Abstract
Rehabilitation after stroke is imperative for patients with spatial neglect as it can help improve behavioural, social and cognitive outcomes in these patients, and therefore reduce the financial burden on public health services. The main aim of this review is to investigate the effectiveness of active pursuit eye movements for rehabilitation interventions in patients with spatial neglect following stroke. Potential papers for inclusion were gathered by searching key terms in four main databases (AMED, Global Health, PubMed/Medline and PsychInfo) in addition to screening relevant reference lists. Two reviewers independently selected papers for inclusion based on agreed inclusion criteria (n = 9 with 147 participants). Risk of bias was assessed using the QUADAS-2 tool. All papers reported a statistically significant result in patients who received an intervention which used pursuit eye movements, and this was reported both as a short-term (immediate) effect and as a sustained effect up to 8 weeks after treatment. These effects were also reported in comparison with interventions using saccadic eye movements. One study also reported increased neural activity in a number of brain regions following pursuit-based intervention. Overall, there is good evidence in support of pursuit intervention used in the rehabilitation of stroke and spatial neglect over and above traditional interventions based on saccadic eye movements. Future research should aim to increase sample sizes, provide information on statistical power, record accurate eye movement responses and use randomised designs to reduce selection bias.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the major causes for mortality and morbidity globally is damage to the brain as a result of an ischemic stroke or haemorrhage [30]. The majority of strokes are ischemic caused by a blood clot in a blood vessel (artery) supplying the brain, which causes a lack of blood flow and tissue death (known as an infarction). A haemorrhage is the result of a leak or bursting artery in the brain as the result of a trauma or spontaneous bleeding. This damage can lead to differing effects on patients depending on the localisation and severity of the lesion. Damage localised in the right hemisphere can lead to impairments in processing visual stimuli within a particular part of the visual field, a phenomenon referred to as spatial neglect. Patients are more likely to suffer from spatial neglect if the stroke damages specific areas within the brain such as the white matter tracts in the prefrontal cortex [11], the temporo-parietal junction and surrounding cortical areas [28] or the perisylvian areas [16].
Between three and five million patients experience spatial neglect as a result of stroke every year worldwide [3] with a third of these still experiencing symptoms of neglect a year after the stroke [39]. The most common cause of neglect in humans is large infarctions resulting from ischemic stroke in the right or middle cerebral artery [18]. The presence and severity of spatial neglect is typically assessed using a standardised battery of perceptual tasks. Stroke patients are assessed on their performance on pen-and-paper-based tests including cancellation tasks (using lines, letters, digits or stars), figure copying or drawing (such as producing a clock face) and line bisection [46]. In patients with left visual neglect, impaired performance on these tasks is manifested by rightward bias in the line bisection task, errors in performance on reading tasks and number of omissions in cancellation tasks [13].
Psychological and clinical research has utilised these tests in order to assess the effectiveness of rehabilitation techniques and improve motor and behavioural outcomes in patients with spatial neglect following stroke. Rehabilitation techniques including neck-proprioceptive training [40], prism adaptation [31] and transcutaneous nerve stimulation [36] have been developed to improve various motor and behavioural outcomes in patients after stroke. Although spontaneous recovery is possible in some neglect patients [12], acute stroke can have long-term impairments on an individual’s ability to engage in activities of daily living [15] so effective rehabilitation is essential.
Recently, research has investigated the role of eye movements (specifically saccades and smooth pursuit) in rehabilitating patients with spatial neglect. Saccadic eye movements are rapid ballistic movements (velocity between 400 and 800°/s) which are completed in 15–20 ms and are typically used to shift the fovea onto new targets of interest [27]. The neural circuitry involved in performing saccadic eye movements spans across a large part of the brain, with visual information being relayed via occipital, parietal and frontal lobes to the basal ganglia (including the caudate nucleus) as well as to structures within the brain stem including the superior colliculus and the interconnected nuclei in the reticular formation [24]. The majority of saccades we make typically involve bottom-up processing of information, where an eye movement is made in response to a visual stimulus in the environment [23]. In the context of rehabilitation techniques for patients with spatial neglect, bottom-up treatments involve using sensory stimulation in order to improve a patient’s visual perception of stimuli in the contralesional space [17].
In contrast to saccades, smooth pursuit eye movements are used to track moving stimuli in the environment. These eye movements are much slower than saccades with average velocities of 30–100°/s [47]. In addition, pursuit relies on the ability to place the high acuity region of the retina (fovea) onto the moving target. This process requires prediction in order to compensate for the inherent neural lag we have within our visual processing stream [2, 4]. For this reason, pursuit eye movements are thought to use top-down processing (internally driven). In rehabilitation, top-down procedures are used to direct a patient’s attention towards their neglected hemispace [17]. In stroke patients, damage to the parietal or frontal lobes can lead to deficits in performing smooth pursuit eye movements [14] as these regions, in combination with areas such as the cerebellum, medial superior temporal and middle temporal areas, are most involved in processing, prediction and relaying visual information [24].
Dong et al. [10] suggested that the assessment of the functionality of the ocular motor system can provide a marker for both cognitive and motor recovery in patients who are mildly affected by stroke. The role of active eye movements in rehabilitation has been increasingly investigated in the literature; however, systematic reviews to date have focused on treatments using saccadic eye movements [25]. A systematic review of research findings on active pursuit eye movements has yet to be published. Hence, this systematic research review aims to establish whether rehabilitation interventions using active (pursuit) eye movements can improve behavioural outcomes in stroke patients. In addition, we will investigate whether there is an optimal intervention strategy to decrease severity of neglect (assessed by behavioural outcomes) in stroke patients, and will discuss the feasibility that improvements in pursuit eye movements from interventions translate into improvements in neuronal activity in these patients.
Method
Types of Studies
This review included all controlled studies which used active pursuit eye movements (active being defined as non-reflexive and requires cognitive control, in contrast to passive optokinetic reflexive type responses) as a rehabilitation intervention in stroke patients with neglect. Two randomised controlled studies were included in the review; however, these studies were assessed as being of lower quality (see “Quality Assessment”), two longitudinal studies were included, and three within subject design from the nine studies were included.
Participant Demographics
Participants exhibited visual neglect symptoms following a stroke. Studies were excluded from the review if participants’ visual neglect had resulted from other forms of damage to the brain such as a brain tumour or major head trauma. Additionally, only patients who had experienced a single stroke were included in the review.
Search Terms and Intervention Types
This review included any forms of rehabilitation which used active pursuit eye movements. Active pursuit involves following a stimulus with your eyes as opposed to keeping your eye’s still in more passive observation. Studies were also included if active eye movement interventions were used in a subgroup or as a control within a study. In order to include alternative definitions of key terms, additional search terms were used in conjunction with stroke and neglect. For instance, the key term of stroke also included lesion and ischemic. The key term neglect encompassed the alternative terms of spatial neglect, visual neglect, unilateral neglect, hemispatial neglect, hemineglect and hemiagnosia. Additional search terms were used to capture alternative terminologies; for example, eye movement* was used in combination with a pre-existing active eye movement intervention (optokinetic stimulation).
Outcome Measures
Behavioural outcome measures that assessed the ability to perform active eye movements were included. The main outcome was patient performance on a battery of standardised assessment measures of neglect including the following: line bisection (perceptual or visuomotor), tactile search, cancellation tests (single or double tasks using digits, lines, shapes or letters) and paragraph or line reading tasks as assessed by error rates, number of leftward omissions or percentage of rightward bias. This review also included outcomes with drawing tasks, such as figure copying (e.g. a clock face), line drawing, and freehand drawing. Papers using neural or cognitive outcomes in these patients were also included.
Identification Process
In order to establish reliability in the identification process search terms were computed in multiple databases by two reviewers independently. The results from the database search were then discussed and confirmed. The key terms were used in the following databases: AMED, Global Health (including in process and non-indexed citations), PubMed/Medline and PsychInfo. Limits were added to the searches in order to exclude non-English papers (for interpretation purposes). Review papers were included in the identification process to allow for screening of reference lists for potential papers which were not retrieved through the database search process.
As previously mentioned, the key terms of neglect and stroke also included commonly used synonyms for these terms (see “Search Terms and Intervention Types” for further details). A total of 337 papers were retrieved using combinations of key terms in each database which were compiled in Excel for filtering and processing.
Methods of Review
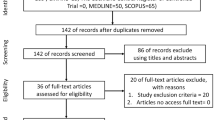
Two reviewers independently selected papers for inclusion in the review based on inclusion criteria (types of studies, participant demographics, intervention type and outcome measures). Eligibility was independently assessed before selecting and cross-checking the data for qualitative and quantitative synthesis. Discussions were held to overcome any differences in opinion and to confirm data for inclusion in the review (see Fig. 1).
After synthesising the relevant papers, the main characteristics and variables were extracted using forms in accordance with the QUADAS-2 tool [45]. These variables included the study design, sample size, information regarding the patient’s lesion (location, severity and time after stroke), the rehabilitation procedure and associated outcome measures, and the reported results. Studies were excluded if key data for more than two of these outcome measures were not reported or were unclear. In papers where active eye movements were not the main manipulation of the study (for example [37]) only data from the relevant subgroup or control condition were used. The methods of assessing neglect were also extracted to enable comparisons to be made between participants across the papers included in this review. The principle summary measures used in the papers were the difference in means whereby a participant’s score on a test at baseline was compared to their score on the same test after the rehabilitation or control procedure.
Quality Assessment
Methodological quality was assessed for studies included in the review using the Physiotherapy Evidence Database (PEDro) scale [42]. Papers were assigned a quality score from 1 to 10 where studies scoring 6 or above are deemed to have good methodological quality. This scale has good reliability [26] and validity [29] and is specifically for use on clinical and experimental research investigating the rehabilitation of stroke patients.
Results
From a total of 340 papers retrieved via database searches and other sources, 9 papers were included in this review. Figure 1 illustrates the number of articles received at each of the stages in the review from identification, screening, eligibility assessment to the final total. Nearly half the papers which were initially identified were found to be duplicates. Of the remaining, many did not meet the inclusion criteria (see “Method”) for example using passive rather than active eye movements or were a non-research paper (for example, a review article). The papers which were removed in the qualitative synthesis were excluded due to the lack of a control condition or due to unclear reporting of outcome measures (this was particularly seen in papers where neglect patients were included as a subgroup). The main study characteristics and variables of those retained in the review are presented in a table format in Appendix item 1.
Neglect Assessment and Outcome Measures
All studies (n = 9) with the exception of one [35] provided some information regarding participants lesions, including the period of time since the stroke occurred. All studies used either a standardised test of neglect or had adapted tests from standardised versions; for example, one study [19] had adapted the standardised tests to allow for administration by the bedside. All studies used these neglect tests as an outcome measure. In the studies using reading tasks, performance on the task was associated with patients’ ability to perform activities of daily living due to its importance in everyday life.
Participant Demographics
There were a total of 147 participants across experimental and control conditions in the nine studies, with an age range of 29–83 years and 30.1 % female. Of these participants, 22 patients had had a haemorrhage, 106 experienced visual neglect following an ischemic stroke or infarction and 3 had experienced bleeding. Information regarding cause of lesion was not provided for 16 participants.
Type of Rehabilitation Intervention
The main types of active eye movement interventions identified in the papers were smooth pursuit training (SPT) and leftwards moving optokinetic stimulation (OKS).
Smooth Pursuit Training
Two studies (69 participants) used smooth pursuit training (SPT) as an intervention, assessing patient outcomes immediately and two weeks after treatment [19, 22]. Both studies were of high quality (receiving a PEDro score of 7) using a randomised design where the intervention was administered to patients no more than a month after stroke. The first study used 5 × 50-min sessions (total 250 min) of SPT with standardised neglect tests as an outcome measure [22]. This study reported a significant main effect on paragraph reading (p < 0.001), significant reductions in rightward bias in both perceptual and motor line bisection tasks (p < 0.05), and finally a significant reduction in leftward omissions in the single and double digit cancellation tasks (p < 0.001) compared to baseline performance (pre-intervention). All these findings were sustained 2 weeks following treatment. No significant results were reported for the saccadic eye movement intervention of visual scanning therapy (VST) on any of the neglect tests (p = 0.11–0.37). Effect sizes (Cohen’s d) were reported based on neglect severity, with small effect sizes in mild neglect patients in the SPT group (0.1 to 2.2) and moderate effects sizes in the VST group (−0.5 to 0.5). In patients with severe neglect, large effect sizes were reported in the SPT group (0.7–0.9) and small to moderate effect sizes in the VST group (−0.3 to 0.5).
The second study by Kerkhoff et al. [19] used 20 × 30-min (total 600 min) sessions of SPT with adapted neglect tests as an outcome measure so they could be used by a patient’s bedside. They reported significant differences in scores on the Unawareness Behavioural Neglect Scale (UBNS) compared to baseline after SPT (mean difference = 0.37, p = .001) which was sustained at follow-up 2 weeks after intervention. The study also found significant differences in participant scores on the Functional Neglect Index (FNI) compared to baseline which was also sustained at follow-up (mean difference = 7.0, p < 0.001). VST did not significantly influence scores on the UBNI tests (smallest p = 1.0); however, VST did significantly improve outcomes on the FNI which was sustained at follow up (mean difference = 2.83, p = 0.01).
Optokinetic Stimulation Intervention
The remaining seven studies (78 participants) used OKS as an intervention in stroke patients. These have been split up by study design and will be discussed in order of methodological quality (highest to lowest).
Randomised Controlled Design (N = 2)
Only two of the seven studies used a randomised controlled design. The first [20] investigated the effects of 20 50-min sessions (total 1000 min) of OKS on a reading task and two neglect tests. They reported that OKS reduced directional biases, mainly rightward bias in horizontal line bisection (p < 0.001) and leftward omissions in digit cancellation (p < 0.05) and reading task (p < 0.05) compared to baseline scores. All OKS-associated improvements in neglect tests remained stable at 2 months follow up. VST was reported to have significantly reduced rightward bias in line bisection compared to baseline (p < 0.001). A PEDro score of 7 was given to this study based on its methodological quality.
The second randomised controlled design involved 20 sessions of OKS which was combined with saccadic exploration training for between 25 and 40 min long (total 500–800 min) with neglect tests, freehand drawing, reading and writing as outcome measures [41]. This study reported a significant improvement in performance overall on the neglect (mean = 5.11, SD = 0.53) measures compared to baseline (mean = 3.20, SD = 1.23) which was sustained at follow-up 1 week post-intervention. They reported no significant changes in neglect tests outcome scores in the VST condition (mean = 3.55, SD = 1.12) compared to baseline (mean = 3.15, SD = 1.38). OKS also significantly improved paragraph reading and writing outcomes (mean = 1.70, SD = 0.24) compared to baseline (mean = 1.20, SD = 0.29). This study was given a PEDro quality assessment score of 6.
Longitudinal Design (N = 2)
There was one longitudinal study which did not use a randomised controlled design; instead, patients were matched by neglect severity [21]. This study used four neglect measures to assess the effect of five 40-min sessions (total 200 min) of OKS. The study reported improvements in the reading task (p < 0.001), decreased percentage of leftward omissions in the cancellation task (p < 0.001) and reduced rightward bias in both perceptual and motor line bisection (p < 0.001) following OKS. These findings were maintained 2 weeks post-intervention (p < 0.05). VST was reported to significantly reduced rightward bias in perceptual line bisection compared to baseline (p < 0.001); however, there was no significant improvement on the other three neglect tests (p > 0.05). Based on its methodological quality, this study was given a PEDro score of 5. Another longitudinal study involved 14 sessions of OKS for 45 min (total 630 min) over a 3-week period [43]. This study used seven different outcome measures and reported that OKS led to improvements in performance on at least one of the neglect assessment tests compared to baseline scores. The PEDro quality assessment score for this study was 5.
Within Groups Design (N = 3)
One paper adopted a cross-sectional design using a single 30-min session of OKS on four neglect tests [17]. This study reported that OKS was more effective in improving behavioural outcomes on the line bisection, reading, cancellation and tactile search tasks (p < 0.05) compared to the control group which received VST. However, only a short-term effect was reported as no subsequent follow-up was made. A PEDro score of 4 was given to this study based on its methodological quality.
Finally, two papers included in the review were considered to be relatively poor in terms of methodological quality. The first study failed to report information regarding patients’ lesions and the duration of OKS treatment, which was measured using only one neglect test [35]. They reported that OKS led to significant improvements in performance on a visual line bisection task compared to patients with either mixed or rightward OKS (p < 0.05). Likewise, the final study in the review failed to report duration of OKS and used only a reading task to measure outcomes [38]. They reported that patients who had received OKS has a significantly fewer omissions during a reading task compared to their baseline scores (p < 0.05). These papers both received a PEDro score of 4 based on their methodological quality.
Cognitive and Neural Outcomes
Of the nine papers included in the review, one also included a cognitive outcome by mapping brain functioning before and after the active eye movement therapy. Using fMRI, Thimm et al. [43] reported that reductions in severity of neglect symptoms after OKS was associated with increased activity (bilaterally) in the precuneus and the middle frontal gyrus during a spatial attention task. They also reported increased neural activity in several areas of the left hemisphere, specifically the occipital cortex, middle temporal gyrus, cingulate gyrus and the angular gyrus.
QUADAS-2 Score
We found that 8/9 studies have low risk of bias for the patient flow ensuring consistency in testing between control and patient groups and also in timeliness of testing, with one study being unclear in this domain (see Fig. 2). In addition 2/9 studies had low risk of bias in their patient selection (generally consecutive), but the other 7 studies did not report this recruitment method. The index tests for neglect are lesion mapping with the reference standard revealing clear visual field (right sided) neglect in patient groups. It seems that most studies reported knowledge of the patient status prior to the recording of index (5/9 studies) and reference tests (7/9 studies) with other studies being unclear about this.
Discussion
The findings from this review favour pursuit-based rehabilitation interventions in improving outcomes in patients with visual neglect over traditional scanning interventions using saccadic eye movements. Patient improvements in at least one standardised test assessing neglect symptoms (including reading, cancellation, and line bisection tasks) were reported in all nine studies, with both immediate effects noted following a single 30-min session to sustained improvements 2 months after repeated OKS interventions (which amounted to a total of 1000 min of therapy). Only one study reported the effect sizes for the key findings which indicated that although statistically significant improvements were found in patients with visual neglect after receiving OKS therapy, the effect sizes were small to moderate and differed depending on the severity of patients neglect symptoms. Although a few of the papers were deemed to have low methodological quality, evidence indicates that interventions based on pursuit eye movements can improve patients’ symptoms of neglect as assessed by standardised neglect tests.
These findings highlighting the effectiveness of pursuit eye movements can have practical implications for the management and rehabilitation of patients with visual neglect. For example, public health services could combine rehabilitation interventions using pursuit eye movements with the assessment of neglect in order to improve outcomes in stroke patients. In addition to the potential benefits at a patient level, using these rehabilitation interventions can reduce the length of time patients spend in hospital following a stroke [9] which can create significant costs for the public health service [33].
However, there is a caveat to these findings which is worth noting: the effectiveness of any eye movement intervention is dependent on extent of the damage in the right hemisphere as a consequence of stroke [15] as well as the visual acuity of the patients [8]. Symptoms often vary from patient to patient due to the heterogeneous nature of visual neglect and its symptoms [7]. The findings from Kerkhoff et al. [22] demonstrated this as effect sizes were greater in those with severe neglect following visual scanning therapy compared to those with mild neglect. Furthermore, these studies excluded patients with any psychological problems such as dementia, limiting the generalizability of the findings from this systematic research review to a specific and potentially very small population. The prevalence of psychological problems such as dementia increase with old age [1]; therefore, future research should consider including such patients in order to assess the effectiveness of pursuit eye movement interventions on a wider population which would consequently increase the generalizability of findings.
Unfortunately, as yet there has been little research investigating the neural and cognitive outcomes of active eye movement therapies. One paper included in this review which did investigate neural activity, reported increased bilateral activity in three brain areas following OKS intervention, as well as increased activity in areas of the left hemisphere [43]. This pattern of activity can be placed in the context of top-down and bottom-up neural processing networks for pursuit and saccadic eye movements, respectively, a distinction which has been demonstrated in studies using fMRI brain scans [34].
Recent research has highlighted the usefulness of eye movement training as a natural and non-invasive intervention to increase visual awareness through strengthening connections and plasticity in key brain areas involved in producing eye movements and attentional processing, in particular the frontal eye field [44]. As suggested by Thimm et al. [43] using pursuit eye movements as a rehabilitation intervention in patients with neglect may help to re-wire the stroke damaged brain in a way that allows for compensatory strategies to be employed through the recruitment of other brain areas. Indeed, a study by Baumann and colleagues [5] also revealed a significant decrease in BOLD activity (and eye movement performance) in the frontal eye fields, the intraparietal sulcus and the cuneus in patients with cerebellar lesions. These findings also indicate that pursuit training could also provide benefit to patients that have suffered lesions sub-cortically to help restore functioning of this cortical network.
A number of limitations were observed with the studies and the outcomes used in the papers included in this systematic research review. Firstly, there is a lack of randomised controlled trials in research investigating neglect and rehabilitation, with some studies opting for a matched group design. Although this method may be useful in assessing outcomes by categorising patients by the severity of their neglect, using a non-randomised method can introduce selection bias. This was quantified by the QUADAS tool with over 50 % of the studies revealing a high risk of bias (five studies). The presence of selection bias in research can alter the contribution of unstated factors which is especially important to consider in healthcare research and clinical trials [32]. Therefore, future studies should aim to use a randomised design when allocating participants to experimental and control groups. We also revealed that in all of the studies, the experimenters were either not blinded or were unclear about this when administering reference and/or index tests for spatial neglect, again introducing significant bias into the interpretation of the results (77 % for reference standard and 55 % for the index test). Finally, 89 % of the studies reveal low bias in the flow and timing domain, suggesting that studies did well in ensuring the timing and consistency of the tests were appropriate.
Another limitation which was highlighted during this review was the lack of reference to statistical power regarding the sample. The reported sample sizes ranged from a single case study up to 45 participants; however, these numbers are unlikely to produce adequate statistical power. Therefore, the sample sizes used in the nine papers included in this review are indicative of a reduced likelihood that the significant results reported are reflective of a true effect [6]. Future research should attempt to address this by increasing sample sizes where possible and providing information on statistical power.
In conclusion, based on the findings reported in this review, pursuit eye movement therapies may be more effective than traditional, saccadic-based therapies such as VST in improving behavioural and neural outcomes in patients with spatial neglect following stroke. More studies using a randomised controlled design, larger sample sizes and omissions of experimenter bias are required to further confirm this finding, in addition to using brain imaging techniques (such as fMRI) to investigate the neural effects of active eye movement rehabilitation techniques for spatial neglect. A new direction for future research could be using cognitive and neural brain activity as an outcome to rehabilitation interventions. The theoretical frameworks involved in eye movements (top-down and bottom-up processing) can then be mapped in terms of neural activity involved in patients with visual neglect and performance both pre- and post-intervention.
References
Abbott A. Dementia: a problem for our age. Nature. 2011;475(7355):S2–4.
Andersen RA, Snyder LH, Bradley DC, Xing J. Multimodal representation of space in the posterior parietal cortex and its use in planning movements. Annu Rev Neurosci. 1997;20(1):303–30.
Appelros P, Karlsson GM, Seiger A, Nydevik I. Neglect and anosognosia after first-ever stroke: incidence and relationship to disability. J Rehabil Med. 2002;34(5):215–20.
Barnes GR, Asselman PT. The mechanism of prediction in human smooth pursuit eye movements. J Physiol. 1991;439:439–61.
Baumann O, Ziemus B, Luerding R, Schuierer G, Bogdahn U, Greenlee MW. Differences in cortical activation during smooth pursuit and saccadic eye movements following cerebellar lesions. Exp Brain Res. 2007;181:237–47.
Button KS, Ioannidis JP, Mokrysz C, Nosek BA, Flint J, Robinson ES, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14(5):365–76.
Buxbaum LJ, Ferraro MK, Veramonti T, Farne A, Whyte J, Ladavas E, et al. Hemispatial neglect Subtypes, neuroanatomy, and disability. Neurology. 2004;62(5):749–56.
Dieterich M, Bucher SF, Seelos KC, Brandt T. Cerebellar activation during optokinetic stimulation and saccades. Neurology. 2000;54(1):148.
Di Monaco M, Schintu S, Dotta M, Barba S, Tappero R, Gindri P. Severity of unilateral spatial neglect is an independent predictor of functional outcome after acute inpatient rehabilitation in individuals with right hemispheric stroke. Arch Phys Med Rehabil. 2011;92(8):1250–6.
Dong W, Yan B, Johnson BP, Millist L, Davis S, Fielding J, et al. Ischaemic stroke: the ocular motor system as a sensitive marker for motor and cognitive recovery. J Neurol Neurosurg Psychiatry. 2013;84(3):337–41.
Doricchi F, Thiebaut de Schotten M, Tomaiuolo F, Bartolomeo P. White matter(dis)connections and gray matter (dys)functions in visual neglect: gaining insights into the brain networks of spatial awareness. Cortex. 2008;44:983–95.
Farne A, Buxbaum LJ, Ferraro M, Frassinetti F, Whyte J, Veramonti T, et al. Patterns of spontaneous recovery of neglect and associated disorders in acute right brain-damaged patients. J Neurol Neurosurg Psychiatry. 2004;75(10):1401–10.
Halligan PW, Cockburn J, Wilson BA. The behavioural assessment of visual neglect. Neuropsychol Rehabil. 1991;1(1):5–32.
Heide W, Kurzidim K, Kömpf D. Deficits of smooth pursuit eye movements after frontal and parietal lesions. Brain. 1996;119(6):1951–69.
Jehkonen M, Ahonen JP, Dastidar P, Koivisto AM, Laippala P, Vilkki J, et al. Visual neglect as a predictor of functional outcome one year after stroke. Acta Neurol Scand. 2000;101(3):195–201.
Karnath HO, Fruhmann BM, Kuker W, Rorden C. The anatomy of spatial neglect based on voxelwise statistical analysis: a study of 140 patients. Cereb Cortex. 2004;14:1164–72.
Keller I, Lefin-Rank G, Lösch J, Kerkhoff G. Combination of pursuit eye movement training with prism adaptation and arm movements in neglect therapy: a pilot study. Neurorehabil Neural Repair. 2009;23(1):58–66.
Kerkhoff G. Hemispatial neglect in man. Prog Neurobiol. 2001;63:1–27.
Kerkhoff G, Bucher L, Brasse M, Leonhart E, Holzgraefe M, Völzke V, et al. Smooth Pursuit “Bedside” Training Reduces Disability and Unawareness During the Activities of Daily Living in Neglect A Randomized Controlled Trial. Neurorehabil Neural Repair. 2014;28:554–63.
Kerkhoff G, Keller I, Artinger F, Hildebrandt H, Marquardt C, Reinhart S, et al. Recovery from auditory and visual neglect after optokinetic stimulation with pursuit eye movements-Transient modulation and enduring treatment effects. Neuropsychologia. 2012;50(6):1164–77.
Kerkhoff G, Keller I, Ritter V, Marquardt C. Repetitive optokinetic stimulation induces lasting recovery from visual neglect. Restor Neurol Neurosci. 2006;24(4):357–69.
Kerkhoff G, Reinhart S, Ziegler W, Artinger F, Marquardt C, Keller I. Smooth pursuit eye movement training promotes recovery from auditory and visual neglect: a randomized controlled study. Neurorehabil Neural Repair. 2013;27(9):789–98.
Kinchla RA, Wolfe JM. The order of visual processing: “Top-down”, “bottom-up”, or “middle-out”. Percept Psychophys. 1979;25(3):225–31.
Krauzlis RJ. Recasting the smooth pursuit eye movement system. J Neurophysiol. 2004;91(2):591–603.
Lisa LP, Jughters A, Kerckhofs E. The effectiveness of different treatment modalities for the rehabilitation of unilateral neglect in stroke patients: A systematic review. NeuroRehabilitation. 2013;33(4):611–20.
Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther. 2003;83(8):713–21.
Martinez-Conde S, Macknik SL, Hubel DH. The role of fixational eye movements in visual perception. Nat Rev Neurosci. 2004;5(3):229–40.
Mort DJ, Malhotra P, Mannan SK, Rorden C, Pambakian A, Kennard C, et al. The anatomy of visual neglect. Brain. 2003;126:1986–97.
de Morton NA. The PEDro scale is a valid measure of the methodological quality of clinical trials: a demographic study. Aust J Physiother. 2009;55(2):129–33.
Murray CJ, Lopez AD. Mortality by cause for eight regions of the world: Global Burden of Disease Study. Lancet. 1997;349(9061):1269–76.
Nijboer TC, Olthoff L, Van der Stigchel S, Visser-Meily JM. Prism adaptation improves postural imbalance in neglect patients. NeuroReport. 2014;25(5):307–11.
Odgaard-Jensen J, Vist GE, Timmer A, Kunz R, Akl EA, Schünemann H, et al. Randomisation to protect against selection bias in healthcare trials. Cochrane Database System Review. 2011;4:MR000012.
Paolucci S, Antonucci G, Grasso MG, Pizzamiglio L. The role of unilateral spatial neglect in rehabilitation of right brain -damaged ischemic stroke patients: A matched comparison. Arch Phys Med Rehabil. 2001;82(6):743–9.
Petit L, Haxby JV. Functional anatomy of pursuit eye movements in humans as revealed by fMRI. J Neurophysiol. 1999;82(1):463–71.
Pitteri M, Kerkhoff G, Keller I, Meneghello F, Priftis K. Extra-powerful on the visuo-perceptual space, but variable on the number space: different effects of optokinetic stimulation in neglect patients. J Neuropsychol. 2014. doi:10.1111/jnp.12051.
Pitzalis S, Spinelli D, Vallar G, Di Russo F. Transcutaneous electrical nerve stimulation effects on neglect: a visual-evoked potential study. Front Hum Neurosci. 2013;7(111):1–9.
Priftis K, Pitteri M, Meneghello F, Umiltà C, Zorzi M. Optokinetic stimulation modulates neglect for the number space: evidence from mental number interval bisection. Front Hum Neurosci. 2012;6:23.
Reinhart S, Schindler I, Kerkhoff G. Optokinetic stimulation affects word omissions but not stimulus-centered reading errors in paragraph reading in neglect dyslexia. Neuropsychologia. 2011;49(9):2728–35.
Rengachary J, He BJ, Shulman GL, Corbetta M. A behavioral analysis of spatial neglect and its recovery after stroke. Front Hum Neurosci. 2011;5:29.
Schindler I, Kerkhoff G, Karnath HO, Keller I, Goldenberg G. Neck muscle vibration induces lasting recovery in spatial neglect. J Neurol Neurosurg Psychiatry. 2002;73(4):412–9.
Schröder A, Wist ER, Hömberg V. TENS and optokinetic stimulation in neglect therapy after cerebrovascular accident: a randomized controlled study. Eur J Neurol. 2008;15(9):922–7.
Sherrington C, Herbert RD, Maher CG, Moseley AM. PEDro. A database of randomized trials and systematic reviews in physiotherapy. Man Ther. 2000;5(4):223–6.
Thimm M, Fink GR, Küst J, Karbe H, Willmes K, Sturm W. Recovery from hemineglect: differential neurobiological effects of optokinetic stimulation and alertness training. Cortex. 2009;45(7):850–62.
Vernet M, Quentin R, Chanes L, Mitsumasu A, Valero-Cabré A. Frontal eye field, where art thou? Anatomy, function, and non-invasive manipulation of frontal regions involved in eye movements and associated cognitive operations. Front Integr Neurosci. 2014;8.
Whiting PF, Rutjes AW, Westwood ME, Mallett S, Deeks JJ, Reitsma JB, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med. 2011;155(8):529–36.
Wilson B, Cockburn J, Halligan P. Development of a behavioral test of visuospatial neglect. Arch Phys Med Rehabil. 1987;68(2):98–102.
Wright KW, Spiegel PH, Thompson LS, editors. Handbook of Pediatric Strabismus and Amblyopia (Vol. 5). New York: Springer; 2006.
Compliance with Ethical Standards
Conflict of Interest
Dr. M Burke declares that she has no conflict of interest; Ms. D Hill declares that she has no conflict of interest; Ms. M Halstead declares that she has no conflict of interest; and Dr. R Coats declares that she has no conflict of interest. This article does not contain any studies with human participants or animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Rights and permissions
About this article
Cite this article
Hill, D., Coats, R.O., Halstead, A. et al. A Systematic Research Review Assessing the Effectiveness of Pursuit Interventions in Spatial Neglect Following Stroke. Transl. Stroke Res. 6, 410–420 (2015). https://doi.org/10.1007/s12975-015-0420-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-015-0420-z