Abstract
Non-ossifying fibroma (NOF) is a common benign bone tumor with a high probability of occurrence in children and adolescents. It is commonly seen in the metaphysis of long bones, eccentrically located, and can coexist with other malignant tumors such as neuroendocrine tumors (NET). To date, plain radiographs play a major role in the diagnosis of these benign bone tumors. Herein, we report the case of a 13-year-old male patient who was diagnosed with pulmonary NET and underwent right lung lobectomy for a hilar mass which later revealed a well-differentiated NET. The follow-up 68Ga DOTA-TOC PET/CT showed a focal somatostatin receptor expression in the left distal femur, with corresponding CT component findings of a well-defined osteolytic bone lesion located within the medial aspect of the left distal femoral metaphysis, strongly indicative of NOF. To the best of our knowledge, this is the first reported case of such an occurrence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Non-ossifying fibroma (NOF), also known as cortical desmoid, fibromatosis, fibrous cortical defect, and fibroxanthoma, is a common benign bone tumor, considered to be the most common non-neoplastic fibrous bone lesion with high incidence in children and adolescents. NOF is commonly seen in the metaphysis of long bones with an eccentric location, and it occurs more frequently in males (M:F ratio is 2:1). NOF can coexist with other malignant tumors simultaneously such as neuroendocrine tumors (NET) [1,2,3,4]. Histologically, NOF is characterized by a benign fibroblastic proliferation mixed with osteoclast-type giant cells [1, 4].
To date, NOF is mainly diagnosed via plain radiographs, while conventional imaging modalities such as computed tomography (CT) and magnetic resonance imaging (MRI) are inappropriate in young children and rarely needed in selected cases [1, 4].
Positron emission tomography and computed tomography (PET/CT) nowadays have become significant molecular-based modalities used in oncology in the detection and characterization of many malignant disorders. Used particularly in NET are 68Ga-labeled somatostatin receptor (SSTR) analogs including 68Ga DOTA-TOC, 68Ga DOTA-NOC, and 68Ga DOTA-TATE. These have proven their utility in staging, assessment of response to therapy, follow-up, prognostication purposes, and increasingly in the assessment of eligibility for peptide receptor radionuclide therapy (PRRT) [5,6,7].
Here, we report the case of a patient with pulmonary well-differentiated NET, where 68Ga somatostatin receptor PET/CT has been useful in detecting a coexisting NOF.
Case Report
A 13-year-old male patient diagnosed with pulmonary NET underwent right lobectomy for a right hilar mass in September 2017. Histopathological examination revealed a primary well-differentiated NET in the right lung mass.
The condition had originally started in March 2017, when the patient experienced high-grade fever and no other remarkable symptoms. After admission to the hospital and evaluation with a full workup (including chest X-ray, laboratory investigations, chest CT scan, and bronchoscopy) that revealed a lung mass, a surgery was performed. The patient was later referred to our nuclear medicine for the evaluation of metastases and residual right hilar disease.
During further follow-up, 68Ga DOTA-TOC PET/CT (Biograph mCT Flow Siemens) was performed from vertex to toes, 110 min after intravenous injection of 143 MBq of 68Ga DOTA-TOC. The scan demonstrated an incidental SSTR-positive, albeit mildly, a focal lesion with a maximum standardized uptake value (SUVmax) measures up to 2.52, located in the medial aspect of the left distal thigh, corresponding to well-defined osteolytic bone lesion located within the medial aspect of the distal metaphysis of the left femur which was strongly indicative of NOF (Figs. 1, 2, 3, 4 and 5). In the chest, there was a stable mildly SSTR-positive (SUVmax = 1.61) soft tissue thickening within the right sub-hilar region (surgical bed, not shown) which was indicative of chronic inflammatory process due to low somatostatin receptor density and stability. Additionally, there was no evidence of disease recurrence or metastases within the lung or elsewhere in the scan.
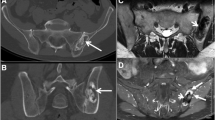
Transaxial images of 68Ga DOTA-TOC PET/CT. A focal somatostatin receptor-positive lesion (mild uptake with SUVmax = 2.52) within the distal left femur just above the knee (arrows) in transaxial-fused PET/CT images (a) corresponding CT findings (b) and fused PET/CT images (c) with rainbow PET coloring of the well-defined lytic bone lesion, indicative of non-ossifying fibroma. Notice the small non-ossifying fibroma on the distal right femur demonstrating minimal somatostatin receptor expression (chevron), indicating the usual pattern of non-ossifying fibroma
Coronal images of 68Ga DOTA-TOC PET/CT. A focal somatostatin receptor-positive lesion (mild uptake with SUVmax = 2.52) within the distal left femur just above the knee (arrows) in PET images (a) corresponding CT findings (b) and fused PET/CT image (c) corresponding to well-defined lytic bone lesion, indicative of non-ossifying fibroma. Notice the small non-ossifying fibroma on the distal right femur demonstrating minimal somatostatin receptor expression (chevron)
Given the incidental PET/CT finding, the patient underwent radiographic examination that revealed a well-demarcated radiolucent lesion within the left distal femoral metaphysis with a slightly sclerotic rim, and that was compatible with NOF.
Discussion
Non-ossifying Fibroma
NOF was initially described by Jaffe and Lichtenstein in 1942 [3, 8]; it is considered one of the most common lesions in bones particularly in children and adolescents [9]; and multifocal occurrence is not uncommon in this pathology, as the usual pattern of NOF is bilateral within long bones, however not necessarily the same size or same developmental age. It is estimated that 30–40% of people under the age of 20 years have NOF, and the disorder is most often asymptomatic with a good prognosis and a likelihood of regression over years with skeletal maturity. Usually, NOF is often discovered incidentally on plain X-rays made for other medical reasons, with few patients reporting pain and swelling, particularly in athletes [1, 9].
NOF is almost always benign and asymptomatic; it does not actively grow or destroy bone but may weaken and predispose the bone to fracture and most cases require no treatment and observation would be enough. This can be related to the pathophysiology of NOF, as these lesions often result in thinning and expansion of the overlying cortex and usually there is no periosteal reaction and no overt violation of the cortex. Hence, pain ensues except in cases of complicated pathological fracture requiring therapy (occasionally occurs in lesions > 3 cm in size). The most common locations for NOF are the distal femoral and distal tibial metaphyses and may move into the diaphysis with bone growth.
The majority of NOF, with time and under the guidance of the skeletal maturity, become to heal and, in the second decade of life, become sclerotic lesions [3]. A Jaffe-Campanacci syndrome, which is a rare condition, occurs in children who come with multiple NOFs; this condition occurs with systemic and cutaneous findings as in the patient with type 1 neurofibromatosis [3, 10, 11].
With plain X-ray, which is the imaging of choice for diagnosis of NOF, there are well-defined lytic lesions with peripheral sclerosis, lesions are mostly metaphyseal and eccentric arising from or involving the cortex, and they may appear expansile with sharp margins and sclerotic rims. The lesion’s long axis will be mostly parallel to the long axis of the affected bone, and multiplicity is possible though not the common scenario [1, 2].
On CT scan, which adds no further information above plain radiographs and exposes patients to ionizing radiation, NOF appears as a multiloculated lucent lesion with a sclerotic rim located eccentrically in the metaphysis, and as the patient ages, the lesions migrate away from the physis [1, 2]. CT scan is useful for determining the true extent of the lesion, demonstrating well-defined sclerotic borders, expansile nature, scalloping of bone, and internal trabeculations [1, 2]. On MRI, the lesion has initially a low signal on T1 and intermediate-to-high signal on T2 with peripheral low signal rim corresponding to the sclerotic border. When the lesion matures and ossifies, the signal becomes low on all sequences [10, 11]. On the MDP bone scan, NOF is usually not associated with increased radiotracer uptake; meanwhile, mild to moderate increased activity during NOF healing and ossification occurs, and high MDP avidity occurs with complications, such as pathological fractures [2]. This is the first case discussing the NOF uptake of 68Ga-labeled SSTR in the PET/CT imaging, and quite a few hypotheses mentioned the mechanism of 68Ga-labeled SSTR uptake within benign bone lesions and mostly no one regarding NOF; however, we surmised that the uptake is attributed to the osteoblastic osseous process, since osteoblast express SSTR-2, bone remodeling happened as a result of abnormal osseous resorption.
Imaging of SSTR-expressing tumors has been tremendously ameliorated by the use of combined PET/CT using 68Ga-labeled somatostatin analog such as DOTA-D-Phe1-Tyr3-octreotide (68Ga DOTA-TOC) as the tracer [14]. Nowadays, many papers emphasize the superiority of 68Ga-labeled SSTR tracers to other PET tracers such as 18F-FDG as well as established tracers for planar scintigraphy and SPECT imaging, including 111In-DTPA-octreotide. The sensitivity of such modalities for NETS has been described as superior to that of anatomical imaging modalities such as CT and MRI [11, 12]. The physiological accumulation of 68Ga SSTR analogs has been described in the spleen, kidneys, liver, and head of the pancreas, adrenal glands, thyroid, and pituitary gland [12].
A peculiar and explicit molecular imaging and targeted therapy to these NETs can be granted by the overexpression of somatostatin receptors (SSTRs) within the cell surface of neuroendocrine cells. Somatostatin is a peptide hormone that binds to these receptors (SSTRs), thereby regulating neurotransmission, hormone secretion, and cell proliferation. Inhibitory effects such as suppressing the release of pancreatic hormones or reducing smooth muscle contraction would be employed by these somatostatins [13].
68Ga-labeled peptides have been recognized as a new class of radiopharmaceuticals showing fast target localization and blood clearance. 68Ga DOTA-TOC, 68GaDOTA-TATE, and 68Ga DOTA-NOC are the most frequently used radiopharmaceuticals for imaging and differentiating lesions of various SSTR subtypes, overexpressed in many NETs [14]. 68Ga DOTA-TOC PET/CT scan is used for staging, treatment response evaluation, residual tumor detection, as well as surveillance and detection of recurrence NET with overexpression of SSTR.
Boy and colleagues demonstrated that 68Ga DOTA-TOC imaging narrates the expression of SSTR-2 at the level of mRNA in normal human tissue. They also provided a novel normative database to improve the diagnosis and the treatment monitoring of SSTR-expressing tumors on a molecular basis [15].
Epiphyseal growth plates in children also reveal a low to moderate increase in tracer activity. Overexpression of SSTR has been noted with other conditions, such as inflammatory processes, due to an expression SSTR-2 in white blood cells including leukocytes and macrophages. Some researchers have used this phenomenon to help image inflammatory processes such as atherosclerotic plaques with SSTR PET/CT [13]. Osteoblastic benign bone lesions show increased uptake on 68Ga-labeled SSTR PET/CT imaging, given the overexpression of SSTR-2 in overactive osteoblasts. These benign conditions, such as fractures, degenerative bone disease, fibrous dysplasia, and vertebral hemangiomas, are showing SSTR expression; however, these entities are mostly distinguished from pathologic activity by virtue of their low- or very low-intensity uptake and consistent features on the CT component of the study [13]. To the best of our knowledge, this is the first report demonstrating the possibility of detecting NOF by 68Ga DOTA-TOC PET/CT.
In summary, this case report has shown that a benign bone lesion (NOF) can express SSTR and thus be visualized on 68Ga-labeled SSTR PET/CT. Reviewing both components of this hybrid imaging modality (DOTA-TOC PET and CT scan) is very useful in reaching the rightful impression and avoiding false-positive interpretation.
References
Hetts SW, Hinchey SD, Wilson R, Franc B. Case 110: nonossifying fibroma. Radiology. 2007;243:288–92.
Hod N, Levi Y, Fire G, Ayash D, Somekh M, Horne T. Scintigraphic characteristics of non-ossifying fibroma in military recruits undergoing bone scintigraphy for suspected stress fractures and lower limb pains. Nucl Med Commun. 2007;28:25–33.
Goldin A, Muzykewicz DA, Dwek J, Mubarak SJ. The aetiology of the non-ossifying fibroma of the distal femur and its relationship to the surrounding soft tissues. J Child Orthop. 2017;11:373–9.
Herget GW, Mauer D, Kraus T, El Tayeh A, Uhl M, Norbert PS, et al. Non-ossifying fibroma: natural history with an emphasis on a stage-related growth, fracture risk and the need for follow up. BMC Musculoskelet Disord. 2016;17:147.
Hirmas N, Jadaan R, Al-Ibraheem A. Peptide receptor radionuclide therapy and the treatment of gastroentero-pancreatic neuroendocrine tumors: current findings and future perspectives. Nucl Med Mol Imaging. 2018;52:190–9.
Prasad V, Ambrosini V, Alavi A, Fanti S, Baum RP. PET/CT in neuroendocrine tumors: evaluation of receptor status and metabolism. PET Clin. 2008:3355–79.
Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumors. Semin Nucl Med. 2008;36:228–47.
Jaffe HL, Lichtenstein L. Non-osteogenic fibroma of bone. Am J Pathol. 1942;18:205–21.
Błaż M, Palczewski P, Swiątkowski J, Gołębiowski M. Cortical fibrous defects and non-ossifying fibromas in children and young adults: the analysis of radiological features in 28 cases and a review of literature. Pol J Radiol. 2011;76:32–9.
Jee WH, Choe BY, Kang HS, Suh KJ, Suh JS, Ryu KN, et al. Nonossifying fibroma: characteristics at MR imaging with pathologic correlation. Radiology. 1998;209:197–2.
Stacy GS, Dixon LB. Pitfalls in MR image interpretation prompting referrals to an orthopedic oncology clinic. Radiographics. 2007;27:805–26.
Al Ibraheem A, Bundschuh RA, Notni J, Buck A, Winter A, Wester HJ, et al. Focal uptake of 68Ga-DOTATOC in the pancreas: pathological or physiological correlate in patients with neuroendocrine tumors. Eur J Nucl Med Mol Imaging. 2011;38:2005–13.
Hofman MS, Lau WF, Hicks RJ. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: clinical utility, Normal patterns, pearls, and pitfalls in interpretation. Radiographics. 2015;35:500–16.
Reubi JC, Waser B, Schaer JC, Laissue AJ. Somatostatin receptor sst1-sst5 expression in normal and neoplastic human tissue using receptor autoradiography with subtype selective ligands. Eur J Nucl Med. 2001;28:836–46.
Boy C, Heusner TA, Poeppel TD, Redmann-Bischofs A, Unger N, Jentzen W, et al. Ga68 DOTATOC PET/CT and somatostatin receptor expression in normal human tissue: correlation of sst2 mRNA and SUVmax. Eur J Nucl Med Mol Imaging. 2011;38:1224–36.
Acknowledgments
The authors would like to thank Mr. Stephan Maus for his technical support to establish the peptide labeling process at our institution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Aysar Khalaf, Nader Hirmas, Farah Anwer, and Akram Al-Ibraheem declare that they have no conflict of interest.
Ethical Standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
The institutional review board of our institute approved this case report, and the requirement to obtain informed consent was waived.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khalaf, A., Hirmas, N., Anwer, F. et al. 68Ga DOTA-TOC Uptake in Non-ossifying Fibroma: a Case Report. Nucl Med Mol Imaging 54, 199–203 (2020). https://doi.org/10.1007/s13139-020-00650-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-020-00650-x