springboard for future research, our findings should provide incentive for testing new and innovative approaches for marsh creation in the FRE and similar systems. Most historical project designs contain forms of hard engineering, particularly riprap sills. This approach has been largely effective at meeting regulatory criteria (i.e., target vegetative cover after a 5-year period), but our findings suggest these conventional designs may be limited in their ability to support self-adapting, resilient marshes that can persist in a changing environment (Mitchell and Bilkovic 2019). Novel approaches that shift away from static, hard-engineered designs and instead utilize natural processes, such as sediment accretion, accretion/vegetation feedback loops, and facilitation should be explored further (Silliman et al. 2015; Bilkovic and Mitchell 2017; Mitchell and Bilkovic 2019).
Conclusion
The FRE has been heavily modified by human activity and continues to be threatened by habitat loss and degradation to this day. Marsh creation projects have been perceived as a tool for mitigating losses in fish habitat productivity, but the long-term outcomes of these projects have never been evaluated to this detail. Given the number and scale of proposed offsetting projects on the horizon, we believe this study is timely in providing a preliminary investigation into what influences the long-term success or failure of created marsh projects. Our findings suggest that regardless of project age, a successful long-term outcome is not guaranteed. While some projects have remained resilient to vegetation recession and invasibility after several decades, we observed recession in over half of the study sites, equating to 9.3% of created marsh area, and invasive species such as Typha spp. and P. arundinacea continue to displace created marsh plant communities. Our findings demonstrate that those designing, constructing, and managing these sites can mitigate marsh recession by factors such as project location, elevation, and sound implementation and maintenance, particularly soon after completion. Innovative new design approaches that can utilise existing natural processes such as sediment accretion and facilitation should also be experimentally tested to further mitigate recession. Invasive species management will largely depend on project design and location, with Typha spp. posing the greatest threat to sites near the delta front and in embayments, while other species such as L. salicaria, I. pseudacorus, and P. arundinacea increase with distance upriver. We hope lessons from these investigations will advance the knowledge of tidal marsh creation, inform management approaches, and serve as stepping stones for further research in the FRE and other urbanized estuaries.
Data Availability
The data, R Scripts, and other digitized materials related used in this study are openly available on Github at https://github.com/asarum-ecological/2024_FRECreatedMarsh. The authors request that appropriate credit be given should these materials be used for further study.
References
Adams MA, Williams GL (2004) Tidal marshes of the Fraser River estuary: composition, structure, and a history of marsh creation efforts to 1997. In: Groulx DC, Luternauer JL, Bilderback DE (eds) Fraser River Delta, British Columbia: Issues of an Urban Estuary, Geological Survey of Canada, Bulletin. Geological Survey of Canada, pp 147–172. https://doi.org/10.4095/215772
Alber M, Swenson EM, Adamowicz SC, Mendelssohn IA (2008) Salt marsh dieback: an overview of recent events in the US. Estuarine, Coastal and Shelf Science 80:1–11. https://doi.org/10.1016/j.ecss.2008.08.009
Albrecht G, Biemelt S (1998) A comparative study on carbohydrate reserves and ethanolic fermentation in the roots of two wetland and non-wetland species after commencement of hypoxia. Physiologia Plantarum 104:81–86. https://doi.org/10.1034/j.1399-3054.1998.1040111.x
Alldred M, Baines SB (2016) Effects of wetland plants on denitrification rates: a meta-analysis. Ecological Applications 26:676–685. https://doi.org/10.1890/14-1525
Arias-Ortiz A, Oikawa PY, Carlin J, Masqué P, Shahan J, Kanneg S, Paytan A, Baldocchi DD (2021) Tidal and nontidal marsh restoration: a trade-off between carbon sequestration, methane emissions, and soil accretion. J Geophys Res: Biogeosci 126:e2021JG006573. https://doi.org/10.1029/2021JG006573
Atkins RJ, Tidd M, Ruffo G (2016) Sturgeon Bank, Fraser River Delta, BC, Canada: 150 years of human influences on salt marsh sedimentation. Journal of Coastal Research 75:790–794. https://doi.org/10.2112/SI75-159.1
Avenza Systems Inc (2021) Avenza maps. Avenza Systems Inc., Toronto, Canada. https://www.avenza.com/avenza-maps/
Baldwin AH, Mendelssohn IA (1998) Effects of salinity and water level on coastal marshes: an experimental test of disturbance as a catalyst for vegetation change. Aquatic Botany 61:255–268. https://doi.org/10.1016/S0304-3770(98)00073-4
Balke E (2017) Investigating the role of elevated salinity in the recession of a large brackish marsh in the Fraser River Estuary (Applied Research Project Report). Simon Fraser University & British Columbia Institute of Technology, Burnaby. https://circuit.bcit.ca/repository/islandora/object/repository%3A759/datastream/PDF/download/citation.pdf
Barbier EB, Hacker SD, Kennedy C, Koch EW, Stier AC, Silliman BR (2011) The value of estuarine and coastal ecosystem services. Ecological Monographs 81:169–193. https://doi.org/10.1890/10-1510.1
Barclay AM, Crawford RMM (1982) Plant growth and survival under strict anaerobiosis. Journal of Experimental Botany 33:541–549. https://doi.org/10.1093/jxb/33.3.541
Bartoń K (2020) MuMIn: Multi-model inference. https://CRAN.R-project.org/package=MuMIn
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Software 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bilkovic DM, Mitchell MM (2017) Designing living shoreline salt marsh ecosystems to promote coastal resilience. In: Bilkovic DM, Mitchell MM, La Peyre MK, Toft JD (eds) Living shorelines: the science and management of nature-based coastal protection. CRC Press, Taylor & Francis Group, Boca Raton, pp 293–316. https://doi.org/10.1201/9781315151465-19
Bilkovic DM, Mitchell M, Davis J, Andrews E, King A, Mason P, Herman J, Tahvildari N, Davis J (2017) Review of boat wake wave impacts on shoreline erosion and potential solutions for the Chesapeake Bay (No. STAC Publication 17-002). Edgewater
Bilkovic DM, Mitchell MM, Jennifer D, Herman J, Andrews E, King A, Mason P, Tahvildari N, Jana D, Dixon RL (2019) Defining boat wake impacts on shoreline stability toward management and policy solutions. Ocean & Coastal Management 182:104945. https://doi.org/10.1016/j.ocecoaman.2019.104945
Blossey B, Skinner LC, Taylor J (2001) Impact and management of purple loosestrife (Lythrum salicaria) in North America. Biodiversity & Conservation 10:1787–1807. https://doi.org/10.1023/A:1012065703604
Borde AB, Diefenderfer HL, Cullinan VI, Zimmerman SA, Thom RM (2020) Ecohydrology of wetland plant communities along an estuarine to tidal river gradient. Ecosphere 11:e03185. https://doi.org/10.1002/ecs2.3185
Bornhold BD, British Columbia, Climate Change Branch, British Columbia, Ministry of Environment (2008) Projected sea level changes for British Columbia in the 21st century. British Columbia Ministry of Environment, Victoria
Boyle CA (1997) Changes in land cover and subsequent effects on Lower Fraser Basin ecosystems from 1827 to 1990. Environmental Management 21:185–196. https://doi.org/10.1007/s002679900017
Bradford MJ, Macdonald JS, Levings CD (2017) Monitoring fish habitat compensation in the Pacific region: lessons from the past 30 years (No. 2017/033), DFO Can. Sci. Advis. Sec. Res. Doc. Fisheries and Oceans Canada, Ottawa
Broome SW, Craft CB, Burchell MR (2019) Tidal marsh creation. In: Perillo GME, Wolanski E, Cahoon DR, Hopkinson CS (eds) Coastal wetlands: an integrated ecosystem approach. Elsevier, Amsterdam, pp 789–816
Brophy LS, Greene CM, Hare VC, Holycross B, Lanier A, Heady WN, O’Connor K, Imaki H, Haddad T, Dana R (2019) Insights into estuary habitat loss in the western United States using a new method for mapping maximum extent of tidal wetlands. PLoS ONE 14:e0218558. https://doi.org/10.1371/journal.pone.0218558
Bruno JF, Stachowicz JJ, Bertness MD (2003) Inclusion of facilitation into ecological theory. Trends in Ecology & Evolution 18:119–125. https://doi.org/10.1016/S0169-5347(02)00045-9
Butler RW, Campbell RW, 1987. The birds of the Fraser River Delta: populations, ecology, and international significance, Occasional paper / Canadian Wildlife Service. Environment Canada, Canadian Wildlife Service, Ottawa
Chalifour L, Scott DC, MacDuffee M, Iacarella JC, Martin TG, Baum JK (2019) Habitat use by juvenile salmon, other migratory fish, and resident fish species underscores the importance of estuarine habitat mosaics. Marine Ecology Progress Series 625:145–162. https://doi.org/10.3354/meps13064
Community Mapping Network (CMN) (2021) BIEAP - FREMP Atlas [Website]. Burrard Inlet Environmental Action Program & Fraser River Estuary Management Program. https://cmnmaps.ca/dfo_fremp/. Accessed September 2021
Coops H, Geilen N, Verheij HJ, Boeters R, van der Velde G (1996) Interactions between waves, bank erosion and emergent vegetation: an experimental study in a wave tank. Aquatic Botany 53:187–198. https://doi.org/10.1016/0304-3770(96)01027-3
Correa RE, Xiao K, Conrad SR, Wadnerkar PD, Wilson AM, Sanders CJ, Santos IR (2021) Groundwater carbon exports exceed sediment carbon burial in a salt marsh. Estuaries and Coasts. https://doi.org/10.1007/s12237-021-01021-1
Crain CM, Silliman BR, Bertness SL, Bertness MD (2004) Physical and biotic drivers of plant distribution across estuarine salinity gradients. Ecology 85:2539–2549. https://doi.org/10.1890/03-0745
Cronk JK, Fennessy MS (2001) Wetland plants: biology and ecology. Lewis Publishers, Boca Raton
Dawe NK, Bradfield GE, Boyd WS, Trethewey DEC, Zolbrod AN (2000) Marsh creation in a Northern Pacific Estuary: is thirteen years of monitoring vegetation dynamics enough? Conserv Ecol 4:12. https://doi.org/10.5751/ES-00214-040212
Dawe NK, Boyd WS, Buechert R, Stewart AC (2011) Recent, significant changes to the native marsh vegetation of the Little Qualicum River estuary, British Columbia; a case of too many Canada Geese (Branta canadensis)? British Columbia Birds 21:11–31
Day JW, Gunn JD, Folan WJ, Yáñez-Arancibia A, Horton BP (2012) The influence of enhanced post-glacial coastal margin productivity on the emergence of complex societies. The Journal of Island and Coastal Archaeology 7:23–52. https://doi.org/10.1080/15564894.2011.650346
Deegan LA, Johnson DS, Warren RS, Peterson BJ, Fleeger JW, Fagherazzi S, Wollheim WM (2012) Coastal eutrophication as a driver of salt marsh loss. Nature 490:388–392. https://doi.org/10.1038/nature11533
Diefenderfer HL, Sinks IA, Zimmerman SA, Cullinan VI, Borde AB (2018) Designing topographic heterogeneity for tidal wetland restoration. Ecological Engineering 123:212–225. https://doi.org/10.1016/j.ecoleng.2018.07.027
El Safty H, Marsooli R (2020) Ship wakes and their potential impacts on salt marshes in Jamaica Bay, New York. Journal of Marine Science and Engineering 8:325. https://doi.org/10.3390/jmse8050325
Engelhardt KAM, Ritchie ME (2002) The Effect of aquatic plant species richness on wetland ecosystem processes. Ecology 83:2911–2924. https://doi.org/10.1890/0012-9658(2002)083[2911:TEOAPS]2.0.CO;2
Engels JG, Jensen K (2009) Patterns of wetland plant diversity along estuarine stress gradients of the Elbe (Germany) and Connecticut (USA) Rivers. Plant Ecology & Diversity 2:301–311. https://doi.org/10.1080/17550870903508947
Farrer EC, Goldberg DE (2009) Litter drives ecosystem and plant community changes in cattail invasion. Ecological Applications 19:398–412. https://doi.org/10.1890/08-0485.1
Fitzgerald M, Gonzalez K, Funk JL, Whitcraft CR, Allen BJ (2021) Recovering ecosystem functions in a restored salt marsh by leveraging positive effects of biodiversity. Ecosphere 12:e03664. https://doi.org/10.1002/ecs2.3664
Gedan KB, Kirwan ML, Wolanski E, Barbier EB, Silliman BR (2011) The present and future role of coastal wetland vegetation in protecting shorelines: answering recent challenges to the paradigm. Climatic Change 106:7–29. https://doi.org/10.1007/s10584-010-0003-7
Glamore WC (2008) A decision support tool for assessing the impact of boat wake waves on inland waterways. Presented at the International Conference on Coastal and Port Engineering in Developing Countries, pp 1–20
GRASS Development Team (2012) Geographic resources analysis support system (GRASS). https://grass.osgeo.org
Grout JA, Levings CD, Richardson JS (1997) Decomposition rates of purple loosestrife (Lythrum salicaria) and Lyngbyei’s Sedge (Carex lyngbyei) in the Fraser River Estuary. Estuaries 20:96–102
Handa IT, Jefferies RL (2000) Assisted revegetation trials in degraded salt-marshes. Journal of Applied Ecology 37:944–958. https://doi.org/10.1046/j.1365-2664.2000.00531.x
Harper DJ, Quigley JT (2005) No net loss of fish habitat: a review and analysis of habitat compensation in Canada. Environmental Management 36:343–355. https://doi.org/10.1007/s00267-004-0114-x
Hitchcock CL, Cronquist A, Giblin D, Legler B, Zika PF, Olmstead RG (2018) Flora of the Pacific Northwest: an illustrated manual, 2nd edn. University of Washington Press, Seattle
Hladik C, Schalles J, Alber M (2013) Salt marsh elevation and habitat mapping using hyperspectral and LIDAR data. Remote Sensing of Environment 139:318–330. https://doi.org/10.1016/j.rse.2013.08.003
Hong P, Schmid B, De Laender F, Eisenhauer N, Zhang X, Chen H, Craven D, De Boeck HJ, Hautier Y, Petchey OL, Reich PB, Steudel B, Striebel M, Thakur MP, Wang S (2022) Biodiversity promotes ecosystem functioning despite environmental change. Ecology Letters 25:555–569. https://doi.org/10.1111/ele.13936
Hood WG (2007) Scaling tidal channel geometry with marsh island area: A tool for habitat restoration, linked to channel formation process. Water Resour Res 43:W03409. https://doi.org/10.1029/2006WR005083
Hood WG (2020) Applying tidal landform scaling to habitat restoration planning, design, and monitoring. Estuarine, Coastal and Shelf Science 244:106060. https://doi.org/10.1016/j.ecss.2018.12.017
Hood WG (2022) Distribution of large wood in River Delta tidal marshes: implications for habitat restoration. Estuaries and Coasts. https://doi.org/10.1007/s12237-022-01122-5
Houser C (2010) Relative importance of vessel-generated and wind waves to salt marsh erosion in a restricted fetch environment. Journal of Coastal Research 26:230–240
James-Pirri M-J, Roman CT, Heltshe JF (2007) Power analysis to determine sample size for monitoring vegetation change in salt marsh habitats. Wetlands Ecol Manage 15:335–345. https://doi.org/10.1007/s11273-007-9034-x
Janus D (2022) Introduced Canada Geese in the Fraser River Estuary: a conservation challenge, UBC Sustainability Hub. University of British Columbia, Vancouver. https://sustain.ubc.ca/sites/default/files/2022-044_Canada%20Geese%20in%20the%20Fraser%20Estuary_Janus_0.pdf
Jessop J, Spyreas G, Pociask GE, Benson TJ, Ward MP, Kent AD, Matthews JW (2015) Tradeoffs among ecosystem services in restored wetlands. Biological Conservation 191:341–348. https://doi.org/10.1016/j.biocon.2015.07.006
Kehoe LJ, Lund J, Chalifour L, Asadian Y, Balke E, Boyd S, Carlson D, Casey JM, Connors B, Cryer N, Drever MC, Hinch S, Levings C, MacDuffee M, McGregor H, Richardson J, Scott DC, Stewart D, Vennesland RG, Wilkinson CE, Zevit P, Baum JK, Martin TG (2021) Conservation in heavily urbanized biodiverse regions requires urgent management action and attention to governance. Conserv Sci Pract 3:e310. https://doi.org/10.1111/csp2.310
Kennish MJ (2002) Environmental threats and environmental future of estuaries. Environmental Conservation 29:78–107. https://doi.org/10.1017/S0376892902000061
Kentula ME (2000) Perspectives on setting success criteria for wetland restoration. Ecological Engineering 15:199–209. https://doi.org/10.1016/S0925-8574(00)00076-8
Kirwan ML, Murray AB (2008) Ecological and morphological response of brackish tidal marshland to the next century of sea level rise: Westham Island, British Columbia. Global and Planetary Change 60:471–486. https://doi.org/10.1016/j.gloplacha.2007.05.005
Kistritz RU (1995) Habitat compensation, restoration and creation in the Fraser River Estuary: are we achieving a no-net-loss of fish habitat? (Can. Tech. Rept. No. 2349). Fish. Aquat. Sci. https://publications.gc.ca/collections/collection_2007/dfo-mpo/Fs97-4-2349E.pdf
Kistritz RU, Scott KJ (1992) Historical changes in fish habitat of selected reaches in the lower Fraser River, 1859 to 1990 (Canadian Technical Report). Prepared for Department of Fisheries and Oceans Biological Sciences Branch, West Vancouver
Kistritz RU, Hall KJ, Yesaki I (1983) Productivity, detritus flux, and nutrient cycling in a Carex lyngbyei tidal marsh. Estuaries 6:227–236. https://doi.org/10.2307/1351514
Kistritz R, Williams G, Scott J (1992) Inspection of red-coded habitat: Fraser River Estuary summer of 1992. Fraser River Estuary Management Program, New Westminster
KML Tools Project (2023) KML Tools [Website]. University of New Hampshire Cooperative Extension. https://ceinfo.unh.edu/kmlTools/. Accessed June 202
Kondoh M (2003) Habitat fragmentation resulting in overgrazing by herbivores. Journal of Theoretical Biology 225:453–460. https://doi.org/10.1016/S0022-5193(03)00279-0
Larkin DJ, Madon SP, West JM, Zedler JB (2008) Topographic heterogeneity influences fish use of an experimentally restored tidal marsh. Ecological Applications 18:483–496
Lee JJ (2021) The impacts of exotic Typha on benthic invertebrate communities in the South Arm of the Fraser River Estuary (Applied Research Project Report). Simon Fraser University & British Columbia Institute of Technology, Burnaby. https://circuit.bcit.ca/repository/islandora/object/repository%3A1901/datastream/PDF/download/citation.pdf
Levings CD (2000) An overview assessment of compensation and mitigation techniques used to assist fish habitat management in British Columbia estuaries. In: Knudsen EE, Steward CR, MacDonald DD, Williams JE, Reiser DW (eds) sustainable fisheries management. CRC Press, Boca Raton, pp 341–347
Levings CD, Nishimura DJH eds (1996) Created and restored sedge marshes in the lower Fraser River and estuary: an evaluation of their functioning as fish habitat (Canadian Technical Report No. 2126), Fisheries and Aquatic Sciences. https://waves-vagues.dfo-mpo.gc.ca/library-bibliotheque/209017.pdf
Levy DA, Northcote TG (1982) Juvenile Salmon Residency in a Marsh Area of the Fraser River Estuary. Can J Fish Aquat Sci 39:270–276. https://doi.org/10.1139/f82-038
Lievesley M, Stewart D, Knight R, Mason B (2016) Assessing habitat compensation and examining limitations to native plant establishment in the lower Fraser River Estuary (Technical Report). BC Conservation Foundation & Community Mapping Network. https://www.cmnbc.ca/wp-content/uploads/2018/11/Lievesley-et-al-2016_Marsh-riparian-habitat-compensation-in-the-Fraser-River-Estuary.pdf
Loreau M, de Mazancourt C (2013) Biodiversity and ecosystem stability: a synthesis of underlying mechanisms. Ecology Letters 16:106–115. https://doi.org/10.1111/ele.12073
Marijnissen R (2017) Potential mechanisms for the salt marsh recession on Sturgeon Bank (Master’s Thesis). Delft University of Technology, Delft, Netherlands. http://resolver.tudelft.nl/uuid:4fb116d6-39d4-46f9-ba4c-ac8f59e74549
Mitchell M, Bilkovic DM (2019) Embracing dynamic design for climate-resilient living shorelines. Journal of Applied Ecology 56:1099–1105. https://doi.org/10.1111/1365-2664.13371
Mitsch WJ, Zhang L, Stefanik KC, Nahlik AM, Anderson CJ, Bernal B, Hernandez M, Song K (2012) Creating wetlands: primary succession, water quality changes, and self-design over 15 years. BioScience 62:237–250. https://doi.org/10.1525/bio.2012.62.3.5
Moody AI (1993) A report concerning the “provision of baseline maps and quantitative data on species composition and cover at marshes developed at the Fraser River Estuary.” Prepared for Fisheries and Oceans Canada, West Vancouver
Morris JT, Sundareshwar PV, Nietch CT, Kjerfve B, Cahoon DR (2002) Responses of coastal wetlands to rising sea level. Ecology 83:2869–2877. https://doi.org/10.1890/0012-9658(2002)083[2869:ROCWTR]2.0.CO;2
Mossman HL, Davy AJ, Grant A (2012) Does managed coastal realignment create saltmarshes with ‘equivalent biological characteristics’ to natural reference sites? Journal of Applied Ecology 49:1446–1456. https://doi.org/10.1111/j.1365-2664.2012.02198.x
Mudd SM, D’Alpaos A, Morris JT (2010) How does vegetation affect sedimentation on tidal marshes? Investigating particle capture and hydrodynamic controls on biologically mediated sedimentation. Journal of Geophysical Research 115, 2009JF001566. https://doi.org/10.1029/2009JF001566
Nakagawa S, Johnson PCD, Schielzeth H (2017) The coefficient of determination R 2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. Journal of The Royal Society Interface 14:20170213. https://doi.org/10.1098/rsif.2017.0213
O’Meara TA, Hillman JR, Thrush SF (2017) Rising tides, cumulative impacts and cascading changes to estuarine ecosystem functions. Scientific Reports 7:10218. https://doi.org/10.1038/s41598-017-11058-7
Peterson CH, Able KW, DeJong CF, Piehler MF, Simenstad CA, Zedler JB (2008) Practical proxies for tidal marsh ecosystem services. In: Advances in marine biology. Elsevier, pp 221–266. https://doi.org/10.1016/S0065-2881(08)00004-7
QGIS.org (2021) QGIS Geographic Information System. Open Source Geospatial Foundation Project. http://qgis.org
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/
Rigby RA, Stasinopoulos DM (2005) Generalized additive models for location, scale and shape. Journal of the Royal Statistical Society Series C: Applied Statistics 54:507–554. https://doi.org/10.1111/j.1467-9876.2005.00510.x
Rolando JL, Hodges M, Garcia KD, Krueger G, Williams N, Carr J Jr, Robinson J, George A, Morris J, Kostka JE (2023) Restoration and resilience to sea level rise of a salt marsh affected by dieback events. Ecosphere 14:e4467. https://doi.org/10.1002/ecs2.4467
Russell SJ, Windham-Myers L, Stuart-Haëntjens EJ, Bergamaschi BA, Anderson F, Oikawa P, Knox SH (2023) Increased salinity decreases annual gross primary productivity at a Northern California brackish tidal marsh. Environmental Research Letters 18:034045. https://doi.org/10.1088/1748-9326/acbbdf
Sanderson EW, Ustin SL, Foin TC (2000) The Influence of Tidal Channels on the Distribution of Salt Marsh Plant Species in Petaluma Marsh, CA, USA. Plant Ecology 146:29–41
Santini NS, Lovelock CE, Hua Q, Zawadzki A, Mazumder D, Mercer TR, Muñoz-Rojas M, Hardwick SA, Madala BS, Cornwell W, Thomas T, Marzinelli EM, Adam P, Paul S, Vergés A (2019) Natural and regenerated saltmarshes exhibit similar soil and belowground organic carbon stocks, root production and soil respiration. Ecosystems 22:1803–1822. https://doi.org/10.1007/s10021-019-00373-x
Scott KJ, Susanto R (1993) Analysis of fish occurrence in restored, unvegetated and natural habitats in the Fraser River Estuary, March through August, 1992. Department of Fisheries and Oceans Biological Sciences Branch, West Vancouver
Silliman BR, Schrack E, He Q, Cope R, Santoni A, Van Der Heide T, Jacobi R, Jacobi M, Van De Koppel J (2015) Facilitation shifts paradigms and can amplify coastal restoration efforts. Proceedings of the National Academy of Sciences of the United States of America 112:14295–14300. https://doi.org/10.1073/pnas.1515297112
Sinks IA, Borde AB, Diefenderfer HL, Karnezis JP (2021) Assessment of methods to control invasive reed canarygrass (Phalaris arundinacea) in tidal freshwater wetlands. Natur Areas J 41:172–185. https://doi.org/10.3375/043.041.0303
Small C, Nichols RJ (2003) A global analysis of human settlement in coastal zones. Journal of Coastal Research 19:17
Smith DW (2000) Management of Canada Geese in the Lower Fraser Valley, southwestern British Columbia. In: Dickson K (eds) Towards Conservation of the Diversity of Canada Geese. Canadian Wildlife Service, pp 151–158. https://publications.gc.ca/collections/Collection/CW69-1-103E.pdf
Stagg CL, Osland MJ, Moon JA, Feher LC, Laurenzano C, Lane TC, Jones WR, Hartley SB (2021) Extreme precipitation and flooding contribute to sudden vegetation dieback in a Coastal Salt Marsh. Plants 10:1841. https://doi.org/10.3390/plants10091841
Steudel B, Hautier Y, Hector A, Kessler M (2011) Diverse marsh plant communities are more consistently productive across a range of different environmental conditions through functional complementarity. Journal of Applied Ecology 48:1117–1124. https://doi.org/10.1111/j.1365-2664.2011.01986.x
Stewart D (2021) Undetected but widespread: the cryptic invasion of non-native cattail (Typha) in the Fraser River Estuary (Master’s Thesis). University of British Columbia, Vancouver. https://open.library.ubc.ca/soa/cIRcle/collections/ubctheses/24/items/1.0397016
Stewart D, Hood WG, Martin TG (2023) Undetected but widespread: the cryptic invasion of non-native cattail (Typha) in a Pacific Northwest estuary. Estuaries and Coasts 46:802–817. https://doi.org/10.1007/s12237-023-01171-4
Stronach JA (1995) An analysis of plant and benthic invertebrate communities in marshes on the Lower Fraser River. Prepared for Department of Fisheries and Oceans West Vancouver Laboratory by Scaconsult Marine Research Ltd.
Vaccaro LE, Bedford BL, Johnston CA (2009) Litter accumulation promotes dominance of invasive species of cattails (Typha spp.) in Lake Ontario wetlands. Wetlands 29:1036–1048. https://doi.org/10.1672/08-28.1
van Belzen J, van de Koppel J, Kirwan ML, van der Wal D, Herman PMJ, Dakos V, Kéfi S, Scheffer M, Guntenspergen GR, Bouma TJ (2017) Vegetation recovery in tidal marshes reveals critical slowing down under increased inundation. Nature Communications 8:15811. https://doi.org/10.1038/ncomms15811
Varty AK, Zedler JB (2008) How waterlogged microsites help an annual plant persist among salt marsh perennials. Estuaries and Coasts: J CERF 31:300–312. https://doi.org/10.1007/s12237-007-9019-2
Watson EB, Byrne R (2009) Abundance and diversity of tidal marsh plants along the salinity gradient of the San Francisco Estuary: implications for global change ecology. Plant Ecology 205:113–128. https://doi.org/10.1007/s11258-009-9602-7
Wilcox DA, Kowalski KP, Hoare HL, Carlson ML, Morgan HN (2008) Cattail Invasion of sedge/grass meadows in Lake Ontario: photointerpretation analysis of sixteen wetlands over five decades. Journal of Great Lakes Research 34:301–323. https://doi.org/10.3394/0380-1330(2008)34[301:CIOGMI]2.0.CO;2
Williams HFL, Hamilton TS (1995) Sedimentary dynamics of an eroding tidal marsh derived from stratigraphic records of 137Cs Fallout, Fraser Delta, British Columbia, Canada. Journal of Coastal Research 11:1145–1156
Williams G, Baumann J, Goldstone R, Kucy V, Lim P, Mather W, Moore K, Murray T (2004) Discovery and Management of Spartina anglica in the Fraser River Estuary, British Columbia, Canada. In: Ayres DR, Kerr DW, Ericson SD, Olofson PR (eds) Proceedings of the Third International Conference on Invasive Spartina. Presented at the International Conference on Invasive Spartina, San Francisco Estuary Invasive Spartina Project of the California State Coastal Conservancy, Oakland, CA, pp 235–242
Woo I, Zedler JB (2002) Can nutrients alone shift a sedge meadow towards dominance by the invasive Typha × glauca. Wetlands 22:509–521. https://doi.org/10.1672/0277-5212(2002)022[0509:CNASAS]2.0.CO;2
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proceedings of the National Academy of Sciences of the United States of America 96:1463–1468. https://doi.org/10.1073/pnas.96.4.1463
Yan J, Zhu Z, Zhou J, Chu X, Sui H, Cui B, Van Der Heide T (2021) Saltmarsh resilience controlled by patch size and plant density of habitat-forming species that trap shells. Science of the Total Environment 778:146119. https://doi.org/10.1016/j.scitotenv.2021.146119
Zedler JB, Kercher S (2004) Causes and consequences of invasive plants in wetlands: opportunities, opportunists, and outcomes. Critical Reviews in Plant Sciences 23:431–452. https://doi.org/10.1080/07352680490514673
Zedler JB, Callaway JC, Sullivan G (2001) Declining biodiversity: why species matter and how their functions might be restored in Californian Tidal Marshes. Bioscience 51:1005–1017. https://doi.org/10.1641/0006-3568(2001)051[1005:DBWSMA]2.0.CO;2
Acknowledgements
This paper presents research from two studies conducted in 2015 and 2021, each differing in their funding sources. The National Wetland Conservation Fund funded 2015 research, with logistical support from the Community Mapping Network and British Columbia Conservation Foundation. Funding for 2021 work came from the British Columbia Wildlife Federation Wetlands Workforce project. This project was supported through the Healthy Watersheds Initiative and delivered by the Real Estate Foundation of B.C. and Watersheds B.C., with financial support from the province of British Columbia. The authors extend thanks to landowners who allowed crews to cross their lands to access the marshes. With gratitude we wish to acknowledge the time and expertise offered by reviewers at various stages of this manuscript.
Funding
This work was funded by the National Wetland Conservation Fund (2015) and the British Columbia Wildlife Federation Wetlands Workforce project (2021). The latter was supported through the Healthy Watersheds Initiative and delivered by the Real Estate Foundation of B.C. and Watersheds B.C., with financial support from the Province of British Columbia. The Ducks Unlimited Canada Institute for Wetland and Waterfowl Research provided analytical and writing support for this project. Nature Force provided open access funding for this article.
Author information
Authors and Affiliations
Contributions
Eric Balke and Daniel Stewart contributed to the study conception and design, building upon the 2015 report that was conceived by Brad Mason, Rob Knight, Megan Lievesley, and Daniel Stewart. Field data were collected by Megan Lievesley and Daniel Stewart (2015) and Robyn Ingham, Daniel Hennigar, and Daniel Stewart (2021). Spatial analyses were conducted by Robyn Ingham, Daniel Hennigar, and Daniel Stewart. Data analysis and modelling was performed by James E Paterson and Daniel Stewart. Figures were created by Daniel Stewart. The first draft of the manuscript was written by Daniel Stewart and Daniel Hennigar, with inputs from Robyn Ingham and Eric Balke. Subsequent edits and input were provided by all authors and collated into a final manuscript by Daniel Stewart. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stewart, D., Lievesley, M., Paterson, J.E. et al. Factors Influencing the Resilience of Created Tidal Marshes in the Fraser River Estuary, British Columbia. Wetlands 44, 53 (2024). https://doi.org/10.1007/s13157-024-01802-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-024-01802-x