Abstract
Asian people depend on rice for its significant nutritional value. The depreciating effect of cadmium (Cd) on rice crops is notably impulsive, leading to high changes in field and negotiating consumer health. A regional two-year survey of 30 fields of the district North 24 Parganas was conducted to study soil parameters contributing Cd uptake in rice grains and subsequently to compare phytochemical properties of seeds among 3 common cultivars. Based on soil Cd concentration, rice cultivars grown in high Cd (HCd) and low Cd (LCd) contaminated fields were chosen. The efficacy of grain nutrition status of each cultivar with HCd and LCd was compared. Our results revealed that soil pH, organic matter, carbonate, and minerals pronouncedly affected Cd accumulation in grains. Cd-exposed Khitish HCd grains displayed significant reduction of phytochemicals like sugar, phenol, thiamine, ascorbic acid, and riboflavin contents imposing major nutritional degradation of grains compared to Khitish LCd. Contrarily, Pratiksha grains exhibited moderate levels of degradation while Maharaj HCd showed the least alteration in phytochemical status with minimum-grain-quality degradation compared to other HCd cultivars. The element analysis also confirmed that the highest Cd content in Khitish grains holds this cultivar as Cd accumulator while low Cd in Maharaj grain makes this “Cd safe” cultivar. This investigation may be recommended to avert consumption of such rice cultivars that accumulate high Cd in grains, and precautions should be drawn to limit Cd accumulation in rice grains by implementing Cd remediating agents for better food security.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Increasing occurrences of numerous abiotic stresses together with ever-increasing population are putting major constraints for achieving global food security. Rice (Oryza sativa L.), the most popular staple food, is very much prone to various abiotic stress, among which heavy metal stress is one of the major environmental issues in the world agricultural market (Maksymiec 2007). Cd is a potent toxic, non-biodegradable heavy metal, and hazardous soil contaminant with a long biological half-life (Ismael et al. 2019). Cd is released into the environment by mining, electroplating, sludge dumping, and smelting and gets accumulated in agricultural soil due to overuse of phosphate fertilizers and contaminated irrigated water (Kubier et al. 2019). Cd stress imposes detrimental effects on plants like poor growth, turgor loss, inhibiting respiration, leaf chlorosis, poor seed germination rate, necrosis, genotoxicity, crippled photosynthetic apparatus, dysfunction of protein, membrane rupture, and anti-oxidative disparity through the generation of reactive oxygen species (Arif et al. 2019). Cd accumulation in plants depends on several edaphic factors like soil pH, soil texture, availability of organic matter, presence of other minerals, nitrogen source, soil microorganisms, etc. (Sarwar et al. 2010; Hussain et al. 2020). Jeopardizing effect of Cd stress on rice crops is considerably more challenging and complicated, generating effective impairments in the cropland. According to the report on the National General Survey of Soil Contamination by Chinese officials, 16.1% of China's farmland had soil pollution levels over the threshold for heavy metals, and 7% of it was contaminated with Cd (Niu et al. 2022). Being highly mobile, Cd is easily translocated from soil to grains and consequently compromises consumer health menace via entering food chain (Grant et al. 1998; Paul et al. 2022) (According to WHO, PTMI of Cd is 25 µg/kg BW/month).
In India, 40 out of 424 water quality monitoring stations, Ganga, Kopili, Tungabhadra, Rapti, and Yamuna Rivers are highly contaminated with Cd, with levels exceeding 3 µg/L and going up to 7.8 µg/L. Vautha water quality monitoring station at Sabarmati River (Gujarat) revealed the highest levels of hazardous Cd content (70.51 µg/L). Uttar Pradesh, Assam, Gujarat, Karnataka, Arunachal Pradesh, Madhya Pradesh, Andhra Pradesh, Rajasthan, and Haryana are the major states affected by Cd contamination of water. West Bengal is less affected (Source: Department of Water Resources, River Development and Ganga Rejuvenation, 2019). Based on most recent available data, Bichhri in Rajasthan has no fertile land left for agriculture due to large clusters of industries. However, recent information has revealed that enrichment factor of Cd is 45 in Indian agricultural land (Xiaofang and Dongmei 2019). Within West Bengal, Dhapa-Bantala (Garbage farming), a small quaint locality in EM Bypass, Kolkata, is one of the richest Cd-contaminated areas. Average soil Cd concentration in this site was about 0.721 (µg/g dry weight) recorded by Banerjee et al. (2010).
According to recent reports, overuse of phosphate fertilizers and application of organophosphate pesticides are prominent contributors to Cd contamination in arable soil (Yang et al. 2020; Alengebawy et al. 2021). Wastewater-contaminated irrigation promotes Cd deposition in soil (Lien et al. 2021). Allowable limits of uncontaminated soil Cd are less than 0.5 mg/kg, yet this can be tolerated up to 3.0 mg/kg of soil subject to soil chemistry (Zhang et al. 2021). Cd bioavailability is regulated by soil physicochemical properties (pH, electrical conductivity, and soil organic matter) (Li et al. 2018a) and extractable soil Cd content (Barman et al. 2020) while Cd accumulation is rice genotype-dependent (Li et al. 2018b; Barman et al. 2020). Acidic pH promotes metal solubility, thus facilitating heavy metals (HMs) accumulation in rice grain (Yu et al. 2016) while soil organic matter sequesters HMs, making it less bio-available.
Based on soil Cd concentration and bio-concentration factors, three rice cultivars (Maharaj, Khitish, and Pratiksha) with high and low Cd accumulation abilities were collected from high Cd-contaminated (HCd), and low Cd (LCd) contaminated sites and their grain nutritional status were explored. Rice grains were collected from same fields during the harvesting season in May and November for two consecutive years—2021 and 2022. HCd cultivars are better capable of Cd uptake, translocation, and accumulation (Guo et al. 2018) than LCd cultivars. Rice, the grain of life, is the major source of starch, protein, and micro-nutrients. Rice has more tendency to absorb Cd in comparison to other crops (Lien et al. 2021). Oral intake of Cd-contaminated rice grain increases health risk as humans lack natural Cd disposal pathway other than urine, which is not enough as Cd is sequestered by metallothionein and reabsorbed in renal tubule (ATSDR 2006). Long-term exposure to Cd affects the nervous system, brain, kidney, gastrointestinal tract, and bone density (Jehan et al. 2018). Rice carcinogenicity is a well-known fact (Lien et al. 2021). Hence for health risk assessment study, the carcinogenic risk has been estimated in terms of cancer risk (CR) factor and weekly oral intake.
According to FMO, rice supplies 20% of the world’s dietary energy. Asian people depend on rice mostly for its caloric value (45%), total protein (40%), nutrients (thiamine, riboflavin, and niacin), etc. (Wordu et al. 2021). In recent years, Chinese and Korean people are quite aware of the health benefits of wild rice grain because of its protein, minerals, and vitamins providing antihypertensive, anti-allergic, anti-cholesterolemic, anti-inflammatory, and immunomodulatory properties (Yu et al. 2020). Furthermore, it was observed that nutritional compositions of rice cultivars vary from one another depending on climate, irrigation, and fertilizer utilization (Surendran et al. 2021). Since each cultivar has distinct nutritional distribution, it is imperative to compare varietal differences in grain nutritional values in response to field Cd contamination and to assess the degree of nutritional degradation, which will be helpful in selecting Cd safe cultivars for human consumption. In this context, this study evaluated nutrient and anti-nutrient (phytic acid) compositions in rice grains of 3 cultivars collected from low Cd-contaminated (average Cd concentrations in soil < 1 mg kg−1) and high Cd-contaminated (average Cd concentrations in soil > 1 mg kg−1) fields.
Materials and methods
Study area
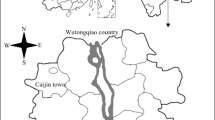
North 24 Parganas District in West Bengal was selected for this study. The area of the district is 4094 km2. Rice, jute, oilseeds, wheat, pulses, and potato are mainly cultivated in this area. The average yield of rice within this district is 3540 kg/ha. Though water of River Ganga is primarily used for irrigation, wastewater irrigation is a common practice for 8–10% land of our study area, mainly mouzas of Barrackpore II block. The entire sampling location map is given in Fig. 1 and Table 1. Total Cd-contaminated study area is classified into two zones based on Cd concentration in soil (Table 2). Details of 3 locally available rice cultivars are specified in Table 3.
Collection of grain and soil samples
Rice grains and corresponding rice field soil samples were collected directly from the field during harvesting season (May and November of 2021 and 2022) via direct communication with the local farmers and the district administration office of N 24 Parganas, West Bengal. Several grains from individual plants were randomly collected from the sampling area and then mixed to give a composite grain sample of each field. In order to eliminate possibility of incorrect sampling, soil samples were collected in zip lock packets, 5 sub-samples were taken from each field of each site at a depth of 10–20 cm using a spud. All sub-samples were mixed to prepare a composite sample of each field. Soil samples were air-dried for 5–7 days and ground to pass through a 1 mm nylon mesh. Metal analyses of the plant and soil samples were carried out by acid digestion (3:1 Conc. HNO3 and Conc. HClO4 v/v) of accurately 1 g of plant tissue and soil samples, respectively.
Riboflavin content
To assay riboflavin content, about 1 g powdered rice grain was homogenized with 50% ethanol, following filtration collected supernatant was mixed with 2.5 ml 5% KMnO4 and 30% H2O2 solution. The whole extract is placed in water bath at 80 °C for 30 min. Then, 1 ml 40% Na2SO4 was added, and riboflavin contents were estimated by measuring absorbance at 510 nm as described previously by Nandagoapalan et al. (2016). Riboflavin content was expressed as μg/Kg DW.
Thiamine content
The thiamine contents in grains were estimated by the method proposed by Bhattacharyya and Roy (2018). Thiamine was extracted from 1 g of crude powdered rice grain by blending with 10 ml of 20% ethanolic NaOH. After that, assay mixture contained 1 ml filtrate and 1 ml of 2% K2Cr2O7 solution, and absorbance was monitored at 360 nm. The result of thiamine content was expressed as μg/Kg DW.
Assay of soluble sugar and starch
Total soluble sugar content of grain was measured by following the details of Dubois et al. (1956) protocol. About 1 g of powdered rice grain sample was homogenized with 80% ethanol followed by centrifugation for 20 min at 2000 rpm. Alcoholic extract was done by phenol–sulfuric acid reagent and expressed as mg/g DW. These end residues collected after centrifugation were suspended in 2.5 ml d.H2O. 3.25 ml HClO4 was added to suspension and centrifuged. Final volume of resultant supernatant was made upto 50 ml with d.H2O, and starch content was measured from filtrate by phenol–sulfuric acid reagent at 490 nm according to method of McCready et al. (1950). The amount of total starch was expressed as g/g DW.
Determination of total protein content
The protein content from 1 g powdered sample has been estimated by the method previously described by Lowry et al. (1951). Total protein contents were expressed as mg/g DW.
Ascorbic acid (AsA) estimation
The method of Okolie et al. (2014) was followed to estimate AsA content. About 100 mg powdered sample was blended with 1.5 ml of 10% TCA and centrifuged. The final reaction mixture was obtained by adding 0.5 ml supernatant with 0.5 ml DNPH reagent. After 3 h of incubation period at room temperature (RT), 2.5 ml of 85% H2SO4 was added slowly, and absorbance was taken at 530 nm after 30 min of incubation. AsA content was expressed as μg/Kg DW.
Phenol estimation
Phenol extraction was carried out using the Folin–Ciocâlteu reagent using gallic acid as standard. About 1 g of powdered grain samples was homogenized with 10 ml of 80% ethanol and centrifuged. After that, 200 μl of supernatant was added with 800 μl diluted Folin–Ciocâlteu reagent and 2 ml of sodium carbonate (7.5%). The resulting reaction mixture was incubated for 30 min in dark. The absorbance was determined at 765 nm using UV/VIS spectrophotometer (Su et al. 2007). Total phenolics were expressed as mg/g DW.
Flavonoid estimation
Total flavonoid concentration was quantified according to the method of Lamaison and Carnet (1990). The methanolic extract of powdered grain was evaporated to dryness in hot air oven. The end residue was mixed with 10 ml of aluminum chloride solution and kept at RT for 30 min. The intensity of resultant yellow color sample was estimated at 420 nm. The result of flavonoid content was expressed as μg quercetin/g DW.
Phytic acid content
The phytic acid content was estimated following the method of Vaintraub and Laptera (1988). About 500 mg powdered rice grain samples were homogenized with 10 mL of 3.5% HCl. After stirring for 1 h, mixture was cold-centrifuged at 10,000 g for 10 min. Next 1 mL of supernatant was diluted with 2 mL of 3.5% HCl and 1 mL Wade reagent, and centrifuged again. The absorbance was taken at the wavelength of 500 nm using spectrophotometer and expressed as g/Kg.
Fatty acid analysis
Fatty acids in rice grains were estimated by the method of Garces and Mancha (1993). For methylation of fatty acid, 1 ml of methylation reaction mixer carrying methanol: n-heptane: benzene: DMP: H2SO4 (37:36:20:5:2) was mixed to 100 mg rice powder taken in 2 ml microfuge tube and incubated for 2 h at 80 °C in water bath. Next, the tube was cooled at RT, mixture was allowed to separate into two phases, and the upper layer containing fatty acid methyl ester was analyzed using Gas Chromatograph with high-resolution Mass Spectrometer (GC-HRMS) (AccuTOF GCV).
Quantification of macro- and micro‑nutrient composition by ICP‑AES
Rice grains were dried in hot air oven at 80 °C for 72 h prior to estimation of nutrient contents. The oven-dried samples were then ground into fine powder following digestion with a tri-acid mixture of HNO3: HCl: HClO4 (4:2:1), and the solutions were adjusted to 25 ml with deionized water. The concentration of macronutrients (Ca, K, and Mg) and micro-nutrients (Zn, Fe, and Na) and Cd in digested solution was determined using Inductively Coupled Plasma-Optical Emission Spectroscopy instrument (ARCOS, Spectro, Germany).
Estimation of bio-available Cd in the soil samples
Bio-available Cd contents in soil samples were determined according to the procedure of Mohamadiun et al. (2018). First, collected soil samples from each field were oven-dried at 60 °C for 48 h. About 1 g of fully dried soil samples was dissolved in 15 ml d.H2O and the soluble fraction was collected after incubation for 24 h using a shaker overnight. After every process, sample was quantified for Cd concentration by the UV/VIS spectrophotometric method. The soil precipitation was washed with d.H20 repeatedly and again dried overnight at 60◦C. Cd content in soil fractions was determined by referencing a standard curve of Cd. Soil parameters, such as pH and EC, were also measured.
Statistical data analysis
All lab experiments were conducted in triplicates and all data were represented as an average of triplicates. All experimental data were statistically analyzed using two-way analysis of variance (ANOVA) test. Heat map was generated to determine the degree of nutritional degradation among 3 cultivars. The results of statistical analysis were performed in SPSS 23 and Past3 software.
Results
Grain qualities
Vitamin B complexes like riboflavin and thiamine have already been recognized in a wide range of rice landraces throughout countries (Asaduzzaman et al. 2013). As shown in Fig. 2A, the percentage degradation of riboflavin content in Khitish HCd collected in 2021 (12.64%) and 2022 (14.52%) was significantly higher and Pratiksha HCd with no significant alteration in riboflavin content relative to that of Pratiksha LCd. In grains collected in 2021, the data recorded significant depletion of thiamine content (Fig. 2B) in Khitish (30.93%) and Pratiksha (17.59%) HCd than Maharaj HCd (6.86%). Similarly, in grains collected in 2022, thiamine content was reduced in the following order—Khitish (36.94%), Pratiksha (12.82%) and Maharaj (7.34%).
Effects of Cd on rice grain qualities—riboflavin (A), thiamine content (B), total soluble sugar (C) and starch content (D) in LCd and HCd soil for years 2021 and 2022. The error bars in this figure indicate standard deviation obtained from the mean value of three individual replicates. Bars followed by (*), (**), (***) and (****) represent significant differences at P ≤ 0.05, P ≤ 0.01, P ≤ 0.001, P ≤ 0.0001 level, respectively. (ns): P > 0.05
All the rice HCd sets had significantly declined soluble sugar contents (Fig. 2C) than their LCd counterparts. Reduction of sugar concentration in ‘Khitish’ was 18.93% and 26.57%, in Pratiksha was 5.07% and 4.6% in the years 2021 and 2022, respectively, while in Maharaj HCd, sugar concentration was elevated by 1.25% in 2021. The percentage of starch content was not uniform in both seasons. Prominent reduction of starch content (Fig. 2D) was observed in Khitish (7.06% and 6.02%) in both years, and enhancement was noted in Pratiksha (6.83%) and Maharaj (4.5%) in 2021 grains, while grains in 2022, starch content declined significantly in Maharaj (16.64%) and Pratiksha (17.56%). In 2021 and 2022, a significant decrease in total protein content (Fig. 3A) of Khitish HCd (40.75%) and (21.14%) was documented, respectively.
Effects of Cd on rice grain qualities—total protein (A), ascorbic acid content (B) in LCd and HCd soil for years 2021 and 2022. The error bars in this figure indicate standard deviation obtained from the mean value of three individual replicates. Bars followed by (*), (**), (***) and (****) represent significant differences at P ≤ 0.05, P ≤ 0.01, P ≤ 0.001, P ≤ 0.0001 level, respectively. (ns): P > 0.05
In contrast, total protein (Fig. 3A) of Pratiksha and Maharaj HCd was statistically insignificant from their LCd set. In comparison to Maharaj HCd, AsA content (Fig. 3B) was found to be decreased significantly in Khitish (35.33%, 24.8%) and Pratiksha (23.86%, 13.94%) in both seasons, respectively. A decreasing trend of the phenol and flavonoid content of all HCd was observed (Fig. 4). The study revealed significant reduction of flavonoid (Fig. 4B) (28.73% and 24.27%), (6.11% and 6.47%) and (1.29% and 2.2%) and phenol content (Fig. 4A) of grains (29.1% and 18.56%), (28.07% and 14.14%) and (3.87% and 4.48%) of Khitish, Pratiksha and Maharaj in the year 2021 and 2022, respectively. Phytic acid content (Fig. 4C) was mentioned to be significantly increased in Khitish HCd (57.14%, 51.93%) and Pratiksha HCd (30.17%, 28.18%), whereas in Maharaj, the value remained the least altered in both seasons.
Effects of Cd on rice grain qualities—phenol (A), flavonoid (B) and phytic acid content (C) in LCd and HCd cultivars for the years 2021 and 2022. Error bars in this figure indicate standard deviation obtained from the mean value of three individual replicates. Bars followed by (*), (**), (***) and (****) represent significant differences at P ≤ 0.05, P ≤ 0.01, P ≤ 0.001, P ≤ 0.0001 level, respectively. (ns): P > 0.05
In both years, due to soil Cd contamination, fatty acid profile (Fig. 5) of Khitish and Pratiksha was affected significantly in comparison with Maharaj grain.
Cd accumulation has an impact on mineral distribution and uptake in grains. In both seasons, the nutritional degradation was approximately uniform, which was evident from significant depletion of some nutrients in grains like Fe, Na, Ca, and K, but very minimal increase in Mg concentration in all 3 cultivars was noted (Table 4).
For health risk assessment, the rate of oral Cd intake through consumption of contaminated rice grains was calculated. Results of Cd content, bio-concentration factor (BCF), oral weekly intake and cancer risk factor in low Cd and high Cd-contaminated cultivars collected in the years 2021 and 2022 were encapsulated in Table 5. It was noticed that maximum level of Cd in grain was in Khitish, which was followed by higher levels of oral weekly intake of Cd. The increment in weekly intake of Cd positively increased CR factor. Moderate level of grain Cd was found in Pratiksha. On the contrary, the lowest Cd accumulation was found in Maharaj, and also dietary Cd intake risk and CR factor were compressed.
The significant differences in pH and EC values of soil were estimated, which are given in Fig. 6. Here, more acidic pH was recorded in the field of Khitish (Fig. 6A) than in other 2 fields. High Cd-contained Khitish soil had relatively higher amounts of Cd in soluble, exchangeable and carbonate fractions and lower level of non-bio-available Cd content (residual, organic, and oxide-hydroxide) relative to Khitish LCd soil which is listed in Table 6. Also, Pratiksha HCd contained moderate amounts of Cd for soluble, exchangeable, carbonate fraction, oxide–hydroxide, residual, and organic matter, which is also mentioned in Table 6. But Maharaj soil contained higher residual Cd, organic matter and pretty negligible bio-available Cd.
Effect of Cd on soil pH (A), soil EC (B) in LCd and HCd fields for the years 2021 and 2022. The error bars in this figure indicate standard deviation obtained from the mean value of three individual replicates. Bars followed by (*), (**), (***) and (****) represent significant differences at P ≤ 0.05, P ≤ 0.01, P ≤ 0.001, P ≤ 0.0001 level, respectively. (ns): P > 0.05
Discussion
Heavy metal toxicity and nutritional value monitoring in rice grain have been recommended as oral consumption of contaminated grains may have significant long-term health risks. Maharaj, Pratiksha, and Khitish are Cd-accumulating rice cultivars. Thus, if cultivated in Cd-contaminated agricultural lands, grain Cd accumulation will be facilitated, and intake of such type of contaminated nutritionally compromised grains may cause serious health problems. To address this global issue, nutritional quality alteration of low Cd-accumulating grains and high Cd-accumulating grains of each cultivar have been investigated.
Phytochemicals are significant bioactive non-nutrient chemical compounds with antioxidant capabilities, found in plant parts like vegetables, fruits, and whole grains, which reduce the risk of chronic diseases (hypertension, heart disease, diabetes, cancer, and other undesirable clinical disorders) (Liu 2013; Arendt and Zannini 2013; Guha et al. 2021).
Riboflavin content was found to be barely degraded in Maharaj maybe because of the cultivar restoring nutritional profile, mitigating free radicals via glutathione redox cycle and under Cd stress (Zeddies et al. 2014; Acosta-Estrada et al. 2019). Thus, Maharaj may likely be safer for consumption. Banerjee and Roychowdhury (2019) have reported that riboflavin content gradually dropped in fluoride-stressed rice seedlings. Interest in thiamine content has grown in the past decade as its deficiency leads to beriberi (Acosta-Estrada et al. 2019). Health-promoting properties of thiamine in controlling metabolism, nerve cell damage, maintaining a healthy cardiovascular function, boosting immunity, and alcoholism treatment are well known (Khalifa et al. 2016; DiNicolantonio et al. 2018). The thiamine content in Maharaj HCd is pretty less compromised than Pratiksha and Khitish HCd which indicates that Maharaj is likely nutritionally richer than Khitish and Pratiksha. Besides this, thiamine content in Maharaj breaks carbohydrate from rice grains to produce sufficient energy (Bhattacharya and Roy 2018). The sugar content was downgraded due to high Cd toxicity in grains. But greater loss of sugar content in Khitish HCd and Pratiksha HCd indicates poor grain quality with compromised nutritional value for consumption and disables to regulate blood glucose level and insulin metabolism, fatty acid metabolism, energy supply, bile acid dehydroxylation, protein glycosylation, etc. (Wylie-Rosett et al. 2013), whereas no remarkable reduction of sugar contents in Maharaj grain indicates its better contribution to release of brain chemicals like serotonin (Singh et al. 2018). The lesser decay of protein content in Maharaj HCd than other HCd grains may be possible as the cultivar is competent enough to withstand Cd genotoxicity and possesses long chain amino acids that catalyze important biochemical reactions (Singh et al. 2018). Being a primary nutrient in rice, protein accomplishes vital functions throughout human body system and validates health benefits of Maharaj for consumption (Singh et al. 2018). Ascorbic acid is another appreciated ingredient of our diet chart. The content of AsA was not very much affected in Maharaj HCd grain which could be linked to possessing effective antioxidant system, which scavenges free radicals generated by Cd toxicity. AsA is efficiently employed in collagen, skin, tendons, ligaments, blood vessels and certain neurotransmitters synthesis for better functioning in human body. So the effects of AsA on human health and nutrition are substantially considerable (Bhattacharya and Roy 2018; Acosta-Estrada et al. 2019). ROS generation in plants is directly correlated to stress. Flavonoids are polyphenolic compounds and potent antioxidants (Asaduzzaman et al. 2013). The minimal degradation of flavonoid content in Maharaj grain marks its better Cd tolerance and hence capable of scavenging free radicals, mitigating oxidative cell damage, controlling inflammation, allergic and carcinogenic activity (Wordu et al. 2021; Bhattacharyya and Roy 2018). Phenolic compounds are key phytochemicals in plants for having hydroxyl groups to confer scavenging ability and control chronic inflammatory diseases in human beings (Eleazu et al. 2020). Limited degradation of phenol in Maharaj grain suggested better management of Cd stress via retardation of lipid peroxidation and acts as immune enhancer in humans, whereas compromised phenol and flavonoid production in Khitish HCd could be attributed to inferior PAL activity, unable to withstand stress with exposure to pathogens resulting severe damage in grain nutritional value (Bhattacharyya and Roy 2018). As a result of fluoride stress, sensitive rice cultivar fails to accumulate the antioxidant, resulting in declined rate of flavonoids in rice grains (Banerjee and Roychowdhury 2019). Phytic acid, familiar as an anti-nutrient, is a major storage form of phosphorus in grains and chelates calcium, zinc, and iron and exerts inhibitory action in proteolytic digestion of monogastric animals, including humans (Wordu et al. 2021). Here pronounced phytic content in Khitish and Pratiksha impedes the bioavailability of these minerals, leading to impairment of Fe-catalyzed oxidative reactions, rendering iron in its inert form (Goufo and Trindade 2014). In our data, phytic acid declined in Maharaj grain, which is desirable and suitable for consumption. This result agreed with the previous finding, which mentioned the anti-nutrient effect of phytic acid in rice grains (Khan et al. 2019).
Fatty acids are the fundamental building units of fats and lipids. It has diverse functions including establishment of healthy cell membranes, proper working of the brain and nervous system, blood pressure regulation, and hormone secretion (Andersson et al. 2015; Ristić-Medić et al. 2013). Interestingly, our results revealed positive correlation between fatty acid composition of Khitish and Pratiksha, which is congruent with the finding that these cultivars can be considered as Cd accumulator (Majumder et al. 2022).
The exposure to high Cd caused alteration in grain macro- (K, Ca, and Mg) and micro- (Fe, Zn, and Na) nutrients, perhaps due to restricted translocation to the shoot and accumulation in grains. This can be supported by the fact that Cd and mineral nutrients not only share physical and chemical similarities but also use same transporters for uptake (Hussain et al. 2021). Cd substitutes Ca2+ in Ca-calmodulin signaling pathway by restricting enzymatic activity and gene expression that are involved in signal transduction pathway (de Araujo et al. 2017). The correlation of Fe content under Cd stress has implicated synergistic interactions between absorption and translocation of Cd and Fe, which is congruent to the previous findings in rice plants (Liu et al. 2003). Here, ample macro- and micro-mineral deficiencies in Khitish and Pratiksha cultivars pose severe health threats which are not preferable. This data is further supported by the report of Raghuvanshi et al. (2022) where they have revealed that Arsenic and Cd toxicity disrupted the balance of minerals in rice grains, reducing crop output.
Our results manifested disparity in Cd accumulation, which can be substantiated by the fact that cultivars may possess variability in Cd transporter, and grain accumulation acts as a prime source of Cd that enters our body through the food chain (Takahashi et al. 2011). According to CODEX, the recommended dietary Cd level in rice grain must be below 0.2 mg/kg. According to the report of WHO, provisional tolerable monthly intake (PTMI) of Cd is near about 7 μg/kg body weights per week. Our evaluation suggests that oral weekly intake of Cd upon consumption of Khitish and Pratiksha grain (high Cd-accumulating cultivars) exceeded 15 μg kg−1 body weight. Likewise, these 2 cultivars highly correlated with the increased level of CR that can be approved by the results of Majumder et al. (2022). The value of CR of Maharaj grain (Cd safe) is out of concern.
The assessment of soil Cd content makes our data more obvious. Our results revealed an indirect linear relationship between soil pH and Cd content in grains due to low soil pH influencing metal solubility and Cd bioavailability by altering the chemical speciation, which is in accordance with previous reports (Guo et al. 2020; Daulta et al. 2021). Increased amount of soil pH in field area of Maharaj led to Cd immobilization through better adsorption (Zhao et al. 2014; Mu et al. 2020). This data was similar to the previous finding by Li et al. (2020), where Cd-polluted calcareous paddy soil improved nutrient profile of soil by lowering soil Cd content. Fahad et al (2015) reported that bio-availability of Cd in soil governs the degree of toxicity rendered by Cd on croplands, as bio-available Cd is only conveyed to whole plant parts. Cd forms complexes with different soil particles like soil geological elements, oxides, organic matter, inorganic components, and soil solid particles (Simmler et al. 2013). It is reported that increased organic matter levels in LCd soils are believed to be key factors in lowering soluble Cd phyto-availability (Saengwilai and Meeinkuirt 2021). From our experiments, the exchangeable Cd fraction has a lofty capability to emigrate and consequently make direct availability for plants, whereas residual Cd has high stability, so non-bio-available for plants (Memoli et al. 2018). Barman et al. (2020) also demonstrated an increase in bio-available Cd fraction (soluble, exchangeable, and carbonate fractions) in high Cd-containing soil. A higher rate of Cd binding with the organic matter was noted in all LCd cultivars which is associated with the least amount of available Cd content with respect to HCd cultivars. Moreover, Maharaj HCd shows better efficiency to diminish phyto-available Cd in soil than other Cd accumulator cultivars and sequentially reduces the translocation of Cd to the grain which is established in our results. To understand the variation in Cd accumulation in rice cropping zones due to seasonal rainfall, we collected rainfall data. Surprisingly, the average annual rainfall of LCd field sites was about 1800 mm with high precipitation triggering Cd ions to submerge into deeper soil, thereby reducing Cd ion transport from roots and shoots to grains by dropping transpiration rate. Rather, HCd fields that received low annual rainfall (average 1600 mm) might be accelerating transpiration rate of rice plants, assisting Cd transport from deep soil layers to vicinity of the root, from roots to shoots and grains (Gill et al. 2014; Liu et al. 2017). Hence, water levels in paddy fields are predominantly controlled by different rainfall amounts and they are a salient factor for generating variation in Cd availability and accumulation in plants.
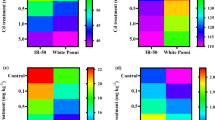
A clustered heat map was created with contrasting color intensity to visualize all considered nutritional parameters, where it was obvious that grain nutritional quality degradation was higher in Khitish and Pratiksha than Maharaj in every aspect and indicates poor efficiency of these two cultivars to resist Cd toxicity (Fig. 7).
To best of our knowledge, this is the first reported attempt where Cd-inflicted nutritional degradation of rice grains has been studied, justifying the potential beneficial role of Maharaj cultivar compared to Pratiksha and Khitish in nutrition of people within the study location.
Conclusion
Cd accumulation in rice grains affects nutritional quality and makes it unsafe for consumption. Therefore, it becomes crucial to have an idea of the nutritional quality degradation of rice cultivars due to Cd contamination in fields. Based on nutritional quality deterioration of rice grains, we concluded that the Maharaj cultivar was superior to Khitish cultivar. Maharaj grain was offered with minimal nutritional degradation while Khitish rice cultivar displayed significant reduction of phytochemicals along with high content of anti-nutritional phytic acid. Therefore, this study envisages urgent need to reduce consumption of such rice cultivars which accumulate high Cd, and precautions should be taken by farmers to build global food security.
Data availability
All data analyzed or generated during this study are included in this published article.
References
Acosta-Estrada BA, Gutiérrez-Uribe JA, Serna-Saldivarso (2019) Minor constituents and phytochemicals of the kernel. Corn. AACC International Press, Washington, D.C., pp 369–403. https://doi.org/10.1016/B978-0-12-811971-6.00014-0
Alengebawy A, Abdelkhalek ST, Qureshi SR, Wang MQ (2021) Heavy metals and pesticides toxicity in agricultural soil and plants: ecological risks and human health implications. Toxics 9(3):42. https://doi.org/10.3390/toxics9030042
Andersson MN, Wang HL, Nord A, Salmón P, Isaksson C (2015) Composition of physiologically important fatty acids in great tits differs between urban and rural populations on a seasonal basis. Front Ecol Evol 3:93. https://doi.org/10.3389/fevo.2015.00093
Arendt EK, Zannini E (2013) Cereal grains for the food and beverage industries. Elsevier, Amsterdam
Arif N, Sharma NC, Yadav V, Ramawat N, Dubey NK, Tripathi DK, Chauhan DK, Sahi S (2019) Understanding heavy metal stress in a rice crop: toxicity, tolerance mechanisms, and amelioration strategies. J Plant Biol 62:239–253. https://doi.org/10.1007/s12374-019-0112-4
Asaduzzaman M, Haque ME, Rahman J, Hasan SK, Ali MA, Akter MS, Ahmed M (2013) Comparisons of physiochemical, total phenol, flavanoid content and functional properties in six cultivars of aromatic rice in Bangladesh. Afr J Food Sci 7(8):198–203. https://doi.org/10.5897/AJFS2013.1001
Banerjee A, Roychoudhury A (2019) Differential regulation of defence pathways in aromatic and non-aromatic indica rice cultivars towards fluoride toxicity. Plant Cell Rep 38:1217–1233. https://doi.org/10.1007/s00299-019-02438-6
Banerjee D, Bairagi H, Mukhopadhyay S, Pal A, Bera D, Ray L (2010) Heavy metal contamination in fruits and vegetables in two districts of West Bengal, India. Elec J Env Agricult Food Chem Title 9(9):1423–1432
Barman F, Majumdar S, Arzoo SH, Kundu R (2020) Genotypic variation among 20 rice cultivars/landraces in response to cadmium stress grown locally in West Bengal, India. Plant Physiol Biochem 148:193–206. https://doi.org/10.1016/j.plaphy.2020.01.019
Bhattacharyya S, Roy S (2018) Qualitative and quantitative assessment of bioactive phytochemicals in gobindobhog and black rice, cultivated in West Bengal, India. Int J Pharm Sci Res 9(9):3845–3851. https://doi.org/10.13040/IJPSR.0975-8232.9(9).3845-51
Daulta R, Sridevi T, Garg VK (2021) Spatial distribution of heavy metals in rice grains, rice husk, and arable soil, their bioaccumulation and associated health risks in Haryana. India Toxin Reviews 40(4):859–871. https://doi.org/10.1080/15569543.2020.1793781
de Araújo RP, de Almeida AAF, Pereira LS, Mangabeira PA, Souza JO, Pirovani CP, Baligar VC (2017) Photosynthetic, antioxidative, molecular and ultrastructural responses of young cacao plants to Cd toxicity in the soil. Ecotoxicol Environ Saf 144:148–157. https://doi.org/10.1016/j.ecoenv.2017.06.006
DiNicolantonio JJ, Liu J, O’Keefe JH (2018) Thiamine and cardiovascular disease: a literature review. Prog Cardiovasc Dis 61(1):27–32. https://doi.org/10.1016/j.pcad.2018.01.009
Dubois M, Gilles KA, Hamilton JK, Rebers PT, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Eleazu CO, Eleazu KF, Ukamaka G, AdeoluT EV, Ezeorah B, Ilom J (2020) Nutrient and antinutrient composition and heavy metal and phenolic profiles of maize (zea mays) as affected by different processing techniques. ACS Food Sci Technol 1(1):113–123. https://doi.org/10.1021/acsfoodscitech.0c00045
Fahad S, Hussain S, Khan F, Wu C, Saud S, Hassan S, Huang J (2015) Effects of tire rubber ash and zinc sulfate on crop productivity and cadmium accumulation in five rice cultivars under field conditions. Environ Sci Pollut Res 22(16):12424–12434. https://doi.org/10.1007/s11356-015-4518-3
Garcés R, Mancha M (1993) One-step lipid extraction and fatty acid methyl esters preparation from fresh plant tissues. Anal Biochem 211(1):139–143. https://doi.org/10.1006/abio.1993.1244
Gill LW, Ring P, Higgins NM, Johnston PM (2014) Accumulation of heavy metals in a constructed wetland treating road runoff. Ecol Eng 70:133–139. https://doi.org/10.1016/j.ecoleng.2014.03.056
Goufo P, Trindade H (2014) Rice antioxidants: phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, γ-oryzanol, and phytic acid. Food Sci Nutr 2(2):75–104. https://doi.org/10.1002/fsn3.86
Grant CA, Buckley WT, Bailey LD, Selles F (1998) Cadmium accumulation in crops. Can J Plant Sci 78(1):1–17. https://doi.org/10.4141/P96-100
Guha T, Mukherjee A, Kundu R (2021) Nano-scale zero valent iron (nZVI) priming enhances yield, alters mineral distribution and grain nutrient content of Oryza sativa L. cv. Gobindobhog: a field study. J Plant Growth Regul. https://doi.org/10.1007/s00344-021-10335-0
Guo JJ, Tan x, Fu HL, Chen JX, Lin XX, Ma Y, Yang ZY (2018) Selection for Cd pollution- safe cultivars of Chinese kale (Brassica alboglabra L. H. Bailey) and biochemical mechanisms of the cultivar-dependent Cd accumulation involving in Cd subcellular distribution. J Agric Food Chem 66(8):1923–1934
Guo B, Hong C, Tong W, Xu M, Huang C, Yin H, Fu Q (2020) Health risk assessment of heavy metal pollution in a soil-rice system: a case study in the Jin-Qu Basin of China. Sci Rep 10(1):1–11. https://doi.org/10.1038/s41598-020-68295-6
Hussain B, Lin Q, Hamid Y, Sanaullah M, Di L, Khan MB, He Z, Yang X (2020) Foliage application of selenium and silicon nanoparticles alleviates Cd and Pb toxicity in rice (Oryza sativa L.). Sci Total Environ 712:136497. https://doi.org/10.1016/j.scitotenv.2020.136497
Hussain B, Ashraf MN, Abbas A, Li J, Farooq M (2021) Cadmium stress in paddy fields: effects of soil conditions and remediation strategies. Sci Total Environ 754:142188. https://doi.org/10.1016/j.scitotenv.2020.142188
Ismael MA, Elyamine AM, Moussa MG, Cai M, Zhao X, Hu C (2019) Cadmium in plants: uptake, toxicity, and its interactions with selenium fertilizers. Metallomics 11(2):255–277. https://doi.org/10.1039/c8mt00247a
Jehan S, Khattak SA, Muhammad S, Ahmad R, Farooq M, Khan S, Ali L (2018) Ecological and health risk assessment of heavy metals in the Hattar industrial estate, Pakistan. Toxin Rev. https://doi.org/10.1080/15569543.2018.1478858
Khalifa F, El-Halwagy ME, Hussein RH (2016) Clinical role of dietary thiamine on regulation of renal response to metabolic acidosis in adult rats. Biosci J (online) 32(2):543–549
Khan MSS, Basnet R, Islam SA, Shu Q (2019) Mutational analysis of OsPLDα1 reveals its involvement in phytic acid biosynthesis in rice grains. J Agric Food Chem 67(41):11436–11443. https://doi.org/10.1021/acs.jafc.9b05052
Kubier A, Wilkin RT, Pichler T (2019) Cadmium in soils and groundwater: a review. Appl Geochem 108:104388. https://doi.org/10.1016/j.apgeochem.2019.104388
Lamaison JLC, Carnet A (1990) Contents in main flavonoid compounds of Crataegus monogyna Jacq and Crataegus laevigata (Poiret) DC flowers at different development stages. Pharmaceutica Acta Helvetica 65(1):315–320
Dq Li, Chen G, Lu Q, Li Y, Wang J, Li H (2018a) Responses of two kidney bean (Phaseolus vulgaris) cultivars to the combined stress of sulfur deficiency and cadmium toxicity. Biometals 31:17–28. https://doi.org/10.1007/s10534-017-0052-8
Li DQ, Li WY, Lu Q, Li YT, Li N, Xu HJ, Ren ZL, Zhang YL, Wang JJ (2018b) Cadmium bioavailability well assessed by DGT and factors influencing cadmium accumulation in rice grains from paddy soils of three parent materials. J Soil Sediments 18(7):2552–2561. https://doi.org/10.1007/s11368-018-1950-2
Li N, Feng A, Liu N, Jiang Z, Wei S (2020) Silicon application improved the yield and nutritional quality while reduced cadmium concentration in rice. Environ Sci Pollut Res 27:20370–20379. https://doi.org/10.1007/s11356-020-08357-4
Lien KW, Pan MH, Ling MP (2021) Levels of heavy metal cadmium in rice (Oryza sativa L.) produced in Taiwan and probabilistic risk assessment for the Taiwanese population. Environ Sci Pollut Res 28:28381–28390. https://doi.org/10.1007/s11356-020-11902-w
Liu RH (2013) Health-promoting components of fruits and vegetables in the diet. Adv Nutr 4(3):384S-392S. https://doi.org/10.3945/an.112.003517
Liu J, Li K, Xu J, Liang J, Lu X, Yang J, Zhu Q (2003) Interaction of Cd and five mineral nutrients for uptake and accumulation in different rice cultivars and genotypes. Field Crop Res 83(3):271–281. https://doi.org/10.1016/S0378-4290(03)00077-7
Liu Y, Zhang C, Zhao Y, Sun S, Liu Z (2017) Effects of growing seasons and genotypes on the accumulation of cadmium and mineral nutrients in rice grown in cadmium contaminated soil. Sci Total Environ 579:1282–1288. https://doi.org/10.1016/j.scitotenv.2016.11.115
Lowry OH, Rosebrough NJ, Farr AL et al (1951) Protein measurement with the Folin phenol reagent. J BiolChem 193:265–275
Majumdar S, Barman F, Guha T, Kundu R (2022) Selection of cadmium non-accumulating rice genotypes to reduce health risk. Plant Archiv 22(1):364–374. https://doi.org/10.51470/PLANTARCHIVES.2022.v22.no1.057
Maksymiec W (2007) Signaling responses in plants to heavy metal stress. Acta Physiol Plant 29(3):177–187. https://doi.org/10.1007/s11738-007-0036-3
McCready RM, Guggolz J, Silviera V, Owens HS (1950) Determination of starch and amylose in vegetables. Anal Chem 22(9):1156–1158. https://doi.org/10.1021/ac60045a016
Memoli V, Eymar E, García-Delgado C, Esposito F, Santorufo L, De Marco A, Maisto G (2018) Total and fraction content of elements in volcanic soil: natural or anthropogenic derivation. Sci Total Environ 625:16–26. https://doi.org/10.1016/j.scitotenv.2017.12.223
Mohamadiun M, Dahrazma B, Saghravani SF, Darban AK (2018) Removal of cadmium from contaminated soil using iron (III) oxide nanoparticles stabilized with polyacrylic acid. J Environ Eng Landsc Manag 26(2):98–106. https://doi.org/10.3846/16486897.2017.1364645
Mu T, Zhou T, Li Z, Hu P, Luo Y, Christie P, Wu L (2020) Prediction models for rice cadmium accumulation in Chinese paddy fields and the implications in deducing soil thresholds based on food safety standards. Environ Pollut 258:113879. https://doi.org/10.1016/j.envpol.2019.113879
Nandagoapalan V, Doss A, Marimuthu C (2016) Phytochemical analysis of some traditional medicinal plants. BiosciDiscov 7:17–20
Niu Z, Ma J, Fang X, Xue Z, Ye Z (2022) Effects of application of rice husk biochar and limestone on cadmium accumulation in wheat under glasshouse and field conditions. Sci Rep 12(1):21929. https://doi.org/10.1038/s41598-022-25927-3
Okolie NP, Falodun A, Davids O (2014) Evaluation of the antioxidant activity of root extract of pepper fruit (Dennetiatripetala), and it’s potential for the inhibition of lipid peroxidation. Afr J Traditional Complement Altern Med 11(3):221–227. https://doi.org/10.4314/ajtcam.v11i3.31
Paul S, Guha T, Dey S, Paul S, Kundu R (2022) Amelioration of cadmium toxicity by enhancing nitrogen assimilation and photosynthetic activity by two different nitrogen supplements in rice (Oryza sativa L.) cv. Lalat. Plant Stress 4:100082. https://doi.org/10.1016/j.stress.2022.100082
Raghuvanshi R, Raut VV, Pandey M, Jeyakumar S, Verulkar S, Suprasanna P, Srivastava AK (2022) Arsenic and cadmium induced macronutrient deficiencies trigger contrasting gene expression changes in rice. Environ Pollut 300:118923. https://doi.org/10.1016/j.envpol.2022.118923
Ristić-Medić DK, Vučić VM, Takić MM, Karadžić I, Glibetić MD (2013) Polyunsaturated fatty acids in health and disease. J Serb Chem Soc 78(9):1269–1289. https://doi.org/10.2298/JSC130402040R
Saengwilai P, Meeinkuirt W (2021) Cadmium (Cd) and zinc (Zn) accumulation by Thai rice varieties and health risk assessment in a Cd–Zn co-contaminated paddy field: effect of soil amendments. Environ Geochem Health. https://doi.org/10.1007/s10653-021-00858-6
Sarwar N, Malhi SS, Zia MH, Naeem A, Bibi S, Farid G (2010) Role of mineral nutrition in minimizing cadmium accumulation by plants. J Sci Food Agric 90(6):925–937. https://doi.org/10.1002/jsfa.3916
Simmler M, Ciadamidaro L, Schulin R, Madejón P, Reiser R, Clucas L, Robinson B (2013) Lignite reduces the solubility and plant uptake of cadmium in pasturelands. Environ Sci Technol 47(9):4497–4504. https://doi.org/10.1021/es303118a
Singh MC, Pandey PK, Singh S, Singh AK, Heisnam P, Shantikumar L, Singh BK (2018) Phyto-chemical and nutritional analysis of Deku and Itanagar indigenous rice cultivars consumed by the Adi tribes of Arunachal Pradesh, India. J Pharmacognosy Phytochem 7(6):64–69
Su L, Yin JJ, Charles D et al (2007) Total phenolic contents, chelating capacities, and radical-scavenging properties of black peppercorn, nutmeg, rosehip, cinnamon and oregano leaf. Food Chem 100(3):990–997. https://doi.org/10.1016/j.foodchem.2005.10.058
Surendran U, Raja P, Jayakumar M, Subramoniam SR (2021) Use of efficient water saving techniques for production of rice in India under climate change scenario: a critical review. J Cleaner Prod 309:127272. https://doi.org/10.1016/j.jclepro.2021.127272
Takahashi R, Ishimaru Y, Nakanishi H, Nishizawa NK (2011) Role of the iron transporter OsNRAMP1 in cadmium uptake and accumulation in rice. Plant Signal Behav 6(11):1813–1816. https://doi.org/10.4161/psb.6.11.17587
Vaintraub IA, Lapteva NA (1988) Colorimetric determination of phytate in unpurified extracts of seeds and the products of their processing. Anal Biochem 175(1):227–230. https://doi.org/10.1016/0003-2697(88)90382-X
Wordu GO, Catherine Orisa A, Ameka Hamilton China M (2021) Nutrient and anti-nutrient composition of four rice varieties in port harcourt metropolis. Asian Food Sci J. https://doi.org/10.9734/AFSJ/2021/v20i1130379
Wylie-Rosett J, Aebersold K, Conlon B, Isasi CR, Ostrovsky NW (2013) Health effects of low-carbohydrate diets: where should new research go? Curr Diabetes Rep 13(2):271–278. https://doi.org/10.1007/s11892-012-0357-5
Xiaofang LI, Dongmei ZHOU (2019) A meta-analysis on phenotypic variation in cadmium accumulation of rice and wheat: implications for food cadmium risk control. Pedosphere 29(5):545–553. https://doi.org/10.1016/S1002-0160(19)60828-3
Yang Y, Li Y, Chen W, Wang M, Wang T, Dai Y (2020) Dynamic interactions between soil cadmium and zinc affect cadmium phytoavailability to rice and wheat: regional investigation and risk modeling. Environ Pollut 267:115613. https://doi.org/10.1016/j.envpol.2020.115613
Yu HY, Li FB, Liu CS, Huang W, Liu TX, Yu WM (2016) Iron redox cycling coupled to transformation and immobilization of heavy metals: implications for paddy rice safety in the red soil of South China. Adv Agron 137:279–317. https://doi.org/10.1016/bs.agron.2015.12.006
Yu X, Chu M, Chu C, Du Y, Shi J, Liu X, Liu Y, Zhang H, Zhang Z, Yan N (2020) Wild rice (Zizania spp.): a review of its nutritional constituents, phytochemicals, antioxidant activities, and health-promoting effects. Food Chem 331:127293. https://doi.org/10.1016/j.foodchem.2020.127293
Zeddies S, De Cuyper IM, van der Meer PF, Daal BB, de Korte D, Gutiérrez L, Thijssen-Timmer DC (2014) Pathogen reduction treatment using riboflavin and ultraviolet light impairs platelet reactivity toward specific agonists in vitro. Transfusion 54(9):2292–2300. https://doi.org/10.1111/trf.12636
Zhang J, Shi Z, Ni S, Wang X, Liao C, Wei F (2021) Source identification of Cd and Pb in typical farmland topsoil in the southwest of China: a case study. Sustainability 13(7):3729. https://doi.org/10.3390/su13073729
Zhao X, Jiang T, Du B (2014) Effect of organic matter and calcium carbonate on behaviors of cadmium adsorption–desorption on/from purple paddy soils. Chemosphere 99:41–48. https://doi.org/10.1016/j.chemosphere.2013.09.030
Acknowledgements
This research work was financially supported by West Bengal Department of Science and Technology and Biotechnology Department [project no. 1925(Sanc.)/STBT-11012(15)/18/2019-STSEC]. AB acknowledges WBDSTBT for providing her fellowship and thankful to Dept. of Botany, Lady Brabourne College for providing laboratory facility. The authors wish to acknowledge SAIF; I.I.T. Bombay for ICP-AES analysis and thankful to the Agriculture and Development Office (ADO) of North 24 Parganas for providing relevant information to accomplish field survey. Authors are also grateful to Prof. Rita Kundu, Head Department of Botany, University of Calcutta, and Miss. Swarnali Dey, Research Scholar, University of Calcutta for their valuable advice. AB designed and carried out all experiments, analyzed data, performed whole statistical analysis and wrote manuscript text. SP1 supervised the work, helped in writing and drafting the manuscript. SP2 provided suggestions and reviewed the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Biswas, A., Pal, S. & Paul, S. Recent status of cadmium-contaminated paddy field and its impact on food safety: a field study over two years in North 24 Parganas, West Bengal, India. J. Crop Sci. Biotechnol. 27, 105–121 (2024). https://doi.org/10.1007/s12892-023-00216-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-023-00216-1