Abstract
Vitamin D reduces myocellular insulin resistance, but the effects of vitamin D on intramyocellular lipid (IMCL) partitioning are unknown. The purpose of this study was to understand how calcitriol, the active vitamin D metabolite, affects insulin sensitivity and lipid partitioning in skeletal muscle cells. C2C12 myotubes were treated with calcitriol (100 nM) or vehicle control for 96 h. Insulin-stimulated Akt phosphorylation (Thr 308) was determined by western blot. Intramyocellular triacylglycerol (IMTG), diacylglycerol (DAG), and ceramide content were measured by LC/MS. IMTG partitioning and lipid droplet accumulation were assessed by oil red O. Expression of genes involved in lipid droplet packaging and lipolysis were measured by RT-PCR. Compared to vehicle-treated myotubes, calcitriol augmented insulin-stimulated pAkt. Calcitriol increased total ceramides and DAG in a subspecies-specific manner. Specifically, calcitriol preferentially increased ceramide 24:1 (1.78 fold) and di-18:0 DAG (46.89 fold). Calcitriol increased total IMTG area as assessed by oil red O, but decreased the proportion of lipid within myotubes. Calcitriol increased mRNA content of genes involved in lipid droplet packaging (perilipin 2; PLIN 2, 2.07 fold) and lipolysis (comparative gene identification-58; CGI-58 and adipose triglyceride lipase; ATGL, ~ 1.80 fold). Calcitriol alters myocellular lipid partitioning and lipid droplet packaging which may favor lipid turnover and partially explain improvements in insulin sensitivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Whole body vitamin D status is inversely related to insulin resistance (IR) [15], and vitamin D repletion reduces whole body IR in humans [4, 39]. Skeletal muscle may account for up to 85% of insulin-stimulated glucose uptake [11] and is, therefore, a major determinant of whole body IR. Zhou et al. [43] showed that calcitriol (1,25(OH)2D3), the active vitamin D metabolite, increases insulin sensitivity in myotubes exposed to high levels of free fatty acids, suggesting vitamin D may reduce whole body IR via effects on skeletal muscle. To date, the physiological mechanism for vitamin D-induced improvements in insulin sensitivity remains unknown.
Intramyocellular lipid (IMCL) content is closely associated with skeletal muscle IR [1, 8, 24]. Partitioning of IMCL between intramuscular triacylglycerols (IMTG), diacylglycerols (DAG), and ceramides [1, 22, 34] appears to be a major determinant of muscle IR. Interestingly, circulating levels of vitamin D have been positively associated with IMCL content in older adults [31]. Furthermore, it has previously been reported that low dose (10 nM) calcitriol increases lipid content of C2C12 myoblasts [32]. Together, these data suggest a paradoxical effect of vitamin D to concomitantly increase IMCL content and reduce myocellular IR. However, it is unknown how calcitriol affects lipid partitioning between IMTG, DAG, and ceramides and lipid droplet packaging within skeletal muscle cells and if changes in partitioning are related to improvements in insulin sensitivity.
Ceramides [1, 34], a family of biologically active sphingolipids, and DAG [22], which are proposed to accumulate during incomplete lipolysis of IMTG, are associated with myocellular IR. Interestingly, fatty acid chain length and saturation appear to be major determinants of ceramide and DAG function [20, 28]. Therefore, ceramide and DAG may affect muscle metabolism in a subspecies-dependent manner. Specifically, ceramides and DAG containing saturated fatty acids tend to increase IR [20, 28]. Therefore, it is possible that shifting subspecies of ceramides and/or DAG to favor those containing unsaturated fatty acids may be associated with improved insulin sensitivity. To date, the effects of vitamin D on expression of specific lipid subspecies remains unknown.
IMTG constitute the majority of IMCL and can serve as a readily available substrate for ATP production [1]. IMTG content is associated with IR [9, 37] in obese, but not endurance-trained [1, 12], individuals, suggesting that total IMTG content alone does not induce IR. IMTG are stored within the highly specialized lipid droplet organelle [1]. It has been postulated that the specific proteins covering the lipid droplet may enhance lipolytic capacity [41] and protect against the development of IR [6, 10]. Perhaps the best described lipid packaging proteins are the perilipin (PLIN) family. PLINs associate with the lipid droplet and promote storage or lipolysis of the constitutive IMTG by repressing or enhancing activity of lipolytic enzymes. Overexpression of PLIN2 promotes increases in IMTG content and improves insulin sensitivity in vitro [6]. PLIN5 overexpression promotes both IMTG accumulation and lipolysis in skeletal muscle [40]. Calcitriol exerts genomic effects via the vitamin D receptor (VDR) to induce or repress gene transcription. To date, the effects of vitamin D on transcription of lipid droplet packaging and lipolytic genes are unknown.
The purpose of this study was to examine the effect of calcitriol on lipid partitioning and expression of genes involved in lipid droplet packaging and lipolysis within skeletal muscle cells as this may mechanistically explain the insulin sensitizing effects of calcitriol. We hypothesized that calcitriol would enhance myocellular insulin sensitivity concomitant with (1) a shift in ceramide and DAG subspecies content to favor those containing unsaturated fatty acids and (2) increased expression of genes involved in lipid droplet packaging (i.e., increased PLIN2 and 5) and lipolysis.
Methods
Cell culture
C2C12 (ATCC type) myoblasts were cultured in DMEM with 10% FBS, 100 IU/ml penicillin/-streptomycin and maintained at 37 °C and 5% CO2. Myoblasts were seeded at a density of 2.5 × 105 cells per well into six-well plates. Once cells were approximately 80% confluent, media was changed to low serum media (DMEM +2% horse serum) to induce differentiation into myotubes. Following 72 h of differentiation, media was supplemented with non-esterified fatty acids (250 μM palmitic acid and 250 μM oleate) conjugated to BSA (Sigma Aldrich). In our hands, failure to supplement media with fatty acids results in negligible accumulation of IMCL. These specific fatty acids were chosen because palmitic acid and oleate have been shown to be the most abundant lipid species found in human adipose tissue, circulating triacylglycerols, and circulating non-esterified fatty acids [21] and are the two most commonly consumed dietary fatty acids [13]. Furthermore, this lipid concentration has previously been shown to be a physiologically relevant concentration of non-esterified free fatty acids in mice [14]. Additionally, myotubes were treated with calcitriol (100 nM) or ethanol as a vehicle control. This dosage of calcitriol has previously been shown to maximally activate VDR in C2C12 myotubes [18]. All experiments were conducted following 96 h exposure to fatty acids and calcitriol.
Immunoblots
To verify that calcitriol improves insulin sensitivity in our model, immunoblots were performed for total and phosphorylated Akt (Thr 308) (cell signaling 9272 and 5110, respectively) following insulin treatment. Myotubes were treated with calcitriol for 96 h and stimulated with 100 μM insulin for 15 min. Myotubes were washed twice with PBS and proteins collected in RIPA buffer with protease and phosphatase inhibitors (Sigma Aldrich). Equal amounts of protein were separated by SDS-PAGE and transferred to nitrocellulose membranes. Membranes were incubated overnight in primary antibodies against pAkt and total Akt. Specific bands were detected by enhanced chemiluminescence following treatment with HRP-conjugated secondary antibodies. Band density was measured using Image J software (NCBI).
Liquid chromatography—mass spectroscopy
Myocellular DAG and ceramide subspecies content were measured following 96 h of calcitriol treatment by liquid chromatography-mass spectroscopy (LC/MS) as previously described [17, 25]. Samples were delivered to the University of Kentucky Small Molecule Mass Spectrometry Laboratory core facility where sample preparation and data collection were performed. The specific DAG subspecies measured were 8:0, 10:0, 14:0, 16:0, 16:0–18:1, 18:0–18:2, 18:0–20:4, 18:1, 18:1–20:0. The specific ceramide subspecies measured were 14:0, 16:0, 18:0, 20:0, 22:0, 24:0, and 24:1. In order to determine whether alterations in ceramide content were due to changes in ceramide synthesis, dihydroceramide (DH-cer) content was also measured. The specific DH-cer subspecies measured were 16:0, 18:0, 20:0, 22:0, 24:0, and 24:1. We also analyzed content of a subset of triacylglycerol subspecies by LC/MS. To control for differences in myocellular seeding density and/or size, LC/MS data were normalized to total phospholipid content as previously described [23].
Oil red O
To determine the effects of calcitriol on lipid droplet size, oil red O (ORO) staining was performed following 96 h of calcitriol treatment as previously described [12, 19]. Briefly, myotubes were washed with PBS, treated with 4% paraformaldehyde, and incubated in 60% oil Red O for 10 min. After washing residual oil red O from myotubes, images were collected using an inverted microscope (Zeiss Observer D1) at 40× magnification. Using Image J software (NCBI), brightfield images were converted to threshold images, and the oil red O positive area was measured. Six images were collected from each condition.
Real-time PCR
Following 96 h of calcitriol treatment, total myocellular RNA was isolated by phenol-chloroform extraction and reverse transcribed to cDNA. Quantitative RT-PCR (ABI 7900, SYBRgreen chemistry) was performed using self-designed primers (NCBI algorithms) for the lipid packaging genes PLIN2 and 5, and the lipolytic genes comparative gene identification-58 (CGI-58), adipose triglyceride lipase (ATGL), and hormone-sensitive lipase (HSL). Primers used in the present study are listed in Table 1. Primer specificity was confirmed by dissociation curve for all genes tested. Relative gene expression was normalized to the endogenous control gene large ribosomal protein (RPLPO), which was unchanged by calcitriol treatment. Relative gene expression was calculated using the ΔΔCt method.
Statistical analysis
All experiments were conducted with n = 3 unless otherwise noted. Data are presented as mean ± SE. Statistical comparisons were made using t test with α = 0.05, using GraphPad Prism 6. Insulin-stimulated pAkt immunoblots were analyzed by two-way-ANOVA with (Sidak post-test) α = 0.05, using GraphPad Prism 6.
Results
Calcitriol increases insulin sensitivity in myotubes exposed to a high fat environment
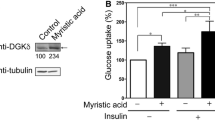
We first wanted to determine how calcitriol affects myocellular insulin sensitivity in myotubes exposed to a high fat environment. Therefore, we performed immunoblots for total and phosphorylated (Thr308) Akt following 15 min of insulin stimulation (Fig. 1a). Under basal conditions, no difference in pAkt/tAkt ratio was noted between vehicle- and calcitriol-treated myotubes. Insulin significantly increased pAkt/tAkt ratio in both vehicle- and calcitriol-treated myotubes (Fig. 1b). Akt phosphorylation (pAkt/tAkt ratio) was significantly (1.93 fold) higher in calcitriol + insulin-treated cells than in vehicle + insulin-treated cells. These results are consistent with enhanced insulin sensitivity following calcitriol treatment.
Calcitriol augments insulin-stimulated Akt phosphorylation in high fat-treated myotubes. C2C12 myotubes were treated with a calcitriol (100 nM) or vehicle control for 96 h and stimulated with 100 μM insulin stimulation for 15 min. a Representative immunoblots for pAkt (Thr308) and total Akt. b pAkt normalized to tAkt content. Data are expressed as mean ± SE of each group (n = 3 per group). Veh vehicle, Cal 100 nM calcitriol. Dagger indicates p < 0.01 v. basal, number sign indicates p < 0.0001 v. veh + insulin
Calcitriol increases ceramide content in a subspecies-specific manner
Since ceramides appear to be major drivers of myocellular IR, we next sought to determine how calcitriol affects ceramide content in cultured myotubes. As shown in Fig. 2a, calcitriol significantly increased total ceramide content (1.22 fold, p = 0.019). Content of ceramides containing saturated fatty acids was not significantly altered by calcitriol treatment (Fig. 2b). However, content of ceramides containing unsaturated fatty acids was significantly increased (1.30 fold, p = 0.013) (Fig. 2c). Examination of specific ceramide subspecies revealed that only Cer-24:1 was significantly altered by calcitriol treatment (1.30 fold increase, p = 0.026) (Fig. 2d). Additionally, we noted that expression of Cer-14:0 (p = 0.107) and Cer-20:0 (p = 0.117) trended toward decreasing with calcitriol treatment. However, Cer-24:0 (p = 0.082) trended toward increasing in response to calcitriol. These data indicate that calcitriol affects ceramide content in a subspecies-specific manner.
Calcitriol increases ceramide content in a subspecies-specific manner. C2C12 myotubes were treated with calcitriol (100 nM) or vehicle control for 96 h and ceramide content measured by LC/MS. a Total ceramide content. b Saturated fatty acid containing ceramide content. c Unsaturated fatty acid containing ceramide content. d Ceramide subspecies content. Veh vehicle-treated, Cal 100 nM calcitriol, Cer ceramide, DHCer dihydroceramide. Data are expressed as mean ± SE (n = 3 per group) and normalized to total phospholipid content. Asterisks indicate p < 0.05, dagger indicates p < 0.01
Calcitriol treatment increases intramyocellular DAG content
Due to previous reports indicating myocellular DAG contribute to IR, we next quantified total and subspecies-specific content of DAG by LC/MS. As shown in Fig. 3, calcitriol significantly increased total DAG (1.87 fold, p = 0.0002, Fig. 3a), saturated DAG (2.17 fold, p = 0.0005, Fig. 3b), and DAG containing one unsaturated and one saturated fatty acid (mixed DAG) (1.49 fold, p = 0.0002, Fig. 3c). As shown in Fig. 3d, calcitriol significantly increased content of several individual DAG subspecies including di-14:0 (1.40 fold), di-16:0 (1.31 fold), di-18:1 (1.69 fold), 16:0–18:1 (1.57 fold), 18:0–18:2 (1.38 fold), 18:0–20:4 (1.73 fold), and 18:1–20:0 (2.69 fold). Interestingly, the relative increase in di-18:0 (46.89 fold) in response to calcitriol was substantially greater than any other subspecies measured. These data indicate that calcitriol increases content of saturated and mixed DAG in cultured myotubes.
Calcitriol increases total, saturated, and mixed diacylglycerol (DAG) content. C2C12 myotubes were treated with calcitriol (100 nM) or vehicle control for 96 h and DAG content measured by LC/MS. a Total DAG content. b Saturated fatty acid containing DAG content. c Content of DAG containing both saturated and unsaturated fatty acids. d DAG subspecies content. Veh vehicle-treated, Cal 100 nM calcitriol. Data are expressed as mean ± SE (n = 3 per group). Asterisk indicates p < 0.05, daggers indicate p < 0.01, number signs indicate p < 0.001
Calcitriol increases IMTG area but not content
To assess IMTG in response to calcitriol, we measured IMTG content (LC/MS) and area (ORO). As shown in Fig. 4a, calcitriol did not significantly alter total IMTG content. Representative ORO images are shown in Fig. 4b. As shown in Fig. 4c, calcitriol significantly increased ORO positive area. However, there appeared to be a divergent response between myoblasts and myotubes. ORO positive area was significantly increased in myoblasts, but decreased in myotubes, treated with calcitriol (Fig. 4d).
Calcitriol increases intramyocellular triacylglycerol (IMTG) area, but not content. C2C12 myotubes were treated with calcitriol (100 nM) or vehicle control for 96 h, and IMTG content assessed by LC/MS and oil red O staining. a Total IMTG content assessed by LC/MS. b Representative oil red O images collected at 40× magnification. c Quantification of total oil red O positive area. d Distribution of oil red O positive area between myotubes and undifferentiated myoblasts. Data are expressed as mean ± SE of each group (n = 3 per group). Veh vehicle-treated, Cal 100 nM calcitriol, IMTG intramyocellular triacylglycerol, TAG triacylglycerol. Asterisks indicate p < 0.05 v. veh, double dagger indicates p < 0.0001 v. corresponding myotube value
Calcitriol increases expression of genes involved lipid droplet packaging and lipolysis
It has previously been suggested that altering lipid droplet packaging and lipolysis may protect against myocellular IR [6]. Since many of the effects of calcitriol are mediated by VDR-induced gene transcription, we next sought to understand how calcitriol affects expression of genes involved in lipid droplet packaging (PLIN2, and PLIN5) and lipolysis (CGI-58, ATGL, HSL). Calcitriol significantly increased mRNA content of the lipolytic genes CGI-58 (1.84 fold) and ATGL (1.80 fold), but not HSL (Fig. 5a). Additionally, calcitriol significantly increased mRNA content of the lipid packaging gene PLIN2 (2.07 fold), but not PLIN5 (Fig. 5b), indicating calcitriol may alter lipid droplet packaging.
Calcitriol increases expression of genes involved in lipid packaging and lipolysis. C2C12 myotubes were treated with calcitriol (100 nM) or vehicle control for 96 h and RT-PC performed. a Lipid droplet packaging genes. b Genes involved in lipolysis. Data are expressed as mean ± SE of each group (n = 4 per group). PLIN perilipin, CGI-58 comparative gene identification-58, ATGL adipose triglyceride lipase, HSL hormone-sensitive lipase, Veh vehicle-treated, Cal 100 nM calcitriol. Asterisks indicate p < 0.05, dagger sign indicates p < 0.01
Discussion
IMCL partitioning, packaging, and metabolism have been linked to myocellular IR. Here, we explored the effect of calcitriol on IMCL partitioning and expression of genes involved in lipid droplet packaging and lipolysis. To our knowledge, this is the first study to specifically examine the effects of calcitriol on lipid partitioning and packaging. The novel findings of this study are that, concomitant with enhanced insulin sensitivity, calcitriol increases myocellular ceramide and DAG content in a subspecies-dependent manner and increases expression of genes involved in lipid droplet packaging and lipolysis in skeletal muscle cells cultured in a high fat environment. These results may begin to broaden our understanding of the insulin sensitizing effects of calcitriol in human skeletal muscle.
Circulating 25(OH)D, an indicator of whole body vitamin D status, is associated with IMCL content in elderly adults [31]. Additionally, Ryan et al. [32] demonstrated that calcitriol augments lipid accumulation within C2C12 myoblasts. Given the association between IMCL content and myocellular IR [27, 38], these findings appear to conflict with data indicating vitamin D reduces IR. However, previous reports indicate that individual DAG and ceramides subspecies have different physiological effects [20, 28]. Therefore, it is possible that individual IMCL subspecies may be a more important determinant of IR than total IMCL content. Our data clearly show that calcitriol increases specific subspecies of DAG and ceramides and alters expression of lipid droplet packaging genes in a manner which does not negatively impact myocellular insulin sensitivity.
In the present study, calcitriol increased total myocellular ceramide content. Ceramides are known to inhibit PI3K-mediated Akt phosphorylation [5, 35, 42]. However, Brown et al. [7] demonstrated that increased ceramide levels in the liver did not impair insulin sensitivity, suggesting that ceramide subspecies composition, rather than total ceramide content, may be a more important determinant of IR than total ceramide content. Indeed, ceramide subspecies have varying physiological effects based on fatty acid chain length and saturation [20]. In the present study, we hypothesized that calcitriol would shift ceramide subspecies content to favor those containing unsaturated fatty acids. Although calcitriol did not decrease the content of ceramides containing saturated fatty acids, calcitriol specifically increased content of Cer-24:1. This shift in ceramide subspecies content partially confirms our hypothesis. Although specific ceramide subspecies have been linked to various disease states [20], the contribution of specific ceramide subspecies, if any, to skeletal muscle IR remains unknown. Argraves et al. [2] found an inverse relationship between Cer-24:1 in serum high density lipoprotein (HDL) and ischemic heart disease and noted that Cer-24:1 may contribute to the protective role of HDL in the disease process. It is possible that, unlike other ceramide subspecies, Cer-24:1 may enhance myocellular insulin sensitivity. Indeed, it has previously been demonstrated that very long chain length ceramides (> 22C) are associated with improved insulin sensitivity and glucose homeostasis in liver cells [29, 30]. A similar mechanism may regulate insulin sensitivity in skeletal muscle cells.
In addition to our observed increase in Cer-24:1, we noted trends for decreased content of Cer-14:0 and Cer-20:0, and concomitantly increase content of Cer-24:0. Although these trends were not statistically significant, the divergent response between short and long fatty acid chain ceramides agrees with recent reports that short and long chain ceramides differently affect insulin responsiveness [26, 29, 30]. Recently, Koch et al. [26] reported that vitamin D therapy specifically increased content of Cer-18:0 in circulation, which supports our finding that calcitriol alters ceramide content in subspecies-dependent manner. Future research should be aimed at determining the specific role ceramide subspecies in calcitriol-induced myocellular insulin sensitivity.
In addition to ceramide accumulation, myocellular DAG have previously been associated with IR [3, 27]. We assessed myocellular DAG content and found that calcitriol treatment increased content of total, saturated, and mixed DAG. Interestingly, in our study, myotubes showed enhanced insulin sensitivity despite increases in DAG. Compared to sedentary individuals, total and mixed DAG content has been shown to be higher in endurance-trained athletes than their sedentary counterparts [1]. Interestingly, these athletes displayed exceptionally high levels of di-18:0 DAG. In the present study, calcitriol preferentially increased content of DAG containing di-18:0. It is possible that increases in DAG, especially, those containing di-18:0, may be one mechanism by which calcitriol improves skeletal muscle metabolic function. Together, these data suggest intramuscular di-18:0 DAG content may be an important determinant of insulin sensitivity. Future research should aim to determine the physiological roles of specific DAG subspecies, particularly regarding IR. Additionally, we found calcitriol did not significantly increase total myocellular IMTG content. Although calcitriol increased total ORO positive area, we observed a non-significant trend for calcitriol to decrease IMTG content assessed by LC/MS. Interestingly, calcitriol decreased ORO positive area of myotubes and increased ORO positive area of the less metabolically active myoblasts, indicating a repartitioning of lipid droplets between multi- and mono-nucleated cells. It is possible that IMTG content, specifically found in myotubes, may have been decreased following calcitriol treatment.
We further report that calcitriol increases gene expression of PLIN2 which is consistent with our finding of increased ORO positive area and supports our hypothesis that calcitriol would increase expression of genes involved in lipid droplet packaging. PLIN2 knockdown increases lipid droplet size, effectively minimizing the surface area-to-volume ratio of available lipid droplets [6]. Therefore, it appears that increasing expression of PLIN2 may reduce lipid droplet size, thus maximizing surface area of lipid droplets. Increased lipid droplet surface area-to-volume may increase access of lipolytic enzymes to enhance IMTG lipolysis and mobilization of fatty acids from constituent IMTG. In addition to its apparent effects on lipid droplet surface area, PLIN2 overexpression has been shown to prevent palmitate-induced IR, despite increased total ceramide content within skeletal muscle [6]. Therefore, it is possible the PLIN2 contributes to calcitriol-induced enhancement of insulin sensitivity in high fat-treated myotubes by controlling lipid droplet size.
Previous reports have shown that exercise-induced increases in IMTG content are due to increased lipid droplet number, but not size [36]. Furthermore, Shepherd et al. [33] found that PLIN2 containing lipid droplets were preferentially used during 1 h of moderate-intensity cycling suggesting that increased PLIN2 expression may increase the ability of these lipid droplets to undergo lipolysis. Therefore, calcitriol-induced increases in PLIN2 may serve to enhance lipolysis and subsequent lipid oxidation. We show here that, in addition to increased PLIN2 expression, calcitriol increased gene expression of the lipolytic genes ATGL and CGI-58. These findings point to both increased lipid storage and lipolysis after calcitriol treatment, suggesting an increase in lipid flux and, by association, a decrease in lipid-mediated pathologies [16]. To confirm this, future research should examine how calcitriol affects the composition and dynamics of lipid droplets in addition to lipid flux and storage in response to lipid-induced cellular stress.
In conclusion, our data demonstrate that calcitriol elicits numerous, distinct effects on myocellular lipid partitioning and lipid packaging potential including altering IMCL subspecies content, partitioning of IMTG between myotubes and myoblasts, and increasing expression of genes involved in lipid droplet packaging and lipolysis. These specific effects on myocellular lipid partitioning and packaging may play a role in enhancing insulin sensitivity during high fat exposure such as in obesity or in aging, disease, and muscle disuse where muscle lipotoxicity contributes to IR.
References
Amati F, Dubé JJ, Alvarez-Carnero E et al (2011) Skeletal muscle triglycerides, diacylglycerols, and ceramides in insulin resistance: another paradox in endurance-trained athletes? Diabetes 60:2588–2597. https://doi.org/10.2337/db10-1221
Argraves KM, Sethi AA, Gazzolo PJ et al (2011) S1P, dihydro-S1P and C24:1-ceramide levels in the HDL-containing fraction of serum inversely correlate with occurrence of ischemic heart disease. Lipids Health Dis 10:70. https://doi.org/10.1186/1476-511X-10-70
Badin PM, Langin D, Moro C (2013) Dynamics of skeletal muscle lipid pools. Trends Endocrinol Metab 24:607–615. https://doi.org/10.1016/j.tem.2013.08.001
Belenchia AM, Tosh AK, Hillman LS, Peterson CA (2013) Correcting vitamin D insufficiency improves insulin sensitivity in obese adolescents: a randomized controlled trial. Am J Clin Nutr 97:774–781. https://doi.org/10.3945/ajcn.112.050013
Bikman BT, Summers SA (2011) Ceramides as modulators of cellular and whole-body metabolism. J Clin Invest 121:4222–4230. https://doi.org/10.1172/JCI57144
Bosma M, Hesselink MKC, Sparks LM et al (2012) Perilipin 2 improves insulin sensitivity in skeletal muscle despite elevated intramuscular lipid levels. Diabetes 61:2679–2690. https://doi.org/10.2337/db11-1402
Brown JM, Betters JL, Lord C et al (2010) CGI-58 knockdown in mice causes hepatic steatosis but prevents diet-induced obesity and glucose intolerance. J Lipid Res 51:3306–3315. https://doi.org/10.1194/jlr.M010256
Coen PM, Dubé JJ, Amati F et al (2010) Insulin resistance is associated with higher intramyocellular triglycerides in type I but not type II myocytes concomitant with higher ceramide content. Diabetes 59:80–88. https://doi.org/10.2337/db09-0988
Corcoran MP, Lamon-fava S, Fielding RA (2007) Skeletal muscle lipid deposition and insulin resistance: effect of dietary fatty acids and exercise 1–3. 662–677
de Wilde J, Smit E, Snepvangers FJM et al (2010) Adipophilin protein expression in muscle—a possible protective role against insulin resistance. FEBS J 277:761–773. https://doi.org/10.1111/j.1742-4658.2009.07525.x
DeFronzo RA, Jacot E, Jequier E, et al (1981) The effect of insulin on the disposal of intravenous glucose: results from indirect calorimetry and hepatic and femoral venous catheterization. Diabetes 30
Dubé JJ, Amati F, Stefanovic-Racic M et al (2008) Exercise-induced alterations in intramyocellular lipids and insulin resistance: the athlete’s paradox revisited. Am J Physiol Endocrinol Metab 294:E882–E888. https://doi.org/10.1152/ajpendo.00769.2007
Ervin RB, Wang C-Y, Wright JD, Kennedy-Stephenson J (2004) Dietary intake of selected minerals for the United States population: 1999-2000. Adv Data 1–5
Fjære E, Aune UL, Røen K et al (2014) Indomethacin treatment prevents high fat diet-induced obesity and insulin resistance but not glucose intolerance in C57BL/6J mice. J Biol Chem 289:16032–16045. https://doi.org/10.1074/jbc.M113.525220
Forouhi NG, Luan J, Cooper A et al (2008) Baseline serum 25-hydroxy vitamin d is predictive of future glycemic status and insulin resistance the medical research council ely prospective study 1990-2000. Diabetes 57:2619–2625. https://doi.org/10.2337/db08-0593
Funai K, Semenkovich CF (2011) Skeletal muscle lipid flux: running water carries no poison. Am J Physiol Endocrinol Metab 301:245–251
Gellett AM, Kharel Y, Sunkara M et al (2012) Biosynthesis of alkyl lysophosphatidic acid by diacylglycerol kinases. Biochem Biophys Res Commun 422:758–763. https://doi.org/10.1016/j.bbrc.2012.05.077
Girgis CM, Clifton-Bligh RJ, Mokbel N et al (2014) Vitamin D signaling regulates proliferation, differentiation, and myotube size in C2C12 skeletal muscle cells. Endocrinology 155:347–357. https://doi.org/10.1210/en.2013-1205
Goodpaster BH, Brown NF (2005) Skeletal muscle lipid and its association with insulin resistance: what is the role for exercise? Exerc Sport Sci Rev 33:150–154. https://doi.org/10.1097/00003677-200507000-00008
Grösch S, Schiffmann S, Geisslinger G (2012) Chain length-specific properties of ceramides. Prog Lipid Res 51:50–62. https://doi.org/10.1016/j.plipres.2011.11.001
Hodson L, Skeaff CM, Fielding BA (2008) Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog Lipid Res 47:348–380. https://doi.org/10.1016/j.plipres.2008.03.003
Itani SI, Ruderman NB, Schmieder F, Boden G (2002) Lipid-induced insulin resistance in human muscle is associated with changes in diacylglycerol, protein kinase C, and IkappaB-alpha. Diabetes 51:2005–2011. https://doi.org/10.2337/diabetes.51.7.2005
Itaya K, Ui M (1966) A new micromethod for the colorimetric determination of inorganic phosphate. Clin Chim Acta 14:361–366. https://doi.org/10.1016/0009-8981(66)90114-8
Kelley DE, Goodpaster BH, Storlien L (2002) Muscle triglyceride and insulin resistance. Annu Rev Nutr 22:325–346. https://doi.org/10.1146/annurev.nutr.22.010402.102912
Kim C, Schneider G, Abdel-Latif A et al (2013) Ceramide-1-phosphate regulates migration of multipotent stromal cells and endothelial progenitor cells—implications for tissue regeneration. Stem Cells 31:500–510. https://doi.org/10.1002/stem.1291
Koch A, Grammatikos G, Trautmann S et al (2017) Vitamin D supplementation enhances C18(Dihydro)ceramide levels in type 2 diabetes patients. Int J Mol Sci 18:1–10. https://doi.org/10.3390/ijms18071532
Li Y, Xu S, Zhang X et al (2015) Skeletal intramyocellular lipid metabolism and insulin resistance. Biophys Rep 1:90–98. https://doi.org/10.1007/s41048-015-0013-0
Montell E, Turini M, Marotta M et al (2001) DAG accumulation from saturated fatty acids desensitizes insulin stimulation of glucose uptake in muscle cells. Am J Physiol Endocrinol Metab 280:E229–E237. https://doi.org/10.1001/archneur.1985.04210090013004
Montgomery MK, Brown SHJ, Lim XY et al (2016) Regulation of glucose homeostasis and insulin action by ceramide acyl-chain length: a beneficial role for very long-chain sphingolipid species. Biochim Biophys Acta Mol Cell Biol Lipids 1861:1828–1839. https://doi.org/10.1016/j.bbalip.2016.08.016
Park J-W, Park W-J, Kuperman Y et al (2013) Ablation of very long acyl chain sphingolipids causes hepatic insulin resistance in mice due to altered detergent-resistant membranes. Hepatology 57:525–532. https://doi.org/10.1002/hep.26015
Redzic M, Powell DK, Thomas DT (2014) Vitamin D status is related to intramyocellular lipid in older adults. Endocrine 47:854–861. https://doi.org/10.1007/s12020-014-0238-6
Ryan KJP, Daniel ZCTR, Craggs LJL et al (2013) Dose-dependent effects of vitamin D on transdifferentiation of skeletal muscle cells to adipose cells. J Endocrinol 217:45–58. https://doi.org/10.1530/JOE-12-0234
Shepherd SO, Cocks M, Tipton KD, et al (2012) Preferential utilization of perilipin 2-associated intramuscular triglycerides during 1 h of moderate-intensity endurance-type exercise. 8:970–980. doi: https://doi.org/10.1113/expphysiol.2012.064592
Straczkowski M, Kowalska I, Baranowski M et al (2007) Increased skeletal muscle ceramide level in men at risk of developing type 2 diabetes. Diabetologia 50:2366–2373. https://doi.org/10.1007/s00125-007-0781-2
Summers SA, Garza LA, Zhou H, Birnbaum MJ (1998) Regulation of insulin-stimulated glucose transporter GLUT4 translocation and Akt kinase activity by ceramide. Mol Cell Biol 18:5457–5464
Tarnopolsky MA, Rennie CD, Robertshaw HA et al (2007) Influence of endurance exercise training and sex on intramyocellular lipid and mitochondrial ultrastructure, substrate use, and mitochondrial enzyme activity. Am J Physiol Regul Integr Comp Physiol 292:R1271–R1278. https://doi.org/10.1152/ajpregu.00472.2006
Van Loon LJC, Goodpaster BH (2006) Increased intramuscular lipid storage in the insulin-resistant and endurance-trained state. Pflugers Arch Eur J Physiol 451:606–616. https://doi.org/10.1007/s00424-005-1509-0
Vogt M, Puntschart A, Howald H et al (2003) Effects of dietary fat on muscle substrates, metabolism, and performance in athletes. Med Sci Sports Exerc 35:952–960. https://doi.org/10.1249/01.MSS.0000069336.30649.BD
von Hurst PR, Stonehouse W, Coad J (2010) Vitamin D supplementation reduces insulin resistance in South Asian women living in New Zealand who are insulin resistant and vitamin D deficient—a randomised, placebo-controlled trial. Br J Nutr 103:549. https://doi.org/10.1017/S0007114509992017
Wolins NE, Quaynor BK, Skinner JR et al (2006) OXPAT/PAT-1 is a PPAR-induced lipid droplet protein that promotes fatty acid utilization. Diabetes 55:3418–3428. https://doi.org/10.2337/db06-0399
Yamaguchi T, Omatsu N, Morimoto E et al (2007) CGI-58 facilitates lipolysis on lipid droplets but is not involved in the vesiculation of lipid droplets caused by hormonal stimulation. J Lipid Res 48:1078–1089. https://doi.org/10.1194/jlr.M600493-JLR200
Zhou H, Summers SA, Birnbaum MJ, Pittman RN (1998) Inhibition of Akt kinase by cell-permeable ceramide and its implications for ceramide-induced apoptosis. J Biol Chem 273:16568–16575. https://doi.org/10.1074/jbc.273.26.16568
Zhou QG, Hou FF, Guo ZJ et al (2008) 1,25-Dihydroxyvitamin D improved the free fatty acid-induced insulin resistance in cultured C2C12 cells. Diabetes Metab Res Rev 24:459–464. https://doi.org/10.1002/dmrr.873
Acknowledgements
The authors would like to thank Charlotte A. Peterson for intellectual contributions. We would also like to thank Dr. Andrew Morris and Manjula Sunkara for their work in collecting the LC/MS data.
Funding
This study was funded in part by start-up funds from the University of Kentucky, College of Education (LMB) and NIH R21 AG046762 (DTT).
Author information
Authors and Affiliations
Contributions
GEJ participated in the experimental procedures, data analysis/interpretation, and manuscript writing. DMS helped in the data analysis/interpretation, manuscript writing/editing, and contributed intellectual contributions. DTT participated in the study design, data interpretation, manuscript review, and contributed financial support. LMB helped in the study design, experimental procedures, data analysis/interpretation, manuscript writing, and contributed financial support.
Corresponding author
Rights and permissions
About this article
Cite this article
Jefferson, G.E., Schnell, D.M., Thomas, D.T. et al. Calcitriol concomitantly enhances insulin sensitivity and alters myocellular lipid partitioning in high fat-treated skeletal muscle cells. J Physiol Biochem 73, 613–621 (2017). https://doi.org/10.1007/s13105-017-0595-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-017-0595-8