Abstract
The hydrological regime determines the structure and diversity of wetland vegetation, and it has been considered as the most relevant determinant for the plant communities in the Paraná River Delta. However, experimental validations explaining the causes of the distribution patterns are scarce. The objective of this article is to determine the effect of flood duration on wetland species germination. For that purpose, the seeds of four species from contrasting hydrogeomorphological environments, three ligneous species and one herbaceous species, were sowed. The 24 germination trays were watered weekly, and undergone four flood duration treatments: zero, one, seven and 13-day flood period every two weeks. We found that the longer the flood duration, the greater the reduction on germination for ligneous species, and the opposite was observed for the herbaceous species. The odds of germination in the longer duration treatment, versus control treatment, can be reduced up to 99% for ligneous species, and can be increased 900% for herbaceous species. Germination in the studied species performed as expected in their natural environments, although the ligneous species showed a broader niche, indicating that other mechanisms are also necessary to understand the distribution of these species. The results provide experimental evidence that flood duration impacts on germination and species composition of wetland species and provide tools for the prediction of anthropic impacts and the ecological restoration of wetlands.
Resumen
El régimen hidrológico determina la estructura y diversidad de la vegetación de humedal y ha sido reconocido como el condicionante más relevante de las comunidades de plantas del delta del río Paraná. Sin embargo, existe una escasez de validaciones experimentales que permitan explicar las causas de los patrones de distribución encontrados. El objetivo del trabajo fue analizar el efecto de la duración de la inundación sobre la germinación de especies de humedal. Para ello se sembraron semillas de cuatro especies, tres leñosas y una herbácea, características de ambientes hidrogeomorfológicos contrastantes. Las 24 bandejas de germinación fueron regadas semanalmente y se sometieron a cuatro tratamientos de duración de inundación: cero, uno, siete y 13 días de inundación cada dos semanas. Se observó una disminución de la germinación a mayor duración de inundación para las especies leñosas y lo contrario para la herbácea. El odds de germinar en el tratamiento de mayor duración, respecto al del control, puede disminuir hasta 99% para las leñosas y aumentar 900% para la herbácea. La germinación de las especies estudiadas respondió según lo esperado a los ambientes donde se desarrollan naturalmente, aunque las especies leñosas mostraron un nicho más amplio indicando que otros mecanismos son también necesarios para entender la distribución de estas especies. Los resultados aportan evidencias experimentales a favor de que la duración de la inundación impacta en la germinación y composición de especies de humedal y aportan herramientas para la predicción de impactos antrópicos y la restauración ecológica de humedales.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The hydrological regime influences the structure and diversity of wetland vegetation, even in the finest spatial scales (Raulings et al. 2010). Duration, frequency, and depth of flooding have been identified as major determinants for the type of plant community that emerges and establishes in a place (Casanova and Brock 2000). In riparian environments, flooding may provide new alluvium for germination, destroy pre-existing vegetation, determine whether seedlings will survive to maturity, and disperse propagules to colonization sites (Bendix and Hupp 2000). In effect, the selection pressure produced by the hydrological regime in the establishment of plants from seed entails evident ecological and evolutionary consequences (Leck and Brock 2000). Flooding produces anaerobiosis, and while some species were documented as able to germinate in such conditions, oxygen is fundamental for plant germination (Zhou et al. 2020). While the seed bank could be a factor in controlling the abundances of species to emerge, the hydrological regime determines which species would become established by stimulating or suppressing the germination of the diverse species (Webb et al. 2006).
The hydrological regime has been recognized as the most relevant determining factor for the Paraná River Delta wetland macromosaic vegetation. In his original description of the vegetal communities, Burkart (1957) noted that the factor impacting vegetation during river flooding is not so much water levels as the flood duration, and the presence or absence of stagnant flood water. Also, Malvárez (1997) concluded that inundability, as well as the origin of the flooding, are the main factors in structuring communities and region species distribution, and in the cases in which anaerobiosis conditions are prevalent, tolerant species are reduced to a few, creating virtually monospecific communities. Accordingly, Kandus et al. (2003) stated that species differential presence corresponds to two main gradients of variance: water permanence and the water source and its hydrodynamics.
The results obtained by Kalesnik et al. (2011) also show that the variables related to the hydrological regime, along with the modifications of landscape elements, could allow to explain vegetation patterns of levee forests, and even invasion patterns by invasive tree species. More recently, Morandeira and Kandus (2016) found a significant association among the effects of flooding frequency, topographic position, and landscape units on herbaceous species functional types. Nonetheless, so far, these regional patterns have not been subject to any experimental validation that could explain, at least partially, its causes.
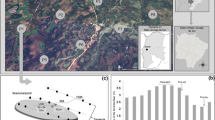
The hydrological regime from the Paraná River Delta region is subject to a main West–East gradient, with a decreasing influence from the Paraná River and a growing influence of the Río de la Plata River; in short, an axis of “fluvial-tidal” influence (Malvárez 1997; Durante and Di Bella 2020). The fluvial influence is defined by the seasonal floods of the Paraná and Uruguay rivers, and the tidal influence, by the daily tides from the Río de la Plata River, which could be significantly increased by wind circulation (Baigún et al. 2008). Due to these variations, the topographic position and the form of deltaic islands determine the frequency and duration of the flooding to which the different species are subject, shaping a prominent environmental heterogeneity with a characteristic and distinctive vegetation (Kandus et al. 1999, 2006). Among the species that characterize the hydrogeomorphological environments of the Lower Paraná River Delta are Terminalia australis Cambess. (Combretaceae), a tree from the levee forests; Erythrina crista-galli L. (Fabaceae), an arboreal species in intermediate topographic positions; Sesbania virgata (Cav.) Pers. (Fabaceae), a bush in environments lower than levees, and adjacent to creeks; and Schoenoplectus californicus (C.A.Mey.) Soják (Cyperaceae), an equisetoid herbaceous plant in lower topographic positions (Fig. 1).
The objective of this study was to analyze the impact of flood duration on wetland species germination. We hypothetize that the flood regime impacts the germination in various species and its response corresponds with the hydrogeomorphological environments they inhabit. Therefore, the prediction was that germinations would be maximum under the following conditions: under short flood durations for T. australis, under intermediate durations for E. crista-galli and S. virgata, and under prolonged durations for S. californicus.
Methods
The four species under study were selected because they are characteristic of contrasting hydrogeomorphological environments and because they have a hydrocorous dispersion (Kandus et al. 2006, Kalesnik and Sirolli 2011). All of them reproduce by seeds although S. californicus can also multiply by division of rhizomes (Quintana et al. 2012). Although they can be recorded in almost the entire Paraná River Delta, T. australis, E. crista-galli and S. californicus are more abundant in the Lower Paraná River Delta and S. virgata in the Upper Paraná River Delta (Burkart 1957).
The fruits of the species under study were collected between March and June of 2018, in the creeks from the Third Section of Paraná River Delta islands (central latitude and longitude coordinates: -34.10, -58.47). The islands of this sector, the Lower Paraná River Delta, are characterized by being basin-shaped and presenting a high edge called “albardón” (levee), only reached by large and brief floods, and a low and swampy interior (Burkart 1957). The fruits were stored in paper envelopes, in a dark storage room at room temperature and humidity until the beginning of the experiment. Before planting, the seeds of E. crista-galli, S. virgata y T. australis were mechanically scarified to break the dormancy (Silva et al. 2006; de Menezes Silva et al. 2011). The germination was carried out on 24 multi-cell plastic trays. Each cell was perforated at its base, and their dimensions were a diameter of 4 cm on the upper part and a depth of 6 cm. Each one of them was filled with 4 cm of sand, and assigned to a species regularly and alternately, resulting in 19 cells per species and tray. The seeds were sowed in the sandy substratum at 1 cm depth, two per cell for S. californicus (N = 38 seeds per tray) and one per cell for the rest of the species (N = 19 seeds per tray).
The flood treatments were generated in the following way: the multi-cell trays were inserted in buckets of 30 by 45 by 10 cm with adjustable drain. The buckets were arranged even over a table in a greenhouse in the experimental campus of Ciudad Universitaria, belonging to the University of Buenos Aires (Argentina). Once every two weeks, the buckets were filled with water until the water level surpassed the sand level in 0.5 cm. The buckets were drained based on the following scheme: “Zero day” treatment (T0), without flooding; “One day” treatment (T1), draining after 24 h of flooding; “Seven day” treatment (T7), draining after 7 days of flooding; “13 day” treatment (T13), draining after 13 days of flooding. Every tray was watered weekly until reaching the soil field capacity.
The experiment was carried out between September 5th and December 20th, 2018. The seeds from the four species were sowed at the same time. The first germinating species, E. crista-galli y S. virgata, reached stabilization in the curve of germination accumulation after six weeks; for that reason, it was decided to establish the duration of the experiment in 6 weeks after the first germination for all the species. The environmental conditions of light, temperature and humidity during the experiment were natural, with absolute minimum, average and absolute maximum for September to December months being 7.0, 19.4 and 31.6 °C respectively. The buckets were rotated on a weekly basis to homogenize the conditions for all experimental units. Data from all cells was logged weekly, and seeds were counted as germinated only if the emergence of any part of the seedling was observed. Upon completion of the experiment, the seedlings were removed, dried in a heater at 50 °C for 24 h, and weighted in an analytical balance. For each tray, the dry weight per seedling at the end of the experiment was the sum of the dry weight of all the emerged seedlings divided by the total number of emerged seedlings in each tray. For each germinated seedling, the germination time was the difference between the germination and sowing dates. The germination time per tray was the average germination time of all the germinated seedlings in each tray.
For the statistical analysis, it was assumed that the Proportion of germinated seed response variable (number of germinated seeds in relation to the total number of sowed seeds per tray), followed a binomial distribution, and it was modeled with generalized linear models using the glm package from the software RStudio (R Core Team 2021). The explanatory variables were Species and Flood duration treatment. The latter was treated as a qualitative variable (four levels: T0, T1, T7, and T13) since the models that took the variable as a quantitative variable did not fulfill the linearity in the response. The assumption of the absence of underdispersion or overdispersion was analyzed with the DHARMa package (Florian Hartig 2021). The overdispersion was significant, consequently, the data was modeled taking the germination tray as a random variable using the glmmTMB package (Brooks et al. 2017). A lack of significant normality was not detected in the residues for said variable, neither when they were graphically analyzed (qqplot) nor with a Shapiro–Wilk test. The model with interaction between species and treatment showed a lower AIC value than the model without interaction and those models considering only one of the two variables. Post-hoc Tukey contrast tests were made using the emmeans package (Lenth 2021). Regarding the Dry weight per seedling variable at the end of the experiment, a normal distribution was assumed, and it was modeled using linear models with the lm package from the aforementioned software. Significant lack of normality and lack of homoscedasticity were detected, consequently the data was modeled with the gls function from the nlme package (Pinheiro et al. 2022). Variance modeling function varIdent showed a lower AIC value than the other two functions analyzed (varPower and varExp). Post-hoc Tukey contrast tests were made using the same package. The significance threshold for all the tests was 0.05.
Results
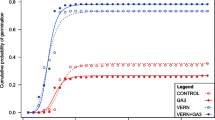
The cumulative number of germinated seeds over time is shown in Fig. 2. The effect of flood duration differed based on the species, showing a significant interaction between these two variables (flood duration and species, p < 0.05, type II Wald chi-square). The greater the flood duration, the greater the germination decrease for ligneous species; and the opposite was observed for the herbaceous species (Fig. 3). Analyzing the simple effects, flood duration impacted the germination of the four wetland species under study (p < 0.05, Tukey). On the contrary, only one significant effect of this variable was observed at the end of the study on the dry weight per seedling in one of the species (E. crista-galli, p < 0.05, Fisher). The time the seeds needed to germinate was (average ± standard deviation) 70 ± 8, 17 ± 7, 17 ± 7 y 56 ± 11 days respectively for T. australis, E. crista-galli, S. virgata, and S. californicus.
Proportion of germinated seeds over time in the four studied wetland species. The four flood duration treatments of this study are indicated in days every two weeks, using different series. The time axis covers six weeks starting at the first germinations, which occurred on different dates for each species. N = 228 sowed seeds per treatment for Schoenoplectus californicus, and 114 for the other three species. Y-axis maximum value is variable for a better visualization
Proportion of germinated seeds based on the flood duration treatment, in days every two weeks, at the end of the study. The observed values per tray are indicated with grey circles, the predicted values are indicated with black squares, and their confidence interval is 95%, based on a generalized linear model. N = 6 trays per treatment. The different letters indicate significant differences among treatments for each species using post hoc Tukey comparisons. Y-axis maximum value is variable for a better visualization
For the species T. australis, 89 seeds germinated, which represents a germination success rate of 20% compared to sowed seeds (Fig. 2). The germination was the highest when they received zero and one day-flood treatment every two weeks and it dropped significatively in the other two more prolonged flood times (p < 0.05, Tukey) (Fig. 3). The model indicates that the probability of germinating versus the probability of not germinating (odds) with a 13-day flood regime every two weeks (T13) drops, in average, 99% compared to the non-flood regime (T0) (Table 1). The dry weight per seedling at the end of the study ranged from 16 to 27 mg, without significant differences among treatments (p > 0.05, Fisher).
The germination success rate of Erythrina crista-galli was 52%, considering the four treatments applied (238 germinated seeds). The germination was the highest in the non-flood treatment, without differences for one and seven day-flood treatments, and the lowest in the more prolonged flooding (p < 0.05, Tukey, Fig. 3). The odds of germination with a regime like T13 drops in average 95% compared to T0 (Table 1). Mean dry weight per seedling at the time of the extraction was 1632, 2510, 1973 and 348 mg respectively for T0, T1, T7 and T13, with significant differences between the longer flooding time and the rest (p < 0.05, Tukey).
For the species Sesbania virgata, a total of 252 seeds germinated, that is, a germination success rate of 55%. A reduction in the germination on longer flood durations was observed, with rates ranging from 78 to 9% (p < 0.05, Tukey, Fig. 3). The model indicates that the odds of germination with a very long flood regime (T13) can decrease, in average, up to 97% compared to the non-flood regime (T0) (Table 1). The dry weight per seedling at the end of the study ranged between 29 and 44 mg without differences among treatments (p > 0.05, Fisher).
The seeds of Schoenoplectus californicus had a germination success rate of 4% (37 seeds). In contrast to the other three species, the germination increased significatively with a longer flood duration, with rates ranging from 1 to 9% (p < 0.05, Tukey, Fig. 3). With the 13-day flood regime every two weeks, the odds of germination increase, in average, 900% compared to the non-flood regime (Table 1). The dry weight per seedling at the extraction ranged between 1 and 2 mg, without differences among treatments (p > 0.05, Fisher).
Discussion
The flood regime had an impact by decreasing or increasing the percentage of germination differentially according to the species. Consequently, this process allows to explain, in part, the vegetation patterns from Paraná River Delta (Burkart 1957). Particularly, in the conditions of the study, without a water deficit due to supplementary irrigation, the longer the duration of the flooding, the biggest inhibitory effect in the germination percentage of ligneous species, and the opposite was true for the herbaceous species. This pattern experimentally underpinned the descriptions by Malvárez (1997), who stated that if the flooding period extends in time, the water in the soil brings to prolonged saturation conditions and a reduced aeration, causing a reduction in the number of tolerant species and in the complexity of communities. In seed bank studies subject to prolonged flooding, lowered community richness dominated by herbaceous species have been reported (Casanova and Brock 2000). Herbaceous species not only show a higher germination rate than ligneous species under longer durations of flooding, but also have better asexual reproduction mechanisms, straightening the predominance of herbaceous dominant communities with little or no representation of ligneous species (Barrett et al. 2010).
Under flooded conditions, most of the seeds absorb water and their coats break, benefiting the germination, although, by contrast, continued soaking results in seed death (Cronk and Fennessy 2001). In this study, water absorption was facilitated by the scarification made to the ligneous species seeds and it wasn’t a limiting factor, as it could occur in nature (Baskin and Baskin 2014). Additionally, this fact would allow to explain the better recruitment of the herbaceous species under more prolonged flooded conditions, since said species hadn’t been scarified. Accordingly, the results of this study agree with Stone et al. (2020), who found differential seedling emergence among trees, shrubs, and herbaceous species under different flood durations.
Once germinated, the dry weight of each seedling at the end of the study wasn’t significatively impacted by flood duration. Various studies on seed banks have found dry weight reductions under a longer flood duration, considering the sum of germinated seedlings (Casanova and Brock 2000). In agreement, experiments about wetland seedling survival have also shown dry weight reductions under flooded conditions (Fraser and Karnezis 2005). Consequently, the results of this study indicate that, in a context without water deficit, the dry weight developed in the first two months post-germination would be determined by the number of germinating and surviving seedlings rather than by a differential growth depending on flood duration. Therefore, the results show that the determination of dry weight found by weighing the dry mass of all individuals, even though less exact than seedling count, is suitable for comparing treatments like those applied to this study.
In three out of the four studied species, experiments have been conducted to assess their response to flooding, and it has been concluded that they are plants with a high tolerance to flooding. In these conditions, E. crista-galli develops a metabolism with a high level of tolerance to flooded conditions, activating the fermentation pathway and increasing the activity of antioxidant enzymes (Ferreira Larré et al. 2016). Likewise, S. virgata can survive in flooded soils for more than 56 days, developing metabolic and anatomic adaptations, such as the increase of intercellular space, defoliation, and the drop of biomass (Davanso-Fabro et al. 1998; Zanandrea et al. 2009). Our results show that these two species germinate to a significant extent, even under flooded conditions during half of the time. Besides, S. californicus has been broadly reported as a tolerant species to flooding, even when compared to other common herbaceous species in wetland ecosystems (Sloey et al. 2015, 2016). Our results show that these adaptations to flooding include early stages of development, such as germination, and they allow us to understand that flooding is an essential factor in the installation of this species on new substrates. Finally, germination studies for T. australis weren’t found, however, our results show that even though it is tolerant to brief flooding periods, its germination declines fast when the flood is prolonged.
The germination of the species under study responds as expected to the flooding duration, if the hydrological regimes associated with the natural topographic positions are considered (Burkart 1957; Malvárez 1997; Kandus et al. 2003; Kalesnik et al. 2011; Sirolli et al. 2021). Nevertheless, in this study it was observed that the ligneous species have a niche that extends to flood values lower and higher than expected, according to their realized niche (Hutchinson 1957). Consequently, as usually happens, other mechanisms such as competence, available nutrients, water quality, sediment texture, etc. are needed to understand the distribution of these species in the studied area. For example, this study results show that E. crista-galli, a species forming monospecific forests in floodable soils, could thrive in topographically higher environments, like the levees where T. australis and other tree species grow, but its abundance is low in the levees, and its population structure pattern is nomadic or remnant (Sirolli et al. 2021). On the contrary, S. californicus is always under flooded conditions, and it is not a part of land communities. The results of this study reveal that this selection occurs from the plant germination.
In a context of lack of knowledge regarding hydrological regime and wetland plants, this study provides experimental evidence supporting flood duration impact on germination and, in consequence, on wetland species composition just as Webb et al. (2012) have stated. It is important to note that this study was limited to a flooding intensity of a few centimeters and to a biweekly flooding frequency; by modifying these variables, the impact of duration could be different, since the three variables act simultaneously (Mitsch and Gosselink 2015). Therefore, the decrease in germination shown in this study with the increase of flood duration should be analyzed in a context of frequent flooding, typical of Paraná River Delta front and other wetlands affected by lunar and/or wind tides. Finally, the results of this study help to predict the impact of wetland loss, or the changes in wetland hydrological regime, on the vegetation, and serve as tools for the management and the ecological restoration of these environments (look deeper into these issues at, for example, Davidson 2014; Sica et al. 2016; Finlayson et al. 2018; An and Verhoeven 2019).
Data Availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
An S, Verhoeven JTA ed. (2019) Wetlands: Ecosystem Services, Restoration and Wise Use. Vol. 238. Ecological Studies. Springer International Publishing Cham https://doi.org/10.1007/978-3-030-14861-4
Baigún CRM, Puig A, Minotti PG, Kandus P, Quintana R, Vicari R, Bo R, Oldani NO, Nestler JA (2008) Resource use in the Paraná River Delta (Argentina): moving away from an ecohydrological approach? Ecohydrology and Hydrobiology 8:245–262. https://doi.org/10.2478/v10104-009-0019-7
Barrett R, Nielsen DL, Croome R (2010) Associations between the plant communities of floodplain wetlands, water regime and wetland type. River Research and Applications 26:866–876. https://doi.org/10.1002/rra.1299
Baskin CC, Baskin JM (2014) Seeds: ecology, biogeography, and evolution of dormancy and germination, 2nd edn. Elsevier/AP, San Diego, CA
Bendix J, Hupp CR (2000) Hydrological and geomorphological impacts on riparian plant communities. Hydrological Processes 14:2977–2990. https://doi.org/10.1002/1099-1085(200011/12)14:16/17%3c2977::AID-HYP130%3e3.0.CO;2-4
Brooks ME, Kristensen K, Koen J, van Benthem A, Magnusson CW, Berg AN, Skaug HJ, Maechler M, Bolker BM (2017) glmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. The R Journal 9(2):378–400
Burkart A (1957) Ojeada sinóptica sobre la vegetación del Delta del Río Paraná. Darwiniana 11:457–561
Casanova MT, Brock MA (2000) How do depth, duration and frequency of flooding influence the establishment of wetland plant communities? Plant Ecology 147:237–250
Cronk JK, Fennessy MS (2001) Wetland plants: biology and ecology. Water Science, Technology and Engineering. Lewis Publ., Boca Raton, Fla
Davanso-Fabro VM, Medri ME, Bianchini E, Pimenta JA, (1998) Tolerância à inundação: aspectos da anatomia ecológica e do desenvolvimento de Sesbania virgata (Cav.) Pers.(Fabaceae). Brazilian Archives of Biology and Technology 41:475–482 SciELO, Brasil
Davidson NC (2014) How much wetland has the world lost? Long-term and recent trends in global wetland area. Marine and Freshwater Research 65:934–941
Durante M, Di Bella CM (2020) A MODIS based tool to assess inundation patterns: an example for the Paraná Delta River. RIA. Revista de Investigaciones Agropecuarias 46(1):96–107. SciELO, Argentina
Ferreira Larré C, Leivas Moraes C, Borella J, do Amarante L, Deune S, Peters JA (2016) Antioxidant activity and fermentative metabolism in the plant Erythrina crista-galli L. under flood conditions. Semina: Ciências Agrárias 37:567–580 Universidade Estadual de Londrina
Finlayson CM, Everard M, Irvine K, McInnes RJ, Middleton BA, van Dam AA, Davidson NC (eds) (2018) The wetland book: I: structure and function, management and methods. Springer Berlin Heidelberg, New York. https://doi.org/10.1007/978-90-481-9659-3
Florian Hartig (2021) DHARMa: Residual Diagnostics for Hierarchical (Multi-Level / Mixed) Regression Models. R package version 0.4.4. https://CRAN.R-project.org/package=DHARMa
Fraser LH, Karnezis JP (2005) A comparative assessment of seedling survival and biomass accumulation for fourteen wetland plant species grown under minor water-depth differences. Wetlands 25:520–530. https://doi.org/10.1672/0277-5212(2005)025[0520:ACAOSS]2.0.CO;2
Hutchinson GE (1957) Concluding Remarks. Cold Spring Harbor Symposia on Quantitative Biology 22:415–427. https://doi.org/10.1101/SQB.1957.022.01.039
Kalesnik FA, Aceñolaza P, Hurtado M, Martínez J (2011) Relationship between vegetation of the levee neo-ecosystems and environmental heterogeneity in the Lower Delta of the Paraná River, Argentina. Water and Environment Journal 25:88–98. https://doi.org/10.1111/j.1747-6593.2009.00196.x
Kalesnik FA, Sirolli H (2011) La vegetación del Bajo Delta Insular del Paraná. Diversidad, situación y uso. In: Quintana RD, Villar MV, Astrada E, et al. (eds) El patrimonio natural y cultural del Bajo Delta Insular. Bases para su conservación y uso sustentable, 1st edn. Aprendelta, Buenos Aires, pp 88–99
Kandus P, Karszenbaum H, Frulla L (1999) Land cover classification system for the Lower Delta of the Paraná River (Argentina): Its relationship with Landsat Thematic Mapper spectral classes. J Coast Res 15(4):909–926
Kandus Patricia, Malvárez AI, Madanes N (2003) Estudio de las comunidades de plantas herbáceas de las islas bonaerenses del Bajo Delta del Río Paraná (Argentina). Darwiniana, Nueva Serie 41:1–16 https://doi.org/10.14522/darwiniana.2014.411-4.201
Kandus P, Quintana RD, Bó RF (2006) Patrones de paisaje y biodiversidad del Bajo Delta del Río Paraná. Mapa de ambientes, 1st edn. Pablo Casamajor Ediciones, Buenos Aires
Leck MA, Brock MA (2000) Ecological and evolutionary trends in wetlands: Evidence from seeds and seed banks in New South Wales, Australia and New Jersey, USA. Plant Species Biology 15:97–112. https://doi.org/10.1046/j.1442-1984.2000.00031.x
Lenth, RV (2021) emmeans: Estimated Marginal Means, aka Least-Squares Means. R package version 1.6.0. https://CRAN.R-project.org/package=emmeans
Malvárez AI (1997) Las comunidades vegetales del Delta del Río Paraná. Su relación con factores ambientales y patrones de paisaje. Tesis de Doctorado. Universidad de Buenos Aires. Facultad de Ciencias Exactas y Naturales. Tesis de Doctorado, Buenos Aires: Universidad de Buenos Aires. Facultad de Ciencias Exactas y Naturales
de Menezes Silva PE, Santiago EF, de Menezes Daloso D, Marques da Silva E, Oliveira Silva J (2011) Quebra de dormência em sementes de Sesbania virgata (Cav.) Pers. Idesia (arica) 29(2):39–45. https://doi.org/10.4067/S0718-34292011000200005
Mitsch WJ, Gosselink JG (2015) Wetlands, 5th edn. John Wiley and Sons Inc., Hoboken, NJ
Morandeira NS, Kandus P (2016) Plant functional types and trait values in the Paraná River floodplain: Modelling their association with environmental features. Flora-Morphology, Distribution, Functional Ecology of Plants 220:63–73
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2022) _nlme: Linear and Nonlinear Mixed Effects Models_. R package version 3.1–155, <URL: https://CRAN.R-project.org/package=nlme>
Quintana RD, Malzof S, Villar MV et al (2012) Plantas, animales y hongos de las islas: una introducción a la biodiversidad del Bajo Delta del Río Paraná, 1st edn. Aprendelta, Buenos Aires
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Raulings EJ, Morris K, Roache MC, Boon PI (2010) The importance of water regimes operating at small spatial scales for the diversity and structure of wetland vegetation. Freshwater Biology 55:701–715. https://doi.org/10.1111/j.1365-2427.2009.02311.x
Sica YV, Quintana RD, Radeloff VC, Gavier-Pizarro GI (2016) Wetland loss due to land use change in the Lower Paraná River Delta, Argentina. Science of The Total Environment 568:967–978. https://doi.org/10.1016/j.scitotenv.2016.04.200
Silva AJC, Carpanezzi AA, Lavoranti OJ (2006) Quebra de Dormência de Sementes de Erythrina crista-galli. Pesquisa Florestal Brasileira 53:65–65
Sirolli H, Torrella S, Kalesnik FA (2021) Composición y estructura de los bosques de albardón del delta frontal del río Paraná, Argentina. Revista Mexicana de Biodiversidad 92:923558 https://doi.org/10.22201/ib.20078706e.2021.92.3558
Sloey TM, Howard RJ, Hester MW (2016) Response of Schoenoplectus acutus and Schoenoplectus californicus at different life-history stages to hydrologic regime. Wetlands 36:37–46 Springer
Sloey TM, Willis JM, Hester MW (2015) Hydrologic and edaphic constraints on Schoenoplectus acutus, Schoenoplectus californicus, and Typha latifolia in tidal marsh restoration. Restoration Ecology 23:430–438 Wiley Online Library
Stone L, Fryirs K, Leishman M (2020) Simulating the effect of environmental flow duration on seedling emergence from riparian seed banks of the Upper Hunter River, New South Wales. River Research and Applications 36:607–619. https://doi.org/10.1002/rra.3376
Webb JA, Wallis EM, Stewardson MJ (2012) A systematic review of published evidence linking wetland plants to water regime components. Aquatic Botany 103:1–14. https://doi.org/10.1016/j.aquabot.2012.06.003
Webb M, Reid M, Capon S, Thoms M, Rayburg S, James C (2006) Are flood plain–wetland plant communities determined by seed bank composition or inundation periods? In: Rowan JS, Duck RW, Werritty A (eds) Sediment Dynamics and the Hydromorphology of Fluvial Systems (Proceedings of a symposium held in Dundee, UK, July 2006), 241–248. 306. IAHS Publications
Zanandrea I, Alves JD, Deuner S, de F. P. Goulart P, de C. Henrique P, Silveira NM (2009) Tolerance of Sesbania virgata plants to flooding. Australian Journal of Botany 57:661https://doi.org/10.1071/BT09144
Zhou W, Chen F, Meng Y, Chandrasekaran U, Luo X, Yang W, Shu K (2020) Plant waterlogging/flooding stress responses: From seed germination to maturation. Plant Physiology and Biochemistry 148:228–236. https://doi.org/10.1016/j.plaphy.2020.01.020
Acknowledgements
The authors express their gratitude to Dr. Natalia Morandeira and an anonymous reviewer for their valuable comments on the manuscript. Funding: PIP 11220150100404CO Grant, Consejo Nacional de Investigaciones Científicas y Técnicas (Argentina), Amela García MT, Kalesnik FA and Aceñolaza P, Directors.
Funding
This work was supported by PIP 11220150100404CO Grant, Consejo Nacional de Investigaciones Científicas y Técnicas (Argentina), Amela García MT, Kalesnik FA and Aceñolaza P, Directors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Horacio Sirolli, Andriy Bazylenko and Mariano Ernesto Ramello. The first draft of the manuscript was written by Horacio Sirolli and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sirolli, H., Bazylenko, A. & Ramello, M.E. Impact of Flood Duration on Germination Success of Paraná River Delta (Argentina) Plants. Wetlands 42, 77 (2022). https://doi.org/10.1007/s13157-022-01599-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-022-01599-7