Abstract
The role of drug-coated balloons (DCBs) in patients with acute myocardial infarction (AMI) remains controversial. We performed a meta-analysis of all published studies comparing the outcomes of DCBs vs. stenting in AMI patients. Four studies with 497 patients (534 lesions) were included (three randomized controlled trials and one observational study). During a mean follow-up of 9 months (range 6–12 months), DCBs were associated with similar risk of major adverse cardiovascular events (5% vs. 4.4%; OR 1.24, 95% CI: [0.34, 4.51], p = 0.74, I2 = 35%), all-cause mortality (0.02% vs. 0.04%; OR 077, 95% CI: [0.15, 3.91], p = 0.75, I2 = 25%), cardiac death (0.01% vs. 0.02%; OR 0.64, 95% CI: [0.16, 2.64], p = 0.54), myocardial infarction (0% vs. 1.4%; OR 0.18, 95% CI: [0.01, 3.56], p = 0.26), and target lesion revascularization (3.7% vs. 2%; OR 1.74, 95% CI: [0.42, 7.13], p = 0.44, I2 = 17%) compared with stenting. During a mean follow-up of 7 months (range 6–9 months), DCBs had similar late lumen loss compared with stenting (mean difference 0.04 mm, 95% CI [– 0.21–0.28], p = 0.77, I2 = 92%). In patients with AMI, there was no statistical difference in the incidence of clinical and angiographic outcomes between AMI patients treated with DCB and DES. Larger studies with longer-term follow-up are needed to assess the clinical utility of DCBs in this setting.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Patients with acute myocardial infarction (AMI) represent one of the highest risk subsets of patients undergoing percutaneous coronary intervention (PCI). Despite advances in technique and medical therapy, AMI patients remain at a high risk of cardiovascular events after initial stabilization. Currently, drug-eluting stents (DES) are recommended for PCI in patients with AMI, given their superiority in reducing the need for reintervention compared with bare-metal stents (BMS) [1, 2]. However, stenting remains limited by neointimal hyperplasia and neoatherosclerosis [3]. Long-term follow-up studies have consistently shown an ongoing risk of adverse cardiovascular events after stenting, even with second-generation DES [4, 5]. Stents are also associated with impairment of the coronary endothelial and vasomotor function [6].
Drug-coated balloons (DCBs) offer the advantage of delivering antiproliferative drugs without leaving metal behind. They provide homogenous distribution of the antiproliferative drug along the vessel wall, potentially leading to positive remodeling and inhibiting neointimal growth with eventual plaque reduction and stabilization [7,8,9]. DCBs have been studied in different anatomical and pathological variants of coronary artery disease (CAD). They have been effective in the treatment of coronary in-stent restenosis (ISR) [10] and de novo CAD, including small vessels, bifurcation lesions, and diabetics [11,12,13,14]. Two early feasibility studies of DCBs in AMI showed favorable short-term rates of major adverse cardiovascular events (MACE) [15, 16].
Given the limitations of stenting in patients with AMI and the limited data on the comparative effectiveness of DCBs vs. DES in AMI patients, we performed a meta-analysis to compare short-term outcomes of both modalities.
Methods
Literature search
The study was conducted according to the proposal for conducting and reporting meta-analyses of observational studies (Moose) [17] and Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (PRISMA) [18]. We performed a computerized search through Medline, Embase, and Cochrane databases from January 2000 to June 2020 using the following search terms separately and in combination; “Drug-eluting balloon,” “DEB,” “drug-coated balloon,” “DCB,” “paclitaxel-coated balloon,” “PCB,” and “acute myocardial infarction.” Bibliographies of the retrieved studies were screened for other relevant studies. The search was limited to the English language.
Study selection
We included randomized controlled trials (RCTs) and observational studies that compared outcomes with DCBs vs. stenting (mainly with DES) in the treatment of AMI [including both ST-elevation myocardial infarction (STEMI) and non-ST-elevation myocardial infarction (NSTEMI)]. In the DCB arm, stenting was allowed only as a bailout strategy in case of suboptimal results, defined as persistent residual stenosis, vessel recoil, or flow-limiting dissection. We excluded studies that included planned DCB + stenting.
Data extraction and quality assessment
The data were extracted by two independent investigators (KB, MS) and confirmed by a third investigator (MM). Discrepancies were settled by consensus. Data were extracted on baseline patient characteristics, procedural details, and outcomes of interest. Bias risk of the included studies was assessed using the Newcastle Ottawa Scale for cohort studies [19], and the Cochrane risk assessment tool for RCTs [20].
Study outcomes
The primary outcome of the current study was the incidence of MACE, defined as a composite endpoint of cardiac death, myocardial infarction (MI), and target lesion revascularization (TLR). Secondary outcomes included all-cause mortality, cardiac death, myocardial infarction (MI), and TLR. Angiographic outcomes included late lumen loss (LLL) defined as the difference between post-procedural and follow-up minimal lumen diameter measured by quantitative coronary analysis (QCA). Definitions of outcomes, inclusion, and exclusion criteria are shown in Table S1.
Results were reported at the latest available follow-up time. The total number of patients was used in the analysis of clinical outcomes, while the total number of lesions was used when angiographic outcomes were assessed.
Data synthesis and statistical analysis
Categorical variables were reported as frequencies while continuous variables as means with standard deviations (SD). Categorical variables were compared using Fisher’s exact or Chi-square tests, while continuous variables were analyzed using the two-sample t-test. Tests were two-tailed, and a p-value of ≤ 0.05 was considered statistically significant. All reported baseline characteristics and outcomes are weighted with the sample size being the weight.
Odds ratios (ORs) and mean differences (MD) with 95% confidence intervals (CIs) are presented as summary statistics. Statistical heterogeneity was assessed by I2 statistic: I2 > 50% was considered substantial, and I2 > 75% was considered considerable [21]. We used the Der-Simonian and Laird random-effects and random-effects generic inverse variance methods to calculate OR and MD, respectively, as we anticipated a high degree of clinical and methodological heterogeneity. Potential publication bias was assessed using the Egger’s test by visual examination of the funnel plots [22]. Outcomes were analyzed for all studies using “as-treated” events from RCTs to decrease heterogeneity when combined with the observational study, and a second analysis was performed for RCTs using an “intention-to-treat” analysis for the primary outcome. The following sensitivity analyses for the primary outcome were conducted: (1) including only RCTs (both intention-to-treat and as-treated analyses), (2) DCBs vs. DES only, (3) including only STEMI patients, (4) including only studies that used second-generation DCB, and (5) excluding one low-quality RCT [23], as the study excluded patients who needed bailout stenting in the DCB arm. Statistical analysis was conducted using Review Manager software (Version 5.3.5. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014).
Results
Studies
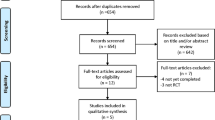
The study selection process is described in Figure S1. Four studies (three RCTs [23,24,25] and one observational study [26]), with 497 patients (534 lesions), met our inclusion criteria (Table 1). Three studies included patients with STEMI [23, 24, 26], and one study included patients with NSTEMI [25]. Three studies used DES [23, 24, 26], while one study included stenting with BMS in 56% of patients [25]. Three studies used second-generation DCBs [23,24,25] while one study used first-generation DCB (DIOR II, Eurocor, Germany) [26] that is no longer commercially available. Publication bias assessment is shown in Figures S2–S7. Bias assessment as per the Cochrane Newcastle Ottawa scale and Cochrane risk assessment tool is illustrated in Tables S2 and S3.
Patients
Patient characteristics are shown in Tables 2 and S4. The DCB group included 244 patients (268 lesions), while the stenting arm included 253 patients (266 lesions). No significant differences in baseline or procedural characteristics were found between either group except greater use of the radial approach in the DCB compared with the stenting arm (86.6% vs. 73.4%, p = 0.028) mainly due to the frequency of radial access in the DCB arm more than DES arm in the included observational study [26].
Of the lesions in the stenting arm, 219 (82.3%) were treated with DES and 170 (64%) were treated with a second-generation DES. The rate of bailout stenting in the DCB arm was 13% (range: 4% to 18%).
Clinical outcomes
Clinical outcomes are summarized in Fig. 1. During a weighted mean follow-up of 9 months (range 6–12 months), DCBs were associated with a similar risk of MACE (5% vs. 4.4%; OR 1.24, 95% CI: [0.34, 4.51], p = 0.74, I2 = 35%), all-cause mortality (0.02% vs. 0.04%; OR 0.77, 95% CI: [0.15, 3.91], p = 0.75, I2 = 25%), cardiac death (0.01% vs. 0.02%; OR 0.64, 95% CI: [0.16, 2.64], p = 0.54), MI (0% vs. 1.4%; OR 0.18, 95% CI: [0.01, 3.56], p = 0.26), and TLR (3.7% vs. 2%; OR 1.74, 95% CI: [0.42, 7.13], p = 0.44, I2 = 17%) (Figure S8).
Both strategies had a similar risk of MACE with different sensitivity analyses when we included only RCTs with intention-to-treat or as-treated analyses, included only DCB vs. DES, included only studies that used second-generation DCBs, included only patients with STEMI, and the low-quality RCT [23] (Figure S9).
Angiographic outcomes
Angiographic follow-up was completed in three studies, all of which studied STEMI patients, including 227 lesions (43% completed follow-up) [23, 24, 26]. During a weighted mean follow-up of 7 months (range 6–9 months), DCB had similar late lumen loss compared with stenting (MD 0.04 mm, 95% CI [– 0.21–0.28], p = 0.77, I2 = 92%) (Figure S8).
A summary of the study results is shown in Fig. 2.
Discussion
The current meta-analysis is the first to compare the clinical and angiographic outcomes in patients with AMI treated with DCBs vs. stenting. We found no difference in the risk of major cardiovascular events or angiographic LLL between groups. Our findings were unchanged when we restricted our analysis to RCTs, and to STEMI patients.
Randomized trials have established that stenting with either BMS or DES results in lower restenosis rates than uncoated balloon angioplasty (BA) [27,28,29]. Some very long-term studies, however, have suggested that, beyond the initial 1–4 years, BA might have a lower risk of TLR compared with BMS and DES over a follow-up period of 9–20 years [30, 31]. These observations have been more pronounced in patients with STEMI, in whom the difference was noted with a landmark analysis after the first year [31]. Moreover, no difference was observed in short- or long-term mortality between BA and stenting in different studies [30,31,32]. Together, these data raise the possibility that balloons could represent an attractive alternative treatment option if their short-term risk of restenosis is similar to that of stents.
In our analysis, DCBs were comparable to stenting in overall MACE and all its components, including TLR, during a mean follow-up of 9 months after PCI. In patients with AMI, there is a higher risk of stent malapposition, uncovered struts, and atherothrombotic prolapse, which is possibly related to residual thrombus burden and incorrect stent sizing in the setting of acute occlusion, increasing the risk of short- and long-term stent-related events [33, 34]. AMI patients also have a higher risk of stent thrombosis and cardiovascular events compared with those without MI in the first 3 months after PCI with stenting [35]. It is plausible that DCBs would provide a similar protective effect in preventing reintervention in the early period after PCI but avoid the longer-term complications of leaving a metallic stent in the coronary arteries and by doing so result in better outcomes in these high-risk patients. These findings support performance of larger and longer-term studies to investigate potential differences in long-term outcomes between the two strategies.
Our results, however, should be interpreted with caution given the limitations of DCB technology. DCBs require optimal lesion preparation to ensure optimal apposition of the drug-eluting surface of the DCB to the endothelium. Therefore, DCBs might not be the first choice in lesions that are challenging to prepare. Like regular BA, DCBs also carry risks of persistent residual stenosis, acute vessel recoil, and dissection, which may require bailout stenting. In our study, the rate of bailout stenting was 13%. DCBs were also shown to adversely affect the endothelial and non-endothelial vasomotor function of the treated segments, but to a lesser extent compared with stenting [26, 36]. Finally, although comparable in terms of hard clinical outcomes including death and MI, DCBs might be associated with higher TLR than DES in some lesion subsets, including coronary ISR [10].
Our study demonstrates that DCBs had comparable angiographic and clinical outcomes when compared with stenting within the year after PCI, a critical period when stenting has been established as superior to BA in preventing reintervention. This suggests that DCBs may be a potential treatment option in patients with AMI, where they may avoid long-term stent-related complications. Larger randomized trials with long-term follow-up would be required to test this hypothesis.
Limitations
Our study has limitations. First, our power to detect differences in clinical outcomes was limited due to the small number of studies, patients, and events. Second, the included studies had a high degree of heterogeneity. Third, one observational study was included, which introduces the risk of selection bias and confounding, although the results did not change when that study was excluded in a sensitivity analysis. Fourth, the included studies of stenting combined the use of BMS and first- and second-generation DES which precluded a comparison between DCB and current-generation DES. Finally, only paclitaxel-coated balloons were used in the included studies. Newer sirolimus-coated balloons have shown promising results [37].
Conclusions
In patients with AMI, there was no statistical difference in the incidence of clinical and angiographic outcomes between AMI patients treated with DCB and DES. Larger studies with longer-term follow-up are needed to assess the clinical utility of DCBs in this setting.
References
Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, Caforio AL, Crea F, Goudevenos JA, Halvorsen S. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2017;39:119–77.
Ogara PT, Kushner FG, Ascheim DD, Casey DE, Chung MK, De Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;61:e78–e140.
Ong AT, McFadden EP, Regar E, de Jaegere PP, van Domburg RT, Serruys PW. Late angiographic stent thrombosis (LAST) events with drug-eluting stents. J Am Coll Cardiol. 2005;45:2088–92.
Vlachojannis GJ, Smits PC, Hofma SH, Togni M, Vázquez N, Valdés M, Voudris V, Slagboom T, Goy J-J, den Heijer P. Biodegradable polymer biolimus-eluting stents versus durable polymer everolimus-eluting stents in patients with coronary artery disease: final 5-year report from the COMPARE II trial (abluminal biodegradable polymer biolimus-eluting stent versus durable polymer everolimus-eluting stent). JACC Cardiovasc Interv. 2017;10:1215–21.
Madhavan MV, Kirtane AJ, Redfors B, Généreux P, Ben-Yehuda O, Palmerini T, Benedetto U, Biondi-Zoccai G, Smits PC, von Birgelen C, Mehran R, McAndrew T, Serruys PW, Leon MB, Pocock SJ, Stone GW. Stent-related adverse events >1 year after percutaneous coronary intervention. J Am Coll Cardiol. 2020;75:590–604.
Hung M-J, Hsu K-H, Chang N-C, Tsimikas S, Hung M-Y. Prevalence of coronary artery spasm after stent placement and its association with inflammation. Int J Cardiol. 2015;179:252–5.
Ann SH, Balbir Singh G, Lim KH, Koo BK, Shin ES. Anatomical and physiological changes after paclitaxel-coated balloon for atherosclerotic de novo coronary lesions: serial IVUS-VH and FFR study. PLoS ONE. 2016;11:e0147057.
Kleber FX, Schulz A, Waliszewski M, Hauschild T, Bohm M, Dietz U, Cremers B, Scheller B, Clever YP. Local paclitaxel induces late lumen enlargement in coronary arteries after balloon angioplasty. Clin Res Cardiol. 2015;104:217–25.
Waksman R, Serra A, Loh JP, Malik F, Torguson R, Stahnke S, Rodriguez A. Drug-coated balloons for de novo coronary lesions: results from the Valentines II trial. EuroIntervention. 2013;9:613–9.
Giacoppo D, Alfonso F, Xu B, Claessen BE, Adriaenssens T, Jensen C, Pérez-Vizcayno MJ, Kang D-Y, Degenhardt R, Pleva L. Paclitaxel-coated balloon angioplasty vs. drug-eluting stenting for the treatment of coronary in-stent restenosis: a comprehensive, collaborative, individual patient data meta-analysis of 10 randomized clinical trials (DAEDALUS study). Eur Heart J 2019.
Megaly M, Rofael M, Saad M, Shishehbor M, Brilakis ES. Outcomes with drug-coated balloons for treating the side branch of coronary bifurcation lesions. J Invasive Cardiol. 2018;30:393–9.
12Megaly M, Ali A, Abraham B, Khalil C, Zordok M, Shaker M, Tawadros M, Hennawy BS, Elbadawi A, Omer M. Outcomes with drug-coated balloons in percutaneous coronary intervention in diabetic patients. Cardiovasc Revasc Med 2019.
Megaly M, Rofael M, Saad M, Rezq A, Kohl LP, Kalra A, Shishehbor M, Soukas P, Abbott J, Brilakis ES. Outcomes with drug-coated balloons in small-vessel coronary artery disease. Catheter Cardiovasc Interv. 2019;93:E277–E286286.
Megaly M, Saad M, Brilakis ES. Role of drug-coated balloons in small-vessel coronary artery disease. US Cardiol Rev. 2019;13:16–20.
Ho HH, Tan J, Ooi YW, Loh KK, Aung TH, Yin NT, Sinaga DA, Jafary FH, Ong PJL. Preliminary experience with drug-coated balloon angioplasty in primary percutaneous coronary intervention. World J Cardiol. 2015;7:311.
Vos NS, Dirksen MT, Vink MA, van Nooijen FC, Amoroso G, Herrman J, Kiemeneij F, Patterson MS, Slagboom T, van der Schaaf RJ. Safety and feasibility of a PAclitaxel-eluting balloon angioplasty in Primary Percutaneous coronary intervention in Amsterdam (PAPPA): one-year clinical outcome of a pilot study. EuroIntervention. 2014;10:584–90.
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA. 2000;283:2008–122.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6:e1000097.
19Wells G. Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. 2011.
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, Savović J, Schulz KF, Weeks L, Sterne JA. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557.
Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34.
Gobić D, Tomulić V, Lulić D, Židan D, Brusich S, Jakljević T, Zaputović L. Drug-coated balloon versus drug-eluting stent in primary percutaneous coronary intervention: a feasibility study. Am J Med Sci. 2017;354:553–60.
24Vos NS, Fagel ND, Amoroso G, Herrman J-PR, Patterson MS, Piers LH, van der Schaaf RJ, Slagboom T, Vink MA. Paclitaxel-coated balloon angioplasty versus drug-eluting stent in acute myocardial infarction: the REVELATION Randomized Trial. JACC Cardiovasc Interv 2019.
Scheller B, Ohlow M-A, Ewen S, Kische S, Rudolph TK, Clever YP, Wagner A, Richter S, El-Garhy M, Böhm M. Bare metal or drug-eluting stent versus drug-coated balloon in non-ST-elevation myocardial infarction: the randomised PEPCAD NSTEMI trial. EuroIntervention. 2020;15:1527.
Nijhoff F, Agostoni P, Belkacemi A, Nathoe HM, Voskuil M, Samim M, Doevendans PA, Stella PR. Primary percutaneous coronary intervention by drug-eluting balloon angioplasty: the nonrandomized fourth arm of the DEB-AMI (drug-eluting balloon in ST-segment elevation myocardial infarction) trial. Catheter Cardiovasc Interv. 2015;86:S34–S44.
Serruys PW, De Jaegere P, Kiemeneij F, Macaya C, Rutsch W, Heyndrickx G, Emanuelsson H, Marco J, Legrand V, Materne P. A comparison of balloon-expandable-stent implantation with balloon angioplasty in patients with coronary artery disease. N Engl J Med. 1994;331:489–95.
Fischman DL, Leon MB, Baim DS, Schatz RA, Savage MP, Penn I, Detre K, Veltri L, Ricci D, Nobuyoshi M. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. N Engl J Med. 1994;331:496–501.
Suryapranata H, Ottervanger J, Nibbering E, van’t Hof A, Hoorntje J, de Boer M, Al M, Zijlstra F. Long term outcome and cost-effectiveness of stenting versus balloon angioplasty for acute myocardial infarction. Heart 2001;85:667–71.
Yamaji K, Kimura T, Morimoto T, Nakagawa Y, Inoue K, Kuramitsu S, Soga Y, Arita T, Shirai S, Ando K. Very long-term (15 to 23 years) outcomes of successful balloon angioplasty compared with bare metal coronary stenting. J Am Heart Assoc. 2012;1:e004085.
Brodie BR, Pokharel Y, Garg A, Kissling G, Hansen C, Milks S, Cooper M, Mcalhany C, Stuckey TD. Very late hazard with stenting versus balloon angioplasty for ST-elevation myocardial infarction: a 16-year single-center experience. J Interv Cardiol. 2014;27:21–8.
De Luca G, Suryapranata H, Stone GW, Antoniucci D, Biondi-Zoccai G, Kastrati A, Chiariello M, Marino P. Coronary stenting versus balloon angioplasty for acute myocardial infarction: a meta-regression analysis of randomized trials. Int J Cardiol. 2008;126:37–44.
Leone AM, Rebuzzi AG, Burzotta F, De Maria GL, Gardi A, Basile E, Cialdella P, Damario D, Paraggio L, Porto I. Stent malapposition, strut coverage and atherothrombotic prolapse after percutaneous coronary interventions in ST-segment elevation myocardial infarction. J Cardiovasc Med. 2019;20:122–30.
Gonzalo N, Barlis P, Serruys PW, Garcia-Garcia HM, Onuma Y, Ligthart J, Regar E. Incomplete stent apposition and delayed tissue coverage are more frequent in drug-eluting stents implanted during primary percutaneous coronary intervention for ST-segment elevation myocardial infarction than in drug-eluting stents implanted for stable/unstable angina: insights from optical coherence tomography. JACC Cardiovasc Interv. 2009;2:445–52.
Yamaji K, Natsuaki M, Morimoto T, Ono K, Furukawa Y, Nakagawa Y, Kadota K, Ando K, Shirai S, Watanabe H. Long-term outcomes after coronary stent implantation in patients presenting with versus without acute myocardial infarction (an Observation from Coronary Revascularization Demonstrating Outcome Study-Kyoto Registry Cohort-2). Am J Cardiol. 2015;116:15–23.
Plass CA, Sabdyusheva-Litschauer I, Bernhart A, Samaha E, Petnehazy O, Szentirmai E, Petrasi Z, Lamin V, Pavo N, Nyolczas N, Jakab A, Murlasits Z, Bergler-Klein J, Maurer G, Gyongyosi M. Time course of endothelium-dependent and -independent coronary vasomotor response to coronary balloons and stents. Comparison of plain and drug-eluting balloons and stents. JACC Cardiovasc Interv 2012;5:741–51.
Ali RM, Kader MASA, Ahmad WAW, Ong TK, Liew HB, Omar A-F, Zuhdi ASM, Nuruddin AA, Schnorr B, Scheller B. Treatment of coronary drug-eluting stent restenosis by a sirolimus-or paclitaxel-coated balloon. JACC Cardiovasc Interv. 2019;12:558–66.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Emmanouil Brilakis: consulting/speaker honoraria from Abbott Vascular, American Heart Association (associate editor Circulation), Amgen, Biotronik, Boston Scientific, Cardiovascular Innovations Foundation (Board of Directors), ControlRad, CSI, Ebix, Elsevier, GE Healthcare, InfraRedx, Medtronic, Siemens, and Teleflex; research support from Regeneron and Siemens. Shareholder: MHI Ventures. M. Nicholas Burke: Shareholder, Egg Medical and MHI Ventures. Speaker, Opsens Medical. Santiago Garcia: Consultant for Surmodics, Osprey Medical, Medtronic, Edwards Lifesciences, and Abbott Vascular. Dr. Institutional grant support from Edwards Lifesciences, Abbott Vascular, and BSCI. Honoraria from the American College of Cardiology. Stephane Rinfret: Consultant, Boston Scientific, Abiomed, Abbott Vascular and Soundbite Medical. J Dawn Abbott: Research consultant for Pfizer and Recork. Herbert Aronow: Consultant, Silk Road Medical, Philips. All other authors have nothing to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Megaly, M., Buda, K.G., Xenogiannis, I. et al. Systematic review and meta-analysis of short-term outcomes with drug-coated balloons vs. stenting in acute myocardial infarction. Cardiovasc Interv and Ther 36, 481–489 (2021). https://doi.org/10.1007/s12928-020-00713-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12928-020-00713-y