Abstract
The impacts of different anthropogenic disturbances on the composition and metabolic function of sediment bacterial community in the largest freshwater lake- Poyang Lake of China were studied. A total of 12 surface sediment samples were collected from four sites, including an undisturbed central lake region (SMS) and three disturbed sites in fishery (NJS), industrial (RH) and tourism (BSZ) areas. Sediment physicochemical properties and heavy metal contents were combined with the results from Illumina Miseq and Biolog-Ecoplates to quantify the impact of environmental factors driving differences in sediment bacterial structure and microbial function. Our results showed that the bacterial community diversity (Shannon index) was higher in disturbed than in undisturbed areas, although the most predominant bacterial phyla in four studied sites were similar. However, the average well color development (AWCD) based on carbon source utilization pattern of the disturbed area was lower than undisturbed areas and the preference of carbon sources differed among the four studied areas. Site contaminants contributed to the distribution of distinct phyla which differentiated bacterial communities among sites. For example, the contents of TN (1.80 g/kg) and TOC (12.34 g/kg) in NJS showed positive correlation with the abundance of Deltaproteobacteria and Chloroflexi; while the concentration of TP (0.43 g/kg), Cu (69.22 mg/kg) and Zn (134.89 mg/kg) in RH was positively correlated with the predominance of Firmicutes and Verrucomicrobia. These results indicated that microbial community carbon metabolic profiles changed with the shift of bacterial community structure, and disturbance-induced pollution enhanced the sediment bacterial diversity and reduced carbon metabolic activity.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Freshwater lakes are important source of domestic water and aquaculture, and also offer important habitat for global biodiversity (Dudgeon et al. 2006; Kumar et al. 2016). Rapid population growth and intensive human activities are contributing to an increase in heavy pollution in many freshwater lakes and making water supply and fish no longer safe (Azizullah et al. 2011). Most pollutants are deposited in lake sediment which often acts as a secondary pollution source (Kim 2003; Haddadchi et al. 2013; Heim and Schwarzbauer 2015). Microorganisms are ubiquitous and play key roles in decomposing, transforming organic matters and remineralizing nutrients (Burkhardt et al. 2014; Landa et al. 2014; Gad et al. 2015). Thus the composition and performance of the sediment microbial community has attracted much attention, and many studies have focused on the relationship between lake pollution and sediment microbes (Smorczewski and Schmidt 1991; Bennett and Metcalfe 1998; Konstantinidis et al. 2003; Devarajan et al. 2015). Previous studies on Dianchi Lake and 13 lakes in Yunnan Plateau suggested that the bacterial abundance was regulated by sediment total organic carbon (TOC) and total nitrogen (TN) (Bai et al. 2012; Zhang et al. 2015). Total phosphorus (TP), copper (Cu), and cadmium (Cd) in sediment were considered to be key factors driving spatial variation of bacterial community in Poyang Lake (Yu et al. 2015). A study demonstrated that the highest diversity of bacterial community appeared in moderately polluted samples, and the abundance of Alphaproteobacteria was increased with the level of heavy metals (Zhu et al. 2013a). These studies demonstrated the influence of sediment properties in shaping microbial community composition in freshwater lakes. However, most studies as mentioned above focused only on heavy metals or nutrient parameters, while contaminated aqueous environments are always complex in which multiple natural terrestrial sources (e.g. leaching, erosion) and anthropogenic origin (e.g. mining, sewage, smelting waste) lead to multi-source pollution (Yuan et al. 2014). Even though these results from previous studies focused on community structures rather than metabolic function, our knowledge about how the sediment bacterial community composition (BCC) respond to multiple source contamination is still far from comprehensive. Because these studies focused on community composition and not metabolic activity, we also have a poor understanding of how environmental factors drive sediment microbial function under different anthropogenic disturbances of lake ecosystems.
Modern biotechnology provides powerful tools for microbial study, making it is easier to conduct more comprehensive research on sediment-associated microorganisms. Molecular methods such as DGGE (Kowalchuk et al. 1998), PLFA (Macalady et al. 2000) and T-RFLP (Paissé et al. 2008), have increased the information availability for bacterial community composition comparing with cultural-dependent methods. However, these techniques have some notable disadvantages: limited amount of data and limited accuracy of species identification (Kircher and Kelso 2010). The latest sequencing method, Illumina Miseq high-throughput sequencing used in this study is more efficient for identifying the complicated profile of microbial community and providing greater throughput (Fang et al. 2015; Li et al. 2015). In addition, the Biolog-Ecoplates, which are suitable for microbial community level physiological profiling, have been widely used in assessing the microbial functional diversity of various environments (Tian et al. 2015).
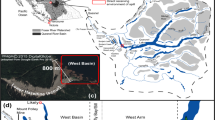
Poyang Lake, the biggest freshwater lake in China, is critical to maintain the aquatic biodiversity and drinking water resource, and also plays a vital role in flood regulation (Wang et al. 2014a). Due to development and excessive utilization of Poyang Lake, different types of pollution were frequently deposited in different regions. A large area of aquaculture activity is found in the Nanjishan (NJS) area, partly separated from the main lake basin in dry seasons. In this region, bait, dead algae, and fish excreta lead to considerable organic pollution in sediment (Hu et al. 2005; Kou et al. 2016). The Raohe (RH) area has been polluted by heavy metals and TP which are attributed to industrial wastewater discharge from upper reaches of the Dexing copper and phosphate mines (He et al. 1998; Yuan et al. 2011; Gao et al. 2014). The Baishazhou (BSZ) region is located near a national wetland park and the contamination mainly comes from tourism. The Songmenshan (SMS) area is in the main water channel of Poyang Lake far away from the shore and less disturbed by anthropogenic activities. In this study, we investigated the variations of sediment bacterial community structure and function in response to different anthropogenic disturbances using Illumina Miseq sequencing and Biolog-Ecoplates. We hypothesized that: (1) different types of pollution associated with different anthropogenic disturbances caused variation in bacterial community structure; and (2) the microbial metabolic function changed and was correlated with the community structure variations.
Materials and Methods
Study Sites and Sample Collection
Surface sediment samples (0-5 cm) were collected in April, 2014 from four selected regions of Poyang Lake (Fig. 1): Songmenshan (SMS, 29°12′26.9″N, 116°11′28″E), Nanjishan (NJS, 28°55′0.1″N, 116°16′44.4″E), Raohe (RH, 29°0′39″N, 116°27′42″E) and Baishazhou (BSZ, 29°10′34″N, 116°37′14″E). Triplicate surface sediment samples were collected at each region using a grab sampler to obtain a total of 12 samples. Samples were temporarily stored in sterile polyethylene ziplock bags, and immediately put on ice and transported to the laboratory. Sediment samples were divided into two aliquots. One aliquot was stored at 4 °C for the measurement of sediment physicochemical parameters and Biolog-Ecoplates; the other aliquot was stored in sterile polypropylene tubes at −80 °C for molecular analysis.
Measurement of Sediment Physicochemical Properties
Sediment pH was detected by testing the sediment paste containing 3 g sediment homogenized in 21 ml distilled waters and using a FE20K pH meter (Wang et al. 2014b). Sediment samples were dried in an oven at 100 °C overnight, homogenized and grounded to fine powder and sieved mechanically using a 0.25 mesh for the following analysis. Sediment ash-free-dry-mass (AFDM) was obtained through calcinations in a BF51800 muffle furnace (Thermal) at 550 °C for 4 h (Sponseller and Fisher 2008). Total nitrogen (TN), total organic carbon (TOC) and total phosphorus (TP) were analyzed using the microkjeldahl method (Lemon et al. 1992), the Walkley-Black wet oxidation procedure (Schumacher 2002) and vanadium-molybdenum-yellow photometric method (Yang et al. 2012), respectively. The concentrations of heavy metals including copper (Cu), zinc (Zn), lead (Pb) and cadmium (Cd) were quantified with the digested samples using AA800 atomic absorption spectrophotometer (Zhao et al. 2010).
DNA Extraction and High-Throughput Sequencing
Microbial genomic DNA was extracted from 0.3 g sediment (wet-weight) using a Power Soil DNA Isolation kit (MoBio) following the manufacturer’s instructions. The quality of extracted DNA was checked in a NanoDrop 2000. Afterwards, extracted DNA samples were sent to BGI Co. Ltd. for Illumina Miseq sequencing of bacterial community. PCR amplicon libraries were constructed with the primers 515F and 806R (Bergmann et al. 2011) that targeted the V4 hypervariable regions of bacterial 16S rRNA genes. Obtained raw sequence reads were processed using the Usearch 3.0 and Python software package for quality control and species annotation. Singleton and chimeric sequences were removed in this step. The remaining sequence reads were clustered into operational taxonomic units (OTUs) at 3% difference, some archaea sequences were abandoned in further analyses. The sequence data generated in this study was deposited in the NCBI Sequence Read Archive and are available under the project number SPR068335.
Biolog-Ecoplates Analysis
The Biolog-EcoPlate method (Christian and Lind 2007) was carried out in this study to measure the carbon source utilization. Specifically, approximate 10 g (dry weight equivalent) of fresh sediment was suspended in 90 ml saline solution (0.85% NaCl, w/v), shaken for 30 min on a rotary shaker, and diluted 100-fold. 150 μl of the diluted samples were added directly into EcoPlates (BIOLOG, Hayward, CA, USA), and then incubated in dark condition at 25 °C for 10 days and measured at 590 nm every 12 h with an EMax Precision Microplate Reader (BIOLOG, Hayward, CA, USA). The first reading was performed immediately after inoculation. The rate of carbon metabolism on Biolog plates over time was determined by calculating the average well color development (AWCD, Average Well Color Development) using the method of Garland (Gomez et al. 2006).
Statistical Analysis
Based on taxonomic annotation, obtained sequences were grouped at phylum level to construct rarefaction curves and diversity indices of Chao1, Simpson and Shannon. NMDS was processed using the Mothur program. The diversity indices of carbon utilization and ANOSIM analysis were calculated using the PRIMER5 program. To investigate the relationship between microbial community and environmental parameters, Monte Carlo tests and Redundancy analysis (RDA) were performed in CANOCO5.0. Differences of sediment parameters among sampling sites was assessed by one-way ANOVA using SPSS 20.0. Pearson coefficient correlations between series of parameters and microbial community datasets were also conducted using SPSS 20.0. P < 0.05 was considered to be statistically significant.
Results
Characterization of Sediment Physicochemical Properties
The detected physicochemical parameters and heavy metal contents from sediment samples were shown in Table 1. Most of the parameters differed significantly among the four sites. All samples showed weak acidity, pH values ranging from 5.67 (NJS) to 6.63 (SMS). NJS samples had the highest contents of AFDM and TN among these four sampling sites, while SMS samples had the lowest content. The contents of TP, Cu and Zn from RH were significantly higher than other three sites, while BSZ was the second highest. The contents of TOC and Pb had no obvious difference among the four sites.
BCCs Revealed by Illumina Miseq Sequencing
A total of 259,216 partial sequences of bacterial 16S rRNA genes with an average length of 252 bp passed the quality control procedures. The OTU numbers of sediment samples ranged between 2350 and 2779, and obvious difference was observed among four sites (P < 0.05). The Shannon-Wiener and Simpson indexes were significantly lower in undisturbed SMS than other disturbed area (RH, BSZ and NJS) (Table 2). We performed rarefaction analysis on 12 samples and none of our samples showed a full saturation in rarefaction curves (Fig.S1).
We obtained a distinct pattern of taxonomic composition among the four sampling sites (Fig. 2). A total of 31 phyla were found in all sediment samples, and the most dominant bacterial lineages present were Proteobacteria(58.60%), Firmicutes (12.62%), Bacteroidetes (5.28%), Chloroflexi (4.62%), Nitrospira (4.21%) and Verrucomicrobia (3.76%) which on behalf of 89% of sequences in total. Proteobacteria was dominated by Deltaproteobacteria (22.50%), Betaproteobacteria (12.98%), Gammaproteobacteria (16.06%) and Alphaproteobacteria (7.02%). NMDS ordination analysis displayed a clear separation of BCCs among the four regions (Fig. 3). ANOSIM (r = 0.867; P < 0.05) analyses further confirmed the robustness of this separation. The distribution of BCCs in NMDS ordination could be divided into three parts. The taxonomic composition of SMS was represented by Gammaproteobacteria (24%), Deltaproteobacteria (18%), and Bacteroidetes (7%) which differentiated SMS with the other three sites. The taxonomic composition of RH and NJS could be represented by Verrucomicrobia, Firmicutes and Delproteotabacteria, Chloroflexi, respectively, and the distribution of BSZ was located between NJS and RH.
Microbial Metabolic Activity Revealed by Biolog-EcoPlate Analysis
Biolog-Ecoplate analysis was used to qualitatively and quantitatively evaluate physiological profile of microbes in community level from different sample sites. The metabolic curves exhibited similar patterns after logarithmic increase state (40-90 h) (Fig.S2). We selected the timing point of 60-h for further analysis on diverse utilize types of carbon source because it showed the highest slope of the logarithmic phase which indicated the rate of carbon source utilization. The AWCD presented a gradient orders as follows: SMS (0.4968) > BSZ (0.3612) > RH (0.2436) > NJS (0.1989) (p < 0.05) at 60-h point. Therefore, the rate of carbon source utilization in disturbed area was lower than undisturbed areas.
Significant differences were detected on the utilization preference of six classes of carbon source from the four sampling sites at 60-h point (Fig. 4). Except for phenolic acids, the utilization efficiency of all carbon sources peaked in SMS. The utilization efficiency of polymer and carbohydrate was lower than other carbon classes, and the lowest efficiency occurred in RH (P < 0.05). The utilization of phenolic acids, carboxylic acids, amino acids and amine showed the lowest efficiency in NJS area (P < 0.05).
The Influence of Sediment Properties on Bacterial Community Structures
Sediment properties had a significant impact on the structure of bacterial communities, and the RDA ordination results displayed their relationship (Fig. 5). It was revealed that the distribution of BCCs was significantly correlated to TP, TOC, pH and TN (p < 0.05). Pearson correlation analysis also revealed that the abundance of dominant phyla was influenced by sediment properties. Alphaproteobacteria, Chloroflexi, Deltaproteobacteria were mainly positively correlated with TN and TOC, while Gammaproteobacteria was negatively correlated with TN and TOC. Nitrospira was mainly positively affected by TP, while Firmicutes and Verrucomicrobia were mainly positively affected by TP and heavy metals (Fig. 6). Nevertheless, the distribution of bacterial phyla was mainly affected by different factors in different sampling sites (TableS1).
The Correlation of Bacterial Composition and Metabolic Activity
As shown in Fig. 6, most abundant phyla had significant correlations with carbon substrates (TableS2). Gammaproteobacteria and Betaproteobacteria were positively associated with carboxylic acids, amino acids or amine (P < 0.05), while Alphaproteobacteria, Chloroflexi and Nitrospira showed negative correlation (P < 0.05). Deltaproteobacteria was negatively correlated with carboxylic acids, and Verrucomicrobia was negatively correlated with polymer (P < 0.05). Furthermore, RDA ordination was performed to explore the relationship between carbon metabolic profile and major bacterial phyla (Fig. 7). Verrucomicrobia and Firmicutes mainly contributed to the carbon metabolic pattern of RH area, Deltaproteobacteria and Chloroflexi mainly functioned in NJS samples. Gammaproteobacteria, Bacteroidetes and Betaproteobacteria collectively functioned in both SMS and BSZ samples, while Nitrospira and Alphaproteobacteria showed negative effects in SMS and BSZ.
As the regression estimate shown in Fig. 8, changes in the sediment bacterial community were significantly correlated with metabolism: the greater community phylogenetic dissimilarity was, the more dissimilarity would display in carbon source metabolisms.
Discussion
This study presents the first assessment of bacterial communities in response to different types of anthropogenic disturbances in the sediment of Poyang Lake. Our results indicated that both physicochemical and microbiological data are impacted by anthropogenic disturbances within the lake ecosystem.
Spatial heterogeneity of sediment characteristics was detected from the areas influenced by different anthropogenic disturbances. Higher contents of TP and Cu were found in RH than in other areas. Wang and Liang (2015) reported that the concentrations of TP in sediments collected from Rao River tributaries were higher than those in the center of Poyang Lake. In this work, copper pollution in sediment from RH sites was found to be at moderate to high levels through calculating the geo-accumulation index of heavy metals (Praveena et al. 2008). High levels of TP, TN, and TOC from aquaculture and human activity were previously documented as sources for organic pollution and eutrophication in the lake ecosystem (Wang et al. 2003; Zhao et al. 2007; Zhu et al. 2013b). Here, the aquaculture activity in NJS area also resulted in the extensive accumulation of TN and TOC.
The observed Poyang Lake sediment bacterial community Shannon index ranged from 5.99 to 6.39 (Fig. 2), which was lower than the results from severe eutrophic water-Dianchi Lake (Bai et al. 2012), but higher than the results from moderate eutrophic water of Vidy Bay (Thevenon et al. 2012; Sauvain et al. 2014). Among the four regions investigated in this work, anthropogenic disturbed area (NJS, RH and BSZ) had higher Shannon index than the uncontaminated area (SMS), and the heavy metal polluted area RH had the highest bacterial Shannon index. Similar results were confirmed in another study where heavy metal pollution could enhance the bacterial Shannon index (Ni et al. 2015). In a word, sediment bacterial diversity could be increased by many pollutants.
In this study, the most dominant bacterial lineages of Poyang Lake sediment were Proteobacteria, Firmicutes, Bacteroidetes, Chloroflexi, Nitrospira and Verrucomicrobia. Similar results were also reported in previous investigations in which the benthic bacterial community of Poyang Lake was dominated by Deltaproteobacteria, Betaproteobacteria and Verrucomicrobia (Kou et al. 2015; Ding et al. 2015).
In SMS area, Gammaproteobacteria, Bacteroidetes and Betaproteobacteria were the phyla which differentiated the BCCs of SMS from other regions (Fig. 3). In a previous study, Gammaproteobacteria showed higher abundance in undisturbed area than disturbed area (Haller et al. 2011). Betaproteobacteria and Bacteroidetes were most frequently detected in sediment samples from the lake without intermittent disturbances (Leon et al. 2012), and Bacteriodetes was also abundantly detected in a pond without mariculture (Li et al. 2015). Therefore, the abundance of Gammaproteobacteria, Betaproteobacteria and Bacteroidetes in SMS sediment may represent an undisturbed or unpolluted environment. Furthermore, pH played an important role in determining the SMS bacterial community according to the redundancy analysis (RDA) (Fig. 5), and this point was frequently reported in other studies (Lauber et al. 2009). The highest carbon source metabolic efficiency (AWCD) was detected in SMS area, which may indicate that less anthropogenic disturbances help to maintain metabolic capacity (Gryta et al. 2014). The typical SMS groups (Gammaproteobacteria, Bacteroidetes and Betaproteobacteria) showed significantly positive correlation with carboxylic acids, amino acids, and amines. Therefore, these results may indicate that the three phyla have potential to utilize the three types of carbon sources, but the direct relation between these phyla and carbon source was still unclear.
In NJS area, only Deltaproteobacteria and Chloroflexi contributed to the community specificity as contrasted with other sites. RDA analysis (Fig. 5) demonstrated that TN and TOC were the main factors influencing bacterial community structure of NJS (Shao et al. 2011; Zhang et al. 2015). Chloroflexi was widely distributed in surface sediment of various freshwater lakes (Kadnikov et al. 2012), especially in eutrophic conditions (Niu et al. 2011). Deltaproteobacteria was found in water and soil environments rich in organic carbon and played a significant role in the global carbon cycles (Rodionov et al. 2004). Thus, Deltaproteobacteria and Chloroflexi were expected to involve in the metabolic process of high organic nutrients. Furthermore, Deltaproteobacteria and Chloroflexi showed negative correlations with carboxylic acids and amino acids. Amino acids and carboxylic acids can be considered as part of the major pool of immobilized N (Liu et al. 2014) and labile pool of organic matter (Pullin et al. 2004) respectively, and they played an important role in metabolism process. The negative correlation may suggest that Deltaproteobacteria and Chloroflexi are unable to utilize these two types of carbon sources or the sources inhibit the utilization process. In terms of the integral metabolic activities, a recent study (Wang et al. 2016) found that soils with organic contamination lead to lower AWCD, which was in consistent with the findings of this study.
The distribution of Verrucomicrobia and Firmicutes was representative for RH area, as the presences of heavy metals (Cu, Zn, Cd) and TP may provide selected pressures for these two phyla. Sauvain et al. (2014) had claimed that the predominance of endospore-forming Firmicutes often occurred in the wastewater polluted by trace metals, and strong correlation between Firmicutes and heavy metals was distinct. Verrucomicrobia was a bacteria phylum often detected in water and soil environments such as Kasumigaura Lake and Taihu Lake (Tamaki et al. 2005; Xi et al. 2007), and appeared to be positively associated with Hg and TN (Zeng et al. 2008; Vishnivetskaya et al. 2011). Furthermore, Verrucomicrobia and Firmicutes also showed apparent influence on metabolic function of sediment bacteria in RH area, and the significant negative correlation with polymers might indicate that these two phyla could not utilize polymers or even were inhibited by high concentration of polymers. This phenomenon could be explained by previous studies, which suggested that Cu could improve the capacity of communities to utilize polymers in low content and inhibit the capacity in high content (Guo et al. 2012). Moreover, our results verified that increasing the contents of heavy metals and TP would reduce the metabolic level (AWCD) of sediment microbial community in freshwater lakes (Wang et al. 2004).
The distribution of Alphaproteobacteria and Nitrospira in BSZ area could discriminate BSZ from other sites. The sediment physicochemical data of BSZ documented the presence of both nutrient and heavy metal pollutants, but no parameter passed the Monte Carlo tests, suggesting BCCs in the BSZ may be synergistically regulated by multiple factors, including heavy metals and nutrients.
To sum up, both bacterial community structure and metabolic function of sediment in Poyang Lake were sensitive to various anthropogenic disturbance-induced contaminations; different types of anthropogenic disturbance consistently increased bacterial diversity but decreased the metabolic activity (AWCD). In addition, different bacterial groups were found to specifically respond to the presence of certain pollutants and these groups are candidates of bio-indicators for lake sediment pollution. For this purpose, further studies should be carried out to quantify the typical proportions of these candidates in sediment bacterial community, and verify the effectiveness in different freshwater lake ecosystems.
References
Azizullah A, Khattak MNK, Richter P, Häder DP (2011) Water pollution in Pakistan and its impact on public health-a review. Environment International 37(2):479–497
Bai Y, Shi Q, Wen D, Li Z, Jefferson WA, Feng C, Tang X (2012) Bacterial communities in the sediments of Dianchi Lake, a partitioned eutrophic waterbody in China. PloS One 7(5):e37796
Bennett ER, Metcalfe CD (1998) Distribution of alkylphenol compounds in Great Lakes sediments, United States and Canada. Environmental Toxicology and Chemistry 17(7):1230–1235
Bergmann GT, Batesa ST, Eilersa KG, Laubera CL, Caporasoc JG, Waltersd WA, Knightc R, Fierer N (2011) The under-recognized dominance of Verrucomicrobia in soil bacterial communities. Soil Biology and Biochemistry 43(7):1450–1455
Burkhardt BG, Watkins-Brandt KS, Defforey D, Paytan A, White AE (2014) Remineralization of phytoplankton-derived organic matter by natural populations of heterotrophic bacteria. Marine Chemistry 163:1–9
Christian BW, Lind OT (2007) Multiple carbon substrate utilization by bacteria at the sediment–water interface: seasonal patterns in a stratified eutrophic reservoir. Hydrobiologia 586(1):43–56
Devarajan N, Laffite A, Graham ND, Meiger M, Prabakar K, Mubedi JI, Elongo V, Mpiana PT, Ibelings BW, Wildi W, Poté J (2015) Accumulation of clinically relevant antibiotic-resistance genes, bacterial load, and metals in freshwater lake sediments in central Europe. Environmental Science & Technology 49(11):6528–6537
Ding X, Peng X, Jin B, Xiao M, Chen J, Li B, Fang C, Nie M (2015) Spatial distribution of bacterial communities driven by multiple environmental factors in a beach wetland of the largest freshwater lake in China. Frontiers in Microbiology 2015(6):129
Dudgeon D, Arthington AH, Gessner MO, Arthington AH, Kawabata ZI, Knowler DJ, Lévêque C, Naiman RJ, Solo D, Stiassny MLJ, Sullivan CA (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biological Reviews 81(02):163–182
Fang H, Lian J, Wang H, Cai L, Yu Y (2015) Exploring bacterial community structure and function associated with atrazine biodegradation in repeatedly treated soils. Journal of Hazardous Materials 286:457–465
Gad H, Wachendorf C, Joergensen RG (2015) Response of maize and soil microorganisms to decomposing poplar root residues after shallow or homogenous mixing into soil. Journal of Plant Nutrition and Soil Science 178(3):507–514
Gao J, Jia J, Kettner AJ, Xing F, Wang Y, Xu X, Yang Y, Zou X, Gao S, Qi S, Liao F (2014) Changes in water and sediment exchange between the Changjiang River and Poyang Lake under natural and anthropogenic conditions, China. Sci Total Environ 481:542–553
Gomez E, Ferreras L, Toresani S (2006) Soil bacterial functional diversity as influenced by organic amendment application. Bioresource Technology 97(13):1484–1489
Gryta A, Frąc M, Oszust K (2014) The application of the Biolog EcoPlate approach in ecotoxicological evaluation of dairy sewage sludge. Applied Biochemistry and Biotechnology 174(4):1434–1443
Guo X, Gu J, Gao H, Qin Q, Chen Z, Shao L, Chen L, Li H, Zhang W, Chen S, Liu J (2012) Effects of cu on metabolisms and enzyme activities of microbial communities in the process of composting. Bioresource Technology 108:140–148
Haddadchi A, Ryder DS, Evrard O, Olley J (2013) Sediment fingerprinting in fluvial systems: review of tracers, sediment sources and mixing models. International Journal of Sediment Research 28(4):560–578
Haller L, Tonolla M, Zopfi J, Peduzzi R, Wildi W, Poté J (2011) Composition of bacterial and archaeal communities in freshwater sediments with different contamination levels (Lake Geneva, Switzerland). Water Research 45(3):1213–1228
He M, Wang Z, Tang H (1998) The chemical, toxicological and ecological studies in assessing the heavy metal pollution in le an river, China. Water Research 2(32):510–518
Heim S, Schwarzbauer J (2015) Pollution history revealed by sedimentary records: a review. Environmental Chemistry Letters 11(3):255–270
Hu M, Wu Z, Zhou H, Zhang A, Song W, Zhang C (2005) The fisheries characters and resource status of Nanjishan natural reserve in Poyang lake. Resources and Environment in the Yangtze Basin 14(5):561–565
Kadnikov VV, Mardanov AV, Beletsky A, Shubenkova OV, Pogodaeva TV, Zemskaya TI, Ravin NV, Skryabin KG (2012) Microbial community structure in methane hydrate-bearing sediments of freshwater Lake Baikal. FEMS Microbiology Ecology 79(2):348–358
Kim JG (2003) Response of sediment chemistry and accumulation rates to recent environmental changes in the clear Lake watershed, California, USA. Wetlands 23(1):95–103
Kircher M, Kelso J (2010) High-throughput DNA sequencing-concepts and limitations. BioEssays 32(6):524–536
Konstantinidis KT, Isaacs N, Fett J, Simpson S, Long DT, Marsh TL (2003) Microbial diversity and resistance to copper in metal-contaminated lake sediment. Microbial Ecology 45(2):191–202
Kou W, Huang Z, Zhang J, Liu Q, Liu F, Liu Y, Wu L (2015) Bacterial community structure and composition in Lake Poyang: a case study in the Songmenshan region, China. Acta Ecologica Sinica 35(23):7608–7614
Kou W, Zhang J, Lu X, Ma Y, Mou X, Wu L (2016) Identification of bacterial communities in sediments of Poyang Lake, the largest freshwater lake in China. Springer Plus 5(1):1
Kowalchuk GA, Bodelier PL, Heilig GHJ, Stephen JR, Laanbroek HJ (1998) Community analysis of ammonia-oxidising bacteria, in relation to oxygen availability in soils and root-oxygenated sediments, using PCR, DGGE and oligonucleotide probe hybridization. FEMS Microbiology Ecology 27(4):339–350
Kumar KCVN, Demudu G, Malini BH, Rao KN, Kubo S (2016) Geospatial analysis of the changing environment of Kolleru Lake, the largest freshwater wetland in India. Wetlands 36(4):745–758
Landa M, Cottrell MT, Kirchman DL, Kaiser K, Medeiros PM, Tremblay L, Batailler N, Caparros J, Catala P, Escoubeyrou K, Oriol L, Blain S (2014) Phylogenetic and structural response of heterotrophic bacteria to dissolved organic matter of different chemical composition in a continuous culture study. Environmental Microbiology 16(6):1668–1681
Lauber CL, Hamady M, Knight R, Fierer N (2009) Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Applied and Environmental Microbiology 75(15):5111–5120
Lemon PW, Tarnopolsky MA, MacDougall JD, Atkinson SA (1992) Protein requirements and muscle mass/strength changes during intensive training in novice body builders. Journal of Applied Physiology 73(2):767–775
Leon C, Campos V, Urrutia R, Mondaca MA (2012) Metabolic and molecular characterization of bacterial community associated to Patagonian Chilean oligotrophic-lakes of quaternary glacial origin. World Journal of Microbiology and Biotechnology 28(4):1511–1521
Li L, Yan B, Li S, Xu J, An X (2015) A comparison of bacterial community structure in seawater pond with shrimp, crab, and shellfish cultures and in non-cultured pond in Ganyu, eastern China. Annales de Microbiologie 2015:1–12
Liu J, Sui Y, Yu Z, Shi Y, Chu H, Jin J, Liu X, Wang G (2014) High throughput sequencing analysis of biogeographical distribution of bacterial communities in the black soils of northeast China. Soil Biology and Biochemistry 70:113–122
Macalady JL, Mack EE, Nelson DC, Scow KM (2000) Sediment microbial community structure and mercury methylation in mercury-polluted clear Lake, California. Applied and Environmental Microbiology 66(4):1479–1488
Ni C, Horton DJ, Rui J, Henson MW, Jiang Y, Huang X, Learman DR (2015) High concentrations of bioavailable heavy metals impact freshwater sediment microbial communities. Annales de Microbiologie 2015:1–10
Niu Y, Shen H, Chen J, Xie P, Yang X, Tao M, Ma Z, Qi M (2011) Phytoplankton community succession shaping bacterioplankton community composition in Lake Taihu, China. Water Research 45(14):4169–4182
Paissé S, Coulon F, Goñi-Urriza M, Peperzak L, McGenity TJ, Duran R (2008) Structure of bacterial communities along a hydrocarbon contamination gradient in a coastal sediment. FEMS Microbiology Ecology 66(2):295–305
Praveena SM, Ahmed A, Radojevic M, Abdullah MH, Aris AZ (2008) Heavy metals in mangrove surface sediment of Mengkabong lagoon, Sabah: multivariate and geo-accumulation index approaches. International Journal of Environmental Research 2(2):139–148
Pullin MJ, Bertilsson S, Goldstone JV, Voelker BM (2004) Effects of sunlight and hydroxyl radical on dissolved organic matter: bacterial growth efficiency and production of carboxylic acids and other substrates. Limnology and Oceanography 49(6):2011–2022
Rodionov DA, Dubchak I, Arkin A, Alm E, Gelfand MS (2004) Reconstruction of regulatory and metabolic pathways in metal-reducing delta-proteobacteria. Genome Biology 5(11):R90
Sauvain L, Bueche M, Junier T, Masson M, Wunderlin T, Kohler-Milleret R, Diez EG, Loizeau JL, Tercier-Waeber ML, Junier P (2014) Bacterial communities in trace metal contaminated lake sediments are dominated by endospore-forming bacteria. Aquatic Sciences 76(1):33–46
Schumacher BA (2002) Methods for the determination of total organic carbon (TOC) in soils and sediments. Ecological Risk Assessment Support Center 2002:1–23
Shao K, Gao G, Qin B, Tang X, Wang Y, Chi K, Dai J (2011) Comparing sediment bacterial communities in the macrophyte-dominated and algae-dominated areas of eutrophic Lake Taihu, China. Canadian Journal of Microbiology 57(4):263–272
Smorczewski WT, Schmidt EL (1991) Numbers, activities, and diversity of autotrophic ammonia-oxidizing bacteria in a freshwater, eutrophic lake sediment. Canadian Journal of Microbiology 37(11):828–833
Sponseller RA, Fisher SG (2008) The influence of drainage networks on patterns of soil respiration in a desert catchment. Ecology 89(4):1089–1100
Tamaki H, Sekiguchi Y, Hanada S, Nakamura K, Nomura N, Matsumura M, Kamagata Y (2005) Comparative analysis of bacterial diversity in freshwater sediment of a shallow eutrophic lake by molecular and improved cultivation-based techniques. Applied and Environmental Microbiology 71(4):2162–2169
Thevenon F, Regier N, Benagli C, Tonolla M, Adatte T, Wildi W, Poté J (2012) Characterization of fecal indicator bacteria in sediments cores from the largest freshwater lake of Western Europe (Lake Geneva, Switzerland). Ecotoxicology and Environmental Safety 78:50–56
Tian J, McCormack L, Wang JY, Guo DL, Wang QF, Zhang XY, Yu GR, Blagodatskaya E, Kuzyakov Y (2015) Linkages between the soil organic matter fractions and the microbial metabolic functional diversity within a broad-leaved Korean pine forest. European Journal of Soil Biology 66:57–64
Vishnivetskaya TA, Mosher JJ, Palumbo AV, Yang ZK, Podar M, Brown SD, Brooks SC, Gu BH, Southworth GR, Drakel MM (2011) Mercury and other heavy metals influence bacterial community structure in contaminated Tennessee streams. Applied and Environmental Microbiology 77(1):302–311
Wang A, Chen J, Crowley DE (2004) Changes in metabolic and structural diversity of a soil bacterial community in response to cadmium toxicity. Biology and Fertility of Soils 39(6):452–456
Wang X, Han J, Xu L, Wan R, Chen Y (2014a) Soil characteristics in relation to vegetation communities in the wetlands of Poyang Lake, China. Wetlands 34(4):829–839
Wang L, Liang T (2015) Distribution characteristics of phosphorus in the sediments and overlying water of Poyang Lake. PloS One 10:e0125859
Wang Z, Liu S, Xu W, Hu Y, Hu Y, Zhang Y (2016) The microbiome and functions of black soils are altered by dibutyl phthalate contamination. Applied Soil Ecology 99:51–61
Wang H, Wang C, Wu W, Mo Z, Wang Z (2003) Persistent organic pollutants in water and surface sediments of Taihu Lake, China and risk assessment. Chemosphere 50(4):557–562
Wang Y, Yin R, Liu R (2014b) Characterization of biochar from fast pyrolysis and its effect on chemical properties of the tea garden soil. Journal of Analytical and Applied Pyrolysis 110:375–381
Xi W, Wu X, Ye W, Yang H (2007) Changes in bacterial community structure during preceding and degraded period of cyanobacterial bloom in a bay of the Taihu Lake. Chinese Journal of Applied & Environmental Biology 13(1):97
Yang L, Su M, Li Y, Liu J (2012) Determination of P content in ferrophorus by vanadium molybdenum yellow differential spectrophotometry. Ferro-alloys 1:019
Yu C, Zhang J, Wu L, Liu Y, Ge G (2015) Effects of heavy metal and nutrients on benthic microbial communities in freshwater sediment of Poyang Lake (China). Journal of Residuals Science & Technology 12(2):105–111
Yuan G, Liu C, Chen L, Yang Z (2011) Inputting history of heavy metals into the inland lake recorded in sediment profiles: Poyang lake in China. Journal of Hazardous Materials 185(1):336–345
Yuan X, Zhang L, Li J, Wang C, Ji J (2014) Sediment properties and heavy metal pollution assessment in the river, estuary and lake environments of a fluvial plain, China. Catena 119:52–60
Zeng J, Yang L, Liang Y, Li J, Xiao L, Jiang L, Zhao D (2008) Spatial distribution of bacterial communities in sediment of a eutrophic lake revealed by denaturing gradient gel electrophoresis and multivariate analysis. Canadian Journal of Microbiology 54(12):1053–1063
Zhang J, Yang Y, Zhao L, Li Y, Xie S, Liu Y (2015) Distribution of sediment bacterial and archaeal communities in plateau freshwater lakes. Applied Microbiology and Biotechnology 99(7):3291–3302
Zhao K, Liu X, Xu J, Selim HM (2010) Heavy metal contaminations in a soil-rice system: identification of spatial dependence in relation to soil properties of paddy fields. Journal of Hazardous Materials 181(1):778–787
Zhao X, Yang L, Yu Z, Fan Q, Zhang Z, Xiao Y (2007) Temporal and spatial distribution of physicochemical characteristics and nutrients in sediments of Lake Taihu. Journal of Lake Science 19(6):698–704
Zhu J, Zhang J, Li Q, Han T, Xie J, Hu Y, Chai L (2013a) Phylogenetic analysis of bacterial community composition in sediment contaminated with multiple heavy metals from the Xiangjiang River in China. Marine Pollution Bulletin 70(1):134–139
Zhu M, Zhu G, Li W, Zhang Y, Zhao L, Gu Z (2013b) Estimation of the algal-available phosphorus pool in sediments of a large, shallow eutrophic lake (Taihu, China) using profiled SMT fractional analysis. Environmental Pollution 173:216–223
Acknowledgements
The authors are grateful to the National Natural Science Foundation of China (No. 31060082, 31660027 and No.31260110) and the Open Foundation of MOE Key laboratory of Poyang Lake Environment and Resource (PYH2015-13) for their financial support. We also express sincere thanks to the anonymous reviewers for their valuable suggestions and comments for improving this manuscript. We also thank Larry Feinstein for his help on English polish.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Additional information
Xiao Jin and Yantian Ma contribute equally to this work.
Electronic supplementary material
ESM 1
(DOC 608 kb)
Rights and permissions
About this article
Cite this article
Jin, X., Ma, Y., Kong, Z. et al. The Variation of Sediment Bacterial Community in Response to Anthropogenic Disturbances of Poyang Lake, China. Wetlands 39 (Suppl 1), 63–73 (2019). https://doi.org/10.1007/s13157-017-0909-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13157-017-0909-1